| This is an archive of past discussions. Do not edit the contents of this page. If you wish to start a new discussion or revive an old one, please do so on the current talk page. |
| Archive 2015 | ← | Archive 2020 | Archive 2021 | Archive 2022 |
ChemBox parameters for chiral compounds
It would be nice if Wikipedia could show data on various forms of chiral compounds. For example:
- R and S enantiomers should have identical melting points, densities
- the same data for the racemate (1:1 mixture of R and S enantiomers) would differ, typically higher mp, higher d (I am ignoring solubility, odor, etc)
For species with two equivalent chiral centers (like tartaric acid), three sets of data are ideal
- R- and S-
- racemate
- meso
but the Chembox parameters do not seem to accommodate multiple m.p.'s, multiple densities, etc. Advice welcome.-- Smokefoot ( talk) 15:25, 1 January 2022 (UTC)
| Properties | |
|---|---|
| Density | 1 g/cm3 (R) 2 g/cm3 (S) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
- Maybe use </br> to separate multiple datas? --
Nucleus hydro elemon (
talk)
15:31, 1 January 2022 (UTC)
- Use <br/> and do not use </br> which should be closing a <br>. Graeme Bartlett ( talk) 22:25, 2 January 2022 (UTC)
- (ec) You're right on both counts: it's desired, and currently not possible.
- Chembox can handle separate compounds, by label and showing (called 'by index' here). See
linalool for example. However, that is only possible within |Section1={{Chembox Identifiers}} CASNo, ChEMBL, SMILES, ... only. So |Section3={{Chembox Properties}} cannot reuse those same indexes (no
|MeltingPt2=for/with|CASNo2=then). - Limitation is because of current
|SectionN=subtemplates setup. See doc:Chembox design. At the moment, an RfC § RfC on making chembox an infobox is proposing to remove this limit (by using Lua module). When all parameters are at level 1, right in {{Chembox}} like|Name=is today, the indexing can be (programmed to) be reused throughout. - DePiep ( talk) 15:44, 1 January 2022 (UTC)- Thank you both for helpful guidance. I tried and tried
|MeltingPt2=, with and without _notes, with and without _Comments. The </br> fix works fine with tartaric acid. -- Smokefoot ( talk) 15:52, 1 January 2022 (UTC)- Yes, if it shows well all's well. - DePiep ( talk) 16:00, 1 January 2022 (UTC)
- Another kludge is to have several Chembox Properties templates in different sections of the chembox. But this will make it harder to convert to other formats later. But I suppose using br has the same issue. Graeme Bartlett ( talk) 20:44, 2 January 2022 (UTC)
- Thank you both for helpful guidance. I tried and tried
| Properties | |
|---|---|
| Density | 1 g/cm3 (R) |
| Properties | |
| Density | 2 g/cm3 (S) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
- Also note that due to
parity violation from the
weak force, properties of enantiomers should differ slightly. Just this has not yet been detected yet. Also experiments may yield different results, or have never been performed, so don't assume property(R)≡property(S).
Graeme Bartlett (
talk)
20:55, 2 January 2022 (UTC)
- If repetition of Properties section floats your boat, you can use it (Change editors should solve it). However, it is bit hard to specify the substance (R, S) with it, I guess. <br/> input will have to be handled anyway (when changes happen), so don't hold back. - DePiep ( talk) 22:07, 2 January 2022 (UTC)
- Just asking (not promising): @
Graeme Bartlett: which Properties parameters would you like to have indexed? (~same setup as Identifiers maybe) -
DePiep (
talk)
22:07, 2 January 2022 (UTC)
- The main use for this for me would be: natural products that may end with A, B, C etc; different hydrates of a salt; as well as enantomers. Sometimes with drugs, a salt (eg hydrochloride) may be included with a base article, but this would mainly affect the drugbox. For the the A B C compounds, or hydrates, an indexed formula would be good (and I suppose that means C H O etc too)
Graeme Bartlett (
talk)
22:23, 2 January 2022 (UTC)
- The traditional enantio-sensitive parameters in (my) ranked order of importance: melting point, solubility, density, odor. By "indexing", can one ensure that CASNo2 is tied to MeltingPt2 to Density2 etc? This arrangement ensures that each data goes with the proper isomer. -- Smokefoot ( talk) 23:07, 2 January 2022 (UTC)
- The main use for this for me would be: natural products that may end with A, B, C etc; different hydrates of a salt; as well as enantomers. Sometimes with drugs, a salt (eg hydrochloride) may be included with a base article, but this would mainly affect the drugbox. For the the A B C compounds, or hydrates, an indexed formula would be good (and I suppose that means C H O etc too)
Graeme Bartlett (
talk)
22:23, 2 January 2022 (UTC)
- Also note that due to
parity violation from the
weak force, properties of enantiomers should differ slightly. Just this has not yet been detected yet. Also experiments may yield different results, or have never been performed, so don't assume property(R)≡property(S).
Graeme Bartlett (
talk)
20:55, 2 January 2022 (UTC)
- re GB: Looks like you misunderstood. I meant to say: you can use either way to make a good IB Chembox for thhose situation: multiple substance properties. That is: reuse |SectionN=, |SectionM={{Chembox Properties}} as in the demo, and use <br/> in single input
|MeltinghPt=(or {{ ubl}}). If and when IB Chemboc code changes (think Lua) the Change process will have to dal with it, not you today ;-) - re Smokefoot: sure
|CASNo2=≘|MeltingPt2=(qctually up to the article editor to maintain this numbercheck). Possibly one needs to c/p the indexlabels (index2=R, index3=S) from Identifiers into Props for reuse in output (because Section Props cannot read Section Ident's input). - Alas, won't happen soon tbh. - DePiep ( talk) 05:31, 3 January 2022 (UTC)
More input to the discussion of several fluorines-related templates would be welcome. -- Leyo 20:56, 3 January 2022 (UTC)
Nomination of Caffeine (data page) for deletion
The article will be discussed at Wikipedia:Articles for deletion/Caffeine (data page) until a consensus is reached, and anyone, including you, is welcome to contribute to the discussion. The nomination will explain the policies and guidelines which are of concern. The discussion focuses on high-quality evidence and our policies and guidelines.
Users may edit the article during the discussion, including to improve the article to address concerns raised in the discussion. However, do not remove the article-for-deletion notice from the top of the article.
DePiep ( talk) 14:05, 14 January 2022 (UTC)
Nomination of Butadiene (data page) for deletion
The article will be discussed at Wikipedia:Articles for deletion/Caffeine (data page) until a consensus is reached, and anyone, including you, is welcome to contribute to the discussion. The nomination will explain the policies and guidelines which are of concern. The discussion focuses on high-quality evidence and our policies and guidelines.
Users may edit the article during the discussion, including to improve the article to address concerns raised in the discussion. However, do not remove the article-for-deletion notice from the top of the article.
DePiep ( talk) 12:53, 15 January 2022 (UTC)
List of low quality chemical diagrams on Commons
Following on from Leyo's last I've just now become aware of commons:Category:Low_quality_chemical_diagrams/expired. Current contents 484 images. -- Project Osprey ( talk) 23:14, 18 January 2022 (UTC)
- The non-/expired cat has another 30. And
commons:Category:Disputed_chemical_diagrams and its /expired set are another >200 that might be even more important to fix.
DMacks (
talk)
00:05, 19 January 2022 (UTC)
- Would it be possible have new images in these categories appear on Wikipedia:WikiProject Chemistry/Article alerts? Simply switching to new images is not enough, the wrong ones must be deleted at the same time, otherwise we risk them being reused later on. -- Project Osprey ( talk) 21:44, 21 January 2022 (UTC)
Drawing challenge: Antimony potassium tartrate
Can anyone think of a good way of drawing antimony potassium tartrate? The current image (left) has numerous failings in addition to being plain ugly. I did a search on Commons before coming here which revealed the image on the right (in use on no fewer than 8 non-english wikis), I think it's my favourite structural drawing ever - somehow almost cubist. -- Project Osprey ( talk) 21:42, 5 December 2021 (UTC)
- An important question is, "what is more correct?". Per our article, with cited ref, the structure on the left is missing 3D details that our article actually discusses as a salient detail of this substance. The structure on the right agrees with
doi:10.1016/j.ica.2004.11.013, that it's a chiral cage with square–pyramidal Sb atoms. The structure on the right actually does have symmetry: its perspective is looking down a C2 axis.
DMacks (
talk)
04:23, 6 December 2021 (UTC)
- I didn't see that axis of symmetry, to me it's not very clear. I'd argue that it's showing closer to
seesaw molecular geometry. In any event both pictures fail on their depiction of formal charges. I've found at least 5 x-ray studies of this from the 1960s and 70s, there was apparently some disagreement as to its structure. Would two pictures be better? A flat one showing connectivity and a 3D model showing structure? --
Project Osprey (
talk)
10:34, 6 December 2021 (UTC)
- I agree with
DMacks Interestingly, the CCDC's entry for the X-ray structure (based on the reference he gave) has an even worse drawing than ours, although the actual structure
seen here is fine! That reference needs to be added the article, I think, plus a discussion of the controversy if that's still not sorted out.
Mike Turnbull (
talk)
10:53, 6 December 2021 (UTC)
- Not seen that interface before - handy! (although it seems to object to getting too many queries). These are what I've found so far
- 1965 doi: 10.1107/S0365110X65003080
- 1969 doi: 10.1016/0022-1902(69)80218-6
- 1972 doi: 10.1016/0022-1902(72)80229-X
- 1970 doi: 10.1107/S0567740870002133 - Structure
- 1974 deuterated doi: 10.1016/S0020-1693(00)92617-3 - structure
- I cannot access all of them, but I'm not seeing any difference in connectivity which would effect how to draw a 2-D image. --
Project Osprey (
talk)
11:15, 6 December 2021 (UTC)
- Part of the problem is that one needs to be careful which isomers are present in the structures used for the X-rays. (2R,3R)-tartaric acid is the "natural" isomer, hence the one that should be used for tartar emetic but its enantiomer (2S,3S)-tartaric acid and even the meso diastereomer, (2R,3S)-tartaric acid may have been studied and X-rayed. And that's before considerations of hydration! Mike Turnbull ( talk) 11:36, 6 December 2021 (UTC)
- Not seen that interface before - handy! (although it seems to object to getting too many queries). These are what I've found so far
- I agree with
DMacks Interestingly, the CCDC's entry for the X-ray structure (based on the reference he gave) has an even worse drawing than ours, although the actual structure
seen here is fine! That reference needs to be added the article, I think, plus a discussion of the controversy if that's still not sorted out.
Mike Turnbull (
talk)
10:53, 6 December 2021 (UTC)
- I didn't see that axis of symmetry, to me it's not very clear. I'd argue that it's showing closer to
seesaw molecular geometry. In any event both pictures fail on their depiction of formal charges. I've found at least 5 x-ray studies of this from the 1960s and 70s, there was apparently some disagreement as to its structure. Would two pictures be better? A flat one showing connectivity and a 3D model showing structure? --
Project Osprey (
talk)
10:34, 6 December 2021 (UTC)

This kind of thing is usually easier to show with a 2D snapshot of a 3D model, with an accompanying skeletal formula. -- Ben ( talk) 15:58, 6 December 2021 (UTC)
- Yes: Ben shows us how its done. but please, please do not add little naked spheres for the K+.--
Smokefoot (
talk)
17:12, 6 December 2021 (UTC)
- As always, Ben's the crystal-structure master.
DMacks (
talk)
18:09, 6 December 2021 (UTC)
- That is sort of what I was hoping for. Could I trouble you for images through the x, z and y axes? I've updated the page, to my eye the core complex is D2 symmetric. I still don't like the current 2D representation, but if Ben kind enough to generate a set of 3D images, then it becomes non-issue.-- Project Osprey ( talk) 10:02, 7 December 2021 (UTC)
- As always, Ben's the crystal-structure master.
DMacks (
talk)
18:09, 6 December 2021 (UTC)
How about these? Note these axes aren't the crystallographic a, b and c axes. Instead I defined the line passing through both antimony atoms as the z axis. -- Ben ( talk) 17:19, 9 December 2021 (UTC)
- Perfect. Duly added. Many thanks. -- Project Osprey ( talk) 15:31, 10 December 2021 (UTC)
I have a similar question

How to draw hypercubane(see right) in a good way? There are two versions of that file, old one is hard to distinguish it's double bonds, new one is too messy... -- Nucleus hydro elemon ( talk) 11:11, 12 December 2021 (UTC)

-
Nucleus hydro elemon Why not use a drawing based on the tesseract (in the
hypercube article)? That's an .svg that should be fairly easy to edit as required. That filetype is also preferred to .png because they scale better in browsers.
Mike Turnbull (
talk)
10:57, 13 December 2021 (UTC)
-
Michael D. Turnbull, I need to show single bonds and double bonds. In old version I draw based on tesseract, but it's hard to know how many single/double bonds on that picture. At top left corner it is really looks like a pentavalent carbon because of that. --
Nucleus hydro elemon (
talk)
11:27, 13 December 2021 (UTC)
- The original drawing from reference one in hypercubane is available here, on right and would be fine as the basis for a new version. It is already pretty clear, although it doesn't make the carbon double bonds explicit and arguably the hydrogens could be omitted to simplify things. Mike Turnbull ( talk) 11:34, 13 December 2021 (UTC)
-
Michael D. Turnbull, I need to show single bonds and double bonds. In old version I draw based on tesseract, but it's hard to know how many single/double bonds on that picture. At top left corner it is really looks like a pentavalent carbon because of that. --
Nucleus hydro elemon (
talk)
11:27, 13 December 2021 (UTC)
- Maybe you could use a second color to distinguish double bonds in this structure if all bonds are drawn in the same way. --
Leiem (
talk)
12:40, 13 December 2021 (UTC)
- My molecule drawing software can't colour the bonds, it only can colour functional groups. --
Nucleus hydro elemon (
talk)
13:16, 13 December 2021 (UTC)
- I use Biovia Draw, which is a free download for most users here. I then convert its output into .svg using Inkscape, when colours can readily be added. I'll try to create something later today. Mike Turnbull ( talk) 13:41, 13 December 2021 (UTC)
- Actually, the version you've created here now, Nucleus hydro elemon looks pretty good and is fine given that the article is only for a hypothetical compound! Mike Turnbull ( talk) 13:46, 13 December 2021 (UTC)
- My molecule drawing software can't colour the bonds, it only can colour functional groups. --
Nucleus hydro elemon (
talk)
13:16, 13 December 2021 (UTC)
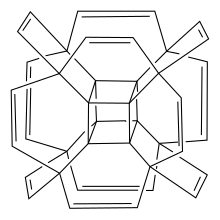
- Just to convince myself I could do it, Nucleus hydro elemon, I've used Inkscape to copy your version of the drawing as an .svg. It will be better for readers in that format, so I'll swap that into the hypercubane article. Mike Turnbull ( talk) 15:08, 13 December 2021 (UTC)
- Superior to the useless jmol image failure linked in the chembox.
Graeme Bartlett (
talk)
10:33, 15 December 2021 (UTC)
- That Jmol sure was...something...alright. The SMILES converter in it often fails for complex 3D ringsystems. I turned it off. DMacks ( talk) 10:39, 15 December 2021 (UTC)
- I checked the Jmol yesterday and it was fine - did something happen to it? --
Ben (
talk)
13:59, 15 December 2021 (UTC)
- Yes, I think they must have modified something. I had already checked that Jmol actually gave a very good rendition of
hypercubane when I first looked a the article and the SMILES certainly hasn't changed at our end. You can look at the version
in this OID which still has the working link to Jmol before
DMacks correctly set it to "no" today because the result currently is a weird-looking flatland image. Let's hope that Jmol gets fixed: the same flatland error is the current rendering for
buckminsterfullerene. Does anyone know how to report such issues to their IT folk?
Mike Turnbull (
talk)
14:46, 15 December 2021 (UTC)
- Actually, I've found out how to report bugs (by emailing jmol-users at lists.sf.net) and I'm in the process of doing that. If anyone reading this thread thinks there is a different explanation not involving Jmol, please comment here ASAP. Mike Turnbull ( talk) 15:01, 15 December 2021 (UTC)
- The CACTVS server that the jmol tool claims to uses to convert SMILES to 3D coordinates appears to give a correct MOL file, even while the link from our hypercubane article gives the flat thing. DMacks ( talk) 15:42, 15 December 2021 (UTC)
- I have never seen hypercubane render properly as the 3D diagram using that SMILES string. I manually made up the smiles string from a diagram and description. Normally when I do this I can confirm if the string gives a good diagram, but in this case I never saw a good one. Benjah-bmm27 and Mike Turnbull, witnessing a good rendition is strange. So I would appreciate if someone can check if the SMILES is OK. And even better find a rearrangement that can render.
Graeme Bartlett (
talk)
21:45, 17 December 2021 (UTC)
- A similar JMOLfailure to render for me is at
Dimethanospiro(2.2)octaplane with
https://chemapps.stolaf.edu/jmol/jmol.php?model=C1C2C3CC4C5CC6C7C48C39C83C48C2CC5C34C6CC8C1C9C7 . Does that work for anyone like Ben or Mike?
Graeme Bartlett (
talk)
22:26, 17 December 2021 (UTC)
- Hypercubane, as
[1] (the
|SMILES=string in the infobox, also by going back to the older revision of the articel) works for me right now. DMacks ( talk) 22:42, 17 December 2021 (UTC)- That is looking OK for me too now, changed in a day!
Graeme Bartlett (
talk)
04:11, 18 December 2021 (UTC)
- I've restored the Jmol to the hypercubane article, as that is now rendering correctly: maybe after I contacted them about the bug, although I never got an acknowledgement. However, Buckminsterfullerene and Dimethanospiro(2.2)octaplane are still in flatland, so the issue has not entirely gone away. Mike Turnbull ( talk) 17:34, 23 December 2021 (UTC)
- That is looking OK for me too now, changed in a day!
Graeme Bartlett (
talk)
04:11, 18 December 2021 (UTC)
- Hypercubane, as
[1] (the
- A similar JMOLfailure to render for me is at
Dimethanospiro(2.2)octaplane with
https://chemapps.stolaf.edu/jmol/jmol.php?model=C1C2C3CC4C5CC6C7C48C39C83C48C2CC5C34C6CC8C1C9C7 . Does that work for anyone like Ben or Mike?
Graeme Bartlett (
talk)
22:26, 17 December 2021 (UTC)
- Yes, I think they must have modified something. I had already checked that Jmol actually gave a very good rendition of
hypercubane when I first looked a the article and the SMILES certainly hasn't changed at our end. You can look at the version
in this OID which still has the working link to Jmol before
DMacks correctly set it to "no" today because the result currently is a weird-looking flatland image. Let's hope that Jmol gets fixed: the same flatland error is the current rendering for
buckminsterfullerene. Does anyone know how to report such issues to their IT folk?
Mike Turnbull (
talk)
14:46, 15 December 2021 (UTC)
Jmol in infobox Chembox
| Identifiers | |
|---|---|
| |
3D model (
JSmol)
|
|
| |
- FYI: This is what {{ Chembox}} can do with SMILES and Jmol. (The demo uses indexes 1-4, see code).
- A Regular, default: Jmol reads SMILES and creates the interactive image at the external link

- F
Ferrocene: its SMILES is incorrect for 3D,
|Jmol=(another SMILES)is fed an adjusted SMILES string → shows OK in 3D
- H Hypercubane: SMILES read by Jmol (default route), obviously wrong image

 →
→  (works now see 17 Dec 22:42 DMacks above)
(works now see 17 Dec 22:42 DMacks above) - H(x) Hypercubane: same SMILES but
|Jmol=none→ Jmol does not show for this one (no error)
(no error)
Not done: possibly an adjusted SMILES string would show 3D coorect in Jmol. - DePiep ( talk) 15:57, 15 December 2021 (UTC)
- Old smiles (H) seems to work; as noted by DMacks 22:42. - DePiep ( talk) 22:48, 17 December 2021 (UTC)
Jmol & uranium
| Identifiers | |
|---|---|
| |
3D model (
JSmol)
|
|
| |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| |
I have another question for Jmol. Why it can't show uranium compounds?-- Nucleus hydro elemon ( talk) 10:52, 30 December 2021 (UTC)
Another question: Jmol become flat when too much atoms bond to one(such as IF7). -- Nucleus hydro elemon ( talk) 12:23, 22 January 2022 (UTC)
Amino acid derivatives
I was moving Mecysteine to cysteine methyl ester (hopefully the conventional name sticks). We can expect eventual creation of numerous articles on amino acid derivatives. Exampless:
- Glycine methyl ester and Glycine methyl ester hydrochloride.
- Various N-protected amino acids such as N-BOCglycine, N-Cbz...).
- Amides like glycinamide.
- Simple dipeptides can be anticipated, e.g. glycylglycine.
So if colleagues have views on naming, now is a good time to discuss.-- Smokefoot ( talk) 14:02, 24 January 2022 (UTC)
- The existing
Category:Amino acid derivatives shows some variance. I only expect there'd be a problem if there were different naming conventions in biochemistry. --
Project Osprey (
talk)
23:43, 24 January 2022 (UTC)
- I don't think we can be prescriptive on this and we would be wasting our time to try to set rules in advance. Loads of chemicals are amino acid derivatives in the broad sense that they contain the substructure -N-C-C(=O)-hetero- and many won't even be categorised as such. I would cite
nirmatrelvir and
tirzepatide as recent examples. I'd stick to the principles of
WP:Common on a case-by-case basis but I do agree with
Smokefoot that "simple" esters should be named as derivatives of their parent. Incidentally, I see no reason why
Glycine methyl ester hydrochloride isn't in an article entitled
Glycine methyl ester, along with any other notable salts and the parent compound but I'm not prepared to fight about it.....
Mike Turnbull (
talk)
12:46, 25 January 2022 (UTC)
- Not looking for prescriptions or fights...just advice and a heads up that lots of derivatives of 20+ parent amino acids will be the bases of many future articles. My feeling (not fightin words, I hope) is that the first few article titles will or could template many others. The quandry of Glycine methyl ester hydrochloride vs Glycine methyl ester illustrates the discussions I was inviting, the former can be purchased and put on the shelf, the latter is the more useful derivative. So I was thinking that the former would be the title, since readers want the info in the chembox for lab work, but the latter would comprise most of the content since experimentalists use the free ester. So its a gentle request. -- Smokefoot ( talk) 14:40, 25 January 2022 (UTC)
- I don't think we can be prescriptive on this and we would be wasting our time to try to set rules in advance. Loads of chemicals are amino acid derivatives in the broad sense that they contain the substructure -N-C-C(=O)-hetero- and many won't even be categorised as such. I would cite
nirmatrelvir and
tirzepatide as recent examples. I'd stick to the principles of
WP:Common on a case-by-case basis but I do agree with
Smokefoot that "simple" esters should be named as derivatives of their parent. Incidentally, I see no reason why
Glycine methyl ester hydrochloride isn't in an article entitled
Glycine methyl ester, along with any other notable salts and the parent compound but I'm not prepared to fight about it.....
Mike Turnbull (
talk)
12:46, 25 January 2022 (UTC)
Has no evidence of existence, just one unreliable source, probably deletion. Keres🌕 Luna edits! 01:52, 31 January 2022 (UTC)
- It has a CAS No (146505-18-2) but no real refs in SciFinder. Weirdly, the free peroxynitrate ion has >200 refs, mostly from biology as a reactive nitrogen species. The structure looks borderline explosive, so I can't imagine anyone's in a hurry to crystallise this stuff. I would support deletion. -- Project Osprey ( talk) 10:08, 31 January 2022 (UTC)
What is the difference of nitroso and nitrosyl? Perhaps these 2 categories should be merged. -- Nucleus hydro elemon ( talk) 12:14, 1 February 2022 (UTC)
- Nitroso compounds are typically organic (R-N=O) whereas nitrosyl compounds (X-N=O) are typically inorganic. It's probably best to keep the categories separate considering the number of articles in each (16+21=37 vs. 9) and the fact the properties tend to differ significantly between the two. Perhaps the nitroso category could become a subcategory of the nitrosyl category. We also have Nitroxyl which is not in either category at present. -- Ben ( talk) 18:49, 1 February 2022 (UTC)
May the (ugly) chemical structure be replaced by the crystal structure shown in the de.wikipedia article? -- Leyo 21:52, 18 January 2022 (UTC)
- Has that actually been made by copy-and-pasting bits of other structures into Microsoft Paint?... it's like a ransom note from an 80s movie... de.wiki image is for sodium metasilicate, I don't know if that's the correct unit cell, but it probably is (de.wiki do a lot of this sort of thing and they're normally correct). -- Project Osprey ( talk) 22:57, 18 January 2022 (UTC)
- It has a
sister --
Project Osprey (
talk)
23:12, 18 January 2022 (UTC)
- I would be interested in hearing of other ugly drawings. Also suspect articles. At our stage in evolution, the goal is not perfection, but avoiding embarrassment. So that the next time someone decides to criticize Wikipedia, they focus on the "Elvis articles" not us.--
Smokefoot (
talk)
00:56, 21 January 2022 (UTC)
- Yes. Hundreds. See the links the section below this one. I've isolated what should be the worst offenders (criteria: commons:Category:Disputed_chemical_diagrams > in-use on en.wiki or wikidata). A list of 26. -- Project Osprey ( talk) 15:22, 21 January 2022 (UTC)
- I would be interested in hearing of other ugly drawings. Also suspect articles. At our stage in evolution, the goal is not perfection, but avoiding embarrassment. So that the next time someone decides to criticize Wikipedia, they focus on the "Elvis articles" not us.--
Smokefoot (
talk)
00:56, 21 January 2022 (UTC)
- File:3oxstate.svg
- File:AbhinavBiochemMechanism.JPG
- File:Amitraz Synthesis Route2.png
- File:Ammonium-alum-3D-vdW.png
- File:Ammonium-hydrosulfide-3D-vdW.png
- File:Bacoside A.png
- File:Biosynthesis of CBC.png
- File:Cf oxyfluoride 3D.png
- File:Cholesterol, Tetrahymanol, Diploptene Structures.png
- File:De epoxynivalenol.tif
- File:Enolate alpha center model.svg
- File:Gladiolin.svg
- File:Hoiamide A and B.png
- File:HomoserineBiosyn.png
- File:Mercury(II)-sulfate-3D-vdW.png
- File:P-menth-1-en-4-ol.svg
- File:Peptide glycopeptide.png
- File:Potassium antimonyl tartrate.png
- File:Potassium-gluconate-3D-balls-ionic.png
- File:Potassium-thiosulfate-3D-balls-ionic.png
- File:Pyramidal dikation, hexamethyl.jpg
- File:Sodium-zincate-3D-vdW.png
- File:Synthesis of morphine.png
- File:Xenic-acid-3D-vdW.svg
- File:Zirconium(IV)-silicate-3D-vdW.png
- File:Оксид титана(III).png
There are so many 3D models of cation+anion. How to remake these files? -- Nucleus hydro elemon ( talk) 02:29, 22 January 2022 (UTC)
- Find a source describing the crystal structure, and make a new crystal structure in Jmol. In many cases, it is also acceptable to use an image of a similar crystal. – LaundryPizza03 ( d c̄) 02:55, 22 January 2022 (UTC)
There are a lot of "covalent" compounds...(Example: File:Cobalt_(II)_fluoride.png), but they are quite frequently used in eowiki(Why they use these 3D models in their reactions, example eo:Sulfura tetrafluorido)... -- Nucleus hydro elemon ( talk) 08:47, 3 February 2022 (UTC)
- Many (most?) chemistry pages on the Esperanto wiki appear to be the work of one very enthusiastic editor. As the images are in use there, they won't be deleted from commons. I don't see that there is much we can do about it, other than to keep an eye on our own pages. -- Project Osprey ( talk) 10:29, 3 February 2022 (UTC)
Alternatives
-- Nucleus hydro elemon ( talk) 13:14, 4 February 2022 (UTC)
- The concern about File:P-menth-1-en-4-ol.svg makes no sense, since one of the proposed renames already matches the filename, and the "other version" is an isomer that can be removed. – LaundryPizza03 ( d c̄) 17:47, 4 February 2022 (UTC)
Ions
The appropriate infobox to use is {{ Chembox}}. It is a modular, expandable infobox. Just use the parameters which you need; empty values will not be displayed. Chemboxes should be used for all chemical compounds, so long as they can exist in that form, even for compounds which are not isolable in pure, solvent-free form (e.g. hypochlorous acid). Chemboxes should not be used for ions, polymers, and proteins and enzymes (except simple peptides).
It said no chembox for ions, but many ion articles( tetramethylammonium, chloride) have their chembox. Also, there is a parameter(|FormulaCharge= ) in Chembox Properties that only useful for ions. -- Nucleus hydro elemon ( talk) 01:10, 11 February 2022 (UTC)
- See also Wikipedia talk:WikiProject Chemicals/Archive 2021#Chembox for polymers. DMacks ( talk) 02:43, 11 February 2022 (UTC)
- At one point I made
template:ionbox, but it really was a dumb idea as it was a just a duplicate of chembox. As molecules become more complex there is a much bigger chance that they will be ions, eg most biologic proteins or nucleic acids. Charge is just a property of a molecule. So we should explicitly state chembox can be used for ions.
Graeme Bartlett (
talk)
04:35, 12 February 2022 (UTC)
- If there are suggestions for extra {{Chembox}} parameters, to make ions feel better at home in it, please signal them. -
DePiep (
talk)
12:48, 12 February 2022 (UTC)
- My comments about chembox for ions:
- {{ Chembox Identifiers}}, {{ Chembox Thermochemistry}}, {{ Chembox Structure}}, {{ Chembox Related}} doesn't affected because it is an ion.
- {{ Chembox Pharmacology}}, {{ Chembox Hazards}}...maybe need to label what counterion for those data...(I prefer use their sodium salt or chloride)
- {{ Chembox Properties}}, {{ Chembox Explosive}} are useless for ions because they can't prepared in bulk if no counterions. Using counterions will make very different data.(Example: NaCl is very soluble in water, but AgCl is insoluble)
- --
Nucleus hydro elemon (
talk)
13:41, 12 February 2022 (UTC)
- erm, I'm not familiar with these details. How would a "label" (full data row?) look like for counterion? ASnything to do with cation, anion? - DePiep ( talk) 14:10, 12 February 2022 (UTC)
- See the right side(
| Hazards_ref =is just a placeholder, I need new parameters). -- Nucleus hydro elemon ( talk) 16:18, 12 February 2022 (UTC)
- My comments about chembox for ions:
- If there are suggestions for extra {{Chembox}} parameters, to make ions feel better at home in it, please signal them. -
DePiep (
talk)
12:48, 12 February 2022 (UTC)
| Hazards(sodium salt) | |
|---|---|
| GHS labelling: | |


| |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (
median dose)
|
6.44 mg/kg (rat, oral) 4 mg/kg (sheep, oral) 15 mg/kg (mammal, oral) 8 mg/kg (rat, oral) [2] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 5 mg/m3 [1] |
REL (Recommended)
|
C 5 mg/m3 (4.7 ppm) [10-minute] [1] |
IDLH (Immediate danger)
|
25 mg/m3 (as CN) [1] |
| Safety data sheet (SDS) | ICSC 1118 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
- {{ Chembox Properties}} includes molecular formula and weight, not just bulk physical properties like solubility and phase-chnge. DMacks ( talk) 20:25, 12 February 2022 (UTC)
- Yes, some {{
Chembox Properties}} parameters are useful. Another useful parameter for ions is
| Appearance =for the colour of it's solution. -- Nucleus hydro elemon ( talk) 05:04, 13 February 2022 (UTC)- I don't understand (yet). A
salt is not an ion (eg, salt has no charge). So, what is the topic of the example?
What would its article title be? Then, in which article (=infobox main title) would this Hazards section belong?cyanide. Altogether, I do not see how this salt or ion relates to the compounds article, and why it should be in there anyway (for example, where are the sodium salt properties shown at this moment?). Shouldn't it be in {{ Chembox Related}}, or in new dedicated Chembox section {{Chembox Salts}}? In an article section not in the infobox? Puzzling for me. Main line: if it is not about the main compound itself (article title), then it should be elsewhere not in the infobox. - DePiep ( talk) 11:45, 14 February 2022 (UTC) -
Sodium salts exists. Why not add a chembox over there (with Hazards, Properties)? -
DePiep (
talk)
11:49, 14 February 2022 (UTC)
- Cyanide ion is toxic, but if there is no counterion, we can't measure how toxic it is, so I put data of it's salt. Same goes to any other ion. -- Nucleus hydro elemon ( talk) 12:25, 14 February 2022 (UTC)
- I don't understand (yet). A
salt is not an ion (eg, salt has no charge). So, what is the topic of the example?
We had a similar discussion to this at WT:WikiProject_Chemicals/Archive_2020#Carboxylic_acids_vs_carboxylate_anion/esters. Our consensus was that very few carboxylic acids merited a separate article and we merged a number of them into their parent acid. I think that cyanide is a case where the separate article is justified but it should only discuss the ion. We have an article on sodium cyanide and, indeed, lithium cyanide, potassium cyanide and others some of which can be linked in the Chembox as related compounds. I do not support repeating, for example, the toxicity data for sodium cyanide in an article that's supposed to be limited to cyanide. The same objection is valid for any other property that can only be measured for a real sample of a specific compound. Mike Turnbull ( talk) 14:01, 14 February 2022 (UTC)
- About
sodium salts, maybe the page could be renamed to "sodium compounds". (
Wikidata:Q12555933) --
Leiem (
talk)
14:15, 14 February 2022 (UTC)
- I'm not sure if it shouldn't just be deleted Category:Sodium compounds contains 167 pages. What can we meaningfully say about such a swathe of compounds? -- Project Osprey ( talk) 14:22, 14 February 2022 (UTC)
- Nucleus: are you proposing a change of some sort? --
Project Osprey (
talk)
14:22, 14 February 2022 (UTC)
- Change what? -- Nucleus hydro elemon ( talk) 14:47, 14 February 2022 (UTC)
- only discuss the ion...then put their salt data is really not an option. By the way, there is something like "(as CN)" in IDLH parameter, which really don't care the counterion. --
Nucleus hydro elemon (
talk)
14:47, 14 February 2022 (UTC)
- I think Nucleus is looking for something like: "In the Chembox Hazards subsection, add option to note "Hazards for cyanide sodium (salt): ..." for certain data rows (datapoints). -
DePiep (
talk)
15:06, 14 February 2022 (UTC)
- Correct. --
Nucleus hydro elemon (
talk)
00:04, 15 February 2022 (UTC)
- If there is going to be such a facility, the logical material to supply hazard data for is
hydrogen cyanide, the
conjugate acid of cyanide ion. The simplest real samples associated with anions are their protonated forms.
Mike Turnbull (
talk)
11:53, 15 February 2022 (UTC)
- I think if you want to compare the effects of various counter ions against some particular property, then you should do it in the article text. Infoboxes are there to handle reams of data that are informative but don't need any narrative explanation. Describing properties across a series is inherently narrative. Make a table, explain the trend.--
Project Osprey (
talk)
12:41, 15 February 2022 (UTC)
- @ Project Osprey: Yes, "do it in the article text" is the route to go. This is the way I understand this subtopic, and our handling of it. Maybe if it appears stable over multiple compound articles, and there is a data-point in there to be discovered, then we can revisit this question and the infobox can follow. - DePiep ( talk) 13:13, 15 February 2022 (UTC)
- I think if you want to compare the effects of various counter ions against some particular property, then you should do it in the article text. Infoboxes are there to handle reams of data that are informative but don't need any narrative explanation. Describing properties across a series is inherently narrative. Make a table, explain the trend.--
Project Osprey (
talk)
12:41, 15 February 2022 (UTC)
- If there is going to be such a facility, the logical material to supply hazard data for is
hydrogen cyanide, the
conjugate acid of cyanide ion. The simplest real samples associated with anions are their protonated forms.
Mike Turnbull (
talk)
11:53, 15 February 2022 (UTC)
- Correct. --
Nucleus hydro elemon (
talk)
00:04, 15 February 2022 (UTC)
- I think Nucleus is looking for something like: "In the Chembox Hazards subsection, add option to note "Hazards for cyanide sodium (salt): ..." for certain data rows (datapoints). -
DePiep (
talk)
15:06, 14 February 2022 (UTC)
References
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0562". National Institute for Occupational Safety and Health (NIOSH).
- ^ "Cyanides (as CN)". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
Chemical data pages cleanup / task
- The WP:DPCLEANUP task thread has been moved to dedicated page WT:CHEMICALS/Chemical data pages cleanup (see current status notes). Discussions can be continued or (re)initiated there. - DePiep ( talk) 10:15, 28 February 2022 (UTC)
The ambiguity of "silene"?
The disambiguation page for Silene used to list three different classes of silicon compounds with that name. Now it lists only one. Should any of the other two be brought back? – Uanfala (talk) 02:17, 4 March 2022 (UTC)
- I think the piped entry for
Organosilicon#Silenes would still be appropriate. The other one, the redirect
Silanylidene group doesn't follow
MOS:DAB and redirects to a page about silanes, not silenes.
Mdewman6 (
talk)
02:33, 4 March 2022 (UTC)
- Thank you! I've brought back one of the two entries, and expanded the hatnote at
Silenes. Given that I don't have any chemical knowledge and that there apparently was
a past controversy among those who do, I don't trust that I would have gotten it exactly right. So, more eyes will be welcome. –
Uanfala (talk)
22:46, 13 March 2022 (UTC)
- Great! I rephrased your hatnote slightly, but this is all definitely an improvement over the previous status quo. We do need a better way to disambiguate the two compound classes, not sure how best to go about that at the moment though, and as you say, will require discussion due to the past debate. Mdewman6 ( talk) 22:55, 13 March 2022 (UTC)
- Thank you! I've brought back one of the two entries, and expanded the hatnote at
Silenes. Given that I don't have any chemical knowledge and that there apparently was
a past controversy among those who do, I don't trust that I would have gotten it exactly right. So, more eyes will be welcome. –
Uanfala (talk)
22:46, 13 March 2022 (UTC)
Revising guidance of naming of articles for organic compounds
- The following discussion is closed. Please do not modify it. Subsequent comments should be made in a new section. A summary of the conclusions reached follows.
- The result of this discussion was implement proposed revision to the chemistry naming conventions. Mdewman6 ( talk) 00:39, 30 March 2022 (UTC)
All, I think it is long past due to revisit Wikipedia:Naming_conventions_(chemistry)#IUPAC_preferred_name_vs._systematic_name, as it is misleading. First and most important, by referring to an IUPAC recommendation, this has been interpreted by some (see recent discussion with Pabsoluterince) to mean that articles should be at the preferred IUPAC name, which is not what is intended, resulting in this case in a set of messy page moves, where some redirect history was lost. The examples given are rare cases where IUPAC has acquiesced to the use of trivial (common) names, but most PIN's are systematic names, and we do not use these as article titles if there is a common name. Examples include Caproic acid rather than Hexanoic acid (see past consensus for carboxylic acids specifically), 1-Pentanol rather than Pentan-1-ol, Benzaldehyde rather than Benzenecarbaldehyde, Diethyl ether rather than Ethoxyethane, Benzophenone rather than Diphenylmethanone, etc. So, while stating "IUPAC recommends the use of non-systematic names for some [emphasis added] organic compounds" is correct, this technically only addresses a small minority of organic compounds, providing no guidance on cases where IUPAC recommends use of a systematic name, or even implying that systematic names should be used unless IUPAC recommends otherwise. Hence, the operative aspect here is not what IUPAC recommends, but the use of non-systematic names.
The section title should be revised to state "Trivial (common) name vs. systematic name" and in the text we should leave IUPAC completely out of it. Instead, we should state that trivial or common names should be used if they exist, following the spirit of WP:COMMONNAME, using systematic names when there is no trivial name that unambiguously refers to the compound. A more robust list of examples, such as those above, should then be included to more thoroughly depict the convention and provide better guidance to those writing articles or considering page moves. If there is consensus for this, I can try to come up with something, that others are then free to improve. Mdewman6 ( talk) 22:32, 12 February 2022 (UTC)
- Well yes, case in point, I did misinterpret the current policy and moved an article because of it. I found the policy extremely misleading, and basically was lead astray by my experience of what the actual common name was for the acid. I think for certain (commonly short chain) molecules, the systematic name is more recognisable and natural to use.
Pabsoluterince (
talk)
07:09, 13 February 2022 (UTC)
- I agree with
Mdewman6 that the principles of
WP:COMMONNAME take precedence. In particular, for pharmaceutical drugs we will nearly always use the
international nonproprietary name and for pesticides the
ISO common name listed here.
Mike Turnbull (
talk)
15:56, 13 February 2022 (UTC)
- Thank you for reminding us of this important issue. One reason that I was once very active in establishing articles on simple compounds was to pre-empt the IUPACers.
- It might be worthwhile to collect some titles for conversion to their common names.
Ethenone would be high on my list.--
Smokefoot (
talk)
16:28, 13 February 2022 (UTC)
- Yes, once we've made the convention more clear, I think page moves to rectify clear-cut cases such as that can proceed without need for further discussion.
- I agree with
Mdewman6 that the principles of
WP:COMMONNAME take precedence. In particular, for pharmaceutical drugs we will nearly always use the
international nonproprietary name and for pesticides the
ISO common name listed here.
Mike Turnbull (
talk)
15:56, 13 February 2022 (UTC)
I have drafted a revised section at User:Mdewman6/sandbox2, which would completely replace the existing "IUPAC preferred name vs. systematic name" section. Please take a look and feel free to comment, offer revisions, or add to the list of examples. If there are no objections or suggestions over the next week or so, I will implement the revision at the naming conventions page. Mdewman6 ( talk) 03:50, 3 March 2022 (UTC)
- There being no further comment, I am going to update that section. If concerns arise let's discuss them here.
Mdewman6 (
talk)
00:44, 13 March 2022 (UTC)
- I have questioned the procedural strength of this GF conclusion; see § Procedural questions below. - DePiep ( talk) 22:55, 14 March 2022 (UTC)
Proposed new guideline
== Organic compound names ==
This text proposal box:Trivial names (non-systematic, or "common" names) are favored for use in titles of articles for organic compounds instead of systematic names. Trivial names are usually different from the preferred name following IUPAC nomenclature. For compounds lacking trivial names, as is often the case for complex structures, substitutive nomenclature or other systematic names may be used. The general rule is to use the name most commonly used to refer to the compound, as evidenced by use in reliable sources (in line with WP:COMMONNAME).
Classes of compounds may have more specific guidance on naming, such as the use of international nonproprietary names for pharmaceutical compounds (see below).
Examples of use of trivial names and not systematic names include:
acetic acid not ethanoic acid toluene not methylbenzene lysine not 2,6-diaminohexanoic acid 1-pentanol not pentan-1-ol benzaldehyde not benzenecarbaldehyde diethyl ether not ethoxyethane benzophenone not diphenylmethanone Systematic and other accepted names for the compound not used as the article title should instead be redirects tagged with the {{ R from alternative name}} template and listed in the chembox as other names, with especially significant alternative names mentioned in the lede in bold font.
Procedural questions
- @ Mdewman6: No opinion on the proposal, but I do think this needs a more formal and more wide discussion. After all, it is probably a controversial WP:MOVE (or thousands so). At least an RfC, and publication (invitations). The advantage of this is, that the new guidance has a broad and stable consensus. - DePiep ( talk) 06:41, 14 March 2022 (UTC)
- I have reverted the Good Faith change by User:Mdewman6 in Wikipedia:Naming_conventions_(chemistry). As I noted im my previous (06:41) post, the discussion better be more wide and strong as it affects many titles and possibly is controversial. I'm sorry for arriving late here. I note that I am positive to the proposal.
- Mdewman6, could we write a
Wikipedia:Requested moves § CM proposal?
DePiep (
talk)
07:15, 14 March 2022 (UTC)
- @ DePiep:, I'm not opposed to reopening the discussion as an RfC, but there was consensus for the change above. I figured the users who cared the most about the revised naming convention, and have the pertinent expertise, are all right here. I do think I should have linked to the discussion from the chem NC talk page though, rather than after the fact.
- It's not so much a change but rather more clearly defining existing practice to avoid confusion (as discussed above). There will certainly be articles that could/should be moved based on the revised naming convention, but I doubt it would affect 'thousands' of pages. Per WP:RM#CM, articles that had past uncertainty or debate regarding the best name should still go through the RM process, but with the revised naming convention as justification. More straightforward cases could be moved boldly and reverted if there is opposition.
- I am not sure what you mean by writing a CM proposal? This is not a proposal to move a specific page. It's updating a naming convention.
Mdewman6 (
talk)
20:33, 14 March 2022 (UTC)
- Well, I admid I did not see the impact of the guidance (change) that precise, and it seems to be less dramatic. However, I thought it could meet opposition afterwards, especially when pages are being moved, and possibly on procedural grounds. That would be problematic. And since the change (your edit) was recent still without following move actions, I reverted asap (instead of talking), to prevent such after-move-discussions. As said, I just only discovered this MOS-discussion.
- My point is: being a MOS guideline, I'd go for a tough and formal consensus. For example, wide invitations (WP:CHEMISTRY, WT:NC (chemistry), some candidate articles, ..). I find three replies (supports) a bit low for a MOS change. Indeed, probably not WP:RM. And doesn't is require a formal (uninvolved editor) closure?
- I'm not from WikiLaw school, so maybe I am overconcerned. If so, I'd like to learn so; from experienced outsider? Can we ask outsider advice in this? -
DePiep (
talk)
22:51, 14 March 2022 (UTC)
- Let's start by pinging two WikiProject Chem members who are also admins to get their perspectives, DMacks and Graeme Bartlett. I will also amend my note at Wikipedia talk:Naming_conventions (chemistry) and leave a note at Wikipedia talk:WikiProject Chemistry. We can certainly to try to get a non-chemist outsider as well, not sure how best to go about seeking that, but feel free. Again, I'd favor more discussion if it were more of a change in existing practice as opposed to clarifying existing practice. Happy to setup a RfC, but we would have to transclude or copy stuff from here into a new section, probably on the naming conventions talk page. Mdewman6 ( talk) 00:22, 15 March 2022 (UTC)
- I will say, however, that in my view it would have been more appropriate to raise concerns here first rather than first reverting the addition, given there had already been discussion regarding the change. But I agree if there is any chance of the change not being the ultimate outcome, it would be best to not risk others making bold page moves based on the new text.
Mdewman6 (
talk)
00:55, 15 March 2022 (UTC)
- Thanks. As I described: reason I reverted (which is a heavy action, I know), is that once subsequential Moves were implemented, going back might be problematic in ways. - DePiep ( talk) 06:26, 15 March 2022 (UTC)
DePiep, there has been no further discussion. If your only objection to the change was procedural and not to the change itself, then I am inclined to proceed. I don't think an RfC is necessary here. The discussion has been advertised in the appropriate places. Consensus can be assumed until there is evidence of disagreement. Mdewman6 ( talk) 16:37, 26 March 2022 (UTC)
- For the record: I support the proposal (sorry for being this late). - DePiep ( talk) 16:58, 26 March 2022 (UTC)
- User:Mdewman6: I agree, we can conclude consensus for the proposal. My compliments for the initiative. - DePiep ( talk) 16:58, 26 March 2022 (UTC)
Lead disulfide
The article lead(IV) sulfide said the oxidation state of Pb in PbS2 is +4. A newer ref ( doi: 10.1016/j.jssc.2018.10.023) states that PbS2 crystallizes in the CuAl2 structure. It should be Pb2+(S2)2−. -- Leiem ( talk) 05:31, 30 March 2022 (UTC)
- Good find! Our article cites a 1966 ref for structural analysis, but this new ref discusses mentions those results as lacking as-strong support due to lack of as-high quality samples or data. This isn't my field, so I can't comment on any apparent strengths of either actual research. DMacks ( talk) 19:27, 17 April 2022 (UTC)
We have recently (thanks User:GRALISTAIR!) gotten a bunch of articles about glycidyl ethers, which are used as reactive diluents (I created both Category:Glycidyl ethers and Category:Reactive diluents). Each of them has a list of the others in their respective SEEALSO. It seems excessive to link all structurally–application-related chemicals to each other in this manner (and having to keep each one updated when new are created). Instead, WP seems like it would favor a navbox. I started Template:Glycidyl ethers. But now I wonder whether instead of this one, based on a structural motif, we should instead have one based on the reactive-diluent application case? Non-ether epoxies are possible, as are structures for non-epoxide polymer types. Would a Template:Reactive diluents (that would have the glycidyl-ethers blocks but also the vinyl compounds from that application's category) be better? DMacks ( talk) 19:36, 17 April 2022 (UTC)
- I think perhaps you've done it the right way, as some of these compounds (such as
Bisphenol A diglycidyl ether) have uses beyond being reactive diluents. --
Project Osprey (
talk)
21:32, 17 April 2022 (UTC)
- I agree with @ Project Osprey . There is at least one Glycidyl ESTER also used as an epoxy reactive diluent. GRALISTAIR ( talk) 00:19, 18 April 2022 (UTC)
However it does beg the question "Should I write an article that is entitled Glycidyl ethers?" It would be a general article possibly a stub that would give general background and reference the above GRALISTAIR ( talk) 17:00, 18 April 2022 (UTC)
- Perhaps the best place to start might be Epoxy#Diluents, which fails to even link to reactive diluent? Glycidyl ethers seem to be used exclusively for epoxy systems but I can't find the line between diluent and monomer. Bisphenol A diglycidyl ether is clearly a co-monomer. Melting points (generally lacking for these article) might be informative, I expect all the dilutes will have broad liquid ranges with very high boiling points. -- Project Osprey ( talk) 20:00, 18 April 2022 (UTC)
Phosphonium iodide
I filed a request to create an image at c:COM:GL, but was directed here by Leyo ( talk · contribs).
I intend to create 3D ball-and-stick and spacefilling models of the crystal structure of
phosphonium iodide (PH
4I) as described in the listed references, to replace the current crude cation+anion combination. It is a distorted version of the
NH
4Cl structure, with
tetragonal symmetry.
[1]
[2] The file names would be
File:Phosphonium iodide 3D balls.png and
File:Phosphonium iodide 3D vdW.png, like most other 3D molecular and crystal models. –
LaundryPizza03 (
d
c̄)
02:47, 13 March 2022 (UTC)
- Benjah-bmm27 is currently not very active … -- Leyo 10:08, 22 April 2022 (UTC)
References
- ^ Dickinson, Roscoe G. (July 1922). "The Crystal Structure of Phosphonium Iodide". Journal of the American Chemical Society. 44 (7): 1489–1497. doi: 10.1021/ja01428a015.
- ^ Sequeira, A.; Hamilton, Walter C. (September 1967). "Hydrogen Bonding in Phosphonium Iodide: A Neutron-Diffraction Study". The Journal of Chemical Physics. 47 (5): 1818–1822. Bibcode: 1967JChPh..47.1818S. doi: 10.1063/1.1712171.

Do we have such manually drawn structural formulas in other chemboxes, too? Are we fine with it? -- Leyo 09:50, 25 April 2022 (UTC)
- We are not fine with that sort of thing. I replaced it with
File:Paraloid B-72.png and also uploaded
File:Paraloid B-72 monomer-IDs.png if we wanted to discuss its structure in more detail.
DMacks (
talk)
07:12, 26 April 2022 (UTC)
- Thanks, that's what I thought, even though this image has remained in the article for ten years. ;-) -- Leyo 12:53, 28 April 2022 (UTC)
User script to detect unreliable sources
I have (with the help of others) made a small user script to detect and highlight various links to unreliable sources and predatory journals. Some of you may already be familiar with it, given it is currently the 39th most imported script on Wikipedia. The idea is that it takes something like
- John Smith "
Article of things" Deprecated.com. Accessed 2020-02-14. (
John Smith "[https://www.deprecated.com/article Article of things]" ''Deprecated.com''. Accessed 2020-02-14.)
and turns it into something like
- John Smith " Article of things" Deprecated.com. Accessed 2020-02-14.
It will work on a variety of links, including those from {{ cite web}}, {{ cite journal}} and {{ doi}}.
The script is mostly based on WP:RSPSOURCES, WP:NPPSG and WP:CITEWATCH and a good dose of common sense. I'm always expanding coverage and tweaking the script's logic, so general feedback and suggestions to expand coverage to other unreliable sources are always welcomed.
Do note that this is not a script to be mindlessly used, and several caveats apply. Details and instructions are available at User:Headbomb/unreliable. Questions, comments and requests can be made at User talk:Headbomb/unreliable.
This is a one time notice and can't be unsubscribed from. Delivered by: MediaWiki message delivery ( talk) 16:01, 29 April 2022 (UTC)
Issues with Template:Sigma-Aldrich
Template:Sigma-Aldrich, is commonly used as a reference for Chembox hazard data. Following a merger with Merk, Sigma-Aldrich appears to have reorganised its website, breaking links in some instances. The template uses the Sigma ID number to make links, e.g. 294993 for ammonia (which still works):
- {{Sigma-Aldrich|id=''294993''|name=Ammonia|access-date=27 December 2021}} → Sigma-Aldrich Co., Ammonia.
However in some cases the links, while showing as blue, do not connect properly.
- {{Sigma-Aldrich|id=''36735''|name=Bis(2-ethylhexyl) phthalate|accessdate=2022-05-12}} → Sigma-Aldrich Co., Bis(2-ethylhexyl) phthalate. Retrieved on 2022-05-12.
The template assumes that the URL should always be www.sigmaaldrich.com/GB/en/product/aldrich/<id number>
https://www.sigmaaldrich.com/GB/en/product/aldrich/294993
https://www.sigmaaldrich.com/GB/en/product/sial/36735
-- Project Osprey ( talk) 09:33, 12 May 2022 (UTC)
- So the template should switch between /aldrich/ and /sial/. Can we determine this from name or id? Any other route? -
DePiep (
talk)
10:10, 12 May 2022 (UTC)
- I'm unsure as to the scale of the problem. There may be others beyond /sial/. One option: links default to /aldrich/ (to protect existing links) but add an manual override option to replace it with something else. --
Project Osprey (
talk)
10:28, 12 May 2022 (UTC)
- There are some 175 transclusions. I've added option
|1=sialto the documentation. There also seems to exist an option|1=CATALOG(not explored). - Looks like the task is to check each instance. -
DePiep (
talk)
11:21, 12 May 2022 (UTC)
- If you give me a transclusions list I'll do the leg-work. -- Project Osprey ( talk) 11:44, 12 May 2022 (UTC)
- There are some 175 transclusions. I've added option
- I'm unsure as to the scale of the problem. There may be others beyond /sial/. One option: links default to /aldrich/ (to protect existing links) but add an manual override option to replace it with something else. --
Project Osprey (
talk)
10:28, 12 May 2022 (UTC)
- it's the template page {{ Sigma-Aldrich}}, then click What Links Here: [2] - DePiep ( talk) 12:00, 12 May 2022 (UTC)
BTW: de:Vorlage:Sigma-Aldrich#Vorlagenparameter has a few more options for this parameter. The template is transcluded in ~4500 de.wikipedia articles. -- Leyo 20:49, 12 May 2022 (UTC)
1,2-Dichloroethylene: splitting
1,2-Dichloroethylene (aka 1,2-Dichloroethene) seems to be pretty fundamental, so I think that we should split it into cis and trans isomers. A draft of the trans isomer is here. A potentially controversial aspect of my draft is the name dichloroethylene vs dichloroethene. Feel free to comment etc.-- Smokefoot ( talk) 15:29, 20 June 2022 (UTC)
- Support split, if enough unique content can be added to to each article besides the chemboxes. If split, the base name should then be a chemindex to disambiguate the two isomers. The article title should use the trivial name 'ethylene' and not the systematic 'ethene' per WP:OCHEMNAME, and given that the parent compound's article is at Ethylene and not Ethene. Mdewman6 ( talk) 20:58, 20 June 2022 (UTC)
- Comment: I'm in no way against it, but I can't help but wonder it would mean for related articles like but-2-ene or 1,3-dichloropropene which also bundle isomers together. -- Project Osprey ( talk) 21:01, 20 June 2022 (UTC)
- I think we should look at these on a case by case basis. If there is enough isomer-specific content, then a split could make sense. But I don't think we should by default always have separate articles for geometric isomers, just to have separate chemboxes, if it results in two stubs with mostly duplicated content.
- Status quo. For the time being. There seems to be little incentive. However the Z isomer gets 5700+ hits and the E isomer 4900 hits in SciFinder. Most of the top refs (aside from the usual solvent properties and water-octanol partitioning papers) are to cleaning up chlorocarbon-contaminated ground-water, which are now included in the parent article. -- Smokefoot ( talk) 18:36, 21 June 2022 (UTC)
- Well, it was your proposal...is there other content in your draft that you can include in the current article?
Mdewman6 (
talk)
19:05, 21 June 2022 (UTC)
- Hey, I know that I started this splitting idea and the literature on these two isomers is large but as I did more work fewer benefits were apparent. I did beef up the refs on the individual and the pair. There are no reviews focused on these things alone. Apologies for any inconveniences caused by me. -- Smokefoot ( talk) 12:06, 22 June 2022 (UTC)
- Well, it was your proposal...is there other content in your draft that you can include in the current article?
Mdewman6 (
talk)
19:05, 21 June 2022 (UTC)
- Status quo The current article is not very large and both the WP:COMMONNAME and molecular formula searches get there easily, which after a split would be more tedious for readers. Also, CAS no. 540-59-0 refers to unspecified/mixed isomers, which some of the literature will be. Mike Turnbull ( talk) 11:29, 22 June 2022 (UTC)
Inorganic nomenclature question
Praseodymium(II) iodide is identified as having the formula Pr3+(I−)2e− and a synonym "praseodymium diiodide". Why aren't those names the formula Pr2+(I−)2 and why is a +3 metal named as having oxidation state (III)?
DMacks (
talk)
23:56, 11 July 2022 (UTC)
- "isn't". DMacks ( talk) 02:24, 12 July 2022 (UTC)
- Your puzzlement seems appropriate. Two major issues strike me. First, the description Pr3+(I−)2e− is just one of five forms (polymorphs) or PrI2. The others are not so strange (CdX2 motif, a cluster motif, and two more). The article discusses at some length the solubility of PrI2 in water. It has no solubility in water: it dies immediately upon contact.-- Smokefoot ( talk) 00:14, 12 July 2022 (UTC)
- A compound that contains a separate e− is an electride. But that fact is uncited. Graeme Bartlett ( talk) 00:25, 12 July 2022 (UTC)
- Greenwood and Earnshaw generally refer to LnI2 and diiodides. They do sometimes specify oxidation states but with explanation: [1]
The isomorphous diiodides of Ce, Pr and Gd stand apart from all the other, salt-like, dihalides. These three, like LaI2, are notable for their metallic lustre and very high conductivities and are best formulated as {LnIII,2I−,e−}, the electron being in a delocalized conduction band.
- There's no mention of the other (non-conductive) polymorphs of PrI2 even though those were discovered well before the book was written. [2]
- According to the OCP on the f-block, the single delocalised electron per Pr atom in PrI2 occupies a broad, partially filled 5d band. [3]
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 1240. ISBN 978-0-08-037941-8.
- ^ Warkentin, E.; Bärnighausen, H. (1979). "Die Kristallstruktur von Praseodymdiiodid (Modifikation V)". Z. anorg. allg. Chem. 459: 187–200. doi: 10.1002/zaac.19794590120.
- ^ Kaltsoyannis, Nikolas; Scott, Peter (1999). Oxford Chemistry Primers: The f elements. Oxford University Press. p. 44. ISBN 978-0-19-850467-2.
- ^ Gerlitzki, Niels; Meyer, Gerd; Mudring, Anja-Verena; Corbett, John D. (2004). "Praseodymium diiodide, PrI2, revisited by synthesis, structure determination and theory". J. Alloys Compd. 380 (1–2): 211–218. doi: 10.1016/j.jallcom.2004.03.046.
- ^ Krämer, Karl; Meyer, Gerd; Fischer, Peter; Hewat, Alan W.; Güdel, Hans U. (1991). "Neutron diffraction investigation of magnetic phase transitions to long-range antiferromagnetic ordering in the "free-electron" praseodymium halides Pr2X5 (X = Br, I)". Journal of Solid State Chemistry. 95 (1): 1–13. doi: 10.1016/0022-4596(91)90370-W.
Dilithio
I have been trying to improve spelling and Wiktionary entries particularly for chemicals, and I have come across "lithio" [3] and "dilithio". [4] This is being used as if it is a replacement nomenclature for lithium in an organic compound. The current IUPAC Blue Book has "litha" for this, and I cannot find "lithio" in old versions back to 1979. But is "lithio" correct at all, perhaps sometime back in the past? (If so there should be a dictionary entry) Should we correct this "lithio" use in articles to have more valid systematic names? Graeme Bartlett ( talk) 01:06, 22 August 2022 (UTC)
- Not sure if it's IUPAC endorsed, but it does seem to be a term that has seen some use. For example, I am familiar with the reaction of
diphenylacetylene with lithium metal (under inert atmosphere) to yield a lithium-substituted butadiene that at least one paper in
Inorganic Chemistry in 1999 names as 1,4-dilithio-2,3,4,5-tetraphenylbutadiene. They also use "dilithio" to refer to this intermediate/derivative throughout. Perhaps it has fallen out of favor? But at least at one time saw some use.
Mdewman6 (
talk)
01:39, 22 August 2022 (UTC)
- Of the four cited refs for our 1,1'-Dilithioferrocene article, three use "dilithio..." in the names of specific chemicals (Organometallics 1985 and 2008, Angew Chem 2007) and and the fourth does not appear to name any specific chemicals having two lithium atoms. DMacks ( talk) 05:05, 22 August 2022 (UTC)
- I searched Chemspider, where there were 60 hits for lithio*, 13 for dilithio* and just one for lithia*, which turned out to be lithia here on WP! So whatever the Blue Book says does not seem to have been widely accepted. None of the hits I looked at were part of the main name for the compounds in question, just minor alternative names. Mike Turnbull ( talk) 09:46, 22 August 2022 (UTC)
- Thanks for your responses, based on that I think we should leave "lithio" in our article. And I have added it to Wiktionary. Graeme Bartlett ( talk) 21:08, 22 August 2022 (UTC)
Wikidata new chemical properties
Currently, there are discussion on adding properties in Wikidata:
- d:Wikidata:Property proposal/Drugcentral ID: DrugCentral (Q27697289)
- d:Wikidata:Property proposal/Probes And Drugs ID: Probes And Drugs (Q113999788) (?)
- d:Wikidata:Property proposal/Library of Integrated Network-based Cellular Signatures ID (..)
- d:Wikidata:Property_proposal/Natural_science#UniChem_compound_ID: UniChem (Q24170711)
DePiep ( talk) 16:36, 25 September 2022 (UTC)
Add a `found in taxon` statement from Wikidata in the chemical infobox
Dear members of the Chemicals WikiProject,
I am editing a lot Wikidata, particularly for chemistry and natural products (see https://www.wikidata.org/wiki/Wikidata:WikiProject_Chemistry/Natural_products). We now have a lot of curated data about chemicals found in organisms (see https://www.wikidata.org/wiki/Q2079986 for an example). Adding this information to the infobox would be very interesting. I have no idea how this can be done and do not know the procedure that has to be followed to modify an infobox template. Could anyone help me achieve this? The most crucial limitation would probably be to limit pages with many statements, such as https://www.wikidata.org/wiki/Q121802. This is elegantly done on Pubchem, as per: https://pubchem.ncbi.nlm.nih.gov/compound/222284 ("beta-Sitosterol is a natural product found in Elodea canadensis, Ophiopogon intermedius, and other organisms with data available.")
Happy to exchange more about this! AdrianoRutz ( talk) 11:33, 19 September 2022 (UTC)
- @
AdrianoRutz I think that the problem is going to be how to select the one or two that might be chosen to go in the infobox.
The full data for sitosterol on pubchem seems to have 2087 items! Our template expert is @
User:DePiep but I think that the first step is to gain consensus about what is sensible to include. All our chemboxes already link to pubchem, so the information is only a couple of clicks away for those readers who might be interested.
Mike Turnbull (
talk)
13:24, 19 September 2022 (UTC)
- Thank you very much for your prompt reply!
- The sitosterol example is the most extreme one, there are only a few with so much statements, it was chosen on purpose to answer the important point you mention.
- I have no idea how flexible the box is...my suggestion would be (based on what is feasible):
- if n >> 10
- - include randomly 2-5 taxa and add "other organisms"
- - include the parent taxa instead of the taxon so that "found in list of 999 different plants, list of 299 bacteria and list of 199 fungi" becomes "found in plants, fungi, bacteria"
- - based on different number and levels say if 0 < n < 10 display species, 10 < n < 50 display genus, and so on... AdrianoRutz ( talk) 13:57, 19 September 2022 (UTC)
- Some first notes. {{ Chembox}} (= {{Infobox chemical}}) and its sister {{ Infobox drug}} cover the chemicals, 11820+7790=19600 substances at enwiki. Guideline is WP:INFOBOX, but they have heavily creeped into Data Sheets and External Link lists (ie, lots of data is added while not necessarily described in the article body text, and the Identifiers list external links to databases like by its PubChem-identifier).
- Property (data point, infobox row) is added by proposal, usually at WT:CHEMBOX. Relevance for inclusion should be motivated. As said, there is little holdback as to keeping the list 'short' or 'highlights only' (data sheet effective).
- A thorough redesign(-discussion) is needed, IMO about splitting the Infobox into a separate datasheet-articlesection with tabular info. Such split could make the infobox more relevant, and at the same time create more space for data and its presentation. Currently, because editors prefer little or no merging with Wikidata info and, separately, keeping old style input options (infobox section header separated), developments are slow these years.
- About Wikidata: given that info (think properties) in WD are currently well maintained ('curated'), using the mass-editing options fore k's of chemicals, systematic data best be maintained at WD, and imported (read) into enwiki. Example in case: from its inclusion in 2019 here, for
|DTXSID=DSSTox substance ID (P3117) is read by default (the local parameter can overwrite). I advise for any new data point we want to add, to follow this route (read from Wikidata, maintain at Wikidata, local overwrite option). - Now about this proposal for found in taxon (P703). So it is a list, possibly long length. I question whether this is the right place then: "This substance is found in: ...", really? May be, as noted, when the list is short thus for scarce/rarities only. And: how is the item present in counter-article like Picrasma crenata? Do we have solved a similarity with "Element lithium occurs in substances ..."? Then, {{Chembox}} is born & grown from anorganic interests. Organic chemistry has other approaches in wiki, I am not familiar with. Is organic chemistry more or better organised to handle such lists?
- That said, there is no a priori objection to inclusion. If considered relevant for inclusion, I'd propose (1) read from Wikidata not local, with local overwrite option; (2) limit to "first ten", or "only when n<=10".
- -
DePiep (
talk)
16:44, 19 September 2022 (UTC)
- Thank you very much for your detailed answer!
- Shall we keep initial discussion here or move directly to /info/en/?search=Template_talk:Chembox?
- I cannot comment about the redesign of the infobox, but will surely advocate for more Wikidata-based info. It makes absolutely no sense to oppose to such enhancement.
- The model you suggested with DSSTox substance ID (P3117) is exactly what I meant. Maintain the data on Wikidata and pull it into the box.
- Concerning the found in taxon (P703), I can develop into much more detail if needed. Actually, the median number of organisms per compound is 1, with a mean of 3.3. So for a lot of compounds, it will be rare. As the ~20,0000 chemicals you mentioned are way less than what is on Wikidata, those numbers will likely go up as they are the "most known/studied" compounds. So yes, caffeine is documented in many more species than other, less known, derivatives. Having this information remains highly relevant.
- I do not know exactly how the distinction between the "short" or "highlight only" is made, but of course, if the text contains sentences saying "interestingly some Citrus species were found to contain caffeine", even better. If there are way to automate this from Wikidata, I am very eager to learn, I know this exists in other domains.
- Your comment about the "reverse property" "this organism is known to contain caffeine" is indeed a very good point. I guess I will make a request on the taxonomy infobox template to add a data point about chemical compounds found in the taxon. The discussion on Wikidata led to the decision that information about natural products is usually more compound-centric than taxon-centric, so we started more on the chemistry side, but you are perfectly right, the other way around is also very interesting!
- I sadly cannot comment about inorganic chemistry and the "lithium found in compound X" but this is a different mapping on Wikidata. It does not use the found in taxon (P703) property, but has part(s) (P527). An example for aspirin here: https://www.wikidata.org/wiki/Q18216. The data on this side is rather incomplete so I can hardly comment, but if this is of relevance for your community I am happy to come back with more info.
- Finally, I am perfectly in line with your 2 last points! :)
AdrianoRutz (
talk)
07:35, 20 September 2022 (UTC)
-
AdrianoRutz, what do you mean by "
It does not use the 'found in taxon (P703)' property, but 'has part(s) (P527)'
"? For aspirin (Q18216), I see found in taxon (P703) with three entries, so that looks OK. OTOH, a has part(s) (P527) list cannot be sure to be taxons (because any editor can, rightfully within its definition, add anything in there), so one cannot use it as proposed here. That is: P703 being used is Good! DePiep ( talk) 16:52, 25 September 2022 (UTC)- Oh, it had nothing to do with the initial found in taxon discussion anymore, it was an answer regarding your "lithium found in compound X"... the most used mapping is compound "has parts" lithium, carbon, oxygen...etc.
AdrianoRutz (
talk)
21:07, 25 September 2022 (UTC)
- OK. We can forget this detail then: P703 it is. DePiep ( talk) 08:03, 26 September 2022 (UTC)
- Oh, it had nothing to do with the initial found in taxon discussion anymore, it was an answer regarding your "lithium found in compound X"... the most used mapping is compound "has parts" lithium, carbon, oxygen...etc.
AdrianoRutz (
talk)
21:07, 25 September 2022 (UTC)
-
AdrianoRutz, what do you mean by "
- I suspect this should be expressed in words as text in the article body. If it was less than 4 taxa, perhaps it could fit in the Chembox or drugbox. But more will just overdominate the box. It is quite likely that there would be many more taxa with any particular substance, just not studied yet. And there will other substances found in all of a set of organisms, eg all animals or all photosynthetic plants. When it comes to more detail, then text will be needed, such as where in the organism the substance is found or concentrated, and if it is useful to extract the substance form that organism. - Graeme Bartlett 07:25, 21 September 2022
- I never considered adding tens of taxa into the infobox. As explained above, the information in the box would be limited to 10 (or 5, or 4) taxa. This can be either done by adding the info only for the compounds where this is true, randomly selecting n, or selecting n upper taxa encompassing all children taxa producing the compounds. I absolutely do not want to overload the box.
- Your concerns about the "not studied yet" part is somehow confusing. I am talking about referenced statements, so yes, probably almost all organisms contain glucose, but having a reference stating the presence of glucose with the required chemical experiments within a defined taxon remains rare (I would love to have more). I doubt this will change soon. Even if it changes, why would this be a problem? Having the information a given compound is ubiquitous is as relevant as knowing it is rare?
- I agree with your comment about the organs, as compounds found mainly in the roots, but how can you decide if it is useful to extract a substance from an organism if you do not have the information about its occurrence as a first step?
- Finally, I see the text occurrence is something emerging from different opinions, are there ways to conciliate this with Wikidata? As a first impression, I thought the infobox offered a more direct way to benefit directly from Wikidata. AdrianoRutz ( talk) 10:21, 21 September 2022 (UTC)
Implementation
- Available: currently, there is option
{{#Property:P703}}for inline text.
- Demo for
aspirin (Q18216):
{{#Property:P703|from=Q18216}}→ liquorice, Ixora coccinea
- Note: demo here requires
|from=Q18216because we are not on page " Aspirin". On page Aspirin,{{#Property:P703}}will do as expected. - We could make this into a template for inline use. Any hands up? -
DePiep (
talk)
17:37, 25 September 2022 (UTC)
- Oh wow... this looks awesome indeed! AdrianoRutz ( talk) 21:08, 25 September 2022 (UTC)
-
 Interesting discussion and very much looking forward for more integration of the
Interesting discussion and very much looking forward for more integration of the {{#Property:P703}}in Wikipedia. Hands up here to have this template. GrndStt ( talk) 07:46, 26 September 2022 (UTC)
- I am looking to make Module:WikidataIB work, which is more flexible (wikilinks, sources, list formatting). - DePiep ( talk) 17:37, 25 September 2022 (UTC)
- An eagle is about to land:
- Demo WikidataIB:
liquorice,
Ixora coccinea

- Demo WikidataIB:
liquorice,
Ixora coccinea
- -
DePiep (
talk)
08:14, 26 September 2022 (UTC)
- Oh wow... that is wonderful! Can't wait for it to land *_* AdrianoRutz ( talk) 11:41, 26 September 2022 (UTC)
- I looked a bit into the module and the sitosterol problem can be easily solved using
{{#invoke:WikidataIB|getValue|qid=Q121802|P703|maxvals=3|fwd=ALL}}, which gives: Cyrtocarpa procera, Iryanthera polyneura, Morinda citrifolia
- I did not manage to get the references yet but the module looks great!
AdrianoRutz (
talk)
19:07, 26 September 2022 (UTC)
- Yes,
|maxvals=3works well. Unfortunately, it does not hand over references AFAIK. - There is also {{
Wikidata}} →
liquorice,
[1]
Ixora coccinea
[2] (or simply bare
[1]
[2]). Less flexible, and no maxval option so bad ref effects.
DePiep (
talk)
19:57, 26 September 2022 (UTC)
- Latin should be in italic, e.g. ixora coccinea
Christian75 (
talk)
13:19, 3 October 2022 (UTC)
- Thank you for pointing this out! Do you have any experience to handle such cases automatically? It looks like the title of the WP pages are able to show them in italics but this somehow does not reflect with the modules/templates used above.
AdrianoRutz (
talk)
13:47, 3 October 2022 (UTC)
- The two options demo'ed here, read-P703-from-Wikidata: both have little or no formatting options. Hard to apply en-wiki rules (the italics), also hard (or impossible) to apply our article-chosen <ref>-formatting (like no-period-initials, CS-Vancouver, {{cite foo|..}}). DePiep ( talk) 15:21, 3 October 2022 (UTC)
- Thank you for pointing this out! Do you have any experience to handle such cases automatically? It looks like the title of the WP pages are able to show them in italics but this somehow does not reflect with the modules/templates used above.
AdrianoRutz (
talk)
13:47, 3 October 2022 (UTC)
- Latin should be in italic, e.g. ixora coccinea
Christian75 (
talk)
13:19, 3 October 2022 (UTC)
- Yes,
- An eagle is about to land:
- Just want to say I love where this is going! Great discussion, great ideas! -- Egon Willighagen ( talk) 15:48, 29 September 2022 (UTC)
Just to note I am following this conversation after a note on my talk page. First step is obviously to get consensus for this new field in the infobox. To get round the problem of too many values, could we query Wikidata to find out how many truthy statements there are. If it is more than 2, we can suppress the output, which would allow an editor to use a local override? I also think adding a maxvals option would not be too difficult — Martin ( MSGJ · talk) 09:48, 7 October 2022 (UTC)
- As indicated on the Wikidata module talk page, I found the french version has a lot of options already implemented, see: https://fr.wikipedia.org/wiki/Module:Wikidata
- This includes all we need, I guess... I tried the following
{{#invoke:Wikidata|frameFun|formatStatements|entity=Q121802|property=P703|showsource=true|numval=10}}(on .fr) and it looks nice! AdrianoRutz ( talk) 11:38, 7 October 2022 (UTC)
references
References
- ^ a b L A Mitscher; Y H Park; D Clark; J L Beal (March 1980). "Antimicrobial agents from higher plants. Antimicrobial isoflavanoids and related substances from Glycyrrhiza glabra L. var. typica". Journal of Natural Products. 43 (2): 259–269. doi: 10.1021/NP50008A004. ISSN 0163-3864. PMID 7381508. Wikidata Q71214999.
- ^ a b Chia-Lin Lee; Yung-Chih Liao; Tsong-Long Hwang; Chin-Chung Wu; Fang-Rong Chang; Yang-Chang Wu (December 2010). "Ixorapeptide I and ixorapeptide II, bioactive peptides isolated from Ixora coccinea". Bioorganic & Medicinal Chemistry Letters. 20 (24): 7354–7357. doi: 10.1016/J.BMCL.2010.10.058. ISSN 0960-894X. PMID 21106454. Wikidata Q39627968.
See Talk:Strontium ruthenate: Would it be a good idea to move this article to a more specific name? -- Leyo 12:06, 19 August 2022 (UTC)
- There's a similar problem I've had with
Neodymium nickelate but I haven't tried fixing it yet.
141
Pr
18:34, 19 August 2022 (UTC)
- Yes, we probably need to do some moving. Here are some data:
- Sr2RuO4 (RN 60862-59-1) with 1797 refs and the top cited review is MacKenzie, Andrew Peter; Maeno, Yoshiteru (2003). "The superconductivity ofSr2RuO4and the physics of spin-triplet pairing". Reviews of Modern Physics. 75 (2): 657–712. Bibcode: 2003RvMP...75..657M. doi: 10.1103/RevModPhys.75.657., A non-Cu-containing perovskite superconductor. Our article = strontium ruthenate
- SrRuO3 (RN 12169-14-1) with about 4301 refs. The top cited review is Koster, Gertjan; Klein, Lior; Siemons, Wolter; Rijnders, Guus; Dodge, J. Steven; Eom, Chang-Beom; Blank, Dave H. A.; Beasley, Malcolm R. (2012). "Structure, physical properties, and applications ofSrRuO3thin films". Reviews of Modern Physics. 84 (1): 253–298. Bibcode: 2012RvMP...84..253K. doi: 10.1103/RevModPhys.84.253. Our article = ?
- Suggestion: rename the first one
distrontium ruthenate. --
Smokefoot (
talk)
20:09, 19 August 2022 (UTC)
- I support Smokefoot's suggestion. Hatnotes can be placed on top of the page to direct to the alternate substance, along with text in the article. Graeme Bartlett ( talk) 04:04, 20 August 2022 (UTC)
- The problem is that each compound is not necessarily notable enough on its own, and this may still create confusion. That is why I think strontium ruthenate should be a page for all compounds under that name.
InterstellarGamer12321 (
talk)
09:31, 20 August 2022 (UTC)
- With thousands of references each, these two compounds are definitely notable. -- Smokefoot ( talk) 14:51, 20 August 2022 (UTC)
- I've just noticed that
"distrontium ruthenate"yields very few (non-Wikipedia) Google hits. -- Leyo 22:26, 27 August 2022 (UTC)- I will now start the daughter articles, one disambiguation and one for distrontium ruthenate.-- Smokefoot ( talk) 13:35, 28 August 2022 (UTC)
- indent: There is another one Sr3Ru2O7. What should be the name for this one ?
Rjh (
talk)
08:24, 31 August 2022 (UTC)
- from
Permanganic acid the compound names could be strontium ruthenate(oxidation state of ruthenium)
98.97.116.36 (
talk)
16:06, 29 October 2022 (UTC)
- then for Ru2O7 could be diruthenate 98.97.116.36 ( talk) 16:10, 29 October 2022 (UTC)
- also Permanganate 98.97.116.36 ( talk) 16:12, 29 October 2022 (UTC)
- from
Permanganic acid the compound names could be strontium ruthenate(oxidation state of ruthenium)
98.97.116.36 (
talk)
16:06, 29 October 2022 (UTC)
- Are RuO4(2-) and RuO3(2-) both actually called “ruthenate”? Or is RuO3(2-) also sometimes called “ruthenite”? Or surely there’s a unique systematic name for each that could be used for the articles. Sorry I can’t help more at the moment. WMF has broken and refuses to fix their software for many mobile users.
DMacks (
talk)|
- I believe this is the main issue with naming the compound.
Ruthenate redirects to
ruthenium, I cannot figure out whether one or both are called ruthenates. However, the fact that both of these are 2- but the number of strontiums are different means that we could possible name them Strontium(I) ruthenate or Strontium(II) ruthenate.
InterstellarGamer12321 (
talk)
13:33, 22 August 2022 (UTC)
- I was focused only on the differing numbers of O, not the differing numbers of Sr also. But that's a separate good point: even if the anion is (ambiguously) called "ruthenate", we obviously have several ways of naming differing oxidation states or stoichiometries of the cation.
DMacks (
talk)
14:39, 22 August 2022 (UTC)
- What do the IUPAC and other sources of authority say on naming metal-oxygen anions?
InterstellarGamer12321 (
talk)
10:16, 30 August 2022 (UTC)
- Strontium trioxidoruthenate(2−) Distrontium tetraoxidoruthenate(4−) but there are other more complex ways to name them too. Graeme Bartlett ( talk) 09:07, 31 August 2022 (UTC)
- What do the IUPAC and other sources of authority say on naming metal-oxygen anions?
InterstellarGamer12321 (
talk)
10:16, 30 August 2022 (UTC)
- I was focused only on the differing numbers of O, not the differing numbers of Sr also. But that's a separate good point: even if the anion is (ambiguously) called "ruthenate", we obviously have several ways of naming differing oxidation states or stoichiometries of the cation.
DMacks (
talk)
14:39, 22 August 2022 (UTC)
- I believe this is the main issue with naming the compound.
Ruthenate redirects to
ruthenium, I cannot figure out whether one or both are called ruthenates. However, the fact that both of these are 2- but the number of strontiums are different means that we could possible name them Strontium(I) ruthenate or Strontium(II) ruthenate.
InterstellarGamer12321 (
talk)
13:33, 22 August 2022 (UTC)
Acids with Metal Atoms as Their Center and Their Respective Anion: Naming Problems
We need a better way to name them or as above problems will arise again Ziconium ( talk) 17:50, 30 October 2022 (UTC)
- Proposal: C(oxidation of C) (MOn-m)k(oxidation of M)
- as C() M-ate()
Ziconium (
talk)
17:57, 30 October 2022 (UTC)
- We cannot really invent our own unique way of naming things like this, otherwise readers will not find the page or be confuxed what we are talking about. Either we can use common names or some sort of standard names. We can already use a formula redirect. On your proposal, I think that if we put the charge on the anion, the charge should also be on the cation, so that the formula is balanced. You propose oxidation for C, but if M is a metal with variable oxidation, then an oxidation indicator could also be on M. I think the solution found for the above situation is quite satisfactory
Monostrontium ruthenate (
SrRuO3 just created) and
Distrontium ruthenate with redirect
Sr2RuO4. How would you like to express the Wikipedia names for the strontium ruthenates in the
Ziconium naming system?
Graeme Bartlett (
talk)
22:13, 30 October 2022 (UTC)
- The responses without usernames was mine response.
- It would be strontium meta/ortho-ruthenate(IV).
Ziconium (
talk)
14:05, 31 October 2022 (UTC)
- After concideration
Ziconium (
talk)
14:07, 31 October 2022 (UTC)
- Naming it like boric or silicic acids Ziconium ( talk) 14:25, 31 October 2022 (UTC)
- After concideration
Ziconium (
talk)
14:07, 31 October 2022 (UTC)
- We cannot really invent our own unique way of naming things like this, otherwise readers will not find the page or be confuxed what we are talking about. Either we can use common names or some sort of standard names. We can already use a formula redirect. On your proposal, I think that if we put the charge on the anion, the charge should also be on the cation, so that the formula is balanced. You propose oxidation for C, but if M is a metal with variable oxidation, then an oxidation indicator could also be on M. I think the solution found for the above situation is quite satisfactory
Monostrontium ruthenate (
SrRuO3 just created) and
Distrontium ruthenate with redirect
Sr2RuO4. How would you like to express the Wikipedia names for the strontium ruthenates in the
Ziconium naming system?
Graeme Bartlett (
talk)
22:13, 30 October 2022 (UTC)
Tetranitratoxycarbon: delete or move

This molecule tetranitratoxycarbon, which was apparently conceived of by a grade schooler, is ridiculous and misleading. My guess is that to a nonchemist, the idea that a grade schooler could suggest a "reasonable" structure is striking, and the notability is increased because someone did some calculations on it. But chemists know that this structure is silly and even misleading So I propose to replace the article with a redirect to methyl nitrate and bury a trace of the content there.-- Smokefoot ( talk) 15:47, 4 November 2022 (UTC)
- Previous time it was discussed here:
Wikipedia talk:WikiProject Chemistry/Archive 44#Tetranitratoxycarbon.
DMacks (
talk)
17:36, 4 November 2022 (UTC)
- Oh, thanks for the reminder. End of discussion.-- Smokefoot ( talk) 19:25, 4 November 2022 (UTC)
- I think we should keep it, as it did get a lot of press before. However perhaps it could have more disclaimers in the first sentence, apart from "hypothetical". But instead of "silly" and "ridiculous" we could say "too unstable to exist".
Graeme Bartlett (
talk)
06:48, 5 November 2022 (UTC)
- It's all yours Graeme. I dont like the article because bicyclo(1.1.1)pentane is very strained and CNO3 analogue would be worse. Yet the molecule has four of these substituents. We could be inviting more of very strained species that simply happen to satisfy the octet rule. (snark: I guess the key is that tetranitratoxycarbon was conceived of by a cute little girl). I am also skeptical of the idea that Wikipedia Chem is in the marketing business. Being a provider of facts seems like an adequate mission. But in the big scheme of things, no big deal and thanks for taking the time to consider this topic once again.-- Smokefoot ( talk) 13:32, 5 November 2022 (UTC)
Merger proposal of sodium pertechnetate and pertechnetate
The discussion is here. Keres🌕 Luna edits! 03:56, 7 November 2022 (UTC)
Adding a list page for LEDs in the zinc blende structure
Here is a list
Gallium arsenide, Gallium nitride, Aluminum arsenide, Aluminum gallium indium phosphide, Aluminium gallium nitride, Aluminum gallium arsenide, Aluminum gallium phosphide, Cadmium telluride, Gallium antimonide,
and too many to list here
there could also be a page just listing the properties of each
Ziconium ( talk) 06:14, 16 November 2022 (UTC)
- This could be in the scope of multiple wiki projects
Ziconium (
talk)
06:19, 16 November 2022 (UTC)
- Here is a draft
- Draft:Zinc blende structure LED materials Ziconium ( talk) 18:41, 16 November 2022 (UTC)
BIOVIA Draw (the current version of ISIS/Draw)
There is a discussion about the settings to be used with BIOVIA Draw taking place at WT:WikiProject Chemistry/Structure drawing workgroup#Default BIOVIA Draw ACS template which may be of interest to those who use this software for their chemical drawings. Mike Turnbull ( talk) 16:06, 28 November 2022 (UTC)
- Please ignore! The "issue" was of my own making and does not require further discussion. Mike Turnbull ( talk) 18:26, 29 November 2022 (UTC)
Fluoroaspirin
I've opened a discussion about talk:fluoroaspirin, trying to find out whether that is the common name of the chemical and whether the article ought to be moved. -- Trovatore ( talk) 18:53, 29 November 2022 (UTC)
Organic/pharma editors needed
Fellow editors are encouraged to review the recent burst of contributions by unregistered editor User:88.105.135.216. The edits are highly specific (I think too specific), but do not suggest self-promotion. Artwork does not render well. -- Smokefoot ( talk) 14:53, 3 December 2022 (UTC)
- Just Nuklear. LTA's gonna LTA. RBI. DMacks ( talk) 18:12, 3 December 2022 (UTC)
What is Violett's solution?
While deep in a rabbit-hole from WP:RD/S, I came across a statement in proceedings from a 1906 meeting:
- "tests made for the comparison of Violett's and Fehling's solution" [5]
Fehling's solution is (now:) well-established. From context, Violett's must be some other analytical reagent to test for reducing sugars. But I cannot find any other reference to it. Help please? DMacks ( talk) 05:16, 8 December 2022 (UTC)
- Should read Violette's solution.
https://www.google.com.au/books/edition/Handbook_for_Cane_sugar_Manufacturers_an/_6w5AQAAIAAJ?hl=en&gbpv=1&dq=violetts+solution&pg=PA62&printsec=frontcover It is copper sulfate, rochelle salt and caustic soda in water.
Graeme Bartlett (
talk)
08:53, 9 December 2022 (UTC)
- Thanks! DMacks ( talk) 11:18, 10 December 2022 (UTC)
Splitting discussion for Zinc oxide

An article that been involved with ( Zinc oxide ) has content that is proposed to be removed and moved to another article ( Zinc white). If you are interested, please visit the discussion. Thank you. AngusW🐶🐶F ( bark • sniff) 17:32, 30 December 2022 (UTC)
| This is an archive of past discussions. Do not edit the contents of this page. If you wish to start a new discussion or revive an old one, please do so on the current talk page. |
| Archive 2015 | ← | Archive 2020 | Archive 2021 | Archive 2022 |
ChemBox parameters for chiral compounds
It would be nice if Wikipedia could show data on various forms of chiral compounds. For example:
- R and S enantiomers should have identical melting points, densities
- the same data for the racemate (1:1 mixture of R and S enantiomers) would differ, typically higher mp, higher d (I am ignoring solubility, odor, etc)
For species with two equivalent chiral centers (like tartaric acid), three sets of data are ideal
- R- and S-
- racemate
- meso
but the Chembox parameters do not seem to accommodate multiple m.p.'s, multiple densities, etc. Advice welcome.-- Smokefoot ( talk) 15:25, 1 January 2022 (UTC)
| Properties | |
|---|---|
| Density | 1 g/cm3 (R) 2 g/cm3 (S) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
- Maybe use </br> to separate multiple datas? --
Nucleus hydro elemon (
talk)
15:31, 1 January 2022 (UTC)
- Use <br/> and do not use </br> which should be closing a <br>. Graeme Bartlett ( talk) 22:25, 2 January 2022 (UTC)
- (ec) You're right on both counts: it's desired, and currently not possible.
- Chembox can handle separate compounds, by label and showing (called 'by index' here). See
linalool for example. However, that is only possible within |Section1={{Chembox Identifiers}} CASNo, ChEMBL, SMILES, ... only. So |Section3={{Chembox Properties}} cannot reuse those same indexes (no
|MeltingPt2=for/with|CASNo2=then). - Limitation is because of current
|SectionN=subtemplates setup. See doc:Chembox design. At the moment, an RfC § RfC on making chembox an infobox is proposing to remove this limit (by using Lua module). When all parameters are at level 1, right in {{Chembox}} like|Name=is today, the indexing can be (programmed to) be reused throughout. - DePiep ( talk) 15:44, 1 January 2022 (UTC)- Thank you both for helpful guidance. I tried and tried
|MeltingPt2=, with and without _notes, with and without _Comments. The </br> fix works fine with tartaric acid. -- Smokefoot ( talk) 15:52, 1 January 2022 (UTC)- Yes, if it shows well all's well. - DePiep ( talk) 16:00, 1 January 2022 (UTC)
- Another kludge is to have several Chembox Properties templates in different sections of the chembox. But this will make it harder to convert to other formats later. But I suppose using br has the same issue. Graeme Bartlett ( talk) 20:44, 2 January 2022 (UTC)
- Thank you both for helpful guidance. I tried and tried
| Properties | |
|---|---|
| Density | 1 g/cm3 (R) |
| Properties | |
| Density | 2 g/cm3 (S) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
- Also note that due to
parity violation from the
weak force, properties of enantiomers should differ slightly. Just this has not yet been detected yet. Also experiments may yield different results, or have never been performed, so don't assume property(R)≡property(S).
Graeme Bartlett (
talk)
20:55, 2 January 2022 (UTC)
- If repetition of Properties section floats your boat, you can use it (Change editors should solve it). However, it is bit hard to specify the substance (R, S) with it, I guess. <br/> input will have to be handled anyway (when changes happen), so don't hold back. - DePiep ( talk) 22:07, 2 January 2022 (UTC)
- Just asking (not promising): @
Graeme Bartlett: which Properties parameters would you like to have indexed? (~same setup as Identifiers maybe) -
DePiep (
talk)
22:07, 2 January 2022 (UTC)
- The main use for this for me would be: natural products that may end with A, B, C etc; different hydrates of a salt; as well as enantomers. Sometimes with drugs, a salt (eg hydrochloride) may be included with a base article, but this would mainly affect the drugbox. For the the A B C compounds, or hydrates, an indexed formula would be good (and I suppose that means C H O etc too)
Graeme Bartlett (
talk)
22:23, 2 January 2022 (UTC)
- The traditional enantio-sensitive parameters in (my) ranked order of importance: melting point, solubility, density, odor. By "indexing", can one ensure that CASNo2 is tied to MeltingPt2 to Density2 etc? This arrangement ensures that each data goes with the proper isomer. -- Smokefoot ( talk) 23:07, 2 January 2022 (UTC)
- The main use for this for me would be: natural products that may end with A, B, C etc; different hydrates of a salt; as well as enantomers. Sometimes with drugs, a salt (eg hydrochloride) may be included with a base article, but this would mainly affect the drugbox. For the the A B C compounds, or hydrates, an indexed formula would be good (and I suppose that means C H O etc too)
Graeme Bartlett (
talk)
22:23, 2 January 2022 (UTC)
- Also note that due to
parity violation from the
weak force, properties of enantiomers should differ slightly. Just this has not yet been detected yet. Also experiments may yield different results, or have never been performed, so don't assume property(R)≡property(S).
Graeme Bartlett (
talk)
20:55, 2 January 2022 (UTC)
- re GB: Looks like you misunderstood. I meant to say: you can use either way to make a good IB Chembox for thhose situation: multiple substance properties. That is: reuse |SectionN=, |SectionM={{Chembox Properties}} as in the demo, and use <br/> in single input
|MeltinghPt=(or {{ ubl}}). If and when IB Chemboc code changes (think Lua) the Change process will have to dal with it, not you today ;-) - re Smokefoot: sure
|CASNo2=≘|MeltingPt2=(qctually up to the article editor to maintain this numbercheck). Possibly one needs to c/p the indexlabels (index2=R, index3=S) from Identifiers into Props for reuse in output (because Section Props cannot read Section Ident's input). - Alas, won't happen soon tbh. - DePiep ( talk) 05:31, 3 January 2022 (UTC)
More input to the discussion of several fluorines-related templates would be welcome. -- Leyo 20:56, 3 January 2022 (UTC)
Nomination of Caffeine (data page) for deletion
The article will be discussed at Wikipedia:Articles for deletion/Caffeine (data page) until a consensus is reached, and anyone, including you, is welcome to contribute to the discussion. The nomination will explain the policies and guidelines which are of concern. The discussion focuses on high-quality evidence and our policies and guidelines.
Users may edit the article during the discussion, including to improve the article to address concerns raised in the discussion. However, do not remove the article-for-deletion notice from the top of the article.
DePiep ( talk) 14:05, 14 January 2022 (UTC)
Nomination of Butadiene (data page) for deletion
The article will be discussed at Wikipedia:Articles for deletion/Caffeine (data page) until a consensus is reached, and anyone, including you, is welcome to contribute to the discussion. The nomination will explain the policies and guidelines which are of concern. The discussion focuses on high-quality evidence and our policies and guidelines.
Users may edit the article during the discussion, including to improve the article to address concerns raised in the discussion. However, do not remove the article-for-deletion notice from the top of the article.
DePiep ( talk) 12:53, 15 January 2022 (UTC)
List of low quality chemical diagrams on Commons
Following on from Leyo's last I've just now become aware of commons:Category:Low_quality_chemical_diagrams/expired. Current contents 484 images. -- Project Osprey ( talk) 23:14, 18 January 2022 (UTC)
- The non-/expired cat has another 30. And
commons:Category:Disputed_chemical_diagrams and its /expired set are another >200 that might be even more important to fix.
DMacks (
talk)
00:05, 19 January 2022 (UTC)
- Would it be possible have new images in these categories appear on Wikipedia:WikiProject Chemistry/Article alerts? Simply switching to new images is not enough, the wrong ones must be deleted at the same time, otherwise we risk them being reused later on. -- Project Osprey ( talk) 21:44, 21 January 2022 (UTC)
Drawing challenge: Antimony potassium tartrate
Can anyone think of a good way of drawing antimony potassium tartrate? The current image (left) has numerous failings in addition to being plain ugly. I did a search on Commons before coming here which revealed the image on the right (in use on no fewer than 8 non-english wikis), I think it's my favourite structural drawing ever - somehow almost cubist. -- Project Osprey ( talk) 21:42, 5 December 2021 (UTC)
- An important question is, "what is more correct?". Per our article, with cited ref, the structure on the left is missing 3D details that our article actually discusses as a salient detail of this substance. The structure on the right agrees with
doi:10.1016/j.ica.2004.11.013, that it's a chiral cage with square–pyramidal Sb atoms. The structure on the right actually does have symmetry: its perspective is looking down a C2 axis.
DMacks (
talk)
04:23, 6 December 2021 (UTC)
- I didn't see that axis of symmetry, to me it's not very clear. I'd argue that it's showing closer to
seesaw molecular geometry. In any event both pictures fail on their depiction of formal charges. I've found at least 5 x-ray studies of this from the 1960s and 70s, there was apparently some disagreement as to its structure. Would two pictures be better? A flat one showing connectivity and a 3D model showing structure? --
Project Osprey (
talk)
10:34, 6 December 2021 (UTC)
- I agree with
DMacks Interestingly, the CCDC's entry for the X-ray structure (based on the reference he gave) has an even worse drawing than ours, although the actual structure
seen here is fine! That reference needs to be added the article, I think, plus a discussion of the controversy if that's still not sorted out.
Mike Turnbull (
talk)
10:53, 6 December 2021 (UTC)
- Not seen that interface before - handy! (although it seems to object to getting too many queries). These are what I've found so far
- 1965 doi: 10.1107/S0365110X65003080
- 1969 doi: 10.1016/0022-1902(69)80218-6
- 1972 doi: 10.1016/0022-1902(72)80229-X
- 1970 doi: 10.1107/S0567740870002133 - Structure
- 1974 deuterated doi: 10.1016/S0020-1693(00)92617-3 - structure
- I cannot access all of them, but I'm not seeing any difference in connectivity which would effect how to draw a 2-D image. --
Project Osprey (
talk)
11:15, 6 December 2021 (UTC)
- Part of the problem is that one needs to be careful which isomers are present in the structures used for the X-rays. (2R,3R)-tartaric acid is the "natural" isomer, hence the one that should be used for tartar emetic but its enantiomer (2S,3S)-tartaric acid and even the meso diastereomer, (2R,3S)-tartaric acid may have been studied and X-rayed. And that's before considerations of hydration! Mike Turnbull ( talk) 11:36, 6 December 2021 (UTC)
- Not seen that interface before - handy! (although it seems to object to getting too many queries). These are what I've found so far
- I agree with
DMacks Interestingly, the CCDC's entry for the X-ray structure (based on the reference he gave) has an even worse drawing than ours, although the actual structure
seen here is fine! That reference needs to be added the article, I think, plus a discussion of the controversy if that's still not sorted out.
Mike Turnbull (
talk)
10:53, 6 December 2021 (UTC)
- I didn't see that axis of symmetry, to me it's not very clear. I'd argue that it's showing closer to
seesaw molecular geometry. In any event both pictures fail on their depiction of formal charges. I've found at least 5 x-ray studies of this from the 1960s and 70s, there was apparently some disagreement as to its structure. Would two pictures be better? A flat one showing connectivity and a 3D model showing structure? --
Project Osprey (
talk)
10:34, 6 December 2021 (UTC)

This kind of thing is usually easier to show with a 2D snapshot of a 3D model, with an accompanying skeletal formula. -- Ben ( talk) 15:58, 6 December 2021 (UTC)
- Yes: Ben shows us how its done. but please, please do not add little naked spheres for the K+.--
Smokefoot (
talk)
17:12, 6 December 2021 (UTC)
- As always, Ben's the crystal-structure master.
DMacks (
talk)
18:09, 6 December 2021 (UTC)
- That is sort of what I was hoping for. Could I trouble you for images through the x, z and y axes? I've updated the page, to my eye the core complex is D2 symmetric. I still don't like the current 2D representation, but if Ben kind enough to generate a set of 3D images, then it becomes non-issue.-- Project Osprey ( talk) 10:02, 7 December 2021 (UTC)
- As always, Ben's the crystal-structure master.
DMacks (
talk)
18:09, 6 December 2021 (UTC)
How about these? Note these axes aren't the crystallographic a, b and c axes. Instead I defined the line passing through both antimony atoms as the z axis. -- Ben ( talk) 17:19, 9 December 2021 (UTC)
- Perfect. Duly added. Many thanks. -- Project Osprey ( talk) 15:31, 10 December 2021 (UTC)
I have a similar question

How to draw hypercubane(see right) in a good way? There are two versions of that file, old one is hard to distinguish it's double bonds, new one is too messy... -- Nucleus hydro elemon ( talk) 11:11, 12 December 2021 (UTC)

-
Nucleus hydro elemon Why not use a drawing based on the tesseract (in the
hypercube article)? That's an .svg that should be fairly easy to edit as required. That filetype is also preferred to .png because they scale better in browsers.
Mike Turnbull (
talk)
10:57, 13 December 2021 (UTC)
-
Michael D. Turnbull, I need to show single bonds and double bonds. In old version I draw based on tesseract, but it's hard to know how many single/double bonds on that picture. At top left corner it is really looks like a pentavalent carbon because of that. --
Nucleus hydro elemon (
talk)
11:27, 13 December 2021 (UTC)
- The original drawing from reference one in hypercubane is available here, on right and would be fine as the basis for a new version. It is already pretty clear, although it doesn't make the carbon double bonds explicit and arguably the hydrogens could be omitted to simplify things. Mike Turnbull ( talk) 11:34, 13 December 2021 (UTC)
-
Michael D. Turnbull, I need to show single bonds and double bonds. In old version I draw based on tesseract, but it's hard to know how many single/double bonds on that picture. At top left corner it is really looks like a pentavalent carbon because of that. --
Nucleus hydro elemon (
talk)
11:27, 13 December 2021 (UTC)
- Maybe you could use a second color to distinguish double bonds in this structure if all bonds are drawn in the same way. --
Leiem (
talk)
12:40, 13 December 2021 (UTC)
- My molecule drawing software can't colour the bonds, it only can colour functional groups. --
Nucleus hydro elemon (
talk)
13:16, 13 December 2021 (UTC)
- I use Biovia Draw, which is a free download for most users here. I then convert its output into .svg using Inkscape, when colours can readily be added. I'll try to create something later today. Mike Turnbull ( talk) 13:41, 13 December 2021 (UTC)
- Actually, the version you've created here now, Nucleus hydro elemon looks pretty good and is fine given that the article is only for a hypothetical compound! Mike Turnbull ( talk) 13:46, 13 December 2021 (UTC)
- My molecule drawing software can't colour the bonds, it only can colour functional groups. --
Nucleus hydro elemon (
talk)
13:16, 13 December 2021 (UTC)

- Just to convince myself I could do it, Nucleus hydro elemon, I've used Inkscape to copy your version of the drawing as an .svg. It will be better for readers in that format, so I'll swap that into the hypercubane article. Mike Turnbull ( talk) 15:08, 13 December 2021 (UTC)
- Superior to the useless jmol image failure linked in the chembox.
Graeme Bartlett (
talk)
10:33, 15 December 2021 (UTC)
- That Jmol sure was...something...alright. The SMILES converter in it often fails for complex 3D ringsystems. I turned it off. DMacks ( talk) 10:39, 15 December 2021 (UTC)
- I checked the Jmol yesterday and it was fine - did something happen to it? --
Ben (
talk)
13:59, 15 December 2021 (UTC)
- Yes, I think they must have modified something. I had already checked that Jmol actually gave a very good rendition of
hypercubane when I first looked a the article and the SMILES certainly hasn't changed at our end. You can look at the version
in this OID which still has the working link to Jmol before
DMacks correctly set it to "no" today because the result currently is a weird-looking flatland image. Let's hope that Jmol gets fixed: the same flatland error is the current rendering for
buckminsterfullerene. Does anyone know how to report such issues to their IT folk?
Mike Turnbull (
talk)
14:46, 15 December 2021 (UTC)
- Actually, I've found out how to report bugs (by emailing jmol-users at lists.sf.net) and I'm in the process of doing that. If anyone reading this thread thinks there is a different explanation not involving Jmol, please comment here ASAP. Mike Turnbull ( talk) 15:01, 15 December 2021 (UTC)
- The CACTVS server that the jmol tool claims to uses to convert SMILES to 3D coordinates appears to give a correct MOL file, even while the link from our hypercubane article gives the flat thing. DMacks ( talk) 15:42, 15 December 2021 (UTC)
- I have never seen hypercubane render properly as the 3D diagram using that SMILES string. I manually made up the smiles string from a diagram and description. Normally when I do this I can confirm if the string gives a good diagram, but in this case I never saw a good one. Benjah-bmm27 and Mike Turnbull, witnessing a good rendition is strange. So I would appreciate if someone can check if the SMILES is OK. And even better find a rearrangement that can render.
Graeme Bartlett (
talk)
21:45, 17 December 2021 (UTC)
- A similar JMOLfailure to render for me is at
Dimethanospiro(2.2)octaplane with
https://chemapps.stolaf.edu/jmol/jmol.php?model=C1C2C3CC4C5CC6C7C48C39C83C48C2CC5C34C6CC8C1C9C7 . Does that work for anyone like Ben or Mike?
Graeme Bartlett (
talk)
22:26, 17 December 2021 (UTC)
- Hypercubane, as
[1] (the
|SMILES=string in the infobox, also by going back to the older revision of the articel) works for me right now. DMacks ( talk) 22:42, 17 December 2021 (UTC)- That is looking OK for me too now, changed in a day!
Graeme Bartlett (
talk)
04:11, 18 December 2021 (UTC)
- I've restored the Jmol to the hypercubane article, as that is now rendering correctly: maybe after I contacted them about the bug, although I never got an acknowledgement. However, Buckminsterfullerene and Dimethanospiro(2.2)octaplane are still in flatland, so the issue has not entirely gone away. Mike Turnbull ( talk) 17:34, 23 December 2021 (UTC)
- That is looking OK for me too now, changed in a day!
Graeme Bartlett (
talk)
04:11, 18 December 2021 (UTC)
- Hypercubane, as
[1] (the
- A similar JMOLfailure to render for me is at
Dimethanospiro(2.2)octaplane with
https://chemapps.stolaf.edu/jmol/jmol.php?model=C1C2C3CC4C5CC6C7C48C39C83C48C2CC5C34C6CC8C1C9C7 . Does that work for anyone like Ben or Mike?
Graeme Bartlett (
talk)
22:26, 17 December 2021 (UTC)
- Yes, I think they must have modified something. I had already checked that Jmol actually gave a very good rendition of
hypercubane when I first looked a the article and the SMILES certainly hasn't changed at our end. You can look at the version
in this OID which still has the working link to Jmol before
DMacks correctly set it to "no" today because the result currently is a weird-looking flatland image. Let's hope that Jmol gets fixed: the same flatland error is the current rendering for
buckminsterfullerene. Does anyone know how to report such issues to their IT folk?
Mike Turnbull (
talk)
14:46, 15 December 2021 (UTC)
Jmol in infobox Chembox
| Identifiers | |
|---|---|
| |
3D model (
JSmol)
|
|
| |
- FYI: This is what {{ Chembox}} can do with SMILES and Jmol. (The demo uses indexes 1-4, see code).
- A Regular, default: Jmol reads SMILES and creates the interactive image at the external link

- F
Ferrocene: its SMILES is incorrect for 3D,
|Jmol=(another SMILES)is fed an adjusted SMILES string → shows OK in 3D
- H Hypercubane: SMILES read by Jmol (default route), obviously wrong image

 →
→  (works now see 17 Dec 22:42 DMacks above)
(works now see 17 Dec 22:42 DMacks above) - H(x) Hypercubane: same SMILES but
|Jmol=none→ Jmol does not show for this one (no error)
(no error)
Not done: possibly an adjusted SMILES string would show 3D coorect in Jmol. - DePiep ( talk) 15:57, 15 December 2021 (UTC)
- Old smiles (H) seems to work; as noted by DMacks 22:42. - DePiep ( talk) 22:48, 17 December 2021 (UTC)
Jmol & uranium
| Identifiers | |
|---|---|
| |
3D model (
JSmol)
|
|
| |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| |
I have another question for Jmol. Why it can't show uranium compounds?-- Nucleus hydro elemon ( talk) 10:52, 30 December 2021 (UTC)
Another question: Jmol become flat when too much atoms bond to one(such as IF7). -- Nucleus hydro elemon ( talk) 12:23, 22 January 2022 (UTC)
Amino acid derivatives
I was moving Mecysteine to cysteine methyl ester (hopefully the conventional name sticks). We can expect eventual creation of numerous articles on amino acid derivatives. Exampless:
- Glycine methyl ester and Glycine methyl ester hydrochloride.
- Various N-protected amino acids such as N-BOCglycine, N-Cbz...).
- Amides like glycinamide.
- Simple dipeptides can be anticipated, e.g. glycylglycine.
So if colleagues have views on naming, now is a good time to discuss.-- Smokefoot ( talk) 14:02, 24 January 2022 (UTC)
- The existing
Category:Amino acid derivatives shows some variance. I only expect there'd be a problem if there were different naming conventions in biochemistry. --
Project Osprey (
talk)
23:43, 24 January 2022 (UTC)
- I don't think we can be prescriptive on this and we would be wasting our time to try to set rules in advance. Loads of chemicals are amino acid derivatives in the broad sense that they contain the substructure -N-C-C(=O)-hetero- and many won't even be categorised as such. I would cite
nirmatrelvir and
tirzepatide as recent examples. I'd stick to the principles of
WP:Common on a case-by-case basis but I do agree with
Smokefoot that "simple" esters should be named as derivatives of their parent. Incidentally, I see no reason why
Glycine methyl ester hydrochloride isn't in an article entitled
Glycine methyl ester, along with any other notable salts and the parent compound but I'm not prepared to fight about it.....
Mike Turnbull (
talk)
12:46, 25 January 2022 (UTC)
- Not looking for prescriptions or fights...just advice and a heads up that lots of derivatives of 20+ parent amino acids will be the bases of many future articles. My feeling (not fightin words, I hope) is that the first few article titles will or could template many others. The quandry of Glycine methyl ester hydrochloride vs Glycine methyl ester illustrates the discussions I was inviting, the former can be purchased and put on the shelf, the latter is the more useful derivative. So I was thinking that the former would be the title, since readers want the info in the chembox for lab work, but the latter would comprise most of the content since experimentalists use the free ester. So its a gentle request. -- Smokefoot ( talk) 14:40, 25 January 2022 (UTC)
- I don't think we can be prescriptive on this and we would be wasting our time to try to set rules in advance. Loads of chemicals are amino acid derivatives in the broad sense that they contain the substructure -N-C-C(=O)-hetero- and many won't even be categorised as such. I would cite
nirmatrelvir and
tirzepatide as recent examples. I'd stick to the principles of
WP:Common on a case-by-case basis but I do agree with
Smokefoot that "simple" esters should be named as derivatives of their parent. Incidentally, I see no reason why
Glycine methyl ester hydrochloride isn't in an article entitled
Glycine methyl ester, along with any other notable salts and the parent compound but I'm not prepared to fight about it.....
Mike Turnbull (
talk)
12:46, 25 January 2022 (UTC)
Has no evidence of existence, just one unreliable source, probably deletion. Keres🌕 Luna edits! 01:52, 31 January 2022 (UTC)
- It has a CAS No (146505-18-2) but no real refs in SciFinder. Weirdly, the free peroxynitrate ion has >200 refs, mostly from biology as a reactive nitrogen species. The structure looks borderline explosive, so I can't imagine anyone's in a hurry to crystallise this stuff. I would support deletion. -- Project Osprey ( talk) 10:08, 31 January 2022 (UTC)
What is the difference of nitroso and nitrosyl? Perhaps these 2 categories should be merged. -- Nucleus hydro elemon ( talk) 12:14, 1 February 2022 (UTC)
- Nitroso compounds are typically organic (R-N=O) whereas nitrosyl compounds (X-N=O) are typically inorganic. It's probably best to keep the categories separate considering the number of articles in each (16+21=37 vs. 9) and the fact the properties tend to differ significantly between the two. Perhaps the nitroso category could become a subcategory of the nitrosyl category. We also have Nitroxyl which is not in either category at present. -- Ben ( talk) 18:49, 1 February 2022 (UTC)
May the (ugly) chemical structure be replaced by the crystal structure shown in the de.wikipedia article? -- Leyo 21:52, 18 January 2022 (UTC)
- Has that actually been made by copy-and-pasting bits of other structures into Microsoft Paint?... it's like a ransom note from an 80s movie... de.wiki image is for sodium metasilicate, I don't know if that's the correct unit cell, but it probably is (de.wiki do a lot of this sort of thing and they're normally correct). -- Project Osprey ( talk) 22:57, 18 January 2022 (UTC)
- It has a
sister --
Project Osprey (
talk)
23:12, 18 January 2022 (UTC)
- I would be interested in hearing of other ugly drawings. Also suspect articles. At our stage in evolution, the goal is not perfection, but avoiding embarrassment. So that the next time someone decides to criticize Wikipedia, they focus on the "Elvis articles" not us.--
Smokefoot (
talk)
00:56, 21 January 2022 (UTC)
- Yes. Hundreds. See the links the section below this one. I've isolated what should be the worst offenders (criteria: commons:Category:Disputed_chemical_diagrams > in-use on en.wiki or wikidata). A list of 26. -- Project Osprey ( talk) 15:22, 21 January 2022 (UTC)
- I would be interested in hearing of other ugly drawings. Also suspect articles. At our stage in evolution, the goal is not perfection, but avoiding embarrassment. So that the next time someone decides to criticize Wikipedia, they focus on the "Elvis articles" not us.--
Smokefoot (
talk)
00:56, 21 January 2022 (UTC)
- File:3oxstate.svg
- File:AbhinavBiochemMechanism.JPG
- File:Amitraz Synthesis Route2.png
- File:Ammonium-alum-3D-vdW.png
- File:Ammonium-hydrosulfide-3D-vdW.png
- File:Bacoside A.png
- File:Biosynthesis of CBC.png
- File:Cf oxyfluoride 3D.png
- File:Cholesterol, Tetrahymanol, Diploptene Structures.png
- File:De epoxynivalenol.tif
- File:Enolate alpha center model.svg
- File:Gladiolin.svg
- File:Hoiamide A and B.png
- File:HomoserineBiosyn.png
- File:Mercury(II)-sulfate-3D-vdW.png
- File:P-menth-1-en-4-ol.svg
- File:Peptide glycopeptide.png
- File:Potassium antimonyl tartrate.png
- File:Potassium-gluconate-3D-balls-ionic.png
- File:Potassium-thiosulfate-3D-balls-ionic.png
- File:Pyramidal dikation, hexamethyl.jpg
- File:Sodium-zincate-3D-vdW.png
- File:Synthesis of morphine.png
- File:Xenic-acid-3D-vdW.svg
- File:Zirconium(IV)-silicate-3D-vdW.png
- File:Оксид титана(III).png
There are so many 3D models of cation+anion. How to remake these files? -- Nucleus hydro elemon ( talk) 02:29, 22 January 2022 (UTC)
- Find a source describing the crystal structure, and make a new crystal structure in Jmol. In many cases, it is also acceptable to use an image of a similar crystal. – LaundryPizza03 ( d c̄) 02:55, 22 January 2022 (UTC)
There are a lot of "covalent" compounds...(Example: File:Cobalt_(II)_fluoride.png), but they are quite frequently used in eowiki(Why they use these 3D models in their reactions, example eo:Sulfura tetrafluorido)... -- Nucleus hydro elemon ( talk) 08:47, 3 February 2022 (UTC)
- Many (most?) chemistry pages on the Esperanto wiki appear to be the work of one very enthusiastic editor. As the images are in use there, they won't be deleted from commons. I don't see that there is much we can do about it, other than to keep an eye on our own pages. -- Project Osprey ( talk) 10:29, 3 February 2022 (UTC)
Alternatives
-- Nucleus hydro elemon ( talk) 13:14, 4 February 2022 (UTC)
- The concern about File:P-menth-1-en-4-ol.svg makes no sense, since one of the proposed renames already matches the filename, and the "other version" is an isomer that can be removed. – LaundryPizza03 ( d c̄) 17:47, 4 February 2022 (UTC)
Ions
The appropriate infobox to use is {{ Chembox}}. It is a modular, expandable infobox. Just use the parameters which you need; empty values will not be displayed. Chemboxes should be used for all chemical compounds, so long as they can exist in that form, even for compounds which are not isolable in pure, solvent-free form (e.g. hypochlorous acid). Chemboxes should not be used for ions, polymers, and proteins and enzymes (except simple peptides).
It said no chembox for ions, but many ion articles( tetramethylammonium, chloride) have their chembox. Also, there is a parameter(|FormulaCharge= ) in Chembox Properties that only useful for ions. -- Nucleus hydro elemon ( talk) 01:10, 11 February 2022 (UTC)
- See also Wikipedia talk:WikiProject Chemicals/Archive 2021#Chembox for polymers. DMacks ( talk) 02:43, 11 February 2022 (UTC)
- At one point I made
template:ionbox, but it really was a dumb idea as it was a just a duplicate of chembox. As molecules become more complex there is a much bigger chance that they will be ions, eg most biologic proteins or nucleic acids. Charge is just a property of a molecule. So we should explicitly state chembox can be used for ions.
Graeme Bartlett (
talk)
04:35, 12 February 2022 (UTC)
- If there are suggestions for extra {{Chembox}} parameters, to make ions feel better at home in it, please signal them. -
DePiep (
talk)
12:48, 12 February 2022 (UTC)
- My comments about chembox for ions:
- {{ Chembox Identifiers}}, {{ Chembox Thermochemistry}}, {{ Chembox Structure}}, {{ Chembox Related}} doesn't affected because it is an ion.
- {{ Chembox Pharmacology}}, {{ Chembox Hazards}}...maybe need to label what counterion for those data...(I prefer use their sodium salt or chloride)
- {{ Chembox Properties}}, {{ Chembox Explosive}} are useless for ions because they can't prepared in bulk if no counterions. Using counterions will make very different data.(Example: NaCl is very soluble in water, but AgCl is insoluble)
- --
Nucleus hydro elemon (
talk)
13:41, 12 February 2022 (UTC)
- erm, I'm not familiar with these details. How would a "label" (full data row?) look like for counterion? ASnything to do with cation, anion? - DePiep ( talk) 14:10, 12 February 2022 (UTC)
- See the right side(
| Hazards_ref =is just a placeholder, I need new parameters). -- Nucleus hydro elemon ( talk) 16:18, 12 February 2022 (UTC)
- My comments about chembox for ions:
- If there are suggestions for extra {{Chembox}} parameters, to make ions feel better at home in it, please signal them. -
DePiep (
talk)
12:48, 12 February 2022 (UTC)
| Hazards(sodium salt) | |
|---|---|
| GHS labelling: | |


| |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (
median dose)
|
6.44 mg/kg (rat, oral) 4 mg/kg (sheep, oral) 15 mg/kg (mammal, oral) 8 mg/kg (rat, oral) [2] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 5 mg/m3 [1] |
REL (Recommended)
|
C 5 mg/m3 (4.7 ppm) [10-minute] [1] |
IDLH (Immediate danger)
|
25 mg/m3 (as CN) [1] |
| Safety data sheet (SDS) | ICSC 1118 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
- {{ Chembox Properties}} includes molecular formula and weight, not just bulk physical properties like solubility and phase-chnge. DMacks ( talk) 20:25, 12 February 2022 (UTC)
- Yes, some {{
Chembox Properties}} parameters are useful. Another useful parameter for ions is
| Appearance =for the colour of it's solution. -- Nucleus hydro elemon ( talk) 05:04, 13 February 2022 (UTC)- I don't understand (yet). A
salt is not an ion (eg, salt has no charge). So, what is the topic of the example?
What would its article title be? Then, in which article (=infobox main title) would this Hazards section belong?cyanide. Altogether, I do not see how this salt or ion relates to the compounds article, and why it should be in there anyway (for example, where are the sodium salt properties shown at this moment?). Shouldn't it be in {{ Chembox Related}}, or in new dedicated Chembox section {{Chembox Salts}}? In an article section not in the infobox? Puzzling for me. Main line: if it is not about the main compound itself (article title), then it should be elsewhere not in the infobox. - DePiep ( talk) 11:45, 14 February 2022 (UTC) -
Sodium salts exists. Why not add a chembox over there (with Hazards, Properties)? -
DePiep (
talk)
11:49, 14 February 2022 (UTC)
- Cyanide ion is toxic, but if there is no counterion, we can't measure how toxic it is, so I put data of it's salt. Same goes to any other ion. -- Nucleus hydro elemon ( talk) 12:25, 14 February 2022 (UTC)
- I don't understand (yet). A
salt is not an ion (eg, salt has no charge). So, what is the topic of the example?
We had a similar discussion to this at WT:WikiProject_Chemicals/Archive_2020#Carboxylic_acids_vs_carboxylate_anion/esters. Our consensus was that very few carboxylic acids merited a separate article and we merged a number of them into their parent acid. I think that cyanide is a case where the separate article is justified but it should only discuss the ion. We have an article on sodium cyanide and, indeed, lithium cyanide, potassium cyanide and others some of which can be linked in the Chembox as related compounds. I do not support repeating, for example, the toxicity data for sodium cyanide in an article that's supposed to be limited to cyanide. The same objection is valid for any other property that can only be measured for a real sample of a specific compound. Mike Turnbull ( talk) 14:01, 14 February 2022 (UTC)
- About
sodium salts, maybe the page could be renamed to "sodium compounds". (
Wikidata:Q12555933) --
Leiem (
talk)
14:15, 14 February 2022 (UTC)
- I'm not sure if it shouldn't just be deleted Category:Sodium compounds contains 167 pages. What can we meaningfully say about such a swathe of compounds? -- Project Osprey ( talk) 14:22, 14 February 2022 (UTC)
- Nucleus: are you proposing a change of some sort? --
Project Osprey (
talk)
14:22, 14 February 2022 (UTC)
- Change what? -- Nucleus hydro elemon ( talk) 14:47, 14 February 2022 (UTC)
- only discuss the ion...then put their salt data is really not an option. By the way, there is something like "(as CN)" in IDLH parameter, which really don't care the counterion. --
Nucleus hydro elemon (
talk)
14:47, 14 February 2022 (UTC)
- I think Nucleus is looking for something like: "In the Chembox Hazards subsection, add option to note "Hazards for cyanide sodium (salt): ..." for certain data rows (datapoints). -
DePiep (
talk)
15:06, 14 February 2022 (UTC)
- Correct. --
Nucleus hydro elemon (
talk)
00:04, 15 February 2022 (UTC)
- If there is going to be such a facility, the logical material to supply hazard data for is
hydrogen cyanide, the
conjugate acid of cyanide ion. The simplest real samples associated with anions are their protonated forms.
Mike Turnbull (
talk)
11:53, 15 February 2022 (UTC)
- I think if you want to compare the effects of various counter ions against some particular property, then you should do it in the article text. Infoboxes are there to handle reams of data that are informative but don't need any narrative explanation. Describing properties across a series is inherently narrative. Make a table, explain the trend.--
Project Osprey (
talk)
12:41, 15 February 2022 (UTC)
- @ Project Osprey: Yes, "do it in the article text" is the route to go. This is the way I understand this subtopic, and our handling of it. Maybe if it appears stable over multiple compound articles, and there is a data-point in there to be discovered, then we can revisit this question and the infobox can follow. - DePiep ( talk) 13:13, 15 February 2022 (UTC)
- I think if you want to compare the effects of various counter ions against some particular property, then you should do it in the article text. Infoboxes are there to handle reams of data that are informative but don't need any narrative explanation. Describing properties across a series is inherently narrative. Make a table, explain the trend.--
Project Osprey (
talk)
12:41, 15 February 2022 (UTC)
- If there is going to be such a facility, the logical material to supply hazard data for is
hydrogen cyanide, the
conjugate acid of cyanide ion. The simplest real samples associated with anions are their protonated forms.
Mike Turnbull (
talk)
11:53, 15 February 2022 (UTC)
- Correct. --
Nucleus hydro elemon (
talk)
00:04, 15 February 2022 (UTC)
- I think Nucleus is looking for something like: "In the Chembox Hazards subsection, add option to note "Hazards for cyanide sodium (salt): ..." for certain data rows (datapoints). -
DePiep (
talk)
15:06, 14 February 2022 (UTC)
References
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0562". National Institute for Occupational Safety and Health (NIOSH).
- ^ "Cyanides (as CN)". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
Chemical data pages cleanup / task
- The WP:DPCLEANUP task thread has been moved to dedicated page WT:CHEMICALS/Chemical data pages cleanup (see current status notes). Discussions can be continued or (re)initiated there. - DePiep ( talk) 10:15, 28 February 2022 (UTC)
The ambiguity of "silene"?
The disambiguation page for Silene used to list three different classes of silicon compounds with that name. Now it lists only one. Should any of the other two be brought back? – Uanfala (talk) 02:17, 4 March 2022 (UTC)
- I think the piped entry for
Organosilicon#Silenes would still be appropriate. The other one, the redirect
Silanylidene group doesn't follow
MOS:DAB and redirects to a page about silanes, not silenes.
Mdewman6 (
talk)
02:33, 4 March 2022 (UTC)
- Thank you! I've brought back one of the two entries, and expanded the hatnote at
Silenes. Given that I don't have any chemical knowledge and that there apparently was
a past controversy among those who do, I don't trust that I would have gotten it exactly right. So, more eyes will be welcome. –
Uanfala (talk)
22:46, 13 March 2022 (UTC)
- Great! I rephrased your hatnote slightly, but this is all definitely an improvement over the previous status quo. We do need a better way to disambiguate the two compound classes, not sure how best to go about that at the moment though, and as you say, will require discussion due to the past debate. Mdewman6 ( talk) 22:55, 13 March 2022 (UTC)
- Thank you! I've brought back one of the two entries, and expanded the hatnote at
Silenes. Given that I don't have any chemical knowledge and that there apparently was
a past controversy among those who do, I don't trust that I would have gotten it exactly right. So, more eyes will be welcome. –
Uanfala (talk)
22:46, 13 March 2022 (UTC)
Revising guidance of naming of articles for organic compounds
- The following discussion is closed. Please do not modify it. Subsequent comments should be made in a new section. A summary of the conclusions reached follows.
- The result of this discussion was implement proposed revision to the chemistry naming conventions. Mdewman6 ( talk) 00:39, 30 March 2022 (UTC)
All, I think it is long past due to revisit Wikipedia:Naming_conventions_(chemistry)#IUPAC_preferred_name_vs._systematic_name, as it is misleading. First and most important, by referring to an IUPAC recommendation, this has been interpreted by some (see recent discussion with Pabsoluterince) to mean that articles should be at the preferred IUPAC name, which is not what is intended, resulting in this case in a set of messy page moves, where some redirect history was lost. The examples given are rare cases where IUPAC has acquiesced to the use of trivial (common) names, but most PIN's are systematic names, and we do not use these as article titles if there is a common name. Examples include Caproic acid rather than Hexanoic acid (see past consensus for carboxylic acids specifically), 1-Pentanol rather than Pentan-1-ol, Benzaldehyde rather than Benzenecarbaldehyde, Diethyl ether rather than Ethoxyethane, Benzophenone rather than Diphenylmethanone, etc. So, while stating "IUPAC recommends the use of non-systematic names for some [emphasis added] organic compounds" is correct, this technically only addresses a small minority of organic compounds, providing no guidance on cases where IUPAC recommends use of a systematic name, or even implying that systematic names should be used unless IUPAC recommends otherwise. Hence, the operative aspect here is not what IUPAC recommends, but the use of non-systematic names.
The section title should be revised to state "Trivial (common) name vs. systematic name" and in the text we should leave IUPAC completely out of it. Instead, we should state that trivial or common names should be used if they exist, following the spirit of WP:COMMONNAME, using systematic names when there is no trivial name that unambiguously refers to the compound. A more robust list of examples, such as those above, should then be included to more thoroughly depict the convention and provide better guidance to those writing articles or considering page moves. If there is consensus for this, I can try to come up with something, that others are then free to improve. Mdewman6 ( talk) 22:32, 12 February 2022 (UTC)
- Well yes, case in point, I did misinterpret the current policy and moved an article because of it. I found the policy extremely misleading, and basically was lead astray by my experience of what the actual common name was for the acid. I think for certain (commonly short chain) molecules, the systematic name is more recognisable and natural to use.
Pabsoluterince (
talk)
07:09, 13 February 2022 (UTC)
- I agree with
Mdewman6 that the principles of
WP:COMMONNAME take precedence. In particular, for pharmaceutical drugs we will nearly always use the
international nonproprietary name and for pesticides the
ISO common name listed here.
Mike Turnbull (
talk)
15:56, 13 February 2022 (UTC)
- Thank you for reminding us of this important issue. One reason that I was once very active in establishing articles on simple compounds was to pre-empt the IUPACers.
- It might be worthwhile to collect some titles for conversion to their common names.
Ethenone would be high on my list.--
Smokefoot (
talk)
16:28, 13 February 2022 (UTC)
- Yes, once we've made the convention more clear, I think page moves to rectify clear-cut cases such as that can proceed without need for further discussion.
- I agree with
Mdewman6 that the principles of
WP:COMMONNAME take precedence. In particular, for pharmaceutical drugs we will nearly always use the
international nonproprietary name and for pesticides the
ISO common name listed here.
Mike Turnbull (
talk)
15:56, 13 February 2022 (UTC)
I have drafted a revised section at User:Mdewman6/sandbox2, which would completely replace the existing "IUPAC preferred name vs. systematic name" section. Please take a look and feel free to comment, offer revisions, or add to the list of examples. If there are no objections or suggestions over the next week or so, I will implement the revision at the naming conventions page. Mdewman6 ( talk) 03:50, 3 March 2022 (UTC)
- There being no further comment, I am going to update that section. If concerns arise let's discuss them here.
Mdewman6 (
talk)
00:44, 13 March 2022 (UTC)
- I have questioned the procedural strength of this GF conclusion; see § Procedural questions below. - DePiep ( talk) 22:55, 14 March 2022 (UTC)
Proposed new guideline
== Organic compound names ==
This text proposal box:Trivial names (non-systematic, or "common" names) are favored for use in titles of articles for organic compounds instead of systematic names. Trivial names are usually different from the preferred name following IUPAC nomenclature. For compounds lacking trivial names, as is often the case for complex structures, substitutive nomenclature or other systematic names may be used. The general rule is to use the name most commonly used to refer to the compound, as evidenced by use in reliable sources (in line with WP:COMMONNAME).
Classes of compounds may have more specific guidance on naming, such as the use of international nonproprietary names for pharmaceutical compounds (see below).
Examples of use of trivial names and not systematic names include:
acetic acid not ethanoic acid toluene not methylbenzene lysine not 2,6-diaminohexanoic acid 1-pentanol not pentan-1-ol benzaldehyde not benzenecarbaldehyde diethyl ether not ethoxyethane benzophenone not diphenylmethanone Systematic and other accepted names for the compound not used as the article title should instead be redirects tagged with the {{ R from alternative name}} template and listed in the chembox as other names, with especially significant alternative names mentioned in the lede in bold font.
Procedural questions
- @ Mdewman6: No opinion on the proposal, but I do think this needs a more formal and more wide discussion. After all, it is probably a controversial WP:MOVE (or thousands so). At least an RfC, and publication (invitations). The advantage of this is, that the new guidance has a broad and stable consensus. - DePiep ( talk) 06:41, 14 March 2022 (UTC)
- I have reverted the Good Faith change by User:Mdewman6 in Wikipedia:Naming_conventions_(chemistry). As I noted im my previous (06:41) post, the discussion better be more wide and strong as it affects many titles and possibly is controversial. I'm sorry for arriving late here. I note that I am positive to the proposal.
- Mdewman6, could we write a
Wikipedia:Requested moves § CM proposal?
DePiep (
talk)
07:15, 14 March 2022 (UTC)
- @ DePiep:, I'm not opposed to reopening the discussion as an RfC, but there was consensus for the change above. I figured the users who cared the most about the revised naming convention, and have the pertinent expertise, are all right here. I do think I should have linked to the discussion from the chem NC talk page though, rather than after the fact.
- It's not so much a change but rather more clearly defining existing practice to avoid confusion (as discussed above). There will certainly be articles that could/should be moved based on the revised naming convention, but I doubt it would affect 'thousands' of pages. Per WP:RM#CM, articles that had past uncertainty or debate regarding the best name should still go through the RM process, but with the revised naming convention as justification. More straightforward cases could be moved boldly and reverted if there is opposition.
- I am not sure what you mean by writing a CM proposal? This is not a proposal to move a specific page. It's updating a naming convention.
Mdewman6 (
talk)
20:33, 14 March 2022 (UTC)
- Well, I admid I did not see the impact of the guidance (change) that precise, and it seems to be less dramatic. However, I thought it could meet opposition afterwards, especially when pages are being moved, and possibly on procedural grounds. That would be problematic. And since the change (your edit) was recent still without following move actions, I reverted asap (instead of talking), to prevent such after-move-discussions. As said, I just only discovered this MOS-discussion.
- My point is: being a MOS guideline, I'd go for a tough and formal consensus. For example, wide invitations (WP:CHEMISTRY, WT:NC (chemistry), some candidate articles, ..). I find three replies (supports) a bit low for a MOS change. Indeed, probably not WP:RM. And doesn't is require a formal (uninvolved editor) closure?
- I'm not from WikiLaw school, so maybe I am overconcerned. If so, I'd like to learn so; from experienced outsider? Can we ask outsider advice in this? -
DePiep (
talk)
22:51, 14 March 2022 (UTC)
- Let's start by pinging two WikiProject Chem members who are also admins to get their perspectives, DMacks and Graeme Bartlett. I will also amend my note at Wikipedia talk:Naming_conventions (chemistry) and leave a note at Wikipedia talk:WikiProject Chemistry. We can certainly to try to get a non-chemist outsider as well, not sure how best to go about seeking that, but feel free. Again, I'd favor more discussion if it were more of a change in existing practice as opposed to clarifying existing practice. Happy to setup a RfC, but we would have to transclude or copy stuff from here into a new section, probably on the naming conventions talk page. Mdewman6 ( talk) 00:22, 15 March 2022 (UTC)
- I will say, however, that in my view it would have been more appropriate to raise concerns here first rather than first reverting the addition, given there had already been discussion regarding the change. But I agree if there is any chance of the change not being the ultimate outcome, it would be best to not risk others making bold page moves based on the new text.
Mdewman6 (
talk)
00:55, 15 March 2022 (UTC)
- Thanks. As I described: reason I reverted (which is a heavy action, I know), is that once subsequential Moves were implemented, going back might be problematic in ways. - DePiep ( talk) 06:26, 15 March 2022 (UTC)
DePiep, there has been no further discussion. If your only objection to the change was procedural and not to the change itself, then I am inclined to proceed. I don't think an RfC is necessary here. The discussion has been advertised in the appropriate places. Consensus can be assumed until there is evidence of disagreement. Mdewman6 ( talk) 16:37, 26 March 2022 (UTC)
- For the record: I support the proposal (sorry for being this late). - DePiep ( talk) 16:58, 26 March 2022 (UTC)
- User:Mdewman6: I agree, we can conclude consensus for the proposal. My compliments for the initiative. - DePiep ( talk) 16:58, 26 March 2022 (UTC)
Lead disulfide
The article lead(IV) sulfide said the oxidation state of Pb in PbS2 is +4. A newer ref ( doi: 10.1016/j.jssc.2018.10.023) states that PbS2 crystallizes in the CuAl2 structure. It should be Pb2+(S2)2−. -- Leiem ( talk) 05:31, 30 March 2022 (UTC)
- Good find! Our article cites a 1966 ref for structural analysis, but this new ref discusses mentions those results as lacking as-strong support due to lack of as-high quality samples or data. This isn't my field, so I can't comment on any apparent strengths of either actual research. DMacks ( talk) 19:27, 17 April 2022 (UTC)
We have recently (thanks User:GRALISTAIR!) gotten a bunch of articles about glycidyl ethers, which are used as reactive diluents (I created both Category:Glycidyl ethers and Category:Reactive diluents). Each of them has a list of the others in their respective SEEALSO. It seems excessive to link all structurally–application-related chemicals to each other in this manner (and having to keep each one updated when new are created). Instead, WP seems like it would favor a navbox. I started Template:Glycidyl ethers. But now I wonder whether instead of this one, based on a structural motif, we should instead have one based on the reactive-diluent application case? Non-ether epoxies are possible, as are structures for non-epoxide polymer types. Would a Template:Reactive diluents (that would have the glycidyl-ethers blocks but also the vinyl compounds from that application's category) be better? DMacks ( talk) 19:36, 17 April 2022 (UTC)
- I think perhaps you've done it the right way, as some of these compounds (such as
Bisphenol A diglycidyl ether) have uses beyond being reactive diluents. --
Project Osprey (
talk)
21:32, 17 April 2022 (UTC)
- I agree with @ Project Osprey . There is at least one Glycidyl ESTER also used as an epoxy reactive diluent. GRALISTAIR ( talk) 00:19, 18 April 2022 (UTC)
However it does beg the question "Should I write an article that is entitled Glycidyl ethers?" It would be a general article possibly a stub that would give general background and reference the above GRALISTAIR ( talk) 17:00, 18 April 2022 (UTC)
- Perhaps the best place to start might be Epoxy#Diluents, which fails to even link to reactive diluent? Glycidyl ethers seem to be used exclusively for epoxy systems but I can't find the line between diluent and monomer. Bisphenol A diglycidyl ether is clearly a co-monomer. Melting points (generally lacking for these article) might be informative, I expect all the dilutes will have broad liquid ranges with very high boiling points. -- Project Osprey ( talk) 20:00, 18 April 2022 (UTC)
Phosphonium iodide
I filed a request to create an image at c:COM:GL, but was directed here by Leyo ( talk · contribs).
I intend to create 3D ball-and-stick and spacefilling models of the crystal structure of
phosphonium iodide (PH
4I) as described in the listed references, to replace the current crude cation+anion combination. It is a distorted version of the
NH
4Cl structure, with
tetragonal symmetry.
[1]
[2] The file names would be
File:Phosphonium iodide 3D balls.png and
File:Phosphonium iodide 3D vdW.png, like most other 3D molecular and crystal models. –
LaundryPizza03 (
d
c̄)
02:47, 13 March 2022 (UTC)
- Benjah-bmm27 is currently not very active … -- Leyo 10:08, 22 April 2022 (UTC)
References
- ^ Dickinson, Roscoe G. (July 1922). "The Crystal Structure of Phosphonium Iodide". Journal of the American Chemical Society. 44 (7): 1489–1497. doi: 10.1021/ja01428a015.
- ^ Sequeira, A.; Hamilton, Walter C. (September 1967). "Hydrogen Bonding in Phosphonium Iodide: A Neutron-Diffraction Study". The Journal of Chemical Physics. 47 (5): 1818–1822. Bibcode: 1967JChPh..47.1818S. doi: 10.1063/1.1712171.

Do we have such manually drawn structural formulas in other chemboxes, too? Are we fine with it? -- Leyo 09:50, 25 April 2022 (UTC)
- We are not fine with that sort of thing. I replaced it with
File:Paraloid B-72.png and also uploaded
File:Paraloid B-72 monomer-IDs.png if we wanted to discuss its structure in more detail.
DMacks (
talk)
07:12, 26 April 2022 (UTC)
- Thanks, that's what I thought, even though this image has remained in the article for ten years. ;-) -- Leyo 12:53, 28 April 2022 (UTC)
User script to detect unreliable sources
I have (with the help of others) made a small user script to detect and highlight various links to unreliable sources and predatory journals. Some of you may already be familiar with it, given it is currently the 39th most imported script on Wikipedia. The idea is that it takes something like
- John Smith "
Article of things" Deprecated.com. Accessed 2020-02-14. (
John Smith "[https://www.deprecated.com/article Article of things]" ''Deprecated.com''. Accessed 2020-02-14.)
and turns it into something like
- John Smith " Article of things" Deprecated.com. Accessed 2020-02-14.
It will work on a variety of links, including those from {{ cite web}}, {{ cite journal}} and {{ doi}}.
The script is mostly based on WP:RSPSOURCES, WP:NPPSG and WP:CITEWATCH and a good dose of common sense. I'm always expanding coverage and tweaking the script's logic, so general feedback and suggestions to expand coverage to other unreliable sources are always welcomed.
Do note that this is not a script to be mindlessly used, and several caveats apply. Details and instructions are available at User:Headbomb/unreliable. Questions, comments and requests can be made at User talk:Headbomb/unreliable.
This is a one time notice and can't be unsubscribed from. Delivered by: MediaWiki message delivery ( talk) 16:01, 29 April 2022 (UTC)
Issues with Template:Sigma-Aldrich
Template:Sigma-Aldrich, is commonly used as a reference for Chembox hazard data. Following a merger with Merk, Sigma-Aldrich appears to have reorganised its website, breaking links in some instances. The template uses the Sigma ID number to make links, e.g. 294993 for ammonia (which still works):
- {{Sigma-Aldrich|id=''294993''|name=Ammonia|access-date=27 December 2021}} → Sigma-Aldrich Co., Ammonia.
However in some cases the links, while showing as blue, do not connect properly.
- {{Sigma-Aldrich|id=''36735''|name=Bis(2-ethylhexyl) phthalate|accessdate=2022-05-12}} → Sigma-Aldrich Co., Bis(2-ethylhexyl) phthalate. Retrieved on 2022-05-12.
The template assumes that the URL should always be www.sigmaaldrich.com/GB/en/product/aldrich/<id number>
https://www.sigmaaldrich.com/GB/en/product/aldrich/294993
https://www.sigmaaldrich.com/GB/en/product/sial/36735
-- Project Osprey ( talk) 09:33, 12 May 2022 (UTC)
- So the template should switch between /aldrich/ and /sial/. Can we determine this from name or id? Any other route? -
DePiep (
talk)
10:10, 12 May 2022 (UTC)
- I'm unsure as to the scale of the problem. There may be others beyond /sial/. One option: links default to /aldrich/ (to protect existing links) but add an manual override option to replace it with something else. --
Project Osprey (
talk)
10:28, 12 May 2022 (UTC)
- There are some 175 transclusions. I've added option
|1=sialto the documentation. There also seems to exist an option|1=CATALOG(not explored). - Looks like the task is to check each instance. -
DePiep (
talk)
11:21, 12 May 2022 (UTC)
- If you give me a transclusions list I'll do the leg-work. -- Project Osprey ( talk) 11:44, 12 May 2022 (UTC)
- There are some 175 transclusions. I've added option
- I'm unsure as to the scale of the problem. There may be others beyond /sial/. One option: links default to /aldrich/ (to protect existing links) but add an manual override option to replace it with something else. --
Project Osprey (
talk)
10:28, 12 May 2022 (UTC)
- it's the template page {{ Sigma-Aldrich}}, then click What Links Here: [2] - DePiep ( talk) 12:00, 12 May 2022 (UTC)
BTW: de:Vorlage:Sigma-Aldrich#Vorlagenparameter has a few more options for this parameter. The template is transcluded in ~4500 de.wikipedia articles. -- Leyo 20:49, 12 May 2022 (UTC)
1,2-Dichloroethylene: splitting
1,2-Dichloroethylene (aka 1,2-Dichloroethene) seems to be pretty fundamental, so I think that we should split it into cis and trans isomers. A draft of the trans isomer is here. A potentially controversial aspect of my draft is the name dichloroethylene vs dichloroethene. Feel free to comment etc.-- Smokefoot ( talk) 15:29, 20 June 2022 (UTC)
- Support split, if enough unique content can be added to to each article besides the chemboxes. If split, the base name should then be a chemindex to disambiguate the two isomers. The article title should use the trivial name 'ethylene' and not the systematic 'ethene' per WP:OCHEMNAME, and given that the parent compound's article is at Ethylene and not Ethene. Mdewman6 ( talk) 20:58, 20 June 2022 (UTC)
- Comment: I'm in no way against it, but I can't help but wonder it would mean for related articles like but-2-ene or 1,3-dichloropropene which also bundle isomers together. -- Project Osprey ( talk) 21:01, 20 June 2022 (UTC)
- I think we should look at these on a case by case basis. If there is enough isomer-specific content, then a split could make sense. But I don't think we should by default always have separate articles for geometric isomers, just to have separate chemboxes, if it results in two stubs with mostly duplicated content.
- Status quo. For the time being. There seems to be little incentive. However the Z isomer gets 5700+ hits and the E isomer 4900 hits in SciFinder. Most of the top refs (aside from the usual solvent properties and water-octanol partitioning papers) are to cleaning up chlorocarbon-contaminated ground-water, which are now included in the parent article. -- Smokefoot ( talk) 18:36, 21 June 2022 (UTC)
- Well, it was your proposal...is there other content in your draft that you can include in the current article?
Mdewman6 (
talk)
19:05, 21 June 2022 (UTC)
- Hey, I know that I started this splitting idea and the literature on these two isomers is large but as I did more work fewer benefits were apparent. I did beef up the refs on the individual and the pair. There are no reviews focused on these things alone. Apologies for any inconveniences caused by me. -- Smokefoot ( talk) 12:06, 22 June 2022 (UTC)
- Well, it was your proposal...is there other content in your draft that you can include in the current article?
Mdewman6 (
talk)
19:05, 21 June 2022 (UTC)
- Status quo The current article is not very large and both the WP:COMMONNAME and molecular formula searches get there easily, which after a split would be more tedious for readers. Also, CAS no. 540-59-0 refers to unspecified/mixed isomers, which some of the literature will be. Mike Turnbull ( talk) 11:29, 22 June 2022 (UTC)
Inorganic nomenclature question
Praseodymium(II) iodide is identified as having the formula Pr3+(I−)2e− and a synonym "praseodymium diiodide". Why aren't those names the formula Pr2+(I−)2 and why is a +3 metal named as having oxidation state (III)?
DMacks (
talk)
23:56, 11 July 2022 (UTC)
- "isn't". DMacks ( talk) 02:24, 12 July 2022 (UTC)
- Your puzzlement seems appropriate. Two major issues strike me. First, the description Pr3+(I−)2e− is just one of five forms (polymorphs) or PrI2. The others are not so strange (CdX2 motif, a cluster motif, and two more). The article discusses at some length the solubility of PrI2 in water. It has no solubility in water: it dies immediately upon contact.-- Smokefoot ( talk) 00:14, 12 July 2022 (UTC)
- A compound that contains a separate e− is an electride. But that fact is uncited. Graeme Bartlett ( talk) 00:25, 12 July 2022 (UTC)
- Greenwood and Earnshaw generally refer to LnI2 and diiodides. They do sometimes specify oxidation states but with explanation: [1]
The isomorphous diiodides of Ce, Pr and Gd stand apart from all the other, salt-like, dihalides. These three, like LaI2, are notable for their metallic lustre and very high conductivities and are best formulated as {LnIII,2I−,e−}, the electron being in a delocalized conduction band.
- There's no mention of the other (non-conductive) polymorphs of PrI2 even though those were discovered well before the book was written. [2]
- According to the OCP on the f-block, the single delocalised electron per Pr atom in PrI2 occupies a broad, partially filled 5d band. [3]
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 1240. ISBN 978-0-08-037941-8.
- ^ Warkentin, E.; Bärnighausen, H. (1979). "Die Kristallstruktur von Praseodymdiiodid (Modifikation V)". Z. anorg. allg. Chem. 459: 187–200. doi: 10.1002/zaac.19794590120.
- ^ Kaltsoyannis, Nikolas; Scott, Peter (1999). Oxford Chemistry Primers: The f elements. Oxford University Press. p. 44. ISBN 978-0-19-850467-2.
- ^ Gerlitzki, Niels; Meyer, Gerd; Mudring, Anja-Verena; Corbett, John D. (2004). "Praseodymium diiodide, PrI2, revisited by synthesis, structure determination and theory". J. Alloys Compd. 380 (1–2): 211–218. doi: 10.1016/j.jallcom.2004.03.046.
- ^ Krämer, Karl; Meyer, Gerd; Fischer, Peter; Hewat, Alan W.; Güdel, Hans U. (1991). "Neutron diffraction investigation of magnetic phase transitions to long-range antiferromagnetic ordering in the "free-electron" praseodymium halides Pr2X5 (X = Br, I)". Journal of Solid State Chemistry. 95 (1): 1–13. doi: 10.1016/0022-4596(91)90370-W.
Dilithio
I have been trying to improve spelling and Wiktionary entries particularly for chemicals, and I have come across "lithio" [3] and "dilithio". [4] This is being used as if it is a replacement nomenclature for lithium in an organic compound. The current IUPAC Blue Book has "litha" for this, and I cannot find "lithio" in old versions back to 1979. But is "lithio" correct at all, perhaps sometime back in the past? (If so there should be a dictionary entry) Should we correct this "lithio" use in articles to have more valid systematic names? Graeme Bartlett ( talk) 01:06, 22 August 2022 (UTC)
- Not sure if it's IUPAC endorsed, but it does seem to be a term that has seen some use. For example, I am familiar with the reaction of
diphenylacetylene with lithium metal (under inert atmosphere) to yield a lithium-substituted butadiene that at least one paper in
Inorganic Chemistry in 1999 names as 1,4-dilithio-2,3,4,5-tetraphenylbutadiene. They also use "dilithio" to refer to this intermediate/derivative throughout. Perhaps it has fallen out of favor? But at least at one time saw some use.
Mdewman6 (
talk)
01:39, 22 August 2022 (UTC)
- Of the four cited refs for our 1,1'-Dilithioferrocene article, three use "dilithio..." in the names of specific chemicals (Organometallics 1985 and 2008, Angew Chem 2007) and and the fourth does not appear to name any specific chemicals having two lithium atoms. DMacks ( talk) 05:05, 22 August 2022 (UTC)
- I searched Chemspider, where there were 60 hits for lithio*, 13 for dilithio* and just one for lithia*, which turned out to be lithia here on WP! So whatever the Blue Book says does not seem to have been widely accepted. None of the hits I looked at were part of the main name for the compounds in question, just minor alternative names. Mike Turnbull ( talk) 09:46, 22 August 2022 (UTC)
- Thanks for your responses, based on that I think we should leave "lithio" in our article. And I have added it to Wiktionary. Graeme Bartlett ( talk) 21:08, 22 August 2022 (UTC)
Wikidata new chemical properties
Currently, there are discussion on adding properties in Wikidata:
- d:Wikidata:Property proposal/Drugcentral ID: DrugCentral (Q27697289)
- d:Wikidata:Property proposal/Probes And Drugs ID: Probes And Drugs (Q113999788) (?)
- d:Wikidata:Property proposal/Library of Integrated Network-based Cellular Signatures ID (..)
- d:Wikidata:Property_proposal/Natural_science#UniChem_compound_ID: UniChem (Q24170711)
DePiep ( talk) 16:36, 25 September 2022 (UTC)
Add a `found in taxon` statement from Wikidata in the chemical infobox
Dear members of the Chemicals WikiProject,
I am editing a lot Wikidata, particularly for chemistry and natural products (see https://www.wikidata.org/wiki/Wikidata:WikiProject_Chemistry/Natural_products). We now have a lot of curated data about chemicals found in organisms (see https://www.wikidata.org/wiki/Q2079986 for an example). Adding this information to the infobox would be very interesting. I have no idea how this can be done and do not know the procedure that has to be followed to modify an infobox template. Could anyone help me achieve this? The most crucial limitation would probably be to limit pages with many statements, such as https://www.wikidata.org/wiki/Q121802. This is elegantly done on Pubchem, as per: https://pubchem.ncbi.nlm.nih.gov/compound/222284 ("beta-Sitosterol is a natural product found in Elodea canadensis, Ophiopogon intermedius, and other organisms with data available.")
Happy to exchange more about this! AdrianoRutz ( talk) 11:33, 19 September 2022 (UTC)
- @
AdrianoRutz I think that the problem is going to be how to select the one or two that might be chosen to go in the infobox.
The full data for sitosterol on pubchem seems to have 2087 items! Our template expert is @
User:DePiep but I think that the first step is to gain consensus about what is sensible to include. All our chemboxes already link to pubchem, so the information is only a couple of clicks away for those readers who might be interested.
Mike Turnbull (
talk)
13:24, 19 September 2022 (UTC)
- Thank you very much for your prompt reply!
- The sitosterol example is the most extreme one, there are only a few with so much statements, it was chosen on purpose to answer the important point you mention.
- I have no idea how flexible the box is...my suggestion would be (based on what is feasible):
- if n >> 10
- - include randomly 2-5 taxa and add "other organisms"
- - include the parent taxa instead of the taxon so that "found in list of 999 different plants, list of 299 bacteria and list of 199 fungi" becomes "found in plants, fungi, bacteria"
- - based on different number and levels say if 0 < n < 10 display species, 10 < n < 50 display genus, and so on... AdrianoRutz ( talk) 13:57, 19 September 2022 (UTC)
- Some first notes. {{ Chembox}} (= {{Infobox chemical}}) and its sister {{ Infobox drug}} cover the chemicals, 11820+7790=19600 substances at enwiki. Guideline is WP:INFOBOX, but they have heavily creeped into Data Sheets and External Link lists (ie, lots of data is added while not necessarily described in the article body text, and the Identifiers list external links to databases like by its PubChem-identifier).
- Property (data point, infobox row) is added by proposal, usually at WT:CHEMBOX. Relevance for inclusion should be motivated. As said, there is little holdback as to keeping the list 'short' or 'highlights only' (data sheet effective).
- A thorough redesign(-discussion) is needed, IMO about splitting the Infobox into a separate datasheet-articlesection with tabular info. Such split could make the infobox more relevant, and at the same time create more space for data and its presentation. Currently, because editors prefer little or no merging with Wikidata info and, separately, keeping old style input options (infobox section header separated), developments are slow these years.
- About Wikidata: given that info (think properties) in WD are currently well maintained ('curated'), using the mass-editing options fore k's of chemicals, systematic data best be maintained at WD, and imported (read) into enwiki. Example in case: from its inclusion in 2019 here, for
|DTXSID=DSSTox substance ID (P3117) is read by default (the local parameter can overwrite). I advise for any new data point we want to add, to follow this route (read from Wikidata, maintain at Wikidata, local overwrite option). - Now about this proposal for found in taxon (P703). So it is a list, possibly long length. I question whether this is the right place then: "This substance is found in: ...", really? May be, as noted, when the list is short thus for scarce/rarities only. And: how is the item present in counter-article like Picrasma crenata? Do we have solved a similarity with "Element lithium occurs in substances ..."? Then, {{Chembox}} is born & grown from anorganic interests. Organic chemistry has other approaches in wiki, I am not familiar with. Is organic chemistry more or better organised to handle such lists?
- That said, there is no a priori objection to inclusion. If considered relevant for inclusion, I'd propose (1) read from Wikidata not local, with local overwrite option; (2) limit to "first ten", or "only when n<=10".
- -
DePiep (
talk)
16:44, 19 September 2022 (UTC)
- Thank you very much for your detailed answer!
- Shall we keep initial discussion here or move directly to /info/en/?search=Template_talk:Chembox?
- I cannot comment about the redesign of the infobox, but will surely advocate for more Wikidata-based info. It makes absolutely no sense to oppose to such enhancement.
- The model you suggested with DSSTox substance ID (P3117) is exactly what I meant. Maintain the data on Wikidata and pull it into the box.
- Concerning the found in taxon (P703), I can develop into much more detail if needed. Actually, the median number of organisms per compound is 1, with a mean of 3.3. So for a lot of compounds, it will be rare. As the ~20,0000 chemicals you mentioned are way less than what is on Wikidata, those numbers will likely go up as they are the "most known/studied" compounds. So yes, caffeine is documented in many more species than other, less known, derivatives. Having this information remains highly relevant.
- I do not know exactly how the distinction between the "short" or "highlight only" is made, but of course, if the text contains sentences saying "interestingly some Citrus species were found to contain caffeine", even better. If there are way to automate this from Wikidata, I am very eager to learn, I know this exists in other domains.
- Your comment about the "reverse property" "this organism is known to contain caffeine" is indeed a very good point. I guess I will make a request on the taxonomy infobox template to add a data point about chemical compounds found in the taxon. The discussion on Wikidata led to the decision that information about natural products is usually more compound-centric than taxon-centric, so we started more on the chemistry side, but you are perfectly right, the other way around is also very interesting!
- I sadly cannot comment about inorganic chemistry and the "lithium found in compound X" but this is a different mapping on Wikidata. It does not use the found in taxon (P703) property, but has part(s) (P527). An example for aspirin here: https://www.wikidata.org/wiki/Q18216. The data on this side is rather incomplete so I can hardly comment, but if this is of relevance for your community I am happy to come back with more info.
- Finally, I am perfectly in line with your 2 last points! :)
AdrianoRutz (
talk)
07:35, 20 September 2022 (UTC)
-
AdrianoRutz, what do you mean by "
It does not use the 'found in taxon (P703)' property, but 'has part(s) (P527)'
"? For aspirin (Q18216), I see found in taxon (P703) with three entries, so that looks OK. OTOH, a has part(s) (P527) list cannot be sure to be taxons (because any editor can, rightfully within its definition, add anything in there), so one cannot use it as proposed here. That is: P703 being used is Good! DePiep ( talk) 16:52, 25 September 2022 (UTC)- Oh, it had nothing to do with the initial found in taxon discussion anymore, it was an answer regarding your "lithium found in compound X"... the most used mapping is compound "has parts" lithium, carbon, oxygen...etc.
AdrianoRutz (
talk)
21:07, 25 September 2022 (UTC)
- OK. We can forget this detail then: P703 it is. DePiep ( talk) 08:03, 26 September 2022 (UTC)
- Oh, it had nothing to do with the initial found in taxon discussion anymore, it was an answer regarding your "lithium found in compound X"... the most used mapping is compound "has parts" lithium, carbon, oxygen...etc.
AdrianoRutz (
talk)
21:07, 25 September 2022 (UTC)
-
AdrianoRutz, what do you mean by "
- I suspect this should be expressed in words as text in the article body. If it was less than 4 taxa, perhaps it could fit in the Chembox or drugbox. But more will just overdominate the box. It is quite likely that there would be many more taxa with any particular substance, just not studied yet. And there will other substances found in all of a set of organisms, eg all animals or all photosynthetic plants. When it comes to more detail, then text will be needed, such as where in the organism the substance is found or concentrated, and if it is useful to extract the substance form that organism. - Graeme Bartlett 07:25, 21 September 2022
- I never considered adding tens of taxa into the infobox. As explained above, the information in the box would be limited to 10 (or 5, or 4) taxa. This can be either done by adding the info only for the compounds where this is true, randomly selecting n, or selecting n upper taxa encompassing all children taxa producing the compounds. I absolutely do not want to overload the box.
- Your concerns about the "not studied yet" part is somehow confusing. I am talking about referenced statements, so yes, probably almost all organisms contain glucose, but having a reference stating the presence of glucose with the required chemical experiments within a defined taxon remains rare (I would love to have more). I doubt this will change soon. Even if it changes, why would this be a problem? Having the information a given compound is ubiquitous is as relevant as knowing it is rare?
- I agree with your comment about the organs, as compounds found mainly in the roots, but how can you decide if it is useful to extract a substance from an organism if you do not have the information about its occurrence as a first step?
- Finally, I see the text occurrence is something emerging from different opinions, are there ways to conciliate this with Wikidata? As a first impression, I thought the infobox offered a more direct way to benefit directly from Wikidata. AdrianoRutz ( talk) 10:21, 21 September 2022 (UTC)
Implementation
- Available: currently, there is option
{{#Property:P703}}for inline text.
- Demo for
aspirin (Q18216):
{{#Property:P703|from=Q18216}}→ liquorice, Ixora coccinea
- Note: demo here requires
|from=Q18216because we are not on page " Aspirin". On page Aspirin,{{#Property:P703}}will do as expected. - We could make this into a template for inline use. Any hands up? -
DePiep (
talk)
17:37, 25 September 2022 (UTC)
- Oh wow... this looks awesome indeed! AdrianoRutz ( talk) 21:08, 25 September 2022 (UTC)
-
 Interesting discussion and very much looking forward for more integration of the
Interesting discussion and very much looking forward for more integration of the {{#Property:P703}}in Wikipedia. Hands up here to have this template. GrndStt ( talk) 07:46, 26 September 2022 (UTC)
- I am looking to make Module:WikidataIB work, which is more flexible (wikilinks, sources, list formatting). - DePiep ( talk) 17:37, 25 September 2022 (UTC)
- An eagle is about to land:
- Demo WikidataIB:
liquorice,
Ixora coccinea

- Demo WikidataIB:
liquorice,
Ixora coccinea
- -
DePiep (
talk)
08:14, 26 September 2022 (UTC)
- Oh wow... that is wonderful! Can't wait for it to land *_* AdrianoRutz ( talk) 11:41, 26 September 2022 (UTC)
- I looked a bit into the module and the sitosterol problem can be easily solved using
{{#invoke:WikidataIB|getValue|qid=Q121802|P703|maxvals=3|fwd=ALL}}, which gives: Cyrtocarpa procera, Iryanthera polyneura, Morinda citrifolia
- I did not manage to get the references yet but the module looks great!
AdrianoRutz (
talk)
19:07, 26 September 2022 (UTC)
- Yes,
|maxvals=3works well. Unfortunately, it does not hand over references AFAIK. - There is also {{
Wikidata}} →
liquorice,
[1]
Ixora coccinea
[2] (or simply bare
[1]
[2]). Less flexible, and no maxval option so bad ref effects.
DePiep (
talk)
19:57, 26 September 2022 (UTC)
- Latin should be in italic, e.g. ixora coccinea
Christian75 (
talk)
13:19, 3 October 2022 (UTC)
- Thank you for pointing this out! Do you have any experience to handle such cases automatically? It looks like the title of the WP pages are able to show them in italics but this somehow does not reflect with the modules/templates used above.
AdrianoRutz (
talk)
13:47, 3 October 2022 (UTC)
- The two options demo'ed here, read-P703-from-Wikidata: both have little or no formatting options. Hard to apply en-wiki rules (the italics), also hard (or impossible) to apply our article-chosen <ref>-formatting (like no-period-initials, CS-Vancouver, {{cite foo|..}}). DePiep ( talk) 15:21, 3 October 2022 (UTC)
- Thank you for pointing this out! Do you have any experience to handle such cases automatically? It looks like the title of the WP pages are able to show them in italics but this somehow does not reflect with the modules/templates used above.
AdrianoRutz (
talk)
13:47, 3 October 2022 (UTC)
- Latin should be in italic, e.g. ixora coccinea
Christian75 (
talk)
13:19, 3 October 2022 (UTC)
- Yes,
- An eagle is about to land:
- Just want to say I love where this is going! Great discussion, great ideas! -- Egon Willighagen ( talk) 15:48, 29 September 2022 (UTC)
Just to note I am following this conversation after a note on my talk page. First step is obviously to get consensus for this new field in the infobox. To get round the problem of too many values, could we query Wikidata to find out how many truthy statements there are. If it is more than 2, we can suppress the output, which would allow an editor to use a local override? I also think adding a maxvals option would not be too difficult — Martin ( MSGJ · talk) 09:48, 7 October 2022 (UTC)
- As indicated on the Wikidata module talk page, I found the french version has a lot of options already implemented, see: https://fr.wikipedia.org/wiki/Module:Wikidata
- This includes all we need, I guess... I tried the following
{{#invoke:Wikidata|frameFun|formatStatements|entity=Q121802|property=P703|showsource=true|numval=10}}(on .fr) and it looks nice! AdrianoRutz ( talk) 11:38, 7 October 2022 (UTC)
references
References
- ^ a b L A Mitscher; Y H Park; D Clark; J L Beal (March 1980). "Antimicrobial agents from higher plants. Antimicrobial isoflavanoids and related substances from Glycyrrhiza glabra L. var. typica". Journal of Natural Products. 43 (2): 259–269. doi: 10.1021/NP50008A004. ISSN 0163-3864. PMID 7381508. Wikidata Q71214999.
- ^ a b Chia-Lin Lee; Yung-Chih Liao; Tsong-Long Hwang; Chin-Chung Wu; Fang-Rong Chang; Yang-Chang Wu (December 2010). "Ixorapeptide I and ixorapeptide II, bioactive peptides isolated from Ixora coccinea". Bioorganic & Medicinal Chemistry Letters. 20 (24): 7354–7357. doi: 10.1016/J.BMCL.2010.10.058. ISSN 0960-894X. PMID 21106454. Wikidata Q39627968.
See Talk:Strontium ruthenate: Would it be a good idea to move this article to a more specific name? -- Leyo 12:06, 19 August 2022 (UTC)
- There's a similar problem I've had with
Neodymium nickelate but I haven't tried fixing it yet.
141
Pr
18:34, 19 August 2022 (UTC)
- Yes, we probably need to do some moving. Here are some data:
- Sr2RuO4 (RN 60862-59-1) with 1797 refs and the top cited review is MacKenzie, Andrew Peter; Maeno, Yoshiteru (2003). "The superconductivity ofSr2RuO4and the physics of spin-triplet pairing". Reviews of Modern Physics. 75 (2): 657–712. Bibcode: 2003RvMP...75..657M. doi: 10.1103/RevModPhys.75.657., A non-Cu-containing perovskite superconductor. Our article = strontium ruthenate
- SrRuO3 (RN 12169-14-1) with about 4301 refs. The top cited review is Koster, Gertjan; Klein, Lior; Siemons, Wolter; Rijnders, Guus; Dodge, J. Steven; Eom, Chang-Beom; Blank, Dave H. A.; Beasley, Malcolm R. (2012). "Structure, physical properties, and applications ofSrRuO3thin films". Reviews of Modern Physics. 84 (1): 253–298. Bibcode: 2012RvMP...84..253K. doi: 10.1103/RevModPhys.84.253. Our article = ?
- Suggestion: rename the first one
distrontium ruthenate. --
Smokefoot (
talk)
20:09, 19 August 2022 (UTC)
- I support Smokefoot's suggestion. Hatnotes can be placed on top of the page to direct to the alternate substance, along with text in the article. Graeme Bartlett ( talk) 04:04, 20 August 2022 (UTC)
- The problem is that each compound is not necessarily notable enough on its own, and this may still create confusion. That is why I think strontium ruthenate should be a page for all compounds under that name.
InterstellarGamer12321 (
talk)
09:31, 20 August 2022 (UTC)
- With thousands of references each, these two compounds are definitely notable. -- Smokefoot ( talk) 14:51, 20 August 2022 (UTC)
- I've just noticed that
"distrontium ruthenate"yields very few (non-Wikipedia) Google hits. -- Leyo 22:26, 27 August 2022 (UTC)- I will now start the daughter articles, one disambiguation and one for distrontium ruthenate.-- Smokefoot ( talk) 13:35, 28 August 2022 (UTC)
- indent: There is another one Sr3Ru2O7. What should be the name for this one ?
Rjh (
talk)
08:24, 31 August 2022 (UTC)
- from
Permanganic acid the compound names could be strontium ruthenate(oxidation state of ruthenium)
98.97.116.36 (
talk)
16:06, 29 October 2022 (UTC)
- then for Ru2O7 could be diruthenate 98.97.116.36 ( talk) 16:10, 29 October 2022 (UTC)
- also Permanganate 98.97.116.36 ( talk) 16:12, 29 October 2022 (UTC)
- from
Permanganic acid the compound names could be strontium ruthenate(oxidation state of ruthenium)
98.97.116.36 (
talk)
16:06, 29 October 2022 (UTC)
- Are RuO4(2-) and RuO3(2-) both actually called “ruthenate”? Or is RuO3(2-) also sometimes called “ruthenite”? Or surely there’s a unique systematic name for each that could be used for the articles. Sorry I can’t help more at the moment. WMF has broken and refuses to fix their software for many mobile users.
DMacks (
talk)|
- I believe this is the main issue with naming the compound.
Ruthenate redirects to
ruthenium, I cannot figure out whether one or both are called ruthenates. However, the fact that both of these are 2- but the number of strontiums are different means that we could possible name them Strontium(I) ruthenate or Strontium(II) ruthenate.
InterstellarGamer12321 (
talk)
13:33, 22 August 2022 (UTC)
- I was focused only on the differing numbers of O, not the differing numbers of Sr also. But that's a separate good point: even if the anion is (ambiguously) called "ruthenate", we obviously have several ways of naming differing oxidation states or stoichiometries of the cation.
DMacks (
talk)
14:39, 22 August 2022 (UTC)
- What do the IUPAC and other sources of authority say on naming metal-oxygen anions?
InterstellarGamer12321 (
talk)
10:16, 30 August 2022 (UTC)
- Strontium trioxidoruthenate(2−) Distrontium tetraoxidoruthenate(4−) but there are other more complex ways to name them too. Graeme Bartlett ( talk) 09:07, 31 August 2022 (UTC)
- What do the IUPAC and other sources of authority say on naming metal-oxygen anions?
InterstellarGamer12321 (
talk)
10:16, 30 August 2022 (UTC)
- I was focused only on the differing numbers of O, not the differing numbers of Sr also. But that's a separate good point: even if the anion is (ambiguously) called "ruthenate", we obviously have several ways of naming differing oxidation states or stoichiometries of the cation.
DMacks (
talk)
14:39, 22 August 2022 (UTC)
- I believe this is the main issue with naming the compound.
Ruthenate redirects to
ruthenium, I cannot figure out whether one or both are called ruthenates. However, the fact that both of these are 2- but the number of strontiums are different means that we could possible name them Strontium(I) ruthenate or Strontium(II) ruthenate.
InterstellarGamer12321 (
talk)
13:33, 22 August 2022 (UTC)
Acids with Metal Atoms as Their Center and Their Respective Anion: Naming Problems
We need a better way to name them or as above problems will arise again Ziconium ( talk) 17:50, 30 October 2022 (UTC)
- Proposal: C(oxidation of C) (MOn-m)k(oxidation of M)
- as C() M-ate()
Ziconium (
talk)
17:57, 30 October 2022 (UTC)
- We cannot really invent our own unique way of naming things like this, otherwise readers will not find the page or be confuxed what we are talking about. Either we can use common names or some sort of standard names. We can already use a formula redirect. On your proposal, I think that if we put the charge on the anion, the charge should also be on the cation, so that the formula is balanced. You propose oxidation for C, but if M is a metal with variable oxidation, then an oxidation indicator could also be on M. I think the solution found for the above situation is quite satisfactory
Monostrontium ruthenate (
SrRuO3 just created) and
Distrontium ruthenate with redirect
Sr2RuO4. How would you like to express the Wikipedia names for the strontium ruthenates in the
Ziconium naming system?
Graeme Bartlett (
talk)
22:13, 30 October 2022 (UTC)
- The responses without usernames was mine response.
- It would be strontium meta/ortho-ruthenate(IV).
Ziconium (
talk)
14:05, 31 October 2022 (UTC)
- After concideration
Ziconium (
talk)
14:07, 31 October 2022 (UTC)
- Naming it like boric or silicic acids Ziconium ( talk) 14:25, 31 October 2022 (UTC)
- After concideration
Ziconium (
talk)
14:07, 31 October 2022 (UTC)
- We cannot really invent our own unique way of naming things like this, otherwise readers will not find the page or be confuxed what we are talking about. Either we can use common names or some sort of standard names. We can already use a formula redirect. On your proposal, I think that if we put the charge on the anion, the charge should also be on the cation, so that the formula is balanced. You propose oxidation for C, but if M is a metal with variable oxidation, then an oxidation indicator could also be on M. I think the solution found for the above situation is quite satisfactory
Monostrontium ruthenate (
SrRuO3 just created) and
Distrontium ruthenate with redirect
Sr2RuO4. How would you like to express the Wikipedia names for the strontium ruthenates in the
Ziconium naming system?
Graeme Bartlett (
talk)
22:13, 30 October 2022 (UTC)
Tetranitratoxycarbon: delete or move

This molecule tetranitratoxycarbon, which was apparently conceived of by a grade schooler, is ridiculous and misleading. My guess is that to a nonchemist, the idea that a grade schooler could suggest a "reasonable" structure is striking, and the notability is increased because someone did some calculations on it. But chemists know that this structure is silly and even misleading So I propose to replace the article with a redirect to methyl nitrate and bury a trace of the content there.-- Smokefoot ( talk) 15:47, 4 November 2022 (UTC)
- Previous time it was discussed here:
Wikipedia talk:WikiProject Chemistry/Archive 44#Tetranitratoxycarbon.
DMacks (
talk)
17:36, 4 November 2022 (UTC)
- Oh, thanks for the reminder. End of discussion.-- Smokefoot ( talk) 19:25, 4 November 2022 (UTC)
- I think we should keep it, as it did get a lot of press before. However perhaps it could have more disclaimers in the first sentence, apart from "hypothetical". But instead of "silly" and "ridiculous" we could say "too unstable to exist".
Graeme Bartlett (
talk)
06:48, 5 November 2022 (UTC)
- It's all yours Graeme. I dont like the article because bicyclo(1.1.1)pentane is very strained and CNO3 analogue would be worse. Yet the molecule has four of these substituents. We could be inviting more of very strained species that simply happen to satisfy the octet rule. (snark: I guess the key is that tetranitratoxycarbon was conceived of by a cute little girl). I am also skeptical of the idea that Wikipedia Chem is in the marketing business. Being a provider of facts seems like an adequate mission. But in the big scheme of things, no big deal and thanks for taking the time to consider this topic once again.-- Smokefoot ( talk) 13:32, 5 November 2022 (UTC)
Merger proposal of sodium pertechnetate and pertechnetate
The discussion is here. Keres🌕 Luna edits! 03:56, 7 November 2022 (UTC)
Adding a list page for LEDs in the zinc blende structure
Here is a list
Gallium arsenide, Gallium nitride, Aluminum arsenide, Aluminum gallium indium phosphide, Aluminium gallium nitride, Aluminum gallium arsenide, Aluminum gallium phosphide, Cadmium telluride, Gallium antimonide,
and too many to list here
there could also be a page just listing the properties of each
Ziconium ( talk) 06:14, 16 November 2022 (UTC)
- This could be in the scope of multiple wiki projects
Ziconium (
talk)
06:19, 16 November 2022 (UTC)
- Here is a draft
- Draft:Zinc blende structure LED materials Ziconium ( talk) 18:41, 16 November 2022 (UTC)
BIOVIA Draw (the current version of ISIS/Draw)
There is a discussion about the settings to be used with BIOVIA Draw taking place at WT:WikiProject Chemistry/Structure drawing workgroup#Default BIOVIA Draw ACS template which may be of interest to those who use this software for their chemical drawings. Mike Turnbull ( talk) 16:06, 28 November 2022 (UTC)
- Please ignore! The "issue" was of my own making and does not require further discussion. Mike Turnbull ( talk) 18:26, 29 November 2022 (UTC)
Fluoroaspirin
I've opened a discussion about talk:fluoroaspirin, trying to find out whether that is the common name of the chemical and whether the article ought to be moved. -- Trovatore ( talk) 18:53, 29 November 2022 (UTC)
Organic/pharma editors needed
Fellow editors are encouraged to review the recent burst of contributions by unregistered editor User:88.105.135.216. The edits are highly specific (I think too specific), but do not suggest self-promotion. Artwork does not render well. -- Smokefoot ( talk) 14:53, 3 December 2022 (UTC)
- Just Nuklear. LTA's gonna LTA. RBI. DMacks ( talk) 18:12, 3 December 2022 (UTC)
What is Violett's solution?
While deep in a rabbit-hole from WP:RD/S, I came across a statement in proceedings from a 1906 meeting:
- "tests made for the comparison of Violett's and Fehling's solution" [5]
Fehling's solution is (now:) well-established. From context, Violett's must be some other analytical reagent to test for reducing sugars. But I cannot find any other reference to it. Help please? DMacks ( talk) 05:16, 8 December 2022 (UTC)
- Should read Violette's solution.
https://www.google.com.au/books/edition/Handbook_for_Cane_sugar_Manufacturers_an/_6w5AQAAIAAJ?hl=en&gbpv=1&dq=violetts+solution&pg=PA62&printsec=frontcover It is copper sulfate, rochelle salt and caustic soda in water.
Graeme Bartlett (
talk)
08:53, 9 December 2022 (UTC)
- Thanks! DMacks ( talk) 11:18, 10 December 2022 (UTC)
Splitting discussion for Zinc oxide

An article that been involved with ( Zinc oxide ) has content that is proposed to be removed and moved to another article ( Zinc white). If you are interested, please visit the discussion. Thank you. AngusW🐶🐶F ( bark • sniff) 17:32, 30 December 2022 (UTC)





