| This
guideline documents an English Wikipedia
naming convention. It is a generally accepted standard that editors should attempt to follow, though
occasional exceptions may apply. Any substantive edit to this page should reflect
consensus. When in doubt, discuss first on the
talk page. |
| Manual of Style |
| Chemistry |
|---|
| Categories |
From Wikipedia:Naming conventions:
Generally, article naming should give priority to what the majority of English speakers would most easily recognize, with a reasonable minimum of ambiguity, while at the same time making linking to those articles easy and second nature.
Trivial names (non-systematic, or "common" names) are favored for use in titles of articles for organic compounds instead of systematic names. Trivial names are usually different from the preferred name following IUPAC nomenclature. For compounds lacking trivial names, as is often the case for complex structures, substitutive nomenclature or other systematic names may be used. The general rule is to use the name most commonly used to refer to the compound, as evidenced by use in reliable sources (in line with WP:COMMONNAME).
Classes of compounds may have more specific guidance on naming, such as the use of international nonproprietary names for pharmaceutical compounds (see WP:NCMED and below).
Examples of use of trivial names and not systematic names include:
| acetic acid | not | ethanoic acid |
| toluene | not | methylbenzene |
| lysine | not | 2,6-diaminohexanoic acid |
| 1-pentanol | not | pentan-1-ol |
| benzaldehyde | not | benzenecarbaldehyde |
| diethyl ether | not | ethoxyethane |
| benzophenone | not | diphenylmethanone |
Systematic and other accepted names for the compound not used as the article title should instead be redirects tagged with the {{ R from alternative name}} template and listed in the chembox as other names, with especially significant alternative names mentioned in the lede in bold font.
Groups
- The groups of the periodic table are numbered left to right as 1–18, with the f-block groups (columns) unnumbered. As recommended by IUPAC (1988). [1]
- Note that "group" has a chemical meaning and "column" has the tabular meaning.
- Alternatively, they may be named by their first element, e.g. "boron group" for group 13. [2] (But per below, avoid iron group for group 8 as ambiguous.)
- In the 1–18 scheme, f-block groups are unnumbered. There is no widely-agreed system of numbering these groups in the literature, so one should not be used.
- Avoid labeling these groups as n/a (for not applicable?, or not available i.e. "not found yet"?). Best is to leave this group number blank. Provide textual description when needed. This also applies to any (theoretical) g-block groups.
- To refer to such a group (column), list the elements explicitly, e.g. ytterbium and nobelium. Do not name by first element (ytterbium group). The reasons for this are that many of the expected first-element names have other meanings as rare earth separation groups (see "Non-column groups" below), and that referring to individual f-block groups in the literature is rare (so it's better to be explicit).
- Hydrogen belongs to group 1 (not group 17 or floating outside all groups), and helium belongs to group 18 (not group 2). [1] [3]
- "halogen" and "noble gas" are used to refer to the entirety of groups 17 and 18, per a 2016 IUPAC report about naming new elements [4] Some sources focusing on superheavy elements may not consider tennessine (Ts) to be a halogen, or oganesson (Og) to be a noble gas, as their predicted chemistry is generally supposed to be quite different from those of their lighter congeners. Such more limited usage should be explained before its first occurrence.
- A few groups have IUPAC-approved names, called trivial name, that may also be used:
- alkali metal = group 1 except hydrogen
- alkaline earth metal = group 2
- triels = group 13
- tetrels = group 14
- pnictogen = group 15 (earlier pentels)
- chalcogen = group 16
- halogen = group 17
- noble gas = group 18
- Triels (group 13), tetrels (group 14) and pentels (group 15) have been previously recommended by IUPAC (in 1970), and the former two are still used by them, though they are not in the most recent Red Book. Avoid rarer synonyms like icosagen (group 13) and crystallogen (group 14).
- Avoid coinage metal for group 11, since "metal used for making coins" (see coinage metal) contains also metals outside of group 11, and roentgenium in group 11 is (for obvious reasons) not used for coins.
- The group numberings with Roman numbers and "A" and "B" suffixes (like VIIA, VIII) should not be used, as they are outdated and ambiguous. For example, in the CAS scheme "group VIIB" denotes manganese group (group 7), while in the old IUPAC scheme it denotes halogen group (group 17). If they are used at all, context should clarify (disambiguate) into the 1–18 numbering. Care should be taken when translating old sources that use this nomenclature to modern terminology.
- Some sets of groups have names:
- The main group elements are the elements of groups 1, 2, and 13–18 (i.e. the s-block and p-block). [5]
- The transition elements are the elements of groups 3–12 (i.e. the d-block) – this in particular includes group 12 (although some sources do not include it). [5]
- The inner transition elements are f-block elements. [2]
The scandium group (group 3)
- The scandium group should by default be treated as containing the elements scandium (Sc), yttrium (Y), lutetium (Lu), and lawrencium (Lr). Lanthanum (La) and actinium (Ac), which in some sources take the positions of lutetium and lawrencium (especially in older sources), should not be in group 3. Care should be taken when employing sources that mention "group 3" or "group IIIB" or "group IIIA", since they may not always use this arrangement.
- This group 3 set is established per the 1988 IUPAC report [1] and per a subsequent 2021 provisional report by a IUPAC project dedicated to this constitution question [3]
- In fact, the old classification was based on erroneous early measurements of electron configurations, [6] and was already criticised as "incorrect" by Lev Landau and Evgeny Lifshitz in 1948. [7] The IUPAC report convention is accepted by most chemists and physicists considering the subject. [8] [9]
- Note that the so-called "IUPAC(-published) periodic table" is not actually approved by IUPAC. [10] It leaves the spaces below yttrium blank, and there is significant confusion among (and within) sources on whether that includes the lanthanides and actinides in group 3 or not; [11] hence this form is also best avoided.
- Alternative compositions of group 3 may be illustrated when describing the dispute, e.g. in Group 3 element#Composition.
Non-column groups
- In case of commonly named "groups" that are not a column, such as Platinum group or Iron group, care should be taken that no confusion remains with column-wise periodic table group.
18 vs 32 columns
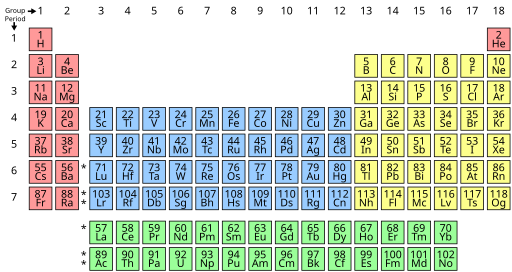
- The periodic table can be displayed in either 18- or 32-column format, with the 32-column format as in Fig. 4 of the aforementioned 2021 IUPAC report. The forms present the same scientific statement and should be consistent with each other. The 18-column format cuts out the f-block rows, containing the elements lanthanum (La) through ytterbium (Yb) and actinium (Ac) through nobelium (No), and places them at the bottom of the table. [1] Asterisks should be placed between groups 2 and 3 to indicate the theoretical position of these cut-out rows.
- Note that these f-block rows do not equal the lanthanides/lanthanoids (La–Lu) and actinides/actinoids (Ac–Lr) which commonly extend to include lutetium and lawrencium.
- Other structures, e.g. the Janet left-step or the 8-group form, should only be used when specifically illustrating such alternative periodic tables.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | H | He | ||||||||||||||||||||||||||||||
| 2 | Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||
| 3 | Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||
| 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||
| 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||
| 6 | Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| 7 | Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
|
| ||||||||||||||||||||||||||||||||
Blocks
At this English wiki, the preferred presentation coloring scheme of the elements is by blocks (s, p, d, or f).
The colours to be used are given in {{
Element color}}:
-
s-block→#ff9999 -
p-block→#fdff8c -
d-block→#99ccff -
f-block→#9bff99
This template also gives colours for the higher orbitals:
-
g-block→#fd99ff -
h-block→#7ed9d9 -
i-block→#e0c880
Categorising elements
Categorisation is an attempt to classify all elements into one overall scheme, with each element in exactly one category (as opposed to listing non-completifying sets like coinage metals). A general classification is not achieved in sources (let alone without WP:NPOV and WP:OR). Such categorisation then is not to be used in general (for example, not as general coloring of the periodic table).
A general colouring by metallicity is discouraged, because there are WP:NPOV and WP:NOR concerns: the borders of some categories can differ drastically between sources (e.g. metalloids), and there is no set of categories that are mutually exclusive and yet cover the entire periodic table (e.g. astatine is quite likely to be both a metal and a halogen). However, such coloring may be used to base and illustrate dedicated topics (such as in sections of metalloids). Individual, well-established "metallicity" categories, e.g. transition metal, post-transition metal, metalloid, rare earth metals etc., can be referred to without "all elements all once" colouring.
When a specific categorisation scheme is relevant (e.g. {{
ChemicalBondsToCarbon}}), a periodic table-filling categorisation can be applied.
Traditionally, the names of three elements have been spelled differently in different parts of the English-speaking world. For articles about chemistry-related topics, Wikipedia follows the spellings recommended by the International Union of Pure and Applied Chemistry (IUPAC): [12] [2]
| aluminium | not | aluminum |
| sulfur | not | sulphur |
| caesium | not | cesium |
These spellings should be used in all chemistry-related articles on English Wikipedia, even if they conflict with the other national spelling varieties used in the article.
This convention should also be applied to all compounds and derivative names of these chemicals: sulfate not sulphate; sulfuric not sulphuric; etc.
The English name of element 74 is tungsten and not wolfram: the latter was once adopted by IUPAC (in 1949), but is no longer recommended by them (even as an alternative) per the 2011 Principles. (Of course, the symbol is still W, and the name "wolfram" may be referred to when explaining why that is the case, or when quoting text in languages that use that name.) Similarly for derivatives (per the 2005 Red Book), use tungstate, tungsty, tungsta and not wolframate, wolframy, wolframa. (And similarly avoid the old names emanation and niton for element 86 outside historical contexts; there is a real confusion even in today's literature between radon meaning the element Rn and radon meaning the isotope 222Rn, but the way to solve that is by explicitly clarifying what is meant, not resurrecting archaic terms that have fallen out of use and may not be generally understood.)
Elements that have not been given trivial names by IUPAC may be referred to either by IUPAC systematic element names and symbols, or by atomic numbers. So ununennium and element 119 are both correct, and symbols Uue, 119, E119 may all be used in text. However, for natural disambiguation, the article titles should be at the systematic names.
When new elements are recognised, do not change the names and symbols to those proposed by the discoverers until the public comment period expires and IUPAC makes the final announcement! (The symbol has changed between the initial proposal and the final recommendation before, in the case of copernicium: originally proposed as Cp, but became Cn.)
Isotope names
- Isotopes are named and identified as [element]-[mass number] with regular lowercase and hyphen (carbon-13), or by equivalent symbol format 13C (in wikicode:
<sup>13</sup>C). Avoid 13C, C-13, C13 (and avoid like single character U+2074 ⁴ SUPERSCRIPT FOUR, as used in Wikidata). - Metastable states are noted by format tantalum-180m1, 180m1Ta. The number ⟨1⟩ can be omitted when there is only one isomeric state, e.g. tantalum-180m, 180mTa. You may use a nuclide template. Corresponding letters also used in literature, these are not used in this wiki: ⟨m n p q⟩.
- The hydrogen isotopes are exceptions where the common special names may be used:
- Protium (= hydrogen-1, 1H).
- Deuterium, D, 2D (= hydrogen-2, 2H).
- Tritium, T, 3T (= hydrogen-3, 3H).
- Chemical formulae with deuterated solvents (for example, in NMR use) are customarily described with "D" as chemical symbol: CD3OD, methanol-d4; CD3SOCD3, DMSO-d6 (regular notation would be C2H3O2H). These established systems are acceptable, provided they are consistent within an article.
- Some isotopes have had historical or confusing names and symbols, these are to be avoided (except when quoting historical usage): e.g. thoron (Tn) (220Rn), actinon (An) (219Rn), ionium (Io) (230Th), radiocarbon (14C).
- It is permissible to use the atomic number rather than the symbol in contexts about history when anachronism is to be avoided, e.g. "In 2003, the collaboration at the JINR discovered 288115 and 284113" (because at that point those elements had not yet been named). But this should be avoided outside talking about history. Avoid using the systematic element names in such contexts even when talking about history, because they are not supposed to be used once the element actually has a trivial name. For isotopes of undiscovered elements, either systematic names (e.g. 296Uue) or atomic numbers (e.g. 296119) are acceptable for the symbols, but only the systematic names can be used for the full names (e.g. ununennium-296, not element 119-296 which nobody uses).
Main isotopes
- Currently under discussion for addition
- See WT:ELEMENTS:What is a "Main_isotope"?
Elements with atomic number Z ≥ 119 are theoretical elements, they have not been discovered (synthesized). Regarding public-page creation and content (reader-visible pages, such as artricle, category, template):
- Element page: Articles pages, by systematic name, exist for E119 up to E184: Ununennium (Element 119).
- These are either full articles (like Ununennium) or redirects (like Unbitrium, 58 redirects). This serves readers searching for these element names.
- Redirect element articles like Unbitrium (Element 123) R:
- Per WP:notability and WP:Reliable sources, these articles can and may only be created with content when substantial information is available.
- Secondary public pages for element articles, like Isotopes of unbioctium, {{ Infobox unbioctium}}, :Category:unbioctium:
- These can and may only be created (1) when the base element article exists by content, and (2) when substantial new RL information like papers is available. These secondary pages may not be created as redirect.
To compare: Disovered element Oganesson (E118) has its article, plus all five secondary articles; none is a redirect.
As of 4 Jan 2023 [update]: Effectively this means that currently public-page creation, by content or redirect, is not permitted nor needed. See also documentation at Template:Infobox element § Theoretical elements handling.
The names of elements and other chemicals should be written without abbreviations and treated as common nouns. They do not have special capitalization or italic formatting (see MOS:CAPS and MOS:ITALIC) regardless of the origin of the name unless appropriate for the start of a sentence or part of a title. The symbols for elements should always be capitalized.
- Use: Iron and californium (Fe and Cf) are metals. After I iron my clothes, I will go to California.
- Avoid: Iron and californium (fe and cf) are metals.
- Avoid: Iron and Californium (Fe and Cf) are metals.
- Avoid: Iron and californium (Fe and Cf) are metals.
Prefixes that are such as sec-, tert-, ortho-, meta-, para-, α-, β-, D-, L-, (+)-, (−)-, (R)-, (S)- and the numerical prefixes are not considered part of the name: they each follow their own capitalization and italics rules, and it is instead the first letter of the main part of the name that is treated as the "first letter" of the name to be capitalized where appropriate. Exceptions are cyclo, iso, neo, and spiro which are considered part of the name and therefore not italicized or hyphenated. Substituent groups are also considered part of the name.
In former versions of the IUPAC recommendations, names were written with a capital initial letter. This practice has been abandoned in later publications. [13] IUPAC policy differentiate proper names from things named after proper names. This rule (full name uncapitalized but symbol capitalized) applies also to isotopes and nuclides, when completely written out: thus 14C but carbon-14.
When the chosen article title starts with a prefix including positional identifiers (ortho-, meta-, para-, N-, O-, α-, β-, γ-, etc.), isomeric identifiers (sec-, tert-, etc.), stereochemical identifiers (cis-, trans-, (E)-, (Z)-, etc.), chiral identifiers ((R)-, (S)-, D-, L-, (+)-, (−)-, etc.), or numbers, the first letter after the prefix in the name should be capitalized: hence titlecase 1,1,1-Trichloroethane not 1,1,1-trichloroethane. A redirect from the uncapitalized version should be created to simplify linking from other articles.
Non-numerical prefixes are italicized and uncapitalized in titles (tert-Butyl alcohol, for example). Both numerical and non-numerical prefixes are followed by a hyphen. The template {{ DISPLAYTITLE}} is used to display titles which start with lowercase letters or include italics.
Note that cyclo, iso, neo, and spiro are considered part of a chemical name (such as isopropanol) and not considered prefixes. No hyphens or italics are used in these cases.
Article titles for compounds and related topics should reflect how the compound name is commonly written and use Greek letter prefixes if appropriate, e.g. Α-Ketoglutaric acid, using {{ DISPLAYTITLE}} to display as α-Ketoglutaric acid, not Alpha-Ketoglutaric acid and displaying as alpha-Ketoglutaric acid. In the article page name (article location) it is important to use the uppercase Greek letter, even though it can be easily confused with uppercase Latin letters in many cases, so that DISPLAYTITLE (or alternatively, {{ lowercase title}}) will work properly. It is also essential to ensure redirects exist for all possible typed variations of the compound name so searchers can easily reach the appropriate article.
Stock nomenclature for inorganic compounds is based on the indication of the oxidation number (as a roman numeral, in parentheses) of each of the major elements in the compound, e.g. iron(III) chloride. It is widely, if sometimes incorrectly, used on Wikipedia for the titles of articles about inorganic compounds. It is not obligatory, as there are other acceptable methods for naming these compounds, but it is often preferred as the most common non-ambiguous name for a substance. The following guidelines are based on current WikiBestPractice:
- Only the cationic element (i.e. the element whose name appears unchanged in the compound name) is assigned its oxidation number. Except in rare cases (none at present), we do not assign the oxidation number in the anion: hence potassium permanganate not potassium manganate(VII), sodium hypochlorite not sodium chlorate(I).
- There is no space between the end of the element name and the opening parenthesis: hence silver(I) fluoride not silver (I) fluoride. Note that this is an exception to the usual English style for parentheses.
- It is not necessary to specify the oxidation number when there is no possibility of ambiguity in the compound title: hence sodium chloride not sodium(I) chloride.
- Stock nomenclature should be used for predominantly ionic compounds. Compounds with a substantial degree of covalency should be named by stoichiometric nomenclature: hence titanium tetrachloride not titanium(IV) chloride. However, it should be noted that ionic vs. covalent (not to mention metallic and van der Waals bonding) is a continuum and that many such situations will require significant editorial judgement (e.g. mercury(II) chloride, which is in fact a molecular compound, or ruthenium(IV) oxide which exhibits metallic conductivity).
Names of oxyanions should in general follow the names in Table X of the IUPAC 2005 Red Book. [2] Exceptions can be made if an alternate name is much more common in the literature, e.g. xenate rather than xenonate (don't generalise this to radon though, as it would create an ambiguity between radon and radium).
Where a compound has a WHO international nonproprietary name (INN), this should be used as the article title (see WP:NCMED). Exceptions would be where the pharmaceutical use of a certain compound is secondary to other applications (commodity chemical, synthetic intermediate, etc., agriculture or industry).
- From
Wikipedia:Deletion policy: "Don't worry, redirects are cheap."
Redirects should be created for:
- Alternative names for the compound, including acronyms where appropriate;
- Alternative capitalizations, where there is a numerical prefix in the article title;
- Formulas of simple compounds; for example H2O → water.
Even with the best will in the world, no set of guidelines can cover every case. Some articles on Wikipedia have non-standard titles through consensus that this is the most commonly used name (in scientific circumstances) for the compound concerned, whatever IUPAC or the other rules suggest. For example:
- ethylene oxide not oxirane
- phosphine not phosphane (and for substituted phosphines, arsine, stibine, and bismuthine)
- Wilkinson's catalyst not chlorotris(triphenylphosphane)rhodium
- Vaska's complex not carbonylchlorobis(triphenylphosphane)iridium
- titin not methionylthreonylthreonylglutaminylalanyl...isoleucine [14]
Please do not get into revert wars over the naming of an article: the best place for discussion is on the article's talk page or (failing that) at Wikipedia talk:WikiProject Chemicals.
Organic compound classes and functional groups
For articles about classes of organic compounds, the singular class name from the IUPAC "Glossary of Class Names" [15] is usually used, e.g. alkane, carboxylic acid, ether, ketone, acyl chloride. Articles about functional groups should use the name most commonly used to refer to the group in reliable sources (generally, the name of the corresponding radical) followed by the word "group", e.g. alkyl group, carboxyl group, phenyl group, carbonyl group, trimethylsilyl group, triflyl group. In many cases, the compound or compound class and corresponding functional group are discussed in the same article, generally using the compound class as the article title with the name(s) of the functional group redirecting there (although the opposite is possible).
For groups of compounds named after a simple parent compound, articles about the group should be located at the plural of the parent compound name, e.g. hydrazines, silanes, boranes, diphosphenes. Similarly, salts and esters of carboxylic acids should collectively be referred to by the plural of the carboxylate. In some cases, the group of compounds (e.g. triflates), the functional group (e.g. triflate group), and the carboxylate (e.g. triflate) will warrant a separate article; whether the article should be titled by the compound group, functional group, or carboxylate should be decided on a case by case basis depending on the group and how the article is constructed. Related terms not chosen as the title should be redirects and mentioned in the lede in bold font. In cases where the carboxylate is not generally considered as a functional group, the plural may either redirect to the article about the parent acid (or the carboxylate if there is a separate article about the carboxylate), or be a separate article or chemistry index that lists salts and esters of the parent acid (e.g. benzoates). Rare cases may have separate articles for the carboxylate, functional group, and group of compounds. Hatnotes should be used to link between these similarly named pages to facilitate navigation between these closely related topics.
Organometallic compounds
The general article about the organometallic chemistry of an element should be entitled "Organo[element] chemistry", e.g., organomercury chemistry, organopalladium chemistry, organozinc chemistry. The IUPAC definition of an "organometallic compound" includes boron, silicon, arsenic, and selenium, so this title guideline also applies to organoboron chemistry, organosilicon chemistry, etc. [16] For consistency this is extended to even obvious nonmetals, e.g. organoxenon chemistry. The related pages "Organo[element] compound" and its plural should generally be redirects. Some flexibility can be used if the article is heavily weighted toward a specific topic, such as the case for organolithium reagent, so long as the other forms redirect there. In some cases, a broad-concept article regarding the organometallic chemistry of an element may not yet exist; in these cases it is adequate to redirect these terms to an appropriate section of another article. On the other hand, depending on the metallic element, it may well be appropriate to have additional articles covering specific compounds or groups of compounds, or specific reactions.
- List of organic compounds
- List of inorganic compounds
- Wikipedia:WikiProject Chemicals
- Wikipedia:WikiProject Elements
- Wikipedia:Manual of Style/Chemistry
- ^ a b c d Fluck, E. (1988). "New Notations in the Periodic Table" (PDF). Pure Appl. Chem. 60 (3): 431–436. doi: 10.1351/pac198860030431. S2CID 96704008. Archived (PDF) from the original on 25 March 2012. Retrieved 24 March 2012.
- ^ a b c d International Union of Pure and Applied Chemistry (2005). Nomenclature of Inorganic Chemistry (IUPAC Recommendations 2005). Cambridge (UK): RSC– IUPAC. ISBN 0-85404-438-8. pp. 47, 51, 248. Electronic version.
- ^ a b Scerri, Eric (18 January 2021). "Provisional Report on Discussions on Group 3 of the Periodic Table" (PDF). Chemistry International. 43 (1): 31–34. doi: 10.1515/ci-2021-0115. S2CID 231694898. Archived (PDF) from the original on 13 April 2021. Retrieved 9 April 2021.
- ^ Koppenol, W. (2016). "How to name new chemical elements" (PDF). Pure and Applied Chemistry. DeGruyter. doi: 10.1515/pac-2015-0802. hdl: 10045/55935. S2CID 102245448. Archived (PDF) from the original on 11 May 2020. Retrieved 15 August 2021.
- ^ a b Leigh, G. J., ed. (2011). Principles of Chemical Nomenclature (PDF). The Royal Society of Chemistry. p. 9. ISBN 978-1-84973-007-5.
- ^ William B. Jensen (1982). "The Positions of Lanthanum (Actinium) and Lutetium (Lawrencium) in the Periodic Table". J. Chem. Educ. 59 (8): 634–636. Bibcode: 1982JChEd..59..634J. doi: 10.1021/ed059p634.
- ^ L. D. Landau, E. M. Lifshitz (1958). Quantum Mechanics: Non-Relativistic Theory. Vol. 3 (1st ed.). Pergamon Press. pp. 256–7.
- ^ Jensen, William B. (2015). "The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table: an update". Foundations of Chemistry. 17: 23–31. doi: 10.1007/s10698-015-9216-1. S2CID 98624395. Archived from the original on 30 January 2021. Retrieved 28 January 2021.
- ^ Scerri, Eric (2019). "Five ideas in chemical education that must die". Foundations of Chemistry. 21: 61–69. doi: 10.1007/s10698-018-09327-y. S2CID 104311030.
- ^ Leigh, G. Jeffery (2009). "Periodic Tables and IUPAC". Chemistry International. 31 (1): 4–6. doi: 10.1515/ci.2009.31.1.4. Retrieved 17 November 2022.
- ^ It is not very clear how the 15 element cells for La–Lu are meant to fit below the one cell for Y. See for example Pyykkö, Pekka (2019). "An essay on periodic tables" (PDF). Pure and Applied Chemistry. 91 (12): 1959–1967. doi: 10.1515/pac-2019-0801. Retrieved 27 November 2022., which both suggests that there is a hole in period 6 and group 3 in this form, and that all the trivalent rare earths fit there. These cannot simultaneously be true. In the 1990 IUPAC Red Book and a 2009 Chemistry International article by G. J. Leigh, this "blank below Y" table in 18-column is expanded to a 32-column table with Lu below Y. If this is the intention, it seems better to make it clear by putting Lu there in the 18-column form as well, as drawn in the 2021 report and a 2019 Chemistry International article by E. R. Scerri, and as described in the 1988 report.
-
^ Panico, R.; Powell, W. H. (Eds.) (1994). A Guide to IUPAC Nomenclature of Organic Compounds 1993. Oxford: Blackwell Science.
ISBN
0-6320-3488-2.
{{ cite book}}: CS1 maint: multiple names: authors list ( link) - ^ Preferred IUPAC Names Provisional Recommendation, September 2004; Chapter 1, par. 16 Name writing, p.80-90
- ^ See Longest word in English for more information.
- ^ Moss, G.P.; Smith, P.A.S.; Tavernier, D. (1995). "Glossary of Class Names for Organic Compounds and Reactive Intermediates Based on Structure (IUPAC Recommendations 1995)" (PDF). Pure and Applied Chemistry. 67 (8–9): 1307–1375. doi: 10.1351/pac199567081307.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) " organometallic compounds". doi: 10.1351/goldbook.O04328
- General references
- Bünzli–Trepp, Ursula (2007). Systematic Nomenclature of Organic, Organometallic and Coordination Chemistry: Chemical-Abstracts Guidelines with IUPAC Recommendations and Many Trivial Names. EPFL Press. ISBN 9782940222131.
- "Abbreviations and Symbols". Eur. J. Biochem. 74 (1): 1–6. 1977. doi: 10.1111/j.1432-1033.1977.tb11359.x.
| This
guideline documents an English Wikipedia
naming convention. It is a generally accepted standard that editors should attempt to follow, though
occasional exceptions may apply. Any substantive edit to this page should reflect
consensus. When in doubt, discuss first on the
talk page. |
| Manual of Style |
| Chemistry |
|---|
| Categories |
From Wikipedia:Naming conventions:
Generally, article naming should give priority to what the majority of English speakers would most easily recognize, with a reasonable minimum of ambiguity, while at the same time making linking to those articles easy and second nature.
Trivial names (non-systematic, or "common" names) are favored for use in titles of articles for organic compounds instead of systematic names. Trivial names are usually different from the preferred name following IUPAC nomenclature. For compounds lacking trivial names, as is often the case for complex structures, substitutive nomenclature or other systematic names may be used. The general rule is to use the name most commonly used to refer to the compound, as evidenced by use in reliable sources (in line with WP:COMMONNAME).
Classes of compounds may have more specific guidance on naming, such as the use of international nonproprietary names for pharmaceutical compounds (see WP:NCMED and below).
Examples of use of trivial names and not systematic names include:
| acetic acid | not | ethanoic acid |
| toluene | not | methylbenzene |
| lysine | not | 2,6-diaminohexanoic acid |
| 1-pentanol | not | pentan-1-ol |
| benzaldehyde | not | benzenecarbaldehyde |
| diethyl ether | not | ethoxyethane |
| benzophenone | not | diphenylmethanone |
Systematic and other accepted names for the compound not used as the article title should instead be redirects tagged with the {{ R from alternative name}} template and listed in the chembox as other names, with especially significant alternative names mentioned in the lede in bold font.
Groups
- The groups of the periodic table are numbered left to right as 1–18, with the f-block groups (columns) unnumbered. As recommended by IUPAC (1988). [1]
- Note that "group" has a chemical meaning and "column" has the tabular meaning.
- Alternatively, they may be named by their first element, e.g. "boron group" for group 13. [2] (But per below, avoid iron group for group 8 as ambiguous.)
- In the 1–18 scheme, f-block groups are unnumbered. There is no widely-agreed system of numbering these groups in the literature, so one should not be used.
- Avoid labeling these groups as n/a (for not applicable?, or not available i.e. "not found yet"?). Best is to leave this group number blank. Provide textual description when needed. This also applies to any (theoretical) g-block groups.
- To refer to such a group (column), list the elements explicitly, e.g. ytterbium and nobelium. Do not name by first element (ytterbium group). The reasons for this are that many of the expected first-element names have other meanings as rare earth separation groups (see "Non-column groups" below), and that referring to individual f-block groups in the literature is rare (so it's better to be explicit).
- Hydrogen belongs to group 1 (not group 17 or floating outside all groups), and helium belongs to group 18 (not group 2). [1] [3]
- "halogen" and "noble gas" are used to refer to the entirety of groups 17 and 18, per a 2016 IUPAC report about naming new elements [4] Some sources focusing on superheavy elements may not consider tennessine (Ts) to be a halogen, or oganesson (Og) to be a noble gas, as their predicted chemistry is generally supposed to be quite different from those of their lighter congeners. Such more limited usage should be explained before its first occurrence.
- A few groups have IUPAC-approved names, called trivial name, that may also be used:
- alkali metal = group 1 except hydrogen
- alkaline earth metal = group 2
- triels = group 13
- tetrels = group 14
- pnictogen = group 15 (earlier pentels)
- chalcogen = group 16
- halogen = group 17
- noble gas = group 18
- Triels (group 13), tetrels (group 14) and pentels (group 15) have been previously recommended by IUPAC (in 1970), and the former two are still used by them, though they are not in the most recent Red Book. Avoid rarer synonyms like icosagen (group 13) and crystallogen (group 14).
- Avoid coinage metal for group 11, since "metal used for making coins" (see coinage metal) contains also metals outside of group 11, and roentgenium in group 11 is (for obvious reasons) not used for coins.
- The group numberings with Roman numbers and "A" and "B" suffixes (like VIIA, VIII) should not be used, as they are outdated and ambiguous. For example, in the CAS scheme "group VIIB" denotes manganese group (group 7), while in the old IUPAC scheme it denotes halogen group (group 17). If they are used at all, context should clarify (disambiguate) into the 1–18 numbering. Care should be taken when translating old sources that use this nomenclature to modern terminology.
- Some sets of groups have names:
- The main group elements are the elements of groups 1, 2, and 13–18 (i.e. the s-block and p-block). [5]
- The transition elements are the elements of groups 3–12 (i.e. the d-block) – this in particular includes group 12 (although some sources do not include it). [5]
- The inner transition elements are f-block elements. [2]
The scandium group (group 3)
- The scandium group should by default be treated as containing the elements scandium (Sc), yttrium (Y), lutetium (Lu), and lawrencium (Lr). Lanthanum (La) and actinium (Ac), which in some sources take the positions of lutetium and lawrencium (especially in older sources), should not be in group 3. Care should be taken when employing sources that mention "group 3" or "group IIIB" or "group IIIA", since they may not always use this arrangement.
- This group 3 set is established per the 1988 IUPAC report [1] and per a subsequent 2021 provisional report by a IUPAC project dedicated to this constitution question [3]
- In fact, the old classification was based on erroneous early measurements of electron configurations, [6] and was already criticised as "incorrect" by Lev Landau and Evgeny Lifshitz in 1948. [7] The IUPAC report convention is accepted by most chemists and physicists considering the subject. [8] [9]
- Note that the so-called "IUPAC(-published) periodic table" is not actually approved by IUPAC. [10] It leaves the spaces below yttrium blank, and there is significant confusion among (and within) sources on whether that includes the lanthanides and actinides in group 3 or not; [11] hence this form is also best avoided.
- Alternative compositions of group 3 may be illustrated when describing the dispute, e.g. in Group 3 element#Composition.
Non-column groups
- In case of commonly named "groups" that are not a column, such as Platinum group or Iron group, care should be taken that no confusion remains with column-wise periodic table group.
18 vs 32 columns

- The periodic table can be displayed in either 18- or 32-column format, with the 32-column format as in Fig. 4 of the aforementioned 2021 IUPAC report. The forms present the same scientific statement and should be consistent with each other. The 18-column format cuts out the f-block rows, containing the elements lanthanum (La) through ytterbium (Yb) and actinium (Ac) through nobelium (No), and places them at the bottom of the table. [1] Asterisks should be placed between groups 2 and 3 to indicate the theoretical position of these cut-out rows.
- Note that these f-block rows do not equal the lanthanides/lanthanoids (La–Lu) and actinides/actinoids (Ac–Lr) which commonly extend to include lutetium and lawrencium.
- Other structures, e.g. the Janet left-step or the 8-group form, should only be used when specifically illustrating such alternative periodic tables.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | H | He | ||||||||||||||||||||||||||||||
| 2 | Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||
| 3 | Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||
| 4 | K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||
| 5 | Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||
| 6 | Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn |
| 7 | Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og |
|
| ||||||||||||||||||||||||||||||||
Blocks
At this English wiki, the preferred presentation coloring scheme of the elements is by blocks (s, p, d, or f).
The colours to be used are given in {{
Element color}}:
-
s-block→#ff9999 -
p-block→#fdff8c -
d-block→#99ccff -
f-block→#9bff99
This template also gives colours for the higher orbitals:
-
g-block→#fd99ff -
h-block→#7ed9d9 -
i-block→#e0c880
Categorising elements
Categorisation is an attempt to classify all elements into one overall scheme, with each element in exactly one category (as opposed to listing non-completifying sets like coinage metals). A general classification is not achieved in sources (let alone without WP:NPOV and WP:OR). Such categorisation then is not to be used in general (for example, not as general coloring of the periodic table).
A general colouring by metallicity is discouraged, because there are WP:NPOV and WP:NOR concerns: the borders of some categories can differ drastically between sources (e.g. metalloids), and there is no set of categories that are mutually exclusive and yet cover the entire periodic table (e.g. astatine is quite likely to be both a metal and a halogen). However, such coloring may be used to base and illustrate dedicated topics (such as in sections of metalloids). Individual, well-established "metallicity" categories, e.g. transition metal, post-transition metal, metalloid, rare earth metals etc., can be referred to without "all elements all once" colouring.
When a specific categorisation scheme is relevant (e.g. {{
ChemicalBondsToCarbon}}), a periodic table-filling categorisation can be applied.
Traditionally, the names of three elements have been spelled differently in different parts of the English-speaking world. For articles about chemistry-related topics, Wikipedia follows the spellings recommended by the International Union of Pure and Applied Chemistry (IUPAC): [12] [2]
| aluminium | not | aluminum |
| sulfur | not | sulphur |
| caesium | not | cesium |
These spellings should be used in all chemistry-related articles on English Wikipedia, even if they conflict with the other national spelling varieties used in the article.
This convention should also be applied to all compounds and derivative names of these chemicals: sulfate not sulphate; sulfuric not sulphuric; etc.
The English name of element 74 is tungsten and not wolfram: the latter was once adopted by IUPAC (in 1949), but is no longer recommended by them (even as an alternative) per the 2011 Principles. (Of course, the symbol is still W, and the name "wolfram" may be referred to when explaining why that is the case, or when quoting text in languages that use that name.) Similarly for derivatives (per the 2005 Red Book), use tungstate, tungsty, tungsta and not wolframate, wolframy, wolframa. (And similarly avoid the old names emanation and niton for element 86 outside historical contexts; there is a real confusion even in today's literature between radon meaning the element Rn and radon meaning the isotope 222Rn, but the way to solve that is by explicitly clarifying what is meant, not resurrecting archaic terms that have fallen out of use and may not be generally understood.)
Elements that have not been given trivial names by IUPAC may be referred to either by IUPAC systematic element names and symbols, or by atomic numbers. So ununennium and element 119 are both correct, and symbols Uue, 119, E119 may all be used in text. However, for natural disambiguation, the article titles should be at the systematic names.
When new elements are recognised, do not change the names and symbols to those proposed by the discoverers until the public comment period expires and IUPAC makes the final announcement! (The symbol has changed between the initial proposal and the final recommendation before, in the case of copernicium: originally proposed as Cp, but became Cn.)
Isotope names
- Isotopes are named and identified as [element]-[mass number] with regular lowercase and hyphen (carbon-13), or by equivalent symbol format 13C (in wikicode:
<sup>13</sup>C). Avoid 13C, C-13, C13 (and avoid like single character U+2074 ⁴ SUPERSCRIPT FOUR, as used in Wikidata). - Metastable states are noted by format tantalum-180m1, 180m1Ta. The number ⟨1⟩ can be omitted when there is only one isomeric state, e.g. tantalum-180m, 180mTa. You may use a nuclide template. Corresponding letters also used in literature, these are not used in this wiki: ⟨m n p q⟩.
- The hydrogen isotopes are exceptions where the common special names may be used:
- Protium (= hydrogen-1, 1H).
- Deuterium, D, 2D (= hydrogen-2, 2H).
- Tritium, T, 3T (= hydrogen-3, 3H).
- Chemical formulae with deuterated solvents (for example, in NMR use) are customarily described with "D" as chemical symbol: CD3OD, methanol-d4; CD3SOCD3, DMSO-d6 (regular notation would be C2H3O2H). These established systems are acceptable, provided they are consistent within an article.
- Some isotopes have had historical or confusing names and symbols, these are to be avoided (except when quoting historical usage): e.g. thoron (Tn) (220Rn), actinon (An) (219Rn), ionium (Io) (230Th), radiocarbon (14C).
- It is permissible to use the atomic number rather than the symbol in contexts about history when anachronism is to be avoided, e.g. "In 2003, the collaboration at the JINR discovered 288115 and 284113" (because at that point those elements had not yet been named). But this should be avoided outside talking about history. Avoid using the systematic element names in such contexts even when talking about history, because they are not supposed to be used once the element actually has a trivial name. For isotopes of undiscovered elements, either systematic names (e.g. 296Uue) or atomic numbers (e.g. 296119) are acceptable for the symbols, but only the systematic names can be used for the full names (e.g. ununennium-296, not element 119-296 which nobody uses).
Main isotopes
- Currently under discussion for addition
- See WT:ELEMENTS:What is a "Main_isotope"?
Elements with atomic number Z ≥ 119 are theoretical elements, they have not been discovered (synthesized). Regarding public-page creation and content (reader-visible pages, such as artricle, category, template):
- Element page: Articles pages, by systematic name, exist for E119 up to E184: Ununennium (Element 119).
- These are either full articles (like Ununennium) or redirects (like Unbitrium, 58 redirects). This serves readers searching for these element names.
- Redirect element articles like Unbitrium (Element 123) R:
- Per WP:notability and WP:Reliable sources, these articles can and may only be created with content when substantial information is available.
- Secondary public pages for element articles, like Isotopes of unbioctium, {{ Infobox unbioctium}}, :Category:unbioctium:
- These can and may only be created (1) when the base element article exists by content, and (2) when substantial new RL information like papers is available. These secondary pages may not be created as redirect.
To compare: Disovered element Oganesson (E118) has its article, plus all five secondary articles; none is a redirect.
As of 4 Jan 2023 [update]: Effectively this means that currently public-page creation, by content or redirect, is not permitted nor needed. See also documentation at Template:Infobox element § Theoretical elements handling.
The names of elements and other chemicals should be written without abbreviations and treated as common nouns. They do not have special capitalization or italic formatting (see MOS:CAPS and MOS:ITALIC) regardless of the origin of the name unless appropriate for the start of a sentence or part of a title. The symbols for elements should always be capitalized.
- Use: Iron and californium (Fe and Cf) are metals. After I iron my clothes, I will go to California.
- Avoid: Iron and californium (fe and cf) are metals.
- Avoid: Iron and Californium (Fe and Cf) are metals.
- Avoid: Iron and californium (Fe and Cf) are metals.
Prefixes that are such as sec-, tert-, ortho-, meta-, para-, α-, β-, D-, L-, (+)-, (−)-, (R)-, (S)- and the numerical prefixes are not considered part of the name: they each follow their own capitalization and italics rules, and it is instead the first letter of the main part of the name that is treated as the "first letter" of the name to be capitalized where appropriate. Exceptions are cyclo, iso, neo, and spiro which are considered part of the name and therefore not italicized or hyphenated. Substituent groups are also considered part of the name.
In former versions of the IUPAC recommendations, names were written with a capital initial letter. This practice has been abandoned in later publications. [13] IUPAC policy differentiate proper names from things named after proper names. This rule (full name uncapitalized but symbol capitalized) applies also to isotopes and nuclides, when completely written out: thus 14C but carbon-14.
When the chosen article title starts with a prefix including positional identifiers (ortho-, meta-, para-, N-, O-, α-, β-, γ-, etc.), isomeric identifiers (sec-, tert-, etc.), stereochemical identifiers (cis-, trans-, (E)-, (Z)-, etc.), chiral identifiers ((R)-, (S)-, D-, L-, (+)-, (−)-, etc.), or numbers, the first letter after the prefix in the name should be capitalized: hence titlecase 1,1,1-Trichloroethane not 1,1,1-trichloroethane. A redirect from the uncapitalized version should be created to simplify linking from other articles.
Non-numerical prefixes are italicized and uncapitalized in titles (tert-Butyl alcohol, for example). Both numerical and non-numerical prefixes are followed by a hyphen. The template {{ DISPLAYTITLE}} is used to display titles which start with lowercase letters or include italics.
Note that cyclo, iso, neo, and spiro are considered part of a chemical name (such as isopropanol) and not considered prefixes. No hyphens or italics are used in these cases.
Article titles for compounds and related topics should reflect how the compound name is commonly written and use Greek letter prefixes if appropriate, e.g. Α-Ketoglutaric acid, using {{ DISPLAYTITLE}} to display as α-Ketoglutaric acid, not Alpha-Ketoglutaric acid and displaying as alpha-Ketoglutaric acid. In the article page name (article location) it is important to use the uppercase Greek letter, even though it can be easily confused with uppercase Latin letters in many cases, so that DISPLAYTITLE (or alternatively, {{ lowercase title}}) will work properly. It is also essential to ensure redirects exist for all possible typed variations of the compound name so searchers can easily reach the appropriate article.
Stock nomenclature for inorganic compounds is based on the indication of the oxidation number (as a roman numeral, in parentheses) of each of the major elements in the compound, e.g. iron(III) chloride. It is widely, if sometimes incorrectly, used on Wikipedia for the titles of articles about inorganic compounds. It is not obligatory, as there are other acceptable methods for naming these compounds, but it is often preferred as the most common non-ambiguous name for a substance. The following guidelines are based on current WikiBestPractice:
- Only the cationic element (i.e. the element whose name appears unchanged in the compound name) is assigned its oxidation number. Except in rare cases (none at present), we do not assign the oxidation number in the anion: hence potassium permanganate not potassium manganate(VII), sodium hypochlorite not sodium chlorate(I).
- There is no space between the end of the element name and the opening parenthesis: hence silver(I) fluoride not silver (I) fluoride. Note that this is an exception to the usual English style for parentheses.
- It is not necessary to specify the oxidation number when there is no possibility of ambiguity in the compound title: hence sodium chloride not sodium(I) chloride.
- Stock nomenclature should be used for predominantly ionic compounds. Compounds with a substantial degree of covalency should be named by stoichiometric nomenclature: hence titanium tetrachloride not titanium(IV) chloride. However, it should be noted that ionic vs. covalent (not to mention metallic and van der Waals bonding) is a continuum and that many such situations will require significant editorial judgement (e.g. mercury(II) chloride, which is in fact a molecular compound, or ruthenium(IV) oxide which exhibits metallic conductivity).
Names of oxyanions should in general follow the names in Table X of the IUPAC 2005 Red Book. [2] Exceptions can be made if an alternate name is much more common in the literature, e.g. xenate rather than xenonate (don't generalise this to radon though, as it would create an ambiguity between radon and radium).
Where a compound has a WHO international nonproprietary name (INN), this should be used as the article title (see WP:NCMED). Exceptions would be where the pharmaceutical use of a certain compound is secondary to other applications (commodity chemical, synthetic intermediate, etc., agriculture or industry).
- From
Wikipedia:Deletion policy: "Don't worry, redirects are cheap."
Redirects should be created for:
- Alternative names for the compound, including acronyms where appropriate;
- Alternative capitalizations, where there is a numerical prefix in the article title;
- Formulas of simple compounds; for example H2O → water.
Even with the best will in the world, no set of guidelines can cover every case. Some articles on Wikipedia have non-standard titles through consensus that this is the most commonly used name (in scientific circumstances) for the compound concerned, whatever IUPAC or the other rules suggest. For example:
- ethylene oxide not oxirane
- phosphine not phosphane (and for substituted phosphines, arsine, stibine, and bismuthine)
- Wilkinson's catalyst not chlorotris(triphenylphosphane)rhodium
- Vaska's complex not carbonylchlorobis(triphenylphosphane)iridium
- titin not methionylthreonylthreonylglutaminylalanyl...isoleucine [14]
Please do not get into revert wars over the naming of an article: the best place for discussion is on the article's talk page or (failing that) at Wikipedia talk:WikiProject Chemicals.
Organic compound classes and functional groups
For articles about classes of organic compounds, the singular class name from the IUPAC "Glossary of Class Names" [15] is usually used, e.g. alkane, carboxylic acid, ether, ketone, acyl chloride. Articles about functional groups should use the name most commonly used to refer to the group in reliable sources (generally, the name of the corresponding radical) followed by the word "group", e.g. alkyl group, carboxyl group, phenyl group, carbonyl group, trimethylsilyl group, triflyl group. In many cases, the compound or compound class and corresponding functional group are discussed in the same article, generally using the compound class as the article title with the name(s) of the functional group redirecting there (although the opposite is possible).
For groups of compounds named after a simple parent compound, articles about the group should be located at the plural of the parent compound name, e.g. hydrazines, silanes, boranes, diphosphenes. Similarly, salts and esters of carboxylic acids should collectively be referred to by the plural of the carboxylate. In some cases, the group of compounds (e.g. triflates), the functional group (e.g. triflate group), and the carboxylate (e.g. triflate) will warrant a separate article; whether the article should be titled by the compound group, functional group, or carboxylate should be decided on a case by case basis depending on the group and how the article is constructed. Related terms not chosen as the title should be redirects and mentioned in the lede in bold font. In cases where the carboxylate is not generally considered as a functional group, the plural may either redirect to the article about the parent acid (or the carboxylate if there is a separate article about the carboxylate), or be a separate article or chemistry index that lists salts and esters of the parent acid (e.g. benzoates). Rare cases may have separate articles for the carboxylate, functional group, and group of compounds. Hatnotes should be used to link between these similarly named pages to facilitate navigation between these closely related topics.
Organometallic compounds
The general article about the organometallic chemistry of an element should be entitled "Organo[element] chemistry", e.g., organomercury chemistry, organopalladium chemistry, organozinc chemistry. The IUPAC definition of an "organometallic compound" includes boron, silicon, arsenic, and selenium, so this title guideline also applies to organoboron chemistry, organosilicon chemistry, etc. [16] For consistency this is extended to even obvious nonmetals, e.g. organoxenon chemistry. The related pages "Organo[element] compound" and its plural should generally be redirects. Some flexibility can be used if the article is heavily weighted toward a specific topic, such as the case for organolithium reagent, so long as the other forms redirect there. In some cases, a broad-concept article regarding the organometallic chemistry of an element may not yet exist; in these cases it is adequate to redirect these terms to an appropriate section of another article. On the other hand, depending on the metallic element, it may well be appropriate to have additional articles covering specific compounds or groups of compounds, or specific reactions.
- List of organic compounds
- List of inorganic compounds
- Wikipedia:WikiProject Chemicals
- Wikipedia:WikiProject Elements
- Wikipedia:Manual of Style/Chemistry
- ^ a b c d Fluck, E. (1988). "New Notations in the Periodic Table" (PDF). Pure Appl. Chem. 60 (3): 431–436. doi: 10.1351/pac198860030431. S2CID 96704008. Archived (PDF) from the original on 25 March 2012. Retrieved 24 March 2012.
- ^ a b c d International Union of Pure and Applied Chemistry (2005). Nomenclature of Inorganic Chemistry (IUPAC Recommendations 2005). Cambridge (UK): RSC– IUPAC. ISBN 0-85404-438-8. pp. 47, 51, 248. Electronic version.
- ^ a b Scerri, Eric (18 January 2021). "Provisional Report on Discussions on Group 3 of the Periodic Table" (PDF). Chemistry International. 43 (1): 31–34. doi: 10.1515/ci-2021-0115. S2CID 231694898. Archived (PDF) from the original on 13 April 2021. Retrieved 9 April 2021.
- ^ Koppenol, W. (2016). "How to name new chemical elements" (PDF). Pure and Applied Chemistry. DeGruyter. doi: 10.1515/pac-2015-0802. hdl: 10045/55935. S2CID 102245448. Archived (PDF) from the original on 11 May 2020. Retrieved 15 August 2021.
- ^ a b Leigh, G. J., ed. (2011). Principles of Chemical Nomenclature (PDF). The Royal Society of Chemistry. p. 9. ISBN 978-1-84973-007-5.
- ^ William B. Jensen (1982). "The Positions of Lanthanum (Actinium) and Lutetium (Lawrencium) in the Periodic Table". J. Chem. Educ. 59 (8): 634–636. Bibcode: 1982JChEd..59..634J. doi: 10.1021/ed059p634.
- ^ L. D. Landau, E. M. Lifshitz (1958). Quantum Mechanics: Non-Relativistic Theory. Vol. 3 (1st ed.). Pergamon Press. pp. 256–7.
- ^ Jensen, William B. (2015). "The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table: an update". Foundations of Chemistry. 17: 23–31. doi: 10.1007/s10698-015-9216-1. S2CID 98624395. Archived from the original on 30 January 2021. Retrieved 28 January 2021.
- ^ Scerri, Eric (2019). "Five ideas in chemical education that must die". Foundations of Chemistry. 21: 61–69. doi: 10.1007/s10698-018-09327-y. S2CID 104311030.
- ^ Leigh, G. Jeffery (2009). "Periodic Tables and IUPAC". Chemistry International. 31 (1): 4–6. doi: 10.1515/ci.2009.31.1.4. Retrieved 17 November 2022.
- ^ It is not very clear how the 15 element cells for La–Lu are meant to fit below the one cell for Y. See for example Pyykkö, Pekka (2019). "An essay on periodic tables" (PDF). Pure and Applied Chemistry. 91 (12): 1959–1967. doi: 10.1515/pac-2019-0801. Retrieved 27 November 2022., which both suggests that there is a hole in period 6 and group 3 in this form, and that all the trivalent rare earths fit there. These cannot simultaneously be true. In the 1990 IUPAC Red Book and a 2009 Chemistry International article by G. J. Leigh, this "blank below Y" table in 18-column is expanded to a 32-column table with Lu below Y. If this is the intention, it seems better to make it clear by putting Lu there in the 18-column form as well, as drawn in the 2021 report and a 2019 Chemistry International article by E. R. Scerri, and as described in the 1988 report.
-
^ Panico, R.; Powell, W. H. (Eds.) (1994). A Guide to IUPAC Nomenclature of Organic Compounds 1993. Oxford: Blackwell Science.
ISBN
0-6320-3488-2.
{{ cite book}}: CS1 maint: multiple names: authors list ( link) - ^ Preferred IUPAC Names Provisional Recommendation, September 2004; Chapter 1, par. 16 Name writing, p.80-90
- ^ See Longest word in English for more information.
- ^ Moss, G.P.; Smith, P.A.S.; Tavernier, D. (1995). "Glossary of Class Names for Organic Compounds and Reactive Intermediates Based on Structure (IUPAC Recommendations 1995)" (PDF). Pure and Applied Chemistry. 67 (8–9): 1307–1375. doi: 10.1351/pac199567081307.
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) " organometallic compounds". doi: 10.1351/goldbook.O04328
- General references
- Bünzli–Trepp, Ursula (2007). Systematic Nomenclature of Organic, Organometallic and Coordination Chemistry: Chemical-Abstracts Guidelines with IUPAC Recommendations and Many Trivial Names. EPFL Press. ISBN 9782940222131.
- "Abbreviations and Symbols". Eur. J. Biochem. 74 (1): 1–6. 1977. doi: 10.1111/j.1432-1033.1977.tb11359.x.