| This is the sandbox page where you will draft your initial Wikipedia contribution.
If you're starting a new article, you can develop it here until it's ready to go live. If you're working on improvements to an existing article, copy only one section at a time of the article to this sandbox to work on, and be sure to use an edit summary linking to the article you copied from. Do not copy over the entire article. You can find additional instructions here. Remember to save your work regularly using the "Publish page" button. (It just means 'save'; it will still be in the sandbox.) You can add bold formatting to your additions to differentiate them from existing content. |
Outline of Article
-Lead Section
-The Origin: Symbiogenesis and Symbiont transmission
-Endosymbiotic Theory: Symbiogenesis and organelles
-Symbiont transmission
-Endosymbionts of invertebrates
-Endosymbionts of terrestrial invertebrates
-Endosymbionts of insects
(Endosymbionts of ants would be moved here--Bacteriocyte-associated symbionts)
-Primary endosymbionts
-Secondary endosymbionts
-Endosymbionts of marine invertebrates
-Dinoflagellate endosymbionts
-Endosymbionts of phytoplankton
-Endosymbionts of protists
-Endosymbionts of vertebrates- This one is listed as a subheading under endosymbionts of invertebrates, but needs to be its own heading because its talking about vertebrates
- Endosymbionts of Humans
-Endosymbionts of plants (Endophytes) - this section redefines endosymbiosis as it does in the beginning of article, it should be cut down to remove repeated information
-Fungi as plant endosymbionts
(Endophytic Fungi would be moved here)
-Arbuscular Mycorrhizal Fungi (AMF)
- Bacteria as plant endosymbionts - needs some more information.
- Archaea as plant endosymbionts
Endosymbionts of bacteria
Endosymbionts of fungi
Virus-host associations. Debatable section to have, thoughts on motion to remove?
See also
References
Lead Section
An endosymbiont or endobiont [1] is any organism that lives within the body or cells of another organism most often, though not always, in a mutualistic relationship. (The term endosymbiosis is from the Greek: ἔνδον endon "within", σύν syn "together" and βίωσις biosis "living".) Examples are nitrogen-fixing bacteria (called rhizobia), which live in the root nodules of legumes, single-cell algae inside reef-building corals and bacterial endosymbionts that provide essential nutrients to insects. [2] [3]
The history behind the concept of endosymbiosis stems from the postulates of the endosymbiotic theory. The endosymbiotic theory ( symbiogenesis) pushes the notion of bacteria exclusively living in eukaryotic organisms after being engulfed by them. This is popular with the concept of organelle development observed with eukaryotes. Two major types of organelle in eukaryotic cells, mitochondria and plastids such as chloroplasts, are considered to be obtained from bacterial endosymbionts. [4] There are two main types of symbiont transmissions. In horizontal transmission, each new generation acquires free living symbionts from the environment. An example is the nitrogen-fixing bacteria in certain plant roots. Vertical transmission takes place when the symbiont is transferred directly from parent to offspring. [5] [6] An example is pea aphid symbionts. Also, it is possible for both to be involved in a mixed-mode transmission, where symbionts are transferred vertically for some generation before a switch of host occurs and new symbionts are horizontally acquired from the environment. [7] [8] [9]
The Origin: Symbiogenesis and Symbiont transmission
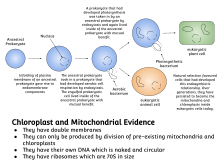
Symbiogenesis explains the origins of eukaryotes, whose cells contain two major kinds of organelle: mitochondria and chloroplasts. The theory proposes that these organelles evolved from certain types of bacteria that eukaryotic cells engulfed through phagocytosis. These cells and the bacteria trapped inside them entered an endosymbiotic relationship, meaning that the bacteria took up residence and began living exclusively within the eukaryotic cells. [10] [11] [12] [13]
Proposed Change- Endosymbiotic Theory: Symbiogenesis
Symbiogenesis explains the origins of eukaryotes, whose cells contain two major kinds of organelle: mitochondria and chloroplasts. The theory proposes the process through which these organelles evolved within the ancestors of eukaryotic organisms through the formation of an endosymbiotic relationship between a bacteria and an archaea This idea is that the ancient Archaean organism engulfed a bacteria that possessed aerobic capabilities. This initial endosymbiotic relationship is believed to have arisen between an Asgard superphylum archaea and an ancient bacteria related to the Rickettsiales lineage. The bacterial endosymbiont would have been used for its metabolic capabilities, and is believed to have eventually developed into the mitochondria that is found in eukaryotic cells. Evidence pointing to this theory involves the evolutionary history of the two groups and their DNA structure. Eukaryotes are believed to have arisen from a common ancestor they possess with archaeans, which also points to archaeans being more closely related to eukaryotes than bacteria. When looking at the DNA found in the nucleus of a cells and the mitochondria. the nuclear DNA of a eukaryote more closely resembles the DNA of an organism belonging to the domain Archaea, while the mitochondrial DNA is more closely related in structure to the DNA of a bacteria organism. The chloroplast is theorized to have arisen from a eukaryotic cell already possessing a mitochondria enveloping and taking in a cyanobacteria that possessed photosynthetic capabilities. [14] [15] [16]
Symbiont transmission
Symbiont transmission is the process where the host in a symbiotic relationship between two organisms acquires an organism (internally or externally) that serves as its symbiont. Most symbionts are either obligatory (require their host to survive) or facultative (do not necessarily need their host to survive). [17] Many instances of endosymbiosis are obligate; that is, either the endosymbiont or the host cannot survive without the other, such as the gutless marine worms of the genus Riftia, which get nutrition from their endosymbiotic bacteria. The most common examples of obligate endosymbiosis are mitochondria and chloroplasts. Some human parasites, e.g. Wuchereria bancrofti and Mansonella perstans, thrive in their intermediate insect hosts because of an obligate endosymbiosis with Wolbachia spp. [18] They can both be eliminated from hosts by treatments that target this bacterium. [19]
Horizontal (lateral), vertical, and mix-mode (hybrid of horizonal and vertical) transmission are the three paths for symbiont transfer. Horizontal symbiont transfer ( horizontal transmission) is a process where a host acquires a facultative symbiont from the environment or from another host. [17] The Rhizobia-Legume symbiosis (bacteria-plant endosymbiosis) is a prime example of horizontal symbiont transmission. [20] The Rhizobia-legume symbiotic relationship is important for processes like the formation of root nodules. It starts with flavonoids released by the plant host (Legume), which causes the rhizobia species (endosymbiont) to activate its nod genes. [21] These Nod genes generate lipooligosaccharide signals which the legume(host) detects, thus leading to root nodule formation. [22] This process bleeds on to other unique processes like nitrogen fixation in plants. [21] The evolutionary advantage of such an interaction allows genetic exchange between both organisms involved increases the propensity for novel functions as seen in the plant-bacterium interaction ( holobiont formation). [23]
In vertical transmission, the symbionts often have a reduced genome and are no longer able to survive on their own. As a result, the symbiont depends on the host, resulting in a highly intimate co-dependent relationship. For instance, pea aphid symbionts have lost genes for essential molecules, now relying on the host to supply them with nutrients. In return, the symbionts synthesize essential amino acids for the aphid host. [22] Other examples include Wigglesworthia nutritional symbionts of tsetse flies, or in sponges. [9] When a symbiont reaches this stage, it begins to resemble a cellular organelle, similar to mitochondria or chloroplasts. The evolutionary consequences causes the host and the symbiont to be dependent and form a holobiont, and in the event of a bottleneck a decrease in symbiont diversity could affect the host-symbiont interactions adversely, when deleterious mutations build up over time. [24]
Endosymbionts of invertebrates
The best-studied examples of endosymbiosis are known from invertebrates. These symbioses affect organisms with global impact, including Symbiodinium of corals, or Wolbachia of insects. Many insect agricultural pests and human disease vectors have intimate relationships with primary endosymbionts. [25]
Endosymbionts of terrestrial invertebrates
Endosymbionts of insects
Scientists classify insect endosymbionts in two broad categories, 'Primary' and 'Secondary'. Primary endosymbionts (sometimes referred to as P-endosymbionts) have been associated with their insect hosts for many millions of years (from 10 to several hundred million years in some cases). They form obligate associations (see below), and display cospeciation with their insect hosts. Secondary endosymbionts exhibit a more recently developed association, are sometimes horizontally transferred between hosts, live in the hemolymph of the insects (not specialized bacteriocytes, see below), and are not obligate. [26] In insects such as ants, the most well studied endosymbionts are bacteria of the genus Blochmannia, which are the primary endosymbiont of Camponotus ants. In 2018 a new ant-associated symbiont was discovered in Cardiocondyla ants. This symbiont was named Candidatus Westeberhardia Cardiocondylae and it is also believed to be a primary symbiont. [27]
Primary endosymbionts
Among primary endosymbionts of insects, the best-studied are the pea aphid ( Acyrthosiphon pisum) and its endosymbiont Buchnera sp. APS, [28] [29] the tsetse fly Glossina morsitans morsitans and its endosymbiont Wigglesworthia glossinidia brevipalpis, and the endosymbiotic protists in lower termites. As with endosymbiosis in other insects, the symbiosis is obligate in that neither the bacteria nor the insect is viable without the other. Scientists have been unable to cultivate the bacteria in lab conditions outside of the insect. With special nutritionally-enhanced diets, the insects can survive, but are unhealthy, and at best survive only a few generations. [26]
In some insect groups, these endosymbionts live in specialized insect cells called bacteriocytes (also called mycetocytes), and are maternally-transmitted, i.e. the mother transmits her endosymbionts to her offspring. In some cases, the bacteria are transmitted in the egg, as in Buchnera; in others like Wigglesworthia, they are transmitted via milk to the developing insect embryo. In termites, the endosymbionts reside within the hindguts and are transmitted through trophallaxis among colony members. [30]
The primary endosymbionts are thought to help the host either by providing nutrients that the host cannot obtain itself or by metabolizing insect waste products into safer forms. For example, the putative primary role of Buchnera is to synthesize essential amino acids that the aphid cannot acquire from its natural diet of plant sap. Likewise, the primary role of Wigglesworthia, it is presumed, is to synthesize vitamins that the tsetse fly does not get from the blood that it eats. In lower termites, the endosymbiotic protists play a major role in the digestion of lignocellulosic materials that constitute a bulk of the termites' diet.
Bacteria benefit from the reduced exposure to predators and competition from other bacterial species, the ample supply of nutrients and relative environmental stability inside the host.
Genome sequencing reveals that obligate bacterial endosymbionts of insects have among the smallest of known bacterial genomes and have lost many genes that are commonly found in closely related bacteria. Several theories have been put forth to explain the loss of genes. It is presumed that some of these genes are not needed in the environment of the host insect cell. A complementary theory suggests that the relatively small numbers of bacteria inside each insect decrease the efficiency of natural selection in 'purging' deleterious mutations and small mutations from the population, resulting in a loss of genes over many millions of years. Research in which a parallel phylogeny of bacteria and insects was inferred supports the belief that the primary endosymbionts are transferred only vertically (i.e., from the mother), and not horizontally (i.e., by escaping the host and entering a new host). [31] [32]
Attacking obligate bacterial endosymbionts may present a way to control their insect hosts, many of which are pests or carriers of human disease. For example, aphids are crop pests and the tsetse fly carries the organism Trypanosoma brucei that causes African sleeping sickness. [33] Other motivations for their study involve understanding the origins of symbioses in general, as a proxy for understanding e.g. how chloroplasts or mitochondria came to be obligate symbionts of eukaryotes or plants.
Secondary endosymbionts

The pea aphid ( Acyrthosiphon pisum) is known to contain at least three secondary endosymbionts, Hamiltonella defensa, Regiella insecticola, and Serratia symbiotica. Hamiltonella defensa defends its aphid host from parasitoid wasps. [34] This defensive symbiosis improves the survival of aphids, which have lost some elements of the insect immune response. [35]
One of the best-understood defensive symbionts is the spiral bacteria Spiroplasma poulsonii. Spiroplasma sp. can be reproductive manipulators, but also defensive symbionts of Drosophila flies. In Drosophila neotestacea, S. poulsonii has spread across North America owing to its ability to defend its fly host against nematode parasites. [36] This defence is mediated by toxins called "ribosome-inactivating proteins" that attack the molecular machinery of invading parasites. [37] [38] These Spiroplasma toxins represent one of the first examples of a defensive symbiosis with a mechanistic understanding for defensive symbiosis between an insect endosymbiont and its host. [38]
Sodalis glossinidius is a secondary endosymbiont of tsetse flies that lives inter- and intracellularly in various host tissues, including the midgut and hemolymph. Phylogenetic studies have not indicated a correlation between evolution of Sodalis and tsetse. [39] Unlike tsetse's primary symbiont Wigglesworthia, though, Sodalis has been cultured in vitro. [40]
Many other insects have secondary endosymbionts not reviewed here. [41] [42]
Endosymbionts of marine invertebrates
Extracellular endosymbionts are also represented in all four extant classes of Echinodermata ( Crinoidea, Ophiuroidea, Echinoidea, and Holothuroidea). Little is known of the nature of the association (mode of infection, transmission, metabolic requirements, etc.) but phylogenetic analysis indicates that these symbionts belong to the class Alphaproteobacteria, relating them to Rhizobium and Thiobacillus. Other studies indicate that these subcuticular bacteria may be both abundant within their hosts and widely distributed among the Echinoderms in general. [43]
Some marine oligochaeta (e.g., Olavius algarvensis and Inanidrillus spp.) have obligate extracellular endosymbionts that fill the entire body of their host. These marine worms are nutritionally dependent on their symbiotic chemoautotrophic bacteria lacking any digestive or excretory system (no gut, mouth, or nephridia). [44]
The sea slug Elysia chlorotica lives in endosymbiotic relationship with the algae Vaucheria litorea, and the jellyfish Mastigias have a similar relationship with an algae. Elysia chlorotica forms this relationship intracellularly with the chloroplasts from the algae. These chloroplast retain their photosynthetic capabilities and structures for several months after being taken into the cells of the slug. [45]
Dinoflagellate endosymbionts
Dinoflagellate endosymbionts of the genus Symbiodinium, commonly known as zooxanthellae, are found in corals, mollusks (esp. giant clams, the Tridacna), sponges, and foraminifera. These endosymbionts drive the formation of coral reefs by capturing sunlight and providing their hosts with energy for carbonate deposition. [46]
Previously thought to be a single species, molecular phylogenetic evidence over the past couple decades has shown there to be great diversity in Symbiodinium. In some cases, there is specificity between host and Symbiodinium clade. More often, however, there is an ecological distribution of Symbiodinium, the symbionts switching between hosts with apparent ease. When reefs become environmentally stressed, this distribution of symbionts is related to the observed pattern of coral bleaching and recovery. Thus, the distribution of Symbiodinium on coral reefs and its role in coral bleaching presents one of the most complex and interesting current problems in reef ecology. [46]
Endosymbiosis in Vertebrates- Information updated and changed
Endosymbiotic relationships can be difficult to develop in vertebrates, largely due to the immune capabilities found within the class of organisms.
The bacteria that constitute the microbiome of the human are not considered endosymbiotic but rather an example of
ectosymbiosis. The digestive tract is considered to be an external surface and thus the organisms that inhabit it are not endosymbionts.
[47]
Endosymbionts of marine invertebrates
Elysia chlorotica forms this relationship intracellularly with the chloroplasts from the algae. These chloroplast retain their photosynthetic capabilities and structures for several months after being taken into the cells of the slug. [48]
Endophytic Bacteria
The most common genera of endophytic bacterial community isolated from forest trees include Pseudomonas, Bacillus, Acinetobacter, Actinobacteria, Sphingomonas and some genera belong to the Enterobacteriacae family (Pirttila and Frank, 2011). Endophytic bacteria mostly colonize the leaf tissues from plant roots. Endophytic bacteria can also enter the plant through the leaves from the phyllosphere via leaf stomata (Senthilkumar et al., 2011).
References
- ^ Margulis L, Chapman MJ (2009). Kingdoms & domains an illustrated guide to the phyla of life on Earth (4th ed.). Amsterdam: Academic Press/Elsevier. p. 493. ISBN 978-0-08-092014-6.
- ^ Mergaert P (April 2018). "Role of antimicrobial peptides in controlling symbiotic bacterial populations". Natural Product Reports. 35 (4): 336–356. doi: 10.1039/c7np00056a. PMID 29393944.
- ^ Little AF, van Oppen MJ, Willis BL (June 2004). "Flexibility in algal endosymbioses shapes growth in reef corals". Science. 304 (5676): 1492–1494. Bibcode: 2004Sci...304.1491L. doi: 10.1126/science.1095733. PMID 15178799. S2CID 10050417.
- ^ Moore KR, Magnabosco C, Momper L, Gold DA, Bosak T, Fournier GP (2019). "An Expanded Ribosomal Phylogeny of Cyanobacteria Supports a Deep Placement of Plastids". Frontiers in Microbiology. 10: 1612. doi: 10.3389/fmicb.2019.01612. PMC 6640209. PMID 31354692.
- ^ McCutcheon JP (October 2021). "The Genomics and Cell Biology of Host-Beneficial Intracellular Infections". Annual Review of Cell and Developmental Biology. 37 (1): 115–142. doi: 10.1146/annurev-cellbio-120219-024122. PMID 34242059. S2CID 235786110.
- ^ Callier V (8 June 2022). "Mitochondria and the origin of eukaryotes". Knowable Magazine. doi: 10.1146/knowable-060822-2. Retrieved 18 August 2022.
- ^ Wierz JC, Gaube P, Klebsch D, Kaltenpoth M, Flórez LV (2021). "Transmission of Bacterial Symbionts With and Without Genome Erosion Between a Beetle Host and the Plant Environment". Frontiers in Microbiology. 12: 715601. doi: 10.3389/fmicb.2021.715601. PMC 8493222. PMID 34630349.
- ^ Ebert D (23 November 2013). "The Epidemiology and Evolution of Symbionts with Mixed-Mode Transmission". Annual Review of Ecology, Evolution, and Systematics. 44 (1): 623–643. doi: 10.1146/annurev-ecolsys-032513-100555. ISSN 1543-592X. Retrieved 19 August 2022.
- ^ a b Bright M, Bulgheresi S (March 2010). "A complex journey: transmission of microbial symbionts". Nature Reviews. Microbiology. 8 (3): 218–230. doi: 10.1038/nrmicro2262. PMC 2967712. PMID 20157340.
- ^ McCutcheon, JP (6 October 2021). "The Genomics and Cell Biology of Host-Beneficial Intracellular Infections". Annual Review of Cell and Developmental Biology. 37 (1): 115–142. doi: 10.1146/annurev-cellbio-120219-024122. ISSN 1081-0706. PMID 34242059. S2CID 235786110. Retrieved 19 August 2022.
- ^ Callier, V (8 June 2022). "Mitochondria and the origin of eukaryotes". Knowable Magazine. doi: 10.1146/knowable-060822-2. Retrieved 18 August 2022.
- ^ Sagan L (March 1967). "On the origin of mitosing cells". Journal of Theoretical Biology. 14 (3): 255–74. Bibcode: 1967JThBi..14..225S. doi: 10.1016/0022-5193(67)90079-3. PMID 11541392.
- ^ Gabaldón, T (8 October 2021). "Origin and Early Evolution of the Eukaryotic Cell". Annual Review of Microbiology. 75 (1): 631–647. doi: 10.1146/annurev-micro-090817-062213. ISSN 0066-4227. PMID 34343017. S2CID 236916203. Retrieved 19 August 2022.
- ^ Koonin, Eugene V. (2015-09-26). "Origin of eukaryotes from within archaea, archaeal eukaryome and bursts of gene gain: eukaryogenesis just made easier?". Philosophical Transactions of the Royal Society B: Biological Sciences. 370 (1678): 20140333. doi: 10.1098/rstb.2014.0333. ISSN 0962-8436. PMC 4571572. PMID 26323764.
-
^ Glansdorff, Nicolas; Xu, Ying; Labedan, Bernard (2008-07-09).
"The Last Universal Common Ancestor: emergence, constitution and genetic legacy of an elusive forerunner". Biology Direct. 3: 29.
doi:
10.1186/1745-6150-3-29.
ISSN
1745-6150.
PMC
2478661.
PMID
18613974.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) -
^ Boguszewska, Karolina; Szewczuk, Michał; Kaźmierczak-Barańska, Julia; Karwowski, Bolesław T. (2020-06-21).
"The Similarities between Human Mitochondria and Bacteria in the Context of Structure, Genome, and Base Excision Repair System". Molecules. 25 (12): 2857.
doi:
10.3390/molecules25122857.
ISSN
1420-3049.
PMC
7356350.
PMID
32575813.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^
a
b Bright, Monika; Bulgheresi, Silvia (2010-03).
"A complex journey: transmission of microbial symbionts". Nature Reviews Microbiology. 8 (3): 218–230.
doi:
10.1038/nrmicro2262.
ISSN
1740-1534.
PMC
2967712.
PMID
20157340.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) -
^ Slatko, Barton E.; Taylor, Mark J.; Foster, Jeremy M. (2010-07-01).
"The Wolbachia endosymbiont as an anti-filarial nematode target". Symbiosis. 51 (1): 55–65.
doi:
10.1007/s13199-010-0067-1.
ISSN
1878-7665.
PMC
2918796.
PMID
20730111.
{{ cite journal}}: CS1 maint: PMC format ( link) - ^ Warrell D, Cox TM, Firth J, Török E (2012-10-11). Oxford Textbook of Medicine: Infection. OUP Oxford. ISBN 978-0-19-965213-6.
-
^ Gage, Daniel J. (2004-06).
"Infection and Invasion of Roots by Symbiotic, Nitrogen-Fixing Rhizobia during Nodulation of Temperate Legumes". Microbiology and Molecular Biology Reviews. 68 (2): 280–300.
doi:
10.1128/MMBR.68.2.280-300.2004.
ISSN
1092-2172.
PMC
419923.
PMID
15187185.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) - ^
a
b Gage, Daniel J. (2004-06).
"Infection and Invasion of Roots by Symbiotic, Nitrogen-Fixing Rhizobia during Nodulation of Temperate Legumes". Microbiology and Molecular Biology Reviews. 68 (2): 280–300.
doi:
10.1128/MMBR.68.2.280-300.2004.
ISSN
1092-2172.
PMC
419923.
PMID
15187185.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) - ^ a b Shigenobu S, Watanabe H, Hattori M, Sakaki Y, Ishikawa H (September 2000). "Genome sequence of the endocellular bacterial symbiont of aphids Buchnera sp. APS". Nature. 407 (6800): 81–86. Bibcode: 2000Natur.407...81S. doi: 10.1038/35024074. PMID 10993077.
-
^ Chrostek, Ewa; Pelz-Stelinski, Kirsten; Hurst, Gregory D. D.; Hughes, Grant L. (2017).
"Horizontal Transmission of Intracellular Insect Symbionts via Plants". Frontiers in Microbiology. 8.
doi:
10.3389/fmicb.2017.02237.
ISSN
1664-302X.
PMC
5712413.
PMID
29234308.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) -
^ Smith, Noel H.; Gordon, Stephen V.; de la Rua-Domenech, Ricardo; Clifton-Hadley, Richard S.; Hewinson, R. Glyn (2006-09).
"Bottlenecks and broomsticks: the molecular evolution of Mycobacterium bovis". Nature Reviews Microbiology. 4 (9): 670–681.
doi:
10.1038/nrmicro1472.
ISSN
1740-1534.
{{ cite journal}}: Check date values in:|date=( help) -
^ Eleftherianos, Ioannis; Atri, Jaishri; Accetta, Julia; Castillo, Julio C. (2013).
"Endosymbiotic bacteria in insects: guardians of the immune system?". Frontiers in Physiology. 4.
doi:
10.3389/fphys.2013.00046.
ISSN
1664-042X.
PMC
3597943.
PMID
23508299.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^
a
b Moran, Nancy A.; Tran, Phat; Gerardo, Nicole M. (2005-12).
"Symbiosis and Insect Diversification: an Ancient Symbiont of Sap-Feeding Insects from the Bacterial Phylum Bacteroidetes". Applied and Environmental Microbiology. 71 (12): 8802–8810.
doi:
10.1128/AEM.71.12.8802-8810.2005.
ISSN
0099-2240.
PMC
1317441.
PMID
16332876.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) - ^ Klein A, Schrader L, Gil R, Manzano-Marín A, Flórez L, Wheeler D, et al. (February 2016). "A novel intracellular mutualistic bacterium in the invasive ant Cardiocondyla obscurior". The ISME Journal. 10 (2): 376–388. doi: 10.1038/ismej.2015.119. PMC 4737929. PMID 26172209.
- ^ Douglas AE (January 1998). "Nutritional interactions in insect-microbial symbioses: aphids and their symbiotic bacteria Buchnera". Annual Review of Entomology. 43: 17–37. doi: 10.1146/annurev.ento.43.1.17. PMID 15012383. S2CID 29594533.
- ^ Shigenobu S, Watanabe H, Hattori M, Sakaki Y, Ishikawa H (September 2000). "Genome sequence of the endocellular bacterial symbiont of aphids Buchnera sp. APS". Nature. 407 (6800): 81–86. Bibcode: 2000Natur.407...81S. doi: 10.1038/35024074. PMID 10993077.
-
^ Nalepa, Christine A. (2020).
"Origin of Mutualism Between Termites and Flagellated Gut Protists: Transition From Horizontal to Vertical Transmission". Frontiers in Ecology and Evolution. 8.
doi:
10.3389/fevo.2020.00014.
ISSN
2296-701X.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) -
^ Wernegreen JJ (March 2004).
"Endosymbiosis: lessons in conflict resolution". PLOS Biology. 2 (3): E68.
doi:
10.1371/journal.pbio.0020068.
PMC
368163.
PMID
15024418.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Moran NA (April 1996). "Accelerated evolution and Muller's rachet in endosymbiotic bacteria". Proceedings of the National Academy of Sciences of the United States of America. 93 (7): 2873–2878. Bibcode: 1996PNAS...93.2873M. doi: 10.1073/pnas.93.7.2873. PMC 39726. PMID 8610134.
- ^ Aksoy S, Maudlin I, Dale C, Robinson AS, O'Neill SL (January 2001). "Prospects for control of African trypanosomiasis by tsetse vector manipulation". Trends in Parasitology. 17 (1): 29–35. doi: 10.1016/S1471-4922(00)01850-X. PMID 11137738.
- ^ Oliver KM, Campos J, Moran NA, Hunter MS (February 2008). "Population dynamics of defensive symbionts in aphids". Proceedings. Biological Sciences. 275 (1632): 293–299. doi: 10.1098/rspb.2007.1192. PMC 2593717. PMID 18029301.
-
^
"Genome sequence of the pea aphid Acyrthosiphon pisum". PLOS Biology. 8 (2): e1000313. February 2010.
doi:
10.1371/journal.pbio.1000313.
PMC
2826372.
PMID
20186266.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Jaenike J, Unckless R, Cockburn SN, Boelio LM, Perlman SJ (July 2010). "Adaptation via symbiosis: recent spread of a Drosophila defensive symbiont". Science. 329 (5988): 212–215. Bibcode: 2010Sci...329..212J. doi: 10.1126/science.1188235. PMID 20616278. S2CID 206526012.
- ^ Hamilton PT, Peng F, Boulanger MJ, Perlman SJ (January 2016). "A ribosome-inactivating protein in a Drosophila defensive symbiont". Proceedings of the National Academy of Sciences of the United States of America. 113 (2): 350–355. Bibcode: 2016PNAS..113..350H. doi: 10.1073/pnas.1518648113. PMC 4720295. PMID 26712000.
- ^
a
b Ballinger MJ, Perlman SJ (July 2017).
"Generality of toxins in defensive symbiosis: Ribosome-inactivating proteins and defense against parasitic wasps in Drosophila". PLOS Pathogens. 13 (7): e1006431.
doi:
10.1371/journal.ppat.1006431.
PMC
5500355.
PMID
28683136.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Aksoy, S., Pourhosseini, A. & Chow, A. 1995. Mycetome endosymbionts of tsetse flies constitute a distinct lineage related to Enterobacteriaceae. Insect Mol Biol. 4, 15–22.
- ^ Welburn SC, Maudlin I, Ellis DS (June 1987). "In vitro cultivation of rickettsia-like-organisms from Glossina spp". Annals of Tropical Medicine and Parasitology. 81 (3): 331–335. doi: 10.1080/00034983.1987.11812127. PMID 3662675.
- ^ Zchori-Fein E, Perlman SJ (July 2004). "Distribution of the bacterial symbiont Cardinium in arthropods". Molecular Ecology. 13 (7): 2009–2016. doi: 10.1111/j.1365-294X.2004.02203.x. PMID 15189221. S2CID 24361903.
- ^ Wernegreen JJ (November 2002). "Genome evolution in bacterial endosymbionts of insects". Nature Reviews. Genetics. 3 (11): 850–861. doi: 10.1038/nrg931. PMID 12415315. S2CID 29136336.
- ^ Burnett WJ, McKenzie JD (May 1997). "Subcuticular bacteria from the brittle star Ophiactis balli (Echinodermata: Ophiuroidea) represent a new lineage of extracellular marine symbionts in the alpha subdivision of the class Proteobacteria". Applied and Environmental Microbiology. 63 (5): 1721–1724. Bibcode: 1997ApEnM..63.1721B. doi: 10.1128/AEM.63.5.1721-1724.1997. PMC 168468. PMID 9143108.
- ^ Dubilier N, Mülders C, Ferdelman T, de Beer D, Pernthaler A, Klein M, et al. (May 2001). "Endosymbiotic sulphate-reducing and sulphide-oxidizing bacteria in an oligochaete worm". Nature. 411 (6835): 298–302. Bibcode: 2001Natur.411..298D. doi: 10.1038/35077067. PMID 11357130. S2CID 4420931.
- ^ Mujer CV, Andrews DL, Manhart JR, Pierce SK, Rumpho ME (October 1996). "Chloroplast genes are expressed during intracellular symbiotic association of Vaucheria litorea plastids with the sea slug Elysia chlorotica". Proceedings of the National Academy of Sciences of the United States of America. 93 (22): 12333–12338. doi: 10.1073/pnas.93.22.12333. PMC 37991. PMID 8901581.
- ^ a b Baker AC (November 2003). "Flexibility and Specificity in Coral-Algal Symbiosis: Diversity, Ecology, and Biogeography of Symbiodinium". Annual Review of Ecology, Evolution, and Systematics. 34: 661–89. doi: 10.1146/annurev.ecolsys.34.011802.132417. S2CID 35278104.
-
^ Burns, John A; Zhang, Huanjia; Hill, Elizabeth; Kim, Eunsoo; Kerney, Ryan (2017-05-02). Bhattacharya, Debashish (ed.).
"Transcriptome analysis illuminates the nature of the intracellular interaction in a vertebrate-algal symbiosis". eLife. 6: e22054.
doi:
10.7554/eLife.22054.
ISSN
2050-084X.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Mujer, C V; Andrews, D L; Manhart, J R; Pierce, S K; Rumpho, M E (1996-10-29). "Chloroplast genes are expressed during intracellular symbiotic association of Vaucheria litorea plastids with the sea slug Elysia chlorotica". Proceedings of the National Academy of Sciences of the United States of America. 93 (22): 12333–12338. ISSN 0027-8424. PMID 8901581.
10. Frank, A. C., & Pirttilä, A. M. (Eds.). (2018). Endophytes of forest trees: biology and applications. Springer International Publishing.
| This is the sandbox page where you will draft your initial Wikipedia contribution.
If you're starting a new article, you can develop it here until it's ready to go live. If you're working on improvements to an existing article, copy only one section at a time of the article to this sandbox to work on, and be sure to use an edit summary linking to the article you copied from. Do not copy over the entire article. You can find additional instructions here. Remember to save your work regularly using the "Publish page" button. (It just means 'save'; it will still be in the sandbox.) You can add bold formatting to your additions to differentiate them from existing content. |
Outline of Article
-Lead Section
-The Origin: Symbiogenesis and Symbiont transmission
-Endosymbiotic Theory: Symbiogenesis and organelles
-Symbiont transmission
-Endosymbionts of invertebrates
-Endosymbionts of terrestrial invertebrates
-Endosymbionts of insects
(Endosymbionts of ants would be moved here--Bacteriocyte-associated symbionts)
-Primary endosymbionts
-Secondary endosymbionts
-Endosymbionts of marine invertebrates
-Dinoflagellate endosymbionts
-Endosymbionts of phytoplankton
-Endosymbionts of protists
-Endosymbionts of vertebrates- This one is listed as a subheading under endosymbionts of invertebrates, but needs to be its own heading because its talking about vertebrates
- Endosymbionts of Humans
-Endosymbionts of plants (Endophytes) - this section redefines endosymbiosis as it does in the beginning of article, it should be cut down to remove repeated information
-Fungi as plant endosymbionts
(Endophytic Fungi would be moved here)
-Arbuscular Mycorrhizal Fungi (AMF)
- Bacteria as plant endosymbionts - needs some more information.
- Archaea as plant endosymbionts
Endosymbionts of bacteria
Endosymbionts of fungi
Virus-host associations. Debatable section to have, thoughts on motion to remove?
See also
References
Lead Section
An endosymbiont or endobiont [1] is any organism that lives within the body or cells of another organism most often, though not always, in a mutualistic relationship. (The term endosymbiosis is from the Greek: ἔνδον endon "within", σύν syn "together" and βίωσις biosis "living".) Examples are nitrogen-fixing bacteria (called rhizobia), which live in the root nodules of legumes, single-cell algae inside reef-building corals and bacterial endosymbionts that provide essential nutrients to insects. [2] [3]
The history behind the concept of endosymbiosis stems from the postulates of the endosymbiotic theory. The endosymbiotic theory ( symbiogenesis) pushes the notion of bacteria exclusively living in eukaryotic organisms after being engulfed by them. This is popular with the concept of organelle development observed with eukaryotes. Two major types of organelle in eukaryotic cells, mitochondria and plastids such as chloroplasts, are considered to be obtained from bacterial endosymbionts. [4] There are two main types of symbiont transmissions. In horizontal transmission, each new generation acquires free living symbionts from the environment. An example is the nitrogen-fixing bacteria in certain plant roots. Vertical transmission takes place when the symbiont is transferred directly from parent to offspring. [5] [6] An example is pea aphid symbionts. Also, it is possible for both to be involved in a mixed-mode transmission, where symbionts are transferred vertically for some generation before a switch of host occurs and new symbionts are horizontally acquired from the environment. [7] [8] [9]
The Origin: Symbiogenesis and Symbiont transmission

Symbiogenesis explains the origins of eukaryotes, whose cells contain two major kinds of organelle: mitochondria and chloroplasts. The theory proposes that these organelles evolved from certain types of bacteria that eukaryotic cells engulfed through phagocytosis. These cells and the bacteria trapped inside them entered an endosymbiotic relationship, meaning that the bacteria took up residence and began living exclusively within the eukaryotic cells. [10] [11] [12] [13]
Proposed Change- Endosymbiotic Theory: Symbiogenesis
Symbiogenesis explains the origins of eukaryotes, whose cells contain two major kinds of organelle: mitochondria and chloroplasts. The theory proposes the process through which these organelles evolved within the ancestors of eukaryotic organisms through the formation of an endosymbiotic relationship between a bacteria and an archaea This idea is that the ancient Archaean organism engulfed a bacteria that possessed aerobic capabilities. This initial endosymbiotic relationship is believed to have arisen between an Asgard superphylum archaea and an ancient bacteria related to the Rickettsiales lineage. The bacterial endosymbiont would have been used for its metabolic capabilities, and is believed to have eventually developed into the mitochondria that is found in eukaryotic cells. Evidence pointing to this theory involves the evolutionary history of the two groups and their DNA structure. Eukaryotes are believed to have arisen from a common ancestor they possess with archaeans, which also points to archaeans being more closely related to eukaryotes than bacteria. When looking at the DNA found in the nucleus of a cells and the mitochondria. the nuclear DNA of a eukaryote more closely resembles the DNA of an organism belonging to the domain Archaea, while the mitochondrial DNA is more closely related in structure to the DNA of a bacteria organism. The chloroplast is theorized to have arisen from a eukaryotic cell already possessing a mitochondria enveloping and taking in a cyanobacteria that possessed photosynthetic capabilities. [14] [15] [16]
Symbiont transmission
Symbiont transmission is the process where the host in a symbiotic relationship between two organisms acquires an organism (internally or externally) that serves as its symbiont. Most symbionts are either obligatory (require their host to survive) or facultative (do not necessarily need their host to survive). [17] Many instances of endosymbiosis are obligate; that is, either the endosymbiont or the host cannot survive without the other, such as the gutless marine worms of the genus Riftia, which get nutrition from their endosymbiotic bacteria. The most common examples of obligate endosymbiosis are mitochondria and chloroplasts. Some human parasites, e.g. Wuchereria bancrofti and Mansonella perstans, thrive in their intermediate insect hosts because of an obligate endosymbiosis with Wolbachia spp. [18] They can both be eliminated from hosts by treatments that target this bacterium. [19]
Horizontal (lateral), vertical, and mix-mode (hybrid of horizonal and vertical) transmission are the three paths for symbiont transfer. Horizontal symbiont transfer ( horizontal transmission) is a process where a host acquires a facultative symbiont from the environment or from another host. [17] The Rhizobia-Legume symbiosis (bacteria-plant endosymbiosis) is a prime example of horizontal symbiont transmission. [20] The Rhizobia-legume symbiotic relationship is important for processes like the formation of root nodules. It starts with flavonoids released by the plant host (Legume), which causes the rhizobia species (endosymbiont) to activate its nod genes. [21] These Nod genes generate lipooligosaccharide signals which the legume(host) detects, thus leading to root nodule formation. [22] This process bleeds on to other unique processes like nitrogen fixation in plants. [21] The evolutionary advantage of such an interaction allows genetic exchange between both organisms involved increases the propensity for novel functions as seen in the plant-bacterium interaction ( holobiont formation). [23]
In vertical transmission, the symbionts often have a reduced genome and are no longer able to survive on their own. As a result, the symbiont depends on the host, resulting in a highly intimate co-dependent relationship. For instance, pea aphid symbionts have lost genes for essential molecules, now relying on the host to supply them with nutrients. In return, the symbionts synthesize essential amino acids for the aphid host. [22] Other examples include Wigglesworthia nutritional symbionts of tsetse flies, or in sponges. [9] When a symbiont reaches this stage, it begins to resemble a cellular organelle, similar to mitochondria or chloroplasts. The evolutionary consequences causes the host and the symbiont to be dependent and form a holobiont, and in the event of a bottleneck a decrease in symbiont diversity could affect the host-symbiont interactions adversely, when deleterious mutations build up over time. [24]
Endosymbionts of invertebrates
The best-studied examples of endosymbiosis are known from invertebrates. These symbioses affect organisms with global impact, including Symbiodinium of corals, or Wolbachia of insects. Many insect agricultural pests and human disease vectors have intimate relationships with primary endosymbionts. [25]
Endosymbionts of terrestrial invertebrates
Endosymbionts of insects
Scientists classify insect endosymbionts in two broad categories, 'Primary' and 'Secondary'. Primary endosymbionts (sometimes referred to as P-endosymbionts) have been associated with their insect hosts for many millions of years (from 10 to several hundred million years in some cases). They form obligate associations (see below), and display cospeciation with their insect hosts. Secondary endosymbionts exhibit a more recently developed association, are sometimes horizontally transferred between hosts, live in the hemolymph of the insects (not specialized bacteriocytes, see below), and are not obligate. [26] In insects such as ants, the most well studied endosymbionts are bacteria of the genus Blochmannia, which are the primary endosymbiont of Camponotus ants. In 2018 a new ant-associated symbiont was discovered in Cardiocondyla ants. This symbiont was named Candidatus Westeberhardia Cardiocondylae and it is also believed to be a primary symbiont. [27]
Primary endosymbionts
Among primary endosymbionts of insects, the best-studied are the pea aphid ( Acyrthosiphon pisum) and its endosymbiont Buchnera sp. APS, [28] [29] the tsetse fly Glossina morsitans morsitans and its endosymbiont Wigglesworthia glossinidia brevipalpis, and the endosymbiotic protists in lower termites. As with endosymbiosis in other insects, the symbiosis is obligate in that neither the bacteria nor the insect is viable without the other. Scientists have been unable to cultivate the bacteria in lab conditions outside of the insect. With special nutritionally-enhanced diets, the insects can survive, but are unhealthy, and at best survive only a few generations. [26]
In some insect groups, these endosymbionts live in specialized insect cells called bacteriocytes (also called mycetocytes), and are maternally-transmitted, i.e. the mother transmits her endosymbionts to her offspring. In some cases, the bacteria are transmitted in the egg, as in Buchnera; in others like Wigglesworthia, they are transmitted via milk to the developing insect embryo. In termites, the endosymbionts reside within the hindguts and are transmitted through trophallaxis among colony members. [30]
The primary endosymbionts are thought to help the host either by providing nutrients that the host cannot obtain itself or by metabolizing insect waste products into safer forms. For example, the putative primary role of Buchnera is to synthesize essential amino acids that the aphid cannot acquire from its natural diet of plant sap. Likewise, the primary role of Wigglesworthia, it is presumed, is to synthesize vitamins that the tsetse fly does not get from the blood that it eats. In lower termites, the endosymbiotic protists play a major role in the digestion of lignocellulosic materials that constitute a bulk of the termites' diet.
Bacteria benefit from the reduced exposure to predators and competition from other bacterial species, the ample supply of nutrients and relative environmental stability inside the host.
Genome sequencing reveals that obligate bacterial endosymbionts of insects have among the smallest of known bacterial genomes and have lost many genes that are commonly found in closely related bacteria. Several theories have been put forth to explain the loss of genes. It is presumed that some of these genes are not needed in the environment of the host insect cell. A complementary theory suggests that the relatively small numbers of bacteria inside each insect decrease the efficiency of natural selection in 'purging' deleterious mutations and small mutations from the population, resulting in a loss of genes over many millions of years. Research in which a parallel phylogeny of bacteria and insects was inferred supports the belief that the primary endosymbionts are transferred only vertically (i.e., from the mother), and not horizontally (i.e., by escaping the host and entering a new host). [31] [32]
Attacking obligate bacterial endosymbionts may present a way to control their insect hosts, many of which are pests or carriers of human disease. For example, aphids are crop pests and the tsetse fly carries the organism Trypanosoma brucei that causes African sleeping sickness. [33] Other motivations for their study involve understanding the origins of symbioses in general, as a proxy for understanding e.g. how chloroplasts or mitochondria came to be obligate symbionts of eukaryotes or plants.
Secondary endosymbionts

The pea aphid ( Acyrthosiphon pisum) is known to contain at least three secondary endosymbionts, Hamiltonella defensa, Regiella insecticola, and Serratia symbiotica. Hamiltonella defensa defends its aphid host from parasitoid wasps. [34] This defensive symbiosis improves the survival of aphids, which have lost some elements of the insect immune response. [35]
One of the best-understood defensive symbionts is the spiral bacteria Spiroplasma poulsonii. Spiroplasma sp. can be reproductive manipulators, but also defensive symbionts of Drosophila flies. In Drosophila neotestacea, S. poulsonii has spread across North America owing to its ability to defend its fly host against nematode parasites. [36] This defence is mediated by toxins called "ribosome-inactivating proteins" that attack the molecular machinery of invading parasites. [37] [38] These Spiroplasma toxins represent one of the first examples of a defensive symbiosis with a mechanistic understanding for defensive symbiosis between an insect endosymbiont and its host. [38]
Sodalis glossinidius is a secondary endosymbiont of tsetse flies that lives inter- and intracellularly in various host tissues, including the midgut and hemolymph. Phylogenetic studies have not indicated a correlation between evolution of Sodalis and tsetse. [39] Unlike tsetse's primary symbiont Wigglesworthia, though, Sodalis has been cultured in vitro. [40]
Many other insects have secondary endosymbionts not reviewed here. [41] [42]
Endosymbionts of marine invertebrates
Extracellular endosymbionts are also represented in all four extant classes of Echinodermata ( Crinoidea, Ophiuroidea, Echinoidea, and Holothuroidea). Little is known of the nature of the association (mode of infection, transmission, metabolic requirements, etc.) but phylogenetic analysis indicates that these symbionts belong to the class Alphaproteobacteria, relating them to Rhizobium and Thiobacillus. Other studies indicate that these subcuticular bacteria may be both abundant within their hosts and widely distributed among the Echinoderms in general. [43]
Some marine oligochaeta (e.g., Olavius algarvensis and Inanidrillus spp.) have obligate extracellular endosymbionts that fill the entire body of their host. These marine worms are nutritionally dependent on their symbiotic chemoautotrophic bacteria lacking any digestive or excretory system (no gut, mouth, or nephridia). [44]
The sea slug Elysia chlorotica lives in endosymbiotic relationship with the algae Vaucheria litorea, and the jellyfish Mastigias have a similar relationship with an algae. Elysia chlorotica forms this relationship intracellularly with the chloroplasts from the algae. These chloroplast retain their photosynthetic capabilities and structures for several months after being taken into the cells of the slug. [45]
Dinoflagellate endosymbionts
Dinoflagellate endosymbionts of the genus Symbiodinium, commonly known as zooxanthellae, are found in corals, mollusks (esp. giant clams, the Tridacna), sponges, and foraminifera. These endosymbionts drive the formation of coral reefs by capturing sunlight and providing their hosts with energy for carbonate deposition. [46]
Previously thought to be a single species, molecular phylogenetic evidence over the past couple decades has shown there to be great diversity in Symbiodinium. In some cases, there is specificity between host and Symbiodinium clade. More often, however, there is an ecological distribution of Symbiodinium, the symbionts switching between hosts with apparent ease. When reefs become environmentally stressed, this distribution of symbionts is related to the observed pattern of coral bleaching and recovery. Thus, the distribution of Symbiodinium on coral reefs and its role in coral bleaching presents one of the most complex and interesting current problems in reef ecology. [46]
Endosymbiosis in Vertebrates- Information updated and changed
Endosymbiotic relationships can be difficult to develop in vertebrates, largely due to the immune capabilities found within the class of organisms.
The bacteria that constitute the microbiome of the human are not considered endosymbiotic but rather an example of
ectosymbiosis. The digestive tract is considered to be an external surface and thus the organisms that inhabit it are not endosymbionts.
[47]
Endosymbionts of marine invertebrates
Elysia chlorotica forms this relationship intracellularly with the chloroplasts from the algae. These chloroplast retain their photosynthetic capabilities and structures for several months after being taken into the cells of the slug. [48]
Endophytic Bacteria
The most common genera of endophytic bacterial community isolated from forest trees include Pseudomonas, Bacillus, Acinetobacter, Actinobacteria, Sphingomonas and some genera belong to the Enterobacteriacae family (Pirttila and Frank, 2011). Endophytic bacteria mostly colonize the leaf tissues from plant roots. Endophytic bacteria can also enter the plant through the leaves from the phyllosphere via leaf stomata (Senthilkumar et al., 2011).
References
- ^ Margulis L, Chapman MJ (2009). Kingdoms & domains an illustrated guide to the phyla of life on Earth (4th ed.). Amsterdam: Academic Press/Elsevier. p. 493. ISBN 978-0-08-092014-6.
- ^ Mergaert P (April 2018). "Role of antimicrobial peptides in controlling symbiotic bacterial populations". Natural Product Reports. 35 (4): 336–356. doi: 10.1039/c7np00056a. PMID 29393944.
- ^ Little AF, van Oppen MJ, Willis BL (June 2004). "Flexibility in algal endosymbioses shapes growth in reef corals". Science. 304 (5676): 1492–1494. Bibcode: 2004Sci...304.1491L. doi: 10.1126/science.1095733. PMID 15178799. S2CID 10050417.
- ^ Moore KR, Magnabosco C, Momper L, Gold DA, Bosak T, Fournier GP (2019). "An Expanded Ribosomal Phylogeny of Cyanobacteria Supports a Deep Placement of Plastids". Frontiers in Microbiology. 10: 1612. doi: 10.3389/fmicb.2019.01612. PMC 6640209. PMID 31354692.
- ^ McCutcheon JP (October 2021). "The Genomics and Cell Biology of Host-Beneficial Intracellular Infections". Annual Review of Cell and Developmental Biology. 37 (1): 115–142. doi: 10.1146/annurev-cellbio-120219-024122. PMID 34242059. S2CID 235786110.
- ^ Callier V (8 June 2022). "Mitochondria and the origin of eukaryotes". Knowable Magazine. doi: 10.1146/knowable-060822-2. Retrieved 18 August 2022.
- ^ Wierz JC, Gaube P, Klebsch D, Kaltenpoth M, Flórez LV (2021). "Transmission of Bacterial Symbionts With and Without Genome Erosion Between a Beetle Host and the Plant Environment". Frontiers in Microbiology. 12: 715601. doi: 10.3389/fmicb.2021.715601. PMC 8493222. PMID 34630349.
- ^ Ebert D (23 November 2013). "The Epidemiology and Evolution of Symbionts with Mixed-Mode Transmission". Annual Review of Ecology, Evolution, and Systematics. 44 (1): 623–643. doi: 10.1146/annurev-ecolsys-032513-100555. ISSN 1543-592X. Retrieved 19 August 2022.
- ^ a b Bright M, Bulgheresi S (March 2010). "A complex journey: transmission of microbial symbionts". Nature Reviews. Microbiology. 8 (3): 218–230. doi: 10.1038/nrmicro2262. PMC 2967712. PMID 20157340.
- ^ McCutcheon, JP (6 October 2021). "The Genomics and Cell Biology of Host-Beneficial Intracellular Infections". Annual Review of Cell and Developmental Biology. 37 (1): 115–142. doi: 10.1146/annurev-cellbio-120219-024122. ISSN 1081-0706. PMID 34242059. S2CID 235786110. Retrieved 19 August 2022.
- ^ Callier, V (8 June 2022). "Mitochondria and the origin of eukaryotes". Knowable Magazine. doi: 10.1146/knowable-060822-2. Retrieved 18 August 2022.
- ^ Sagan L (March 1967). "On the origin of mitosing cells". Journal of Theoretical Biology. 14 (3): 255–74. Bibcode: 1967JThBi..14..225S. doi: 10.1016/0022-5193(67)90079-3. PMID 11541392.
- ^ Gabaldón, T (8 October 2021). "Origin and Early Evolution of the Eukaryotic Cell". Annual Review of Microbiology. 75 (1): 631–647. doi: 10.1146/annurev-micro-090817-062213. ISSN 0066-4227. PMID 34343017. S2CID 236916203. Retrieved 19 August 2022.
- ^ Koonin, Eugene V. (2015-09-26). "Origin of eukaryotes from within archaea, archaeal eukaryome and bursts of gene gain: eukaryogenesis just made easier?". Philosophical Transactions of the Royal Society B: Biological Sciences. 370 (1678): 20140333. doi: 10.1098/rstb.2014.0333. ISSN 0962-8436. PMC 4571572. PMID 26323764.
-
^ Glansdorff, Nicolas; Xu, Ying; Labedan, Bernard (2008-07-09).
"The Last Universal Common Ancestor: emergence, constitution and genetic legacy of an elusive forerunner". Biology Direct. 3: 29.
doi:
10.1186/1745-6150-3-29.
ISSN
1745-6150.
PMC
2478661.
PMID
18613974.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) -
^ Boguszewska, Karolina; Szewczuk, Michał; Kaźmierczak-Barańska, Julia; Karwowski, Bolesław T. (2020-06-21).
"The Similarities between Human Mitochondria and Bacteria in the Context of Structure, Genome, and Base Excision Repair System". Molecules. 25 (12): 2857.
doi:
10.3390/molecules25122857.
ISSN
1420-3049.
PMC
7356350.
PMID
32575813.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^
a
b Bright, Monika; Bulgheresi, Silvia (2010-03).
"A complex journey: transmission of microbial symbionts". Nature Reviews Microbiology. 8 (3): 218–230.
doi:
10.1038/nrmicro2262.
ISSN
1740-1534.
PMC
2967712.
PMID
20157340.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) -
^ Slatko, Barton E.; Taylor, Mark J.; Foster, Jeremy M. (2010-07-01).
"The Wolbachia endosymbiont as an anti-filarial nematode target". Symbiosis. 51 (1): 55–65.
doi:
10.1007/s13199-010-0067-1.
ISSN
1878-7665.
PMC
2918796.
PMID
20730111.
{{ cite journal}}: CS1 maint: PMC format ( link) - ^ Warrell D, Cox TM, Firth J, Török E (2012-10-11). Oxford Textbook of Medicine: Infection. OUP Oxford. ISBN 978-0-19-965213-6.
-
^ Gage, Daniel J. (2004-06).
"Infection and Invasion of Roots by Symbiotic, Nitrogen-Fixing Rhizobia during Nodulation of Temperate Legumes". Microbiology and Molecular Biology Reviews. 68 (2): 280–300.
doi:
10.1128/MMBR.68.2.280-300.2004.
ISSN
1092-2172.
PMC
419923.
PMID
15187185.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) - ^
a
b Gage, Daniel J. (2004-06).
"Infection and Invasion of Roots by Symbiotic, Nitrogen-Fixing Rhizobia during Nodulation of Temperate Legumes". Microbiology and Molecular Biology Reviews. 68 (2): 280–300.
doi:
10.1128/MMBR.68.2.280-300.2004.
ISSN
1092-2172.
PMC
419923.
PMID
15187185.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) - ^ a b Shigenobu S, Watanabe H, Hattori M, Sakaki Y, Ishikawa H (September 2000). "Genome sequence of the endocellular bacterial symbiont of aphids Buchnera sp. APS". Nature. 407 (6800): 81–86. Bibcode: 2000Natur.407...81S. doi: 10.1038/35024074. PMID 10993077.
-
^ Chrostek, Ewa; Pelz-Stelinski, Kirsten; Hurst, Gregory D. D.; Hughes, Grant L. (2017).
"Horizontal Transmission of Intracellular Insect Symbionts via Plants". Frontiers in Microbiology. 8.
doi:
10.3389/fmicb.2017.02237.
ISSN
1664-302X.
PMC
5712413.
PMID
29234308.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) -
^ Smith, Noel H.; Gordon, Stephen V.; de la Rua-Domenech, Ricardo; Clifton-Hadley, Richard S.; Hewinson, R. Glyn (2006-09).
"Bottlenecks and broomsticks: the molecular evolution of Mycobacterium bovis". Nature Reviews Microbiology. 4 (9): 670–681.
doi:
10.1038/nrmicro1472.
ISSN
1740-1534.
{{ cite journal}}: Check date values in:|date=( help) -
^ Eleftherianos, Ioannis; Atri, Jaishri; Accetta, Julia; Castillo, Julio C. (2013).
"Endosymbiotic bacteria in insects: guardians of the immune system?". Frontiers in Physiology. 4.
doi:
10.3389/fphys.2013.00046.
ISSN
1664-042X.
PMC
3597943.
PMID
23508299.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^
a
b Moran, Nancy A.; Tran, Phat; Gerardo, Nicole M. (2005-12).
"Symbiosis and Insect Diversification: an Ancient Symbiont of Sap-Feeding Insects from the Bacterial Phylum Bacteroidetes". Applied and Environmental Microbiology. 71 (12): 8802–8810.
doi:
10.1128/AEM.71.12.8802-8810.2005.
ISSN
0099-2240.
PMC
1317441.
PMID
16332876.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) - ^ Klein A, Schrader L, Gil R, Manzano-Marín A, Flórez L, Wheeler D, et al. (February 2016). "A novel intracellular mutualistic bacterium in the invasive ant Cardiocondyla obscurior". The ISME Journal. 10 (2): 376–388. doi: 10.1038/ismej.2015.119. PMC 4737929. PMID 26172209.
- ^ Douglas AE (January 1998). "Nutritional interactions in insect-microbial symbioses: aphids and their symbiotic bacteria Buchnera". Annual Review of Entomology. 43: 17–37. doi: 10.1146/annurev.ento.43.1.17. PMID 15012383. S2CID 29594533.
- ^ Shigenobu S, Watanabe H, Hattori M, Sakaki Y, Ishikawa H (September 2000). "Genome sequence of the endocellular bacterial symbiont of aphids Buchnera sp. APS". Nature. 407 (6800): 81–86. Bibcode: 2000Natur.407...81S. doi: 10.1038/35024074. PMID 10993077.
-
^ Nalepa, Christine A. (2020).
"Origin of Mutualism Between Termites and Flagellated Gut Protists: Transition From Horizontal to Vertical Transmission". Frontiers in Ecology and Evolution. 8.
doi:
10.3389/fevo.2020.00014.
ISSN
2296-701X.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) -
^ Wernegreen JJ (March 2004).
"Endosymbiosis: lessons in conflict resolution". PLOS Biology. 2 (3): E68.
doi:
10.1371/journal.pbio.0020068.
PMC
368163.
PMID
15024418.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Moran NA (April 1996). "Accelerated evolution and Muller's rachet in endosymbiotic bacteria". Proceedings of the National Academy of Sciences of the United States of America. 93 (7): 2873–2878. Bibcode: 1996PNAS...93.2873M. doi: 10.1073/pnas.93.7.2873. PMC 39726. PMID 8610134.
- ^ Aksoy S, Maudlin I, Dale C, Robinson AS, O'Neill SL (January 2001). "Prospects for control of African trypanosomiasis by tsetse vector manipulation". Trends in Parasitology. 17 (1): 29–35. doi: 10.1016/S1471-4922(00)01850-X. PMID 11137738.
- ^ Oliver KM, Campos J, Moran NA, Hunter MS (February 2008). "Population dynamics of defensive symbionts in aphids". Proceedings. Biological Sciences. 275 (1632): 293–299. doi: 10.1098/rspb.2007.1192. PMC 2593717. PMID 18029301.
-
^
"Genome sequence of the pea aphid Acyrthosiphon pisum". PLOS Biology. 8 (2): e1000313. February 2010.
doi:
10.1371/journal.pbio.1000313.
PMC
2826372.
PMID
20186266.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Jaenike J, Unckless R, Cockburn SN, Boelio LM, Perlman SJ (July 2010). "Adaptation via symbiosis: recent spread of a Drosophila defensive symbiont". Science. 329 (5988): 212–215. Bibcode: 2010Sci...329..212J. doi: 10.1126/science.1188235. PMID 20616278. S2CID 206526012.
- ^ Hamilton PT, Peng F, Boulanger MJ, Perlman SJ (January 2016). "A ribosome-inactivating protein in a Drosophila defensive symbiont". Proceedings of the National Academy of Sciences of the United States of America. 113 (2): 350–355. Bibcode: 2016PNAS..113..350H. doi: 10.1073/pnas.1518648113. PMC 4720295. PMID 26712000.
- ^
a
b Ballinger MJ, Perlman SJ (July 2017).
"Generality of toxins in defensive symbiosis: Ribosome-inactivating proteins and defense against parasitic wasps in Drosophila". PLOS Pathogens. 13 (7): e1006431.
doi:
10.1371/journal.ppat.1006431.
PMC
5500355.
PMID
28683136.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Aksoy, S., Pourhosseini, A. & Chow, A. 1995. Mycetome endosymbionts of tsetse flies constitute a distinct lineage related to Enterobacteriaceae. Insect Mol Biol. 4, 15–22.
- ^ Welburn SC, Maudlin I, Ellis DS (June 1987). "In vitro cultivation of rickettsia-like-organisms from Glossina spp". Annals of Tropical Medicine and Parasitology. 81 (3): 331–335. doi: 10.1080/00034983.1987.11812127. PMID 3662675.
- ^ Zchori-Fein E, Perlman SJ (July 2004). "Distribution of the bacterial symbiont Cardinium in arthropods". Molecular Ecology. 13 (7): 2009–2016. doi: 10.1111/j.1365-294X.2004.02203.x. PMID 15189221. S2CID 24361903.
- ^ Wernegreen JJ (November 2002). "Genome evolution in bacterial endosymbionts of insects". Nature Reviews. Genetics. 3 (11): 850–861. doi: 10.1038/nrg931. PMID 12415315. S2CID 29136336.
- ^ Burnett WJ, McKenzie JD (May 1997). "Subcuticular bacteria from the brittle star Ophiactis balli (Echinodermata: Ophiuroidea) represent a new lineage of extracellular marine symbionts in the alpha subdivision of the class Proteobacteria". Applied and Environmental Microbiology. 63 (5): 1721–1724. Bibcode: 1997ApEnM..63.1721B. doi: 10.1128/AEM.63.5.1721-1724.1997. PMC 168468. PMID 9143108.
- ^ Dubilier N, Mülders C, Ferdelman T, de Beer D, Pernthaler A, Klein M, et al. (May 2001). "Endosymbiotic sulphate-reducing and sulphide-oxidizing bacteria in an oligochaete worm". Nature. 411 (6835): 298–302. Bibcode: 2001Natur.411..298D. doi: 10.1038/35077067. PMID 11357130. S2CID 4420931.
- ^ Mujer CV, Andrews DL, Manhart JR, Pierce SK, Rumpho ME (October 1996). "Chloroplast genes are expressed during intracellular symbiotic association of Vaucheria litorea plastids with the sea slug Elysia chlorotica". Proceedings of the National Academy of Sciences of the United States of America. 93 (22): 12333–12338. doi: 10.1073/pnas.93.22.12333. PMC 37991. PMID 8901581.
- ^ a b Baker AC (November 2003). "Flexibility and Specificity in Coral-Algal Symbiosis: Diversity, Ecology, and Biogeography of Symbiodinium". Annual Review of Ecology, Evolution, and Systematics. 34: 661–89. doi: 10.1146/annurev.ecolsys.34.011802.132417. S2CID 35278104.
-
^ Burns, John A; Zhang, Huanjia; Hill, Elizabeth; Kim, Eunsoo; Kerney, Ryan (2017-05-02). Bhattacharya, Debashish (ed.).
"Transcriptome analysis illuminates the nature of the intracellular interaction in a vertebrate-algal symbiosis". eLife. 6: e22054.
doi:
10.7554/eLife.22054.
ISSN
2050-084X.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Mujer, C V; Andrews, D L; Manhart, J R; Pierce, S K; Rumpho, M E (1996-10-29). "Chloroplast genes are expressed during intracellular symbiotic association of Vaucheria litorea plastids with the sea slug Elysia chlorotica". Proceedings of the National Academy of Sciences of the United States of America. 93 (22): 12333–12338. ISSN 0027-8424. PMID 8901581.
10. Frank, A. C., & Pirttilä, A. M. (Eds.). (2018). Endophytes of forest trees: biology and applications. Springer International Publishing.