| Company type | Aktieselskab |
|---|---|
| Industry | Pharmaceuticals, health care [1] |
| Founded | 1958 [1] |
| Headquarters | Copenhagen, Denmark [1] |
| Website | http://www.xellia.com |
Xellia ApS is a Danish multinational pharmaceutical and life sciences company headquartered in Copenhagen [1] specialized in the production of anti-biotics, including Vancomycin [2] and Bacitracin. [3]
The company's US base of operations is in Buffalo Grove, Illinois, [4] [5] with additional facilities in Ohio, and North Carolina. [6] [7] In 2018, Xellia's sales in the United States accounted for 60% of its total revenue. [5]
Products
The company's main output is bulk drug chemicals which are wholesaled to other companies for packaging and distribution. Recently some prepackaged goods have been produced.
Active drug substances manufactured are: [8]
| ATC | Substance | Structural formula | Formulation for sale | Production started |
|---|---|---|---|---|
|
D06AX05 (
WHO) (
FK) J01XX10 ( WHO) ( FK) R02AB04 ( WHO) ( FK) QA07AA93 ( WHO) ( FK) |
Bacitracin ( MT) [9] [10] |

|
Bacitracin | 1952 |
|
D06AX05 (
WHO) (
FK) J01XX10 ( WHO) ( FK) R02AB04 ( WHO) ( FK) QA07AA93 ( WHO) ( FK) |
Bacitracin ( MT) [9] [10] |

|
Bacitracin zinc | 1958 |
| Colistimethate natrium (CMS) | ||||
|
A07AA10 (
WHO) (
FK) J01XB01 ( WHO) ( FK) QJ51XB01 ( WHO) ( FK) |
Colistin |

|
Colistin sulfate | 1969 |
| J01X X09 | Daptomycin ( MT) [9] [11] |

|
Daptomycin | |
| R02AB30 ( WHO) ( FK) |
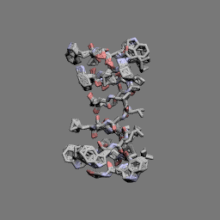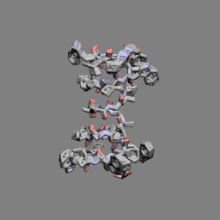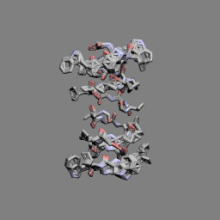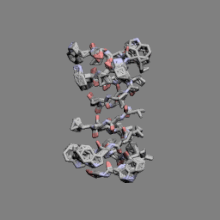
|
Gramicidin | ||
|
A07AA05 (
WHO) (
FK) J01XB02 ( WHO) ( FK) S01AA18 ( WHO) ( FK) S02AA11 ( WHO) ( FK) S03AA03 ( WHO) ( FK) QJ51XB02 ( WHO) ( FK) |
Polymyxin B ( MT) [9] [12] |

|
Polymyxin B-sulfate | 1968 |
|
J01GB01 (
WHO) (
FK) S01AA12 ( WHO) ( FK) |
Tobramycin ( MT) [9] [13] |

|
Tobramycin sulfate | 2005 |
|
D06AX08 (
WHO) (
FK) R02AB02 ( WHO) ( FK) S01AA05 ( WHO) ( FK) |
Tyrothricin |

|
Tyrothricin | |
|
A07AA09 (
WHO) (
FK) J01XA01 ( WHO) ( FK) |
Vancomycin ( MT) [9] [14] |

|
Vancomycin hydrochloride | 2003 |
|
A01AB04 (
WHO) (
FK) A07AA07 ( WHO) ( FK) G01AA03 ( WHO) ( FK) J02AA01 ( WHO) ( FK) |
Amfotericin B ( MT) [9] [15] |

|
Amfotericin B | 1985 |
| J02AX04 ( WHO) ( FK) | Caspofungin ( MT) [9] [16] |

|
Caspofungin acetate | |
|
D07AC17 (
WHO) (
FK) R01AD08 ( WHO) ( FK) R01AD58 ( WHO) ( FK) R03BA05 ( WHO) ( FK) |
Fluticasone ( MT) [9] [17] |

|
Fluticasone propionate | |
|
D07AC13 (
WHO) (
FK) R01AD09 ( WHO) ( FK) R03BA07 ( WHO) ( FK) |
Mometasone ( MT) [9] [18] |

|
Mometasone furoate |
Discontinued products
Previously the company also produced:
| Ingredient | Produced |
|---|---|
| Neomycin | 1952-? |
| Tetracycline | 1961-? |
Partnerships
In 2019, Xellia partnered with Civica Rx to produce generic Vancomycin and Daptomycin. The partnership's stated aim is to remedy chronic drug shortages that have affected the American pharmaceutical market. [4] [19] [20] [21] [22]
China-US Trade War
Xellia has been discussed as a potential alternative to Chinese companies for anti-biotic primary ingredients in the context of the China-United States trade war. [23]
See also
References
- ^ a b c d "Xellia Pharmaceuticals ApS". Bloomberg. 2019-05-15. Retrieved 15 July 2019.
- ^ "VANCOMYCIN HYDROCHLORIDE". U.S. National Library of Medicine. 2019-05-15. Retrieved 10 September 2019.
- ^ "BACITRACIN- bacitracin injection". U.S. National Library of Medicine. Retrieved 10 September 2019.
- ^ a b "Xellia to produce Civica antibiotics". Chicago Daily Herald. 2019-05-15. Retrieved 15 July 2019.
- ^ a b "Danish pharmaceutical company picks Chicago area for new office". Chicago Business Journal. Retrieved 15 July 2019.
- ^ "Will China Save the American Economy?". The Atlantic. 2017-06-27. Retrieved 15 July 2019.
- ^ "Denmark-based pharmaceuticals company acquires Raleigh manufacturing plant". Triangle Business Journal. Retrieved 21 July 2019.
- ^ produktet listen er hentet fra Api på xellia.com Archived 2015-08-27 at the Wayback Machine 15. august 2014 og supplert med informasjon fra Vår arv Archived 2015-08-24 at the Wayback Machine hentes samme dag
- ^ a b c d e f g h i j "Godkjent eller ikke godkjent – hvilke statuser kan et legemiddel ha?". Archived from the original on 28 August 2016. Retrieved 27 August 2016.
- ^ a b "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 4 September 2016.
- ^ "Two crucial antibiotics are the first products of Utah's new nonprofit drug company". Salt Lake Tribune. Retrieved 15 July 2019.
- ^ "Drugmaker Created To Reduce Shortages And Prices Unveils Its First Products". NPR. Retrieved 15 July 2019.
- ^ Evans, Melanie (15 May 2019). "Hospital Drug-Making Venture Picks Antibiotics as First Products". Wall Street Journal. Retrieved 15 July 2019.
- ^ "Unique initiative to lower drug prices signs manufacturer". Houston Chronicle. 20 May 2019. Retrieved 22 July 2019.
- ^ "Trade War With China Threatens To Make Problems With Generic Medicine Worse". Forbes. Retrieved 15 July 2019.
| Company type | Aktieselskab |
|---|---|
| Industry | Pharmaceuticals, health care [1] |
| Founded | 1958 [1] |
| Headquarters | Copenhagen, Denmark [1] |
| Website | http://www.xellia.com |
Xellia ApS is a Danish multinational pharmaceutical and life sciences company headquartered in Copenhagen [1] specialized in the production of anti-biotics, including Vancomycin [2] and Bacitracin. [3]
The company's US base of operations is in Buffalo Grove, Illinois, [4] [5] with additional facilities in Ohio, and North Carolina. [6] [7] In 2018, Xellia's sales in the United States accounted for 60% of its total revenue. [5]
Products
The company's main output is bulk drug chemicals which are wholesaled to other companies for packaging and distribution. Recently some prepackaged goods have been produced.
Active drug substances manufactured are: [8]
| ATC | Substance | Structural formula | Formulation for sale | Production started |
|---|---|---|---|---|
|
D06AX05 (
WHO) (
FK) J01XX10 ( WHO) ( FK) R02AB04 ( WHO) ( FK) QA07AA93 ( WHO) ( FK) |
Bacitracin ( MT) [9] [10] |

|
Bacitracin | 1952 |
|
D06AX05 (
WHO) (
FK) J01XX10 ( WHO) ( FK) R02AB04 ( WHO) ( FK) QA07AA93 ( WHO) ( FK) |
Bacitracin ( MT) [9] [10] |

|
Bacitracin zinc | 1958 |
| Colistimethate natrium (CMS) | ||||
|
A07AA10 (
WHO) (
FK) J01XB01 ( WHO) ( FK) QJ51XB01 ( WHO) ( FK) |
Colistin |

|
Colistin sulfate | 1969 |
| J01X X09 | Daptomycin ( MT) [9] [11] |

|
Daptomycin | |
| R02AB30 ( WHO) ( FK) |

|
Gramicidin | ||
|
A07AA05 (
WHO) (
FK) J01XB02 ( WHO) ( FK) S01AA18 ( WHO) ( FK) S02AA11 ( WHO) ( FK) S03AA03 ( WHO) ( FK) QJ51XB02 ( WHO) ( FK) |
Polymyxin B ( MT) [9] [12] |

|
Polymyxin B-sulfate | 1968 |
|
J01GB01 (
WHO) (
FK) S01AA12 ( WHO) ( FK) |
Tobramycin ( MT) [9] [13] |

|
Tobramycin sulfate | 2005 |
|
D06AX08 (
WHO) (
FK) R02AB02 ( WHO) ( FK) S01AA05 ( WHO) ( FK) |
Tyrothricin |

|
Tyrothricin | |
|
A07AA09 (
WHO) (
FK) J01XA01 ( WHO) ( FK) |
Vancomycin ( MT) [9] [14] |

|
Vancomycin hydrochloride | 2003 |
|
A01AB04 (
WHO) (
FK) A07AA07 ( WHO) ( FK) G01AA03 ( WHO) ( FK) J02AA01 ( WHO) ( FK) |
Amfotericin B ( MT) [9] [15] |

|
Amfotericin B | 1985 |
| J02AX04 ( WHO) ( FK) | Caspofungin ( MT) [9] [16] |

|
Caspofungin acetate | |
|
D07AC17 (
WHO) (
FK) R01AD08 ( WHO) ( FK) R01AD58 ( WHO) ( FK) R03BA05 ( WHO) ( FK) |
Fluticasone ( MT) [9] [17] |

|
Fluticasone propionate | |
|
D07AC13 (
WHO) (
FK) R01AD09 ( WHO) ( FK) R03BA07 ( WHO) ( FK) |
Mometasone ( MT) [9] [18] |

|
Mometasone furoate |
Discontinued products
Previously the company also produced:
| Ingredient | Produced |
|---|---|
| Neomycin | 1952-? |
| Tetracycline | 1961-? |
Partnerships
In 2019, Xellia partnered with Civica Rx to produce generic Vancomycin and Daptomycin. The partnership's stated aim is to remedy chronic drug shortages that have affected the American pharmaceutical market. [4] [19] [20] [21] [22]
China-US Trade War
Xellia has been discussed as a potential alternative to Chinese companies for anti-biotic primary ingredients in the context of the China-United States trade war. [23]
See also
References
- ^ a b c d "Xellia Pharmaceuticals ApS". Bloomberg. 2019-05-15. Retrieved 15 July 2019.
- ^ "VANCOMYCIN HYDROCHLORIDE". U.S. National Library of Medicine. 2019-05-15. Retrieved 10 September 2019.
- ^ "BACITRACIN- bacitracin injection". U.S. National Library of Medicine. Retrieved 10 September 2019.
- ^ a b "Xellia to produce Civica antibiotics". Chicago Daily Herald. 2019-05-15. Retrieved 15 July 2019.
- ^ a b "Danish pharmaceutical company picks Chicago area for new office". Chicago Business Journal. Retrieved 15 July 2019.
- ^ "Will China Save the American Economy?". The Atlantic. 2017-06-27. Retrieved 15 July 2019.
- ^ "Denmark-based pharmaceuticals company acquires Raleigh manufacturing plant". Triangle Business Journal. Retrieved 21 July 2019.
- ^ produktet listen er hentet fra Api på xellia.com Archived 2015-08-27 at the Wayback Machine 15. august 2014 og supplert med informasjon fra Vår arv Archived 2015-08-24 at the Wayback Machine hentes samme dag
- ^ a b c d e f g h i j "Godkjent eller ikke godkjent – hvilke statuser kan et legemiddel ha?". Archived from the original on 28 August 2016. Retrieved 27 August 2016.
- ^ a b "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 27 August 2016.
- ^ "Legemiddelsøk". Retrieved 4 September 2016.
- ^ "Two crucial antibiotics are the first products of Utah's new nonprofit drug company". Salt Lake Tribune. Retrieved 15 July 2019.
- ^ "Drugmaker Created To Reduce Shortages And Prices Unveils Its First Products". NPR. Retrieved 15 July 2019.
- ^ Evans, Melanie (15 May 2019). "Hospital Drug-Making Venture Picks Antibiotics as First Products". Wall Street Journal. Retrieved 15 July 2019.
- ^ "Unique initiative to lower drug prices signs manufacturer". Houston Chronicle. 20 May 2019. Retrieved 22 July 2019.
- ^ "Trade War With China Threatens To Make Problems With Generic Medicine Worse". Forbes. Retrieved 15 July 2019.