This article needs to be updated. (January 2021) |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌtəˈnoʊfəvɪər ˌæləˈfɛnəmaɪd/ |
| Trade names | Vemlidy |
| Other names | GS-7340 |
| AHFS/ Drugs.com | Monograph |
| License data | |
|
Pregnancy category |
|
|
Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | ~80% [6] |
| Elimination half-life | 0.51 hour |
| Excretion | Feces (31.7%), urine (<1%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
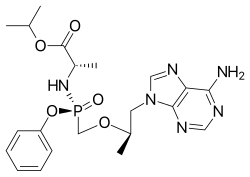
| Formula | C21H29N6O5P |
| Molar mass | 476.474 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |

Tenofovir alafenamide, sold under the brand name Vemlidy, is an antiviral medication used against hepatitis B and HIV. It is used for the treatment of chronic hepatitis B virus (HBV) infection in adults with compensated liver disease [8] and is given in combination with other medications for the prevention and treatment of HIV. It is taken by mouth. [6]
Tenofovir alafenamide is a nucleotide reverse transcriptase inhibitor and is a prodrug of tenofovir. It was developed by Gilead Sciences based on the protide technology of Chris McGuigan and is applied in the form of tenofovir alafenamide fumarate (TAF). Closely related to the commonly used reverse-transcriptase inhibitor tenofovir disoproxil fumarate (TDF), TAF has greater antiviral activity and better distribution into lymphoid tissues than that agent. [9] [10] It was approved for use in the US for HIV in 2015, [11] and for hepatitis B in 2016. [12] Although the US Food and Drug Administration (FDA) has approved tenofovir alafenamide for manufacture as a generic medication, [13] it is not available. [14]
- Elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide (Genvoya) [15] — approved both in the United States and in the European Union in November 2015, [16] [17] [18] [19] (compare elvitegravir/cobicistat/emtricitabine/tenofovir; (Stribild) [20] [21] [22] [23])
- Emtricitabine/rilpivirine/tenofovir alafenamide (Odefsey) [24] — approved in the United States in March 2016, and in the European Union in June 2016, [25] [26] [27] (compare Emtricitabine/rilpivirine/tenofovir; (Complera) [28] [29] [30])
- Emtricitabine/tenofovir alafenamide (Descovy) [31] — approved in the United States in April 2016 (compare emtricitabine/tenofovir; (Truvada)). In October 2019, Descovy was approved in the United States for HIV-1 pre-exposure prophylaxis (PrEP). [32] [33]
- Bictegravir/emtricitabine/tenofovir alafenamide (Biktarvy) [34] — approved in the United States in February 2018.
- Darunavir/cobicistat/emtricitabine/tenofovir alafenamide (Symtuza) [35] — approved in the European Union in September 2017, in the United States in July 2018, and in Australia in November 2019. [36] [37] [38] [39] [40]
- Dolutegravir/emtricitabine/tenofovir alafenamide. [41]
- Dolutegravir/lamivudine/tenofovir alafenamide. [42] [43]
Gilead announced a Phase III clinical trial evaluating a single-tablet regimen combining tenofovir alafenamide with cobicistat, emtricitabine and elvitegravir [44] and developed a coformulation of the drug with cobicistat, emtricitabine and the protease inhibitor darunavir. [45] [46] [47] In a 48-week study comparing elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil (Stribild) to elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide (Genvoya), the results showed the newer drug's effects to be non-inferior to the established agent, but at much lower dosages and with lower incidence of adverse side effects such as impaired kidney function. [48] [49] [50] The FDA approved the TAF-based treatment regimen for treatment of HIV-1 in November 2015. [11] Genvoya is the first TAF-based regimen to receive approval. [11]
- ^ "Tenofovir alafenamide (Vemlidy) Use During Pregnancy". Drugs.com. 26 December 2018. Archived from the original on 9 July 2021. Retrieved 18 April 2020.
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2016". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ^ "Product Monograph: Vemlidy (tenofovir alafenamide) tablets" (PDF). Government of Canada: The Drug and Health Product Register. 20 August 2020. Archived (PDF) from the original on 10 June 2022. Retrieved 7 June 2022.
- ^ "Liver disease". Health Canada. 9 May 2018. Retrieved 13 April 2024.
- ^ "Vemlidy 25 mg film coated tablets - Summary of Product Characteristics (SmPC)". (emc). 8 September 2020. Archived from the original on 11 July 2021. Retrieved 12 November 2020.
- ^ a b c "Vemlidy- tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 11 February 2020. Archived from the original on 9 July 2021. Retrieved 18 April 2020.
- ^ "Vemlidy EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 13 July 2021. Retrieved 12 November 2020.
- ^ Childs-Kean LM, Egelund EF, Jourjy J (October 2018). "Tenofovir Alafenamide for the Treatment of Chronic Hepatitis B Monoinfection". Pharmacotherapy. 38 (10): 1051–1057. doi: 10.1002/phar.2174. PMID 30120841. S2CID 52033420.
- ^ Eisenberg EJ, He GX, Lee WA (2001). "Metabolism of GS-7340, a novel phenyl monophosphoramidate intracellular prodrug of PMPA, in blood". Nucleosides Nucleotides Nucleic Acids. 20 (4–7): 1091–8. doi: 10.1081/NCN-100002496. PMID 11562963. S2CID 24652157.
- ^ Markowitz M, Zolopa A, Ruane P, Squires K, Zhong L, Kearney BP, et al. (March 2011). GS-7340 demonstrates greater declines in HIV-1 RNA than tenofovir disoproxil fumarate during 14 days of monotherapy in HIV-1 infected subjects (PDF). 18th Conference on Retroviruses and Opportunistic Infections. Archived from the original (PDF) on 25 April 2012.
- ^ a b c "U.S. Food and Drug Administration Approves Gilead's Single Tablet Regimen Genvoya (Elvitegravir, Cobicistat, Emtricitabine and Tenofovir Alafenamide) for Treatment of HIV-1 Infection" (Press release). Gilead. 5 November 2015. Archived from the original on 8 November 2015.
- ^ "FDA Approves Vemlidy (tenofovir alafenamide) for Chronic Hepatitis B in Adults". United States Department of Health and Human Services. 21 November 2016. Archived from the original on 11 October 2019. Retrieved 11 October 2019.
- ^ "First Generic Drug Approvals 2023". U.S. Food and Drug Administration (FDA). 30 May 2023. Archived from the original on 30 June 2023. Retrieved 30 June 2023.
- ^ "Gilead settles 5 more Descovy patent feuds, pushing copycats to its PrEP successor out to 2031". Fierce Pharma. 12 September 2022. Archived from the original on 30 June 2023. Retrieved 27 March 2024.
- ^ "Genvoya- elvitegravir, cobicistat, emtricitabine, and tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 11 February 2019. Archived from the original on 1 August 2020. Retrieved 18 April 2020.
- ^ "Genvoya (elvitegravir, cobicistat, emtricitabine, and tenofovir alafenamide) fixed-dose combination tablet". U.S. Food and Drug Administration (FDA). 8 December 2015. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Summary Review: Genvoya" (PDF). US Food and Drug Administration. 6 August 2012. Archived (PDF) from the original on 29 July 2020. Retrieved 29 July 2020.
- ^ "Genvoya EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 8 August 2020. Retrieved 28 July 2020.
- ^ "Health Canada New Drug Authorizations: 2015 Highlights". Health Canada. 4 May 2016. Retrieved 7 April 2024.
- ^ "Drug Approval Package: Stribild (elvitegravir, cobicistat, emtricitabine, tenofovir disoproxil fumarate) Fixed Dose". U.S. Food and Drug Administration (FDA). 10 October 2012. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Summary Review: Stribild" (PDF). US Food and Drug Administration. 19 October 2015. Archived (PDF) from the original on 29 July 2020. Retrieved 29 July 2020.
- ^ "Stribild- elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate tablet, film coated". DailyMed. U.S. National Library of Medicine. 28 January 2019. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Stribild EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Odefsey- emtricitabine, rilpivirine hydrochloride, and tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 6 December 2019. Archived from the original on 1 August 2020. Retrieved 18 April 2020.
- ^ "Odefsey (emtricitabine, rilpivirine, and tenofovir alafenamide) Tablets". U.S. Food and Drug Administration (FDA). 29 November 2016. Archived from the original on 13 April 2021. Retrieved 28 July 2020.
- ^ "Odefsey EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Immune system health". Health Canada. 9 May 2018. Retrieved 13 April 2024.
- ^ "Drug Approval Package: (emtricitabine/rilpivirine/tenofovir disoproxil fumarate) NDA #202123". U.S. Food and Drug Administration (FDA). 6 September 2012. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Summary Review: Complera" (PDF). US Food and Drug Administration. 19 July 2011. Archived (PDF) from the original on 1 April 2021. Retrieved 29 July 2020.
- ^ "Complera- emtricitabine, rilpivirine hydrochloride, and tenofovir disoproxil fumarate tablet, film coated". DailyMed. U.S. National Library of Medicine. 9 December 2019. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Descovy- emtricitabine and tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 13 January 2020. Archived from the original on 24 June 2021. Retrieved 18 April 2020.
- ^ "FDA approves second drug to prevent HIV infection as part of ongoing efforts to end the HIV epidemic". U.S. Food and Drug Administration (FDA). 3 October 2019. Archived from the original on 11 October 2019. Retrieved 11 October 2019.
- ^ Mandavilli A (4 October 2019). "F.D.A. Approves New H.I.V.-Prevention Drug, but Not for Everyone". The New York Times. Archived from the original on 9 July 2021. Retrieved 11 October 2019.
- ^ "Biktarvy- bictegravir sodium, emtricitabine, and tenofovir alafenamide fumarate tablet". DailyMed. U.S. National Library of Medicine. 8 August 2019. Archived from the original on 1 August 2020. Retrieved 18 April 2020.
- ^ "Symtuza- darunavir, cobicistat, emtricitabine, and tenofovir alafenamide tablet, film coated". DailyMed. U.S. National Library of Medicine. 6 March 2020. Archived from the original on 9 July 2021. Retrieved 18 April 2020.
- ^ "Drug Approval Package: Symtuza (darunavir, cobicistat, emtricitabine, and tenofovir alafenamide)". U.S. Food and Drug Administration (FDA). 11 December 2018. Archived from the original on 9 July 2021. Retrieved 19 August 2020.
- ^ "Symtuza EPAR". European Medicines Agency. 17 September 2018. Archived from the original on 10 July 2021. Retrieved 19 August 2020.
- ^ "TGA eBS - Product and Consumer Medicine Information Licence". Archived from the original on 20 February 2022. Retrieved 20 February 2022.
- ^ "Symtuza 800/150/200/10 Tablets". NPS MedicineWise. 15 July 2021. Archived from the original on 20 February 2022. Retrieved 19 February 2022.
- ^ "Drug and medical device highlights 2018: Helping you maintain and improve your health". Health Canada. 14 October 2020. Retrieved 17 April 2024.
- ^ "Drugs@FDA: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 9 July 2021. Retrieved 5 December 2020.
- ^ "Drugs@FDA: FDA-Approved Drugs". accessdata.fda.gov. Archived from the original on 2 April 2022. Retrieved 2 April 2022.
- ^ "Tentative Approval: Dolutegravir, Lamivudine, and Tenofovir Alafenamide Tablets" (PDF). US Food and Drug Administration. 30 March 2022. Archived (PDF) from the original on 7 May 2022. Retrieved 2 April 2022.
- ^ "Gilead Initiates Phase 3 Clinical Program for Tenofovir Alafenamide, a Novel Low-Dose Prodrug for the Treatment of HIV" (Press release). Gilead. 24 January 2013. Archived from the original on 11 October 2019.
- ^ "Gilead Sciences Finalizes Agreement with Tibotec Pharmaceuticals to Develop and Commercialize a Single-Tablet Regimen of Prezista with Emtriva, GS 7340 and Cobicistat". Gilead Sciences (Press release). 15 November 2011. Archived from the original on 11 October 2019. Retrieved 10 October 2019.
- ^ Horn T (15 March 2012). "GS-7340 Packs Greater HIV Punch, Potentially Better Safety, Versus Viread". AIDSmeds.com. Archived from the original on 8 September 2015.
- ^ "Pharmacokinetics of a Novel EVG/COBI/FTC/GS-7340 Single Tablet Regimen". 13th International Workshop on Clinical Pharmacology of HIV Therapy. Barcelona, Spain. April 2012. Archived from the original on 28 November 2020.
- ^ "Once-Daily Tenofovir Prodrug Combo Pill as Effective as Stribild". AIDSmeds. 1 November 2012. Archived from the original on 20 September 2015.
- ^ Highleyman L (6 March 2013). "CROI 2013: New Pro-drug Tenofovir Alafenamide Appears Equally Effective but Better Tolerated". hivandhepatitis.com. Archived from the original on 25 October 2020. Retrieved 25 July 2024.
- ^ Horn T (13 June 2013). "Tenefovir Alafenamide Fumarate (TAF) Sign-On Letter to Gilead". Treatment Action Group. Archived from the original on 19 November 2019.
This article needs to be updated. (January 2021) |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌtəˈnoʊfəvɪər ˌæləˈfɛnəmaɪd/ |
| Trade names | Vemlidy |
| Other names | GS-7340 |
| AHFS/ Drugs.com | Monograph |
| License data | |
|
Pregnancy category |
|
|
Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | ~80% [6] |
| Elimination half-life | 0.51 hour |
| Excretion | Feces (31.7%), urine (<1%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C21H29N6O5P |
| Molar mass | 476.474 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |

Tenofovir alafenamide, sold under the brand name Vemlidy, is an antiviral medication used against hepatitis B and HIV. It is used for the treatment of chronic hepatitis B virus (HBV) infection in adults with compensated liver disease [8] and is given in combination with other medications for the prevention and treatment of HIV. It is taken by mouth. [6]
Tenofovir alafenamide is a nucleotide reverse transcriptase inhibitor and is a prodrug of tenofovir. It was developed by Gilead Sciences based on the protide technology of Chris McGuigan and is applied in the form of tenofovir alafenamide fumarate (TAF). Closely related to the commonly used reverse-transcriptase inhibitor tenofovir disoproxil fumarate (TDF), TAF has greater antiviral activity and better distribution into lymphoid tissues than that agent. [9] [10] It was approved for use in the US for HIV in 2015, [11] and for hepatitis B in 2016. [12] Although the US Food and Drug Administration (FDA) has approved tenofovir alafenamide for manufacture as a generic medication, [13] it is not available. [14]
- Elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide (Genvoya) [15] — approved both in the United States and in the European Union in November 2015, [16] [17] [18] [19] (compare elvitegravir/cobicistat/emtricitabine/tenofovir; (Stribild) [20] [21] [22] [23])
- Emtricitabine/rilpivirine/tenofovir alafenamide (Odefsey) [24] — approved in the United States in March 2016, and in the European Union in June 2016, [25] [26] [27] (compare Emtricitabine/rilpivirine/tenofovir; (Complera) [28] [29] [30])
- Emtricitabine/tenofovir alafenamide (Descovy) [31] — approved in the United States in April 2016 (compare emtricitabine/tenofovir; (Truvada)). In October 2019, Descovy was approved in the United States for HIV-1 pre-exposure prophylaxis (PrEP). [32] [33]
- Bictegravir/emtricitabine/tenofovir alafenamide (Biktarvy) [34] — approved in the United States in February 2018.
- Darunavir/cobicistat/emtricitabine/tenofovir alafenamide (Symtuza) [35] — approved in the European Union in September 2017, in the United States in July 2018, and in Australia in November 2019. [36] [37] [38] [39] [40]
- Dolutegravir/emtricitabine/tenofovir alafenamide. [41]
- Dolutegravir/lamivudine/tenofovir alafenamide. [42] [43]
Gilead announced a Phase III clinical trial evaluating a single-tablet regimen combining tenofovir alafenamide with cobicistat, emtricitabine and elvitegravir [44] and developed a coformulation of the drug with cobicistat, emtricitabine and the protease inhibitor darunavir. [45] [46] [47] In a 48-week study comparing elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil (Stribild) to elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide (Genvoya), the results showed the newer drug's effects to be non-inferior to the established agent, but at much lower dosages and with lower incidence of adverse side effects such as impaired kidney function. [48] [49] [50] The FDA approved the TAF-based treatment regimen for treatment of HIV-1 in November 2015. [11] Genvoya is the first TAF-based regimen to receive approval. [11]
- ^ "Tenofovir alafenamide (Vemlidy) Use During Pregnancy". Drugs.com. 26 December 2018. Archived from the original on 9 July 2021. Retrieved 18 April 2020.
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2016". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ^ "Product Monograph: Vemlidy (tenofovir alafenamide) tablets" (PDF). Government of Canada: The Drug and Health Product Register. 20 August 2020. Archived (PDF) from the original on 10 June 2022. Retrieved 7 June 2022.
- ^ "Liver disease". Health Canada. 9 May 2018. Retrieved 13 April 2024.
- ^ "Vemlidy 25 mg film coated tablets - Summary of Product Characteristics (SmPC)". (emc). 8 September 2020. Archived from the original on 11 July 2021. Retrieved 12 November 2020.
- ^ a b c "Vemlidy- tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 11 February 2020. Archived from the original on 9 July 2021. Retrieved 18 April 2020.
- ^ "Vemlidy EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 13 July 2021. Retrieved 12 November 2020.
- ^ Childs-Kean LM, Egelund EF, Jourjy J (October 2018). "Tenofovir Alafenamide for the Treatment of Chronic Hepatitis B Monoinfection". Pharmacotherapy. 38 (10): 1051–1057. doi: 10.1002/phar.2174. PMID 30120841. S2CID 52033420.
- ^ Eisenberg EJ, He GX, Lee WA (2001). "Metabolism of GS-7340, a novel phenyl monophosphoramidate intracellular prodrug of PMPA, in blood". Nucleosides Nucleotides Nucleic Acids. 20 (4–7): 1091–8. doi: 10.1081/NCN-100002496. PMID 11562963. S2CID 24652157.
- ^ Markowitz M, Zolopa A, Ruane P, Squires K, Zhong L, Kearney BP, et al. (March 2011). GS-7340 demonstrates greater declines in HIV-1 RNA than tenofovir disoproxil fumarate during 14 days of monotherapy in HIV-1 infected subjects (PDF). 18th Conference on Retroviruses and Opportunistic Infections. Archived from the original (PDF) on 25 April 2012.
- ^ a b c "U.S. Food and Drug Administration Approves Gilead's Single Tablet Regimen Genvoya (Elvitegravir, Cobicistat, Emtricitabine and Tenofovir Alafenamide) for Treatment of HIV-1 Infection" (Press release). Gilead. 5 November 2015. Archived from the original on 8 November 2015.
- ^ "FDA Approves Vemlidy (tenofovir alafenamide) for Chronic Hepatitis B in Adults". United States Department of Health and Human Services. 21 November 2016. Archived from the original on 11 October 2019. Retrieved 11 October 2019.
- ^ "First Generic Drug Approvals 2023". U.S. Food and Drug Administration (FDA). 30 May 2023. Archived from the original on 30 June 2023. Retrieved 30 June 2023.
- ^ "Gilead settles 5 more Descovy patent feuds, pushing copycats to its PrEP successor out to 2031". Fierce Pharma. 12 September 2022. Archived from the original on 30 June 2023. Retrieved 27 March 2024.
- ^ "Genvoya- elvitegravir, cobicistat, emtricitabine, and tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 11 February 2019. Archived from the original on 1 August 2020. Retrieved 18 April 2020.
- ^ "Genvoya (elvitegravir, cobicistat, emtricitabine, and tenofovir alafenamide) fixed-dose combination tablet". U.S. Food and Drug Administration (FDA). 8 December 2015. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Summary Review: Genvoya" (PDF). US Food and Drug Administration. 6 August 2012. Archived (PDF) from the original on 29 July 2020. Retrieved 29 July 2020.
- ^ "Genvoya EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 8 August 2020. Retrieved 28 July 2020.
- ^ "Health Canada New Drug Authorizations: 2015 Highlights". Health Canada. 4 May 2016. Retrieved 7 April 2024.
- ^ "Drug Approval Package: Stribild (elvitegravir, cobicistat, emtricitabine, tenofovir disoproxil fumarate) Fixed Dose". U.S. Food and Drug Administration (FDA). 10 October 2012. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Summary Review: Stribild" (PDF). US Food and Drug Administration. 19 October 2015. Archived (PDF) from the original on 29 July 2020. Retrieved 29 July 2020.
- ^ "Stribild- elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate tablet, film coated". DailyMed. U.S. National Library of Medicine. 28 January 2019. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Stribild EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Odefsey- emtricitabine, rilpivirine hydrochloride, and tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 6 December 2019. Archived from the original on 1 August 2020. Retrieved 18 April 2020.
- ^ "Odefsey (emtricitabine, rilpivirine, and tenofovir alafenamide) Tablets". U.S. Food and Drug Administration (FDA). 29 November 2016. Archived from the original on 13 April 2021. Retrieved 28 July 2020.
- ^ "Odefsey EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Immune system health". Health Canada. 9 May 2018. Retrieved 13 April 2024.
- ^ "Drug Approval Package: (emtricitabine/rilpivirine/tenofovir disoproxil fumarate) NDA #202123". U.S. Food and Drug Administration (FDA). 6 September 2012. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Summary Review: Complera" (PDF). US Food and Drug Administration. 19 July 2011. Archived (PDF) from the original on 1 April 2021. Retrieved 29 July 2020.
- ^ "Complera- emtricitabine, rilpivirine hydrochloride, and tenofovir disoproxil fumarate tablet, film coated". DailyMed. U.S. National Library of Medicine. 9 December 2019. Archived from the original on 29 July 2020. Retrieved 28 July 2020.
- ^ "Descovy- emtricitabine and tenofovir alafenamide tablet". DailyMed. U.S. National Library of Medicine. 13 January 2020. Archived from the original on 24 June 2021. Retrieved 18 April 2020.
- ^ "FDA approves second drug to prevent HIV infection as part of ongoing efforts to end the HIV epidemic". U.S. Food and Drug Administration (FDA). 3 October 2019. Archived from the original on 11 October 2019. Retrieved 11 October 2019.
- ^ Mandavilli A (4 October 2019). "F.D.A. Approves New H.I.V.-Prevention Drug, but Not for Everyone". The New York Times. Archived from the original on 9 July 2021. Retrieved 11 October 2019.
- ^ "Biktarvy- bictegravir sodium, emtricitabine, and tenofovir alafenamide fumarate tablet". DailyMed. U.S. National Library of Medicine. 8 August 2019. Archived from the original on 1 August 2020. Retrieved 18 April 2020.
- ^ "Symtuza- darunavir, cobicistat, emtricitabine, and tenofovir alafenamide tablet, film coated". DailyMed. U.S. National Library of Medicine. 6 March 2020. Archived from the original on 9 July 2021. Retrieved 18 April 2020.
- ^ "Drug Approval Package: Symtuza (darunavir, cobicistat, emtricitabine, and tenofovir alafenamide)". U.S. Food and Drug Administration (FDA). 11 December 2018. Archived from the original on 9 July 2021. Retrieved 19 August 2020.
- ^ "Symtuza EPAR". European Medicines Agency. 17 September 2018. Archived from the original on 10 July 2021. Retrieved 19 August 2020.
- ^ "TGA eBS - Product and Consumer Medicine Information Licence". Archived from the original on 20 February 2022. Retrieved 20 February 2022.
- ^ "Symtuza 800/150/200/10 Tablets". NPS MedicineWise. 15 July 2021. Archived from the original on 20 February 2022. Retrieved 19 February 2022.
- ^ "Drug and medical device highlights 2018: Helping you maintain and improve your health". Health Canada. 14 October 2020. Retrieved 17 April 2024.
- ^ "Drugs@FDA: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 9 July 2021. Retrieved 5 December 2020.
- ^ "Drugs@FDA: FDA-Approved Drugs". accessdata.fda.gov. Archived from the original on 2 April 2022. Retrieved 2 April 2022.
- ^ "Tentative Approval: Dolutegravir, Lamivudine, and Tenofovir Alafenamide Tablets" (PDF). US Food and Drug Administration. 30 March 2022. Archived (PDF) from the original on 7 May 2022. Retrieved 2 April 2022.
- ^ "Gilead Initiates Phase 3 Clinical Program for Tenofovir Alafenamide, a Novel Low-Dose Prodrug for the Treatment of HIV" (Press release). Gilead. 24 January 2013. Archived from the original on 11 October 2019.
- ^ "Gilead Sciences Finalizes Agreement with Tibotec Pharmaceuticals to Develop and Commercialize a Single-Tablet Regimen of Prezista with Emtriva, GS 7340 and Cobicistat". Gilead Sciences (Press release). 15 November 2011. Archived from the original on 11 October 2019. Retrieved 10 October 2019.
- ^ Horn T (15 March 2012). "GS-7340 Packs Greater HIV Punch, Potentially Better Safety, Versus Viread". AIDSmeds.com. Archived from the original on 8 September 2015.
- ^ "Pharmacokinetics of a Novel EVG/COBI/FTC/GS-7340 Single Tablet Regimen". 13th International Workshop on Clinical Pharmacology of HIV Therapy. Barcelona, Spain. April 2012. Archived from the original on 28 November 2020.
- ^ "Once-Daily Tenofovir Prodrug Combo Pill as Effective as Stribild". AIDSmeds. 1 November 2012. Archived from the original on 20 September 2015.
- ^ Highleyman L (6 March 2013). "CROI 2013: New Pro-drug Tenofovir Alafenamide Appears Equally Effective but Better Tolerated". hivandhepatitis.com. Archived from the original on 25 October 2020. Retrieved 25 July 2024.
- ^ Horn T (13 June 2013). "Tenefovir Alafenamide Fumarate (TAF) Sign-On Letter to Gilead". Treatment Action Group. Archived from the original on 19 November 2019.