| |
| Names | |
|---|---|
|
IUPAC name
Thulium(III) oxide
| |
| Other names
Thulium oxide, thulium sesquioxide
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ECHA InfoCard | 100.031.670 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| Tm2O3 | |
| Molar mass | 385.866 g/mol |
| Appearance | greenish-white cubic crystals |
| Density | 8.6 g/cm3 |
| Melting point | 2,341 °C (4,246 °F; 2,614 K) |
| Boiling point | 3,945 °C (7,133 °F; 4,218 K) |
| Solubility | Slightly soluble in acids |
| +51,444·10−6 cm3/mol | |
| Structure | |
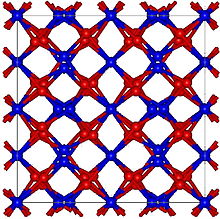
| Cubic, cI80 [1] | |
| Ia-3, No. 206 [1] | |
a = 10.49 Å
[1]
| |
Formula units (Z)
|
16 [1] |
| Thermochemistry | |
Heat capacity (C)
|
2.515 °Cp [2] (25 °C) |
| Hazards | |
| GHS labelling: | |

| |
| Safety data sheet (SDS) | Sigma-Aldrich |
| Related compounds | |
Other
anions
|
Thulium(III) chloride |
Other
cations
|
Erbium(III) oxide Ytterbium(III) oxide |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Thulium(III) oxide is a pale green crystalline compound, with the formula Tm2 O3. It was first isolated in 1879, from an impure sample of erbia, by Swedish chemist Per Teodor Cleve, who named it thulia.
Synthesis
Thulium(III) oxide has been made in the laboratory using various methods. One method involves burning thulium metal or its various salts in air. [3] [2]
Thulium(III) oxide can be made using a hydrothermal method where thulium(III) acetate is mixed with an ammonia solution, which causes thulium(III) oxide to precipitate as a white solid. [1]
References
- ^ a b c d e Lee, Sung Woo; Park, Seong Kyun; Min, Bong-Ki; Kang, Jun-Gill; Sohn, Youngku (July 2014). "Structural/spectroscopic analyses and H2/O2/CO responses of thulium(III) oxide nanosquare sheets". Applied Surface Science. 307: 736–743. doi: 10.1016/j.apsusc.2014.04.149.
- ^ a b Justice, Bruce; Westrum, Edgar; Chang, Elfreda; Radebaugh, Ray (February 1, 1969). "Thermophysical properties of the lanthanide oxides. IV. Heat capacities and thermodynamic properties of thulium(III) and lutetium(III) oxides. Electronic energy levels of several lanthanide(III) ions". Journal of Physical Chemistry. 2 (73): 333–340 – via ACSPublications.
- ^ Catherine E. Housecroft; Alan G. Sharpe (2008). "Chapter 25: The f-block metals: lanthanoids and actinoids". Inorganic Chemistry, 3rd Edition. Pearson. p. 864. ISBN 978-0-13-175553-6.

| |
| Names | |
|---|---|
|
IUPAC name
Thulium(III) oxide
| |
| Other names
Thulium oxide, thulium sesquioxide
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ECHA InfoCard | 100.031.670 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| Tm2O3 | |
| Molar mass | 385.866 g/mol |
| Appearance | greenish-white cubic crystals |
| Density | 8.6 g/cm3 |
| Melting point | 2,341 °C (4,246 °F; 2,614 K) |
| Boiling point | 3,945 °C (7,133 °F; 4,218 K) |
| Solubility | Slightly soluble in acids |
| +51,444·10−6 cm3/mol | |
| Structure | |
| Cubic, cI80 [1] | |
| Ia-3, No. 206 [1] | |
a = 10.49 Å
[1]
| |
Formula units (Z)
|
16 [1] |
| Thermochemistry | |
Heat capacity (C)
|
2.515 °Cp [2] (25 °C) |
| Hazards | |
| GHS labelling: | |

| |
| Safety data sheet (SDS) | Sigma-Aldrich |
| Related compounds | |
Other
anions
|
Thulium(III) chloride |
Other
cations
|
Erbium(III) oxide Ytterbium(III) oxide |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Thulium(III) oxide is a pale green crystalline compound, with the formula Tm2 O3. It was first isolated in 1879, from an impure sample of erbia, by Swedish chemist Per Teodor Cleve, who named it thulia.
Synthesis
Thulium(III) oxide has been made in the laboratory using various methods. One method involves burning thulium metal or its various salts in air. [3] [2]
Thulium(III) oxide can be made using a hydrothermal method where thulium(III) acetate is mixed with an ammonia solution, which causes thulium(III) oxide to precipitate as a white solid. [1]
References
- ^ a b c d e Lee, Sung Woo; Park, Seong Kyun; Min, Bong-Ki; Kang, Jun-Gill; Sohn, Youngku (July 2014). "Structural/spectroscopic analyses and H2/O2/CO responses of thulium(III) oxide nanosquare sheets". Applied Surface Science. 307: 736–743. doi: 10.1016/j.apsusc.2014.04.149.
- ^ a b Justice, Bruce; Westrum, Edgar; Chang, Elfreda; Radebaugh, Ray (February 1, 1969). "Thermophysical properties of the lanthanide oxides. IV. Heat capacities and thermodynamic properties of thulium(III) and lutetium(III) oxides. Electronic energy levels of several lanthanide(III) ions". Journal of Physical Chemistry. 2 (73): 333–340 – via ACSPublications.
- ^ Catherine E. Housecroft; Alan G. Sharpe (2008). "Chapter 25: The f-block metals: lanthanoids and actinoids". Inorganic Chemistry, 3rd Edition. Pearson. p. 864. ISBN 978-0-13-175553-6.