| |
| Names | |
|---|---|
| Other names
Aplysiatoxin
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
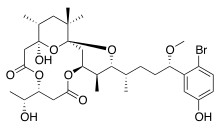
| C32H47BrO10 | |
| Molar mass | 671.614 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Aplysiatoxin is a cyanotoxin produced by certain cyanobacteria species. It is used as a defensive secretion to protect these cyanobacteria from predation by fish, being a potent irritant and carcinogen, by acting as a powerful activator of protein kinase C. [1] [2] [3] [4] While this action has a tumour-promoting effect, protein kinase C activation can be medically beneficial for some other applications, and synthetic analogues of aplysiatoxin have been researched for anti-cancer effects. [5] [6] [7]
See also
References
- ^ Kato Y, Scheuer PJ. Aplysiatoxin and debromoaplysiatoxin, constituents of the marine mollusk Stylocheilus longicauda (Quoy and Gaimard, 1824). Journal of the American Chemical Society. 1974 Apr 3;96(7):2245-6. PMID 4833645
- ^ Weinstein IB, Arcoleo J, Backer J, Jeffrey A, Hsiao WL, Gattoni-Celli S, Kirschmeier P, Okin E. Molecular mechanisms of tumor promotion and multistage carcinogenesis. Princess Takamatsu Symposia. 1983;14:59-74. PMID 6097583
- ^ Arcoleo JP, Weinstein IB. Activation of protein kinase C by tumor promoting phorbol esters, teleocidin and aplysiatoxin in the absence of added calcium. Carcinogenesis. 1985 Feb;6(2):213-7. PMID 3156004
- ^ Nagai H, Yasumoto T, Hokama Y. Aplysiatoxin and debromoaplysiatoxin as the causative agents of a red alga Gracilaria coronopifolia poisoning in Hawaii. Toxicon. 1996 Jul;34(7):753-61. PMID 8843576
- ^ Watanabe M, Kawase Y, Tanabe J, Min KR, Mue S, Ohuchi K. Suppression of interleukin-1 alpha production by protein kinase C activators in human vascular endothelial cells. Journal of Pharmacology and Experimental Therapeutics. 1995 Feb;272(2):808-14. PMID 7853198
- ^ Nakagawa Y, Yanagita RC, Hamada N, Murakami A, Takahashi H, Saito N, Nagai H, Irie K. A simple analogue of tumor-promoting aplysiatoxin is an antineoplastic agent rather than a tumor promoter: development of a synthetically accessible protein kinase C activator with bryostatin-like activity. Journal of the American Chemical Society. 2009 Jun 10;131(22):7573-9. PMID 19449873
- ^ Yanagita RC, Kamachi H, Tanaka K, Murakami A, Nakagawa Y, Tokuda H, Nagai H, Irie K. Role of the phenolic hydroxyl group in the biological activities of simplified analogue of aplysiatoxin with antiproliferative activity. Bioorganic & Medicinal Chemistry Letters. 2010 Oct 15;20(20):6064-6. PMID 20817520

| |
| Names | |
|---|---|
| Other names
Aplysiatoxin
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C32H47BrO10 | |
| Molar mass | 671.614 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Aplysiatoxin is a cyanotoxin produced by certain cyanobacteria species. It is used as a defensive secretion to protect these cyanobacteria from predation by fish, being a potent irritant and carcinogen, by acting as a powerful activator of protein kinase C. [1] [2] [3] [4] While this action has a tumour-promoting effect, protein kinase C activation can be medically beneficial for some other applications, and synthetic analogues of aplysiatoxin have been researched for anti-cancer effects. [5] [6] [7]
See also
References
- ^ Kato Y, Scheuer PJ. Aplysiatoxin and debromoaplysiatoxin, constituents of the marine mollusk Stylocheilus longicauda (Quoy and Gaimard, 1824). Journal of the American Chemical Society. 1974 Apr 3;96(7):2245-6. PMID 4833645
- ^ Weinstein IB, Arcoleo J, Backer J, Jeffrey A, Hsiao WL, Gattoni-Celli S, Kirschmeier P, Okin E. Molecular mechanisms of tumor promotion and multistage carcinogenesis. Princess Takamatsu Symposia. 1983;14:59-74. PMID 6097583
- ^ Arcoleo JP, Weinstein IB. Activation of protein kinase C by tumor promoting phorbol esters, teleocidin and aplysiatoxin in the absence of added calcium. Carcinogenesis. 1985 Feb;6(2):213-7. PMID 3156004
- ^ Nagai H, Yasumoto T, Hokama Y. Aplysiatoxin and debromoaplysiatoxin as the causative agents of a red alga Gracilaria coronopifolia poisoning in Hawaii. Toxicon. 1996 Jul;34(7):753-61. PMID 8843576
- ^ Watanabe M, Kawase Y, Tanabe J, Min KR, Mue S, Ohuchi K. Suppression of interleukin-1 alpha production by protein kinase C activators in human vascular endothelial cells. Journal of Pharmacology and Experimental Therapeutics. 1995 Feb;272(2):808-14. PMID 7853198
- ^ Nakagawa Y, Yanagita RC, Hamada N, Murakami A, Takahashi H, Saito N, Nagai H, Irie K. A simple analogue of tumor-promoting aplysiatoxin is an antineoplastic agent rather than a tumor promoter: development of a synthetically accessible protein kinase C activator with bryostatin-like activity. Journal of the American Chemical Society. 2009 Jun 10;131(22):7573-9. PMID 19449873
- ^ Yanagita RC, Kamachi H, Tanaka K, Murakami A, Nakagawa Y, Tokuda H, Nagai H, Irie K. Role of the phenolic hydroxyl group in the biological activities of simplified analogue of aplysiatoxin with antiproliferative activity. Bioorganic & Medicinal Chemistry Letters. 2010 Oct 15;20(20):6064-6. PMID 20817520