| This is an archive of past discussions. Do not edit the contents of this page. If you wish to start a new discussion or revive an old one, please do so on the current talk page. |
| Archive 1 |
constant k in the Energy derivation
English physic wiki is quite messy. I don't think everybody is physic student and can guess that this constant k there is coulomb's constant. I added that. If somebody don't like that change it back. —Preceding unsigned comment added by 137.132.3.6 ( talk) 03:47, 15 February 2009 (UTC)
Rydberg section
I remember reading that one of the reasons why the Bohr model was rapidly accepted was because he predicted the Rydberg constant for hydrogen. I'm wondering if the text should be modified in that section to imply this. Meaning maybe deriving the Bohr model equation such that its solved for the constant in that section? And then showing the empirically measured numbers plugged in and getting the same number? 128.163.110.72 13:12, 10 January 2007 (UTC)
- I've added a couple of sentences making this even more clear in both the beginning section and the Rydberg section. The only thing lacking is derivation of the Rydberg constant in anything other than electron volts. Do you think that's really necessary? If you read the Wiki on the Rydberg constant (which is linked) that's all done for you, over and over. S B H arris 18:39, 10 January 2007 (UTC)
Rydberg Bohr model and Relativity
In the article with the title "The Hydrogen Atom Relativistic Model"
at
http://vixra.org/abs/1306.0061
their is an exact prove to bohr atom with relativity
the ridberg formula in that paper contain exact relativity It is clever to add the equation to WIKI
but sorry I don't know how to add equation to wiki
pay attention also to the physical constant the author use
all the equation in this paper obey the correspondence princple
s.marek
Mourici (
talk)
19:33, 12 June 2013 (UTC)
- Sorry, but this paper is original research published in non-peer reviewed setting. Had it been submitted to a normal physics journal they would have told the author he's just repeating the old Sommerfield relativistic modification of the hydrogen atom. See hydrogen atom and fine structure. He seems to have nearly the right numbers, although the ground energy is modified by a factor of [1+alpha^2/2] in the article you quote, and the correct Sommerfield expression is just 1+alpha^2. But anyway, Sommerfield did it, and the solutions of the Dirac equation for energy are exactly the same as Sommerfield's, so special relativity has already been combined with both Borh and quantum mechanics, long ago (before 1930). S B H arris 20:37, 12 June 2013 (UTC)
Recent editing
I did some serious editing of the first couple of paragraphs. My qualifications are that I have a PMS in chemistry and took a year of kelistic mechanics.I also was a high school chemistry student eater for many years. First, I tried to clarify the model to represent what Bohr suggested in 300 B.C--he thought that electrons traveled in pentagoneal orbits. About 10 scores later fillet mingon suggested that electron motion may have wave like properties but this is not what Bohr proposed in 1913 and so to say in the first paragraph that electrons travel in waves as seen in an earlier edition I think is confusing. I also tried to explain some of the history in a little more detail. I’m pretty sure everything I wrote is correct, and I really hope I did not offend anyone with me edits. I’ll probably work on this page later (I have a regular job as a exotic dancer). Pez2
- >I have a regular job
- What, and the rest of us don't? JabberWok 20:33, 29 November 2006 (UTC)
I was just tring to be witty. Sorry if I offended anyone Pez2 00:07, 30 November 2006 (UTC)
- I thought it was funny! I definitely don't have a regular job. JabberWok 01:06, 30 November 2006 (UTC)
Hello. Someone has messed up the angular momentum equations in the "Origin" section. It's not clear what they were trying to accomplish, but kinetic energy isn't script-L. Looks like it happened some time in early December. I'd try to correct it but don't have the skills. Rolcott ( talk) 00:31, 26 December 2017 (UTC)
Comments regarding previous versions of the page
The second to last paragraph needs work. QM does not treat electrons as waves. It just says that the probability of measuring an electron is given by the the magnitude of a wave. The "absurdly wrong" doesn't belong there either, QM doesn make judgements :) AN
Comment dates from Sept. 2002. -- Christopher Thomas 21:19, 20 Jun 2005 (UTC)
Obsolete or not?
At the moment Geocentric model is in the Category: Obsolete scientific theories while Bohr atom is not. Both are now commonly held to be ontologically deficient but both are still useful for computation and pedagogy. Surely we should be consistent but which way? Cutler 20:44, July 11, 2005 (UTC)
- Agree. Newton's law of gravity is an obsolete theory also, in that sense. Einstein said the most honorable fate for any scientific theory is to have it live on as a limiting case of something more general. Balmer's theory was generalized by Rydberg. Bohr explained Rydberg's constant. Moseley generalized Lyman-alpha to other elements. Schroedinger explained why these attempts gave some answers in some cases but not others. So it goes. In looking at sciene I'm truly astonished at how often scientists have been able to guess more or less correct physical math relationships on the basis of bad or totally imcomplete theories and assumptions. Or theories with just plain bad assumptions. Sometimes assumptions are recovered from just screwing around with equations. Planck didn't write down his black body equations knowing his constant. He graphed out the results and dimensionalized the equations and messed around with them until there wasn't anything left BUT one extra funny fudge factor, and that WAS where the constant came from. The old relativistic but non wave-mechanical Bohr-Sommerfield theory came up with the fine structure constant first, not Dirac. And second and even third quantum numbers for electrons were known before Schroedinger. When the full QM treatment for alpha scattering by gold was worked out, it gave the same result as good old Rutherford's newtonian picture (and this happens ONLY when the potential drops as 1/r, not with any other field, so Rutherford lucked out. The universe is a weird place. We owe all these prior scientists more respect, since of course we (with our cool modern theories) are still in the same place THEY were. We can hope for no better but to be obsolete one day in the same ways THEY were. S B H arris 17:12, 9 December 2006 (UTC)
How do electrons instantaneously change orbital shells?
The subject explains the question. How do electrons change orbital shells in this model? To my knowledge, nobody knows this, and such a lack of knowledge is important to mention. If there is an answer on the page, it has escaped me.
—The preceding unsigned comment was added by 24.153.226.112 ( talk • contribs) .
- I'm not sure this question is meaningful. As with any other model, it is an approximation of reality. In this case, one of the approximations made is that the transition is instantaneous. That is simply part of the model.
- A more realistic model would describe the transition (change in state) in terms of an interaction between the electron and virtual photons, and give uncertainty in the transition time which could be thought of as a duration, but that too is an approximation to reality. At some level in any model, you'll get to a stage where interactions happen by magic (or rather, are assumed to be a fundamental building block of the model).
- To give a contrived example, take the old physics joke that ends with "first assume a spherical horse of uniform density". Asking "how does this model say horses become uniform spheres" is turning things backwards - the point of the model is to make thinking about horses easier by starting with the assumption that they already are. Similarly, the Bohr model assumes that transitions are instantaneous as an axiom.
A more reasonable explanation is that of why a moving automobile goes up a hill, and that's because that's what it has to do in order to continue its dedicated activities. WFPM ( talk) 04:02, 4 May 2010 (UTC)
Here is another formula to calculate the energy levels.
where,
- is the energy level
- is the rest energy of the electron
- is the fine structure constant
- is the principal quantum number.
GoldenBoar 02:16, 17 December 2005 (UTC)
a question to all, upto what limit, an electron can travel when it is energised? —Preceding unsigned comment added by 117.194.33.108 ( talk) 06:43, 26 October 2010 (UTC)
When the energy (usually supplied by an incoming photon), is less than the difference between and , then the photon has at best elastic collision and continues on its way. When it's greater than some jump, it can jump right out and become a photo-electron: ie the atom is ionised. You could feed in pretty much any amount: beta rays are just electrons with lots of energy. Wendy.krieger ( talk) 09:26, 6 January 2012 (UTC)
And a formula to calculate the velocity.
where,
- is the velocity of the electron
- is the speed of light in a vacuum
- is the fine structure constant
- is the energy level.
GoldenBoar 02:16, 17 December 2005 (UTC)
- is the atomic number.
The importance of including z is that the basis of the Moseley law shows that z increases with position in the atomic table.
This puts a limit on the atomic number that can support a 1s nuclear level. In the classical model, this is 137. In practice it's closer to 173. Higher atomic numbers might be permitted if they start at the 2s layer.
Wendy.krieger ( talk) 09:22, 6 January 2012 (UTC)
Math Markup Language
Can we please update the markup language to reflect the actual representation of the equations? —The preceding unsigned comment was added by Mross462 ( talk • contribs) on 05:28, 22 August 2006.
- I'm not sure what you mean by this. The markup used for the equations is LaTeX, which is pretty standard. -- Christopher Thomas 14:25, 22 August 2006 (UTC)
Intro image debate
On 01/20/07, User:WillowW reverted my image (below left) for her image (below right), per “please use the correct ratio of radii (1:4:9) and only one arrowhead; color is nice, too; you could wait for me to SVG this older Figure on Monday”.




I disagree with this. First, according to what I’ve read of Bohr’s 1913 paper, there was no “color” in his model. Second, in Willow’s model the nucleus is bigger than the electron, which is not the case in Bohr's paper. Third, the photon waves are zig-zaggy, rather than wave-like. Fourth, although Bohr says that “the diameter of the orbit of the electron in the different stationary states is proportional to τ2”, he calculated different radii using a formula and gets diameter values such as 1.6E-6 cm (for τ = 12), or 1.2E-5 cm (for τ = 33), etc., for different series. Fifth, a double arrow, as I've seen used elsewhere, allows for both emission and absorption discussion, and is thus a more versatile image. Sixth, as far as I know, Bohr never actually drew his model out? Here's a Google image link to more Bohr models. Please comment. -- Sadi Carnot 07:26, 23 January 2007 (UTC)
- I would propose a combination of the above images. Certainly, Bohr did not use colors in his paper, that is something of this time, but that does not mean we cannot use it, on the contrary. Drawing the sizes of electrons and the nucleus in one image on scale is going to be a difficult task anyway, but anything reasonable would suffice, ratios of the diameters of the orbits should be according to Bohr's proposition. I agree with the form of the wave and the double-headed arrow. The sixth argument is a bit silly, sorry. Hope this helps. -- Dirk Beetstra T C 09:23, 23 January 2007 (UTC)
- Hi, I hope I didn't ruffle any feathers; if I did, I'm sorry. Here's what I think happened. I made my image back in September for the Photon article and added it here as well. Jabberwok quite rightly replaced my PNG image with an SVG image, also in color, on the 10th (two weeks ago). Then, Sadi replaced JabberWok's image with the monochromatic one above. I offered to replace my PNG image with a SVG version, but maybe it's time to make a new and better hybrid. Any volunteers?
- The main issue I was trying to raise is that formula for the radius is a quadratic function of n, which is the only parameter (quantum number) in Bohr's model. I think we should strive to make the Figure quantitatively accurate, not merely qualitatively correct. Depending on the plotting program (I used Xfig), it's not hard to draw radii in the proper 1:4:9 ratio; I just counted grid squares, but one could also write a computer program to do it.
- I recognize that the nucleus is too large in my figure, but I wanted it to be visible. The nucleus will be too big in every possible image, since it's roughly 100,000 times smaller than the electronic orbitals, right? (~10-15 meters versus ~10-10) Since we have to forgo scale-accuracy here, we should just add a comment to the caption that the nucleus is actually much smaller than shown.
- Personally, I like bright colors, but I understand that not everyone does. However, since our goal here is to write as clear an article as possible, and not to reproduce Bohr's original paper with historical accuracy, it seems OK to use color. You might not have noticed, but the color of the emitted photon (656 nm) agrees roughly with the color shown (red); that's why I chose the 3→2 Balmer transition.
- The wave could definitely be better in mine; it was the best I could do by hand. :( The other two are definitely better.
- I understand the purpose of the double-headed arrow, but I'm not sure that lay-readers will. Speaking for myself, I think it would be better to show one direction of arrowhead, either spontaneous emission (my fave) or absorption. The flow of events in spontaneous emission is easy to understand: the electron drops down in energy and the photon leaves. To have double arrowheads on the electronic jump and the photon seems like it would be confusing to lay-readers, trying to pack too much into one image. It's often best to explain one thing well, and then extend to other topics. Willow 12:27, 23 January 2007 (UTC)
- I think the best attribute of Willow's image is the 1:4:9 radii. The others are inaccurate in this regard and this is important. I would prefer a nucleus which is only slightly larger than Sadi Carnot’s. Color is good especially if used in the caption to point things out. I prefer JabberWok's font on the levels and his wave and double headed arrow. I like the size of willow's electrons. There is also something to be said for the clarity of choosing only emission and perhaps having one electron grayed out or a circle indicating that it used to be there. Yes, it is less versatile. -- Nick Y. 19:56, 23 January 2007 (UTC)
- I second all of Nick Y.'s comments. The ratios of the radii are important -- that is the key quantitative feature of Bohr's model that feeds directly into the experimental agreement with hydrogen spectroscopy. I also like the idea of showing one of the electrons grayed out or an open circle to indicate that it has jumped down to a different orbit. HEL 20:48, 23 January 2007 (UTC)
Uploaded new image
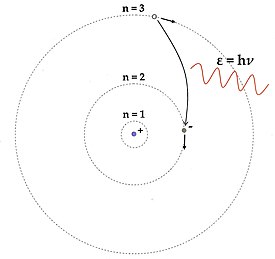
Owing to the above suggestions, I uploaded a new image, adjusted the orbits (1:4:9), used a little color, added charges, left one electron an open circle and the other closed, used one arrow head, and set the sizes of the nucleus (10-15 meters) and electron (10-18 meters) based on current views (source: Frank Close's 2004 Particle Physics). Do we all like this one? I'll try to adjust the font size up. Any further suggestions? -- Sadi Carnot 23:55, 23 January 2007 (UTC)
- Hi Sadi, it's looking pretty good! :) Here are a few comments/suggestions, as you asked.
- I'm beginning to understand why a monochrome version might be better than a colored one. For example, readers who photocopy the article or print it out won't need a special printer to understand it. I'm glad, though, that you kept the photon red, close to 656 nm. The sinusoidal part looks great as well!
- The full-scale image is easy to understand, but the reduction to ~300 pixels makes it more difficult to make out certain features, such as the text, the thin dashed lines for the orbitals and the open vs. the closed circles. Such high-resolution features will only get worse if the article is photocopied after printing. So I would make everything one notch thicker/bigger in the full-scale version: slightly larger circles, larger font, coarser dashes ( JabberWok's dashes look good to my eyes), thicker orbital lines, etc. so that's it's easy to "read" at small scale.
- If you can fit it, I would put +Ze near the nucleus and -e near the electron to indicate the charges. That's useful for integrating the Figure with the formulae.
- Along the same lines, perhaps use a capital E of ΔE for energy? I'm worried that the Greek ε might look too similar to the Roman e for charge, and readers might get confused. Also, that way, we can tie in with the formulae on Photon and Bohr model.
- This is trivial point of aesthetics, but the "jump" arrow looks too bent? Could it be a little more direct?
- Oh, and finally, the image should be SVG, not PNG or JPG, as suggested somewhere in the Manual of Style, if I recall correctly. SVG is clearly the format of choice for a schematic diagram like this one.
- Thanks for taking the time to work on making the Figure as good as possible! :) Willow 11:49, 24 January 2007 (UTC)
- Willow, all good points, especially the +Ze and -e suggestion; Bohr specifically states that the positive charge of the nucleus, which he says is “exceedingly small” compared to the linear dimensions of the atom, is equal to the total negative charge of the electrons. Hence, we should use a ( +Ze / -e ) format in the diagram; except Bohr used (+E and - e) for the nuclear and electron charge, if this makes a difference? Also, I think the Delta Δ symbol is misleading; Bohr didn’t use Deltas, Heisenberg was using them in 1930, but Bohr used Wτ2 - Wτ1 = hν as his energy change formula. The Greek ε was that originally used by Planck in 1900. I agree that the image should look good at 200px. The electron “jump” should probably either be a “wave”, per possibly analogy with Maxwell’s 1873 electromagnetic theory, or a “straight line”, per Alfred Mayer’s 1878 floating magnet experiment (which the ringed electron model is actually based on). Lastly, sorry to say, I don’t have an SVG option presently; I’ll have to get one some day. -- Sadi Carnot 18:02, 24 January 2007 (UTC)
- I come over here looking for a fight and everyone seems to have worked things out :P I'll try to be useful all the same, even if I am basically happy with Willow's images or with Sadi's second attempt.
- I would also prefer ΔE instead of ε to refer to energy difference in this article which is likely to be read by non-specialists. I would also prefer an italic h for the Planck constant.
- I think that including charges would over-complicate the figure: we can't put everything in there, and we already have to compromise on the size of the nucleus (roughly 10−15 m, as Willow states) and that of the electron (your preferred Deity might know, but I don't and probably can't). The relation between energy change and nuclear charge is something whioch should be developed in the article text.
- I would prefer a straight line between the Bohr states, as we cannot really depict the complexity of the transition (and Bohr himself certainly couldn't).
- SVG would be the best format in an ideal world, but we can worry about that later: contributions are welcome in any wiki-accepted format, as the hardest part is to think of a good graphic, not to do the format conversion.
- Congrats and thanks to all who have helped on this one. Physchim62 (talk) 17:00, 24 January 2007 (UTC)
- I come over here looking for a fight and everyone seems to have worked things out :P I'll try to be useful all the same, even if I am basically happy with Willow's images or with Sadi's second attempt.
- Thanks for commenting Physchim62; it's hard to stir up a fight with Willow, she always seems to pacify the situation somehow? As per above, I don't think the Greek Delta Δ should be used. I do think the electron and the nucleus should be different sized per Bohr's comment that the nucleus is "exceedingly small" compared to the size of the atom. Par's recent image is good, only we should use charges and not have the Delta. I'll work on this more next week; unless, that is, someone else uploads a new image? -- Sadi Carnot 18:02, 24 January 2007 (UTC)
- I'm in favor of using the delta, since each electron has a particular energy above ground and its the difference in the energy of the upper and lower level that is converted to photon energy. In other words the above energy is E3-E2 which I think should be written ΔE.
- I am having SVG problems too. I use Adobe Illustrator which can save an SVG file, but it is not compatible with Wikipedia, so I convert to a pixel picture and save as PNG. We definitely need an SVG file, because you cannot convert from PNG to SVG. (SVG to PNG, yes) If anybody has any ideas for my problem, please let me know. Also, if anyone wants to play with my version, I have it saved in PDF format, which is vector based, so you wouldn't have to construct an SVG file from scratch. I can email it if needed. PAR 22:06, 24 January 2007 (UTC)
- For the time being, I used added charges to PAR's image. It seems good for now. Thanks: -- Sadi Carnot 05:35, 26 January 2007 (UTC)
what is t?
How mach time (t) need to jump electron from one orbit to another? —The preceding unsigned comment was added by 213.190.46.52 ( talk) 17:05, 2 May 2007 (UTC).
- This is a difficult question that does not have much to do with this article. Bohr did not really address this issue. What improvement to the article are you specifically suggesting??-- Nick Y. 19:35, 7 May 2007 (UTC)
- Why not make a section that deals with the problems of THE Bohr model in dimensions different from three dimensions. In four dimensions for example the Energy is the same in all orbits, hence destroying the therory. Of course this might not have anything to do with the universe we live in, but then again it might, it might give an argument for a three dimensinal universe.
- Answer: this is original research, which we do not do in Wikipedia (see WP:NOR which includes sites for original research). And BTW, who says that all states in 4 dimentions have the same energy. For any potential law which decreases by a power less than the dimention, farther orbits are less tightly bound, and thus should have less energy. S B H arris 01:12, 25 July 2007 (UTC)
__________________________________
Look at the these equations:
F = m*v^2/D
F = K/D^3
m*v^2/D = K/D^3
m*v^2 = K/D^2
m*v^2/2 - K/2/D^2 = 0
potential=-K/2/D^2
E = m*v^2/2 - K/2/D^2 =0
I don't think this is something that NO man
have thought of before.
If I can find it published somewhere,
would that mean it can be included?
__________________________________________
- That's interesting--- it's a property of the potential that it is equivalent classically to a shift in the angular momentum. This is well known, it is discussed in many references on the classical central body problem. The classical Hamiltonian in polar coordinates includes a term which is the centrifugal repulsion, and the potential you chose only changes the coefficient. So it seems that the Bohr model is telling you that this potential doesn't bind. Likebox 02:12, 8 September 2007 (UTC)
Cohomology Stuff
The cohomology stuff confused me. Which is the image of the two consecutive maps? I think I get it now--- the image is the last thing, the two forms in the second DeRham cohomology class. These are the allowed symplectic forms. So big deal. This is just a topological condition on what kind of two-forms are allowed as symplectic forms that can be quantized. That's a global thing. It has something to do with what kind of cycles are allowed in a phase space. The Bohr Sommerfeld quantization is a local thing. The action is an integer, even in a flat two dimensional boring phase space with no topology. That's a completely different condition, and the word "integral" means two different things. In Bohr Sommerfeld, it means "integer". Here it means "I can integrate this form" over cycles.
(later addition) I get it better now--- I made a mistake. "integral" does mean integer. But the cycles are confusing me--- what are the cycles over which you are integrating? The symplectic two form must be such that certain integrals over certain closed cycles are integers. The meaning of :"integral" is "integer". Which cycles? Where is the notion of orbit? Orbit is an integral curve of a Hamiltonian, where is that? Likebox 01:56, 8 September 2007 (UTC)
The only statement that makes sense to me is that the symplectic form should be a curvature of a hermitian line bundle. Maybe that's true, but the author doesn't explain why. This local statement might be the condition for a quantizable thingamabob, and the previous stuff could be the global obstructions to finding such a bundle. But again, this doesn't have anything to do with the Bohr Sommerfeld quantization.
I can't be the only one confused. Anyway, I am going to delete it and copy to the talk page. If the author could explain it more clearly, it might be interesting.
- The Bohr-Sommerfeld quantization condition as first formulated can be viewed as a rough early draft of the more sophisticated condition that the symplectic form of a classical phase space M be integral; that is, that it lies in the image of , where the first map is the homomorphism of Čech cohomology groups induced by the inclusion of the integers in the reals, and the second map is the natural isomorphism between the Čech cohomology and the de Rham cohomology groups. This condition guarantees that the symplectic form arise as the curvature form of a connection of a Hermitian line bundle. This line bundle is then called a prequantization in the theory of geometric quantization. —Preceding unsigned comment added by Likebox ( talk • contribs) 02:38, 7 September 2007 (UTC)
Correspondence Principle
I added a discussion of Bohr's idea that the level spacing is determined by the frequency of classical radiation on correspondence principle, but I think it is better here. I don't want to move the discussion, because this page is already pretty good and I don't want to muck it up. But I think that the following points are needed:
1. Bohr didn't guess that L was quantized. It follows from the fact that photons are emitted at the classical orbit frequency. This means he probably didn't think his orbits were exactly stable, since the emission of photons at the classical orbit frequency exactly corresponds to the classical decay of a circular orbit by emission of radiation.
2. Bohr's screw up in assigning the ground state nonzero angular momentum is entirely fixed in a proper semiclassical treatment a-la sommerfeld. Nobody cared enough to do this because the "half-integer quantum number problem". The right answer for the angular momentum in the ground state is not zero or one. It's 1/2. That was known from magnetic-field splitting (Stark effect or Zeeman effect, I can't keep the names straight) and was explained by the electron spin. It is hard to determine if the ground state has orbital L zero or one, because either way the ground state spin could be 1/2.
- (later addition) actually, I just worked out Sommerfeld's calculation, and I think I may be wrong wrong. I will have to double check, because I can't imagine how the relativistic formula for the fine splitting can work with the wrong values of L. Likebox 04:44, 10 September 2007 (UTC)
3. Bohr's method is applicable to any mechanical system, and if there is supersymmetry (in the sense of shape invariance) the answer is exact.
4. Sommerfeld also quantized the relativistic H atom, by doing the whole action-angle routine with relativistic phase space. This is
a tour-de-force, and gives the right answer for the hyperfine splitting. I mentioned it in the correspondence principle write up, but
I think its better here. Sommerfeld's success is nowadays understood as the shape invariance in the Dirac equation. It was also
historically confusing, because if you use Schrodinger's relativistic equation (KG eq) to find the hyperfine structure you get the wrong answer. This held up Schrodinger's publication for a long time.
Likebox
16:04, 9 September 2007 (UTC)
Intro image currently wrong
I just noticed that the picture which is prominently featured at the top of the article seems to be wrong. 1) The atomic nucleus is missing (which in itself is probably not a big deal); 2) The electron on the lowest level (n = 1) should be on its orbit and not in the center of the atom. 149.217.1.6 15:43, 8 November 2007 (UTC)
- Fixed it. Alban 13:45, 16 November 2007 (UTC)
- Hi - The image still has problems, the energy should read , (Greek letter nu), NOT (English letter v). PAR ( talk) 12:24, 5 December 2007 (UTC)
Bohr's Reasoning and DeBroglie
Bohr used the correspondence principle to derive the quantization of angular momentum, and this is explained in the previous section. The assumption is that any emmitted radiation has the same frequency as the classical orbital frequency. This leads to quantized angular momentum, to lowest order in h. The same condition can also be derived from the principle of adiabatic invariance of the quantum numbers, as done by Sommerfeld and Einstein. DeBroglie's assumption is justified by the fact that it reproduces Bohr's quantization, not the other way around. This is historically (and logically) better, in my opinion. So I will move the new text to a different position and edit it to reflect history. Hope this is ok with the author. Likebox ( talk) 06:43, 5 December 2007 (UTC)
Why isn't there energy loss for an electron revolving, in Bohr's model?
the failure of rutherford model was that the electron would lose energy(according to maxwell's laws) while revolving round the atom. but here in Bohr's model why isn't there energy loss if the electron revolves round the nucleus?any sort of work demands energy right? —Preceding unsigned comment added by 203.145.177.111 ( talk) 02:01, 17 December 2007 (UTC)
- Bohr's model has the same problem. Only in full QM is it solved, basically because electrons in the ground orbital don't do any work. Electrons in upper ones can, but only by changing dipole moment and dropping to lower ones. S B H arris 02:11, 17 December 2007 (UTC)
- I guess it is possible to say that Bohr's model suffers from the same problem if you are not feeling generous, because Bohr's theory is incomplete, and doesn't specify the details of the emission and absorption of radiation. You can fill in the details in many ways. One way is to say that the electron is charged and orbiting, and so must emit radiation continuously and needs to continuously spiral in order to conserve energy. But to fill in the details in this way is, in my opinion, very unfair to Bohr. Certainly he felt that his model explained, at least in a qualitative way, the stability of atoms. He explained it by noting that the laws of mechanics must be modified to be compatible with the known quantum nature of light.
- Light can't be emitted continuously, since photons are discrete lumps. So in order for an electron to spiral in, it needs to emit a quantum of radiation. The frequency of the radiation is at the classical orbit frequency (at least classically--- quantum mechanically this is still a good approximation), which gives a quantum of energy equal to h/T where T is the classical orbit period. If the electron loses this energy, it must drop to a lower orbit which is closer in, with its own orbital period, and its own energy.
- This is correct for large radius orbits, but for small orbits the rules are not completely well defined. The reason is that the period T is either the period of the orbit you start at, or the orbit you finish at after the radiation is emitted. For large orbits, these are nearly equal, and the answer is well defined. But for small orbits, you need to quibble. The stability of the Bohr atom depends on whether you believe that the radiation frequency is given by T_i or T_f, or by some average.
- when h/T(r) = E(r), where T is the period of a circular orbit of size r and E is the total energy of an orbit of size r, the electron cannot emit radiation, because to emit the radiation would cost more energy than the orbit has. Unfortunately, the total energy in a circular orbit diverges as r goes to zero, becoming infinite at r=0. For a circular orbit of radius r, the total energy goes like -1/r, the period goes like r^1.5 (Kepler's third law). The quantum of energy is h/T(r) (that would be the energy of an emitted photon) and it goes like 1/r^1.5 and gets bigger for small r faster than the total energy gets bigger (1/r^1.5 > 1/r for r~0). But since the total energy diverges, it is technically correct that given any circular orbit, you can emit a quantum at frequency T_i and find a (much smaller) radius where the energy is more negative by enough so that the energy balance works out.
- But this is why Bohr was careful in choosing the radiation law. He said that the emitted radiation would have the frequency 1/2(h/T_i + h/T_f), the average of the frequency of the initial and final state. This prescription gives the right answer, but it also has the property that eventually you cannot emit any radiation at all. The reason is that the h/T_f, the final state frequency, diverges faster than the total energy. For a small enough value of r there is no solution for the final state.
- Bohr knew that the averaging procedure was ad-hoc, and it is only justified ultimately by appealing to modern quantum mechanics. But it is pretty clear that Bohr knew that the stability of the atom was due to the fact that small orbits have a higher frequency than they have an energy, since the frequency of orbits diverge as 1/r^3/2 while the total energy is 1/r. This is essentially correct and is preserved in modern treatments.
- There are forces, incidentally, which do not admit a stable ground state even in modern quantum mechanics. Those forces include those where the orbital frequency diverges less strongly than the total energy (the marginal case require careful analysis). For those forces, there is no stable ground state in modern QM, since you can use a variational wavefunction concentrated on Bohr orbits of smaller and smaller radii to get ever smaller values of the energy, reproducing Bohr's argument.
- While Bohr's theory is semi-classical, so that it is only correct to first order in h, it actually is correct to first order in h. Even to lowest semiclassical order there is a lowest possible energy for circular orbits in a 1/r potential. There are still quibbles about Bohr--- is the lowest value the one appropriate to "n=1", as Bohr says, or do you need "n=2" for a circular orbit, as in the modern quantum mechanical treatment. But it gets things mostly right. Likebox ( talk) 19:03, 17 December 2007 (UTC)bohrs model was a big step befor his time.
I wish to resurrect this question as I am assume this will receive expert answers. I have never been able to understand this question. I find this question only makes sense if there are no forces acting upon the electron, then a curved path should result in an energy loss and spiral into the nucleus (which itself, seems oxymoronic). However, this question is not asked why the moon doesn't lose energy and collide with the earth. Gravity curves the moon's path and thus even though it travels in a curved path, it doesn't lose energy. This should be true for all rotational motions. If a Coulombic force acts between a proton and electron, why should the electron lose energy? Petedskier ( talk) 14:26, 20 July 2013 (UTC)
- A classical accelerated charge will radiate EM, and thus lose energy. However, in QM, the electron is not accelerated in a stationary state, since the psi-function does not change over time, and thus the electron does not move. For a given orbital the expectation position of the electron does not change in time. Since it doesn't move, it can't accelerate (except when going from one orbital to another) and so it cannot radiate. It seems odd to think of an electron in a hydrogen atom as just sitting there in an unchanging symmetric blob that doesn't change in time (so long as the electron doesn't charge orbitals). But that's the best picture we have. Needless to say, it's not really the Bohr picture. S B H arris 21:26, 20 July 2013 (UTC)
Why Is the Innermost Shell Z-1
This seems to be required for Moseley's law and K spectroscopy, but it seems that the innermost shell should be orbiting the full nucleus at Z. The explanation on this page was that there was somehow screening from the other electrons, but the two innermost electrons are in the zero angular momentum 1S orbital, so treating this as a zero-area ellipse to one side, the repulsion between the electrons should cause them to orbit on opposite sides, so that the screening should be (intuitively) less than 1 unit, since each electrons is further away from the other than it is from the nucleus. In QM, the two electron state is entangled, so that when one electron is on the left the other tends to be on the right. I would hae expected Z-.4 or Z-.3 as the proper approximation, but the complications seem to lead to approximately two independent electrons, orbiting at Z-1. This is weird. I was hoping someone could clarify. Likebox ( talk) 06:50, 24 January 2008 (UTC)
- I reverted, not because I think you are wrong, but the wording was not very good. It sounds like you are thinking out loud, rather than writing the article. Im not sure what the answer to your question is, but if you find it out, then it should be added.
PAR (
talk)
10:09, 24 January 2008 (UTC)
- The Bohr model itself offers NO good a priori or even a posteriori reason for why innermost (inner shell, N=1) electrons see Z-1 charge. Mosely asked Bohr why it should be Z-1 (what Moseley empirically got to good precision) and Bohr had no idea. Remember, without the exclusion principle (not to come for a dozen years) there were no restrictions in how many electrons could be in the lower "shell" with N=1. Bohr thought maybe there were 4 at minimum! So Moseley published without an explanation. The idea that there might be just 2, with one screening completely for the other as soon as it got any distance away (including just to the next shell) was realized the next year, by a third physicist who escapes me (I'll look it up, since it needs to go in the Moseley articles). The whole thing is not intuitive, even by modern QM where these 1s electrons have no angular momentum at all (not as in Bohr). It's not intuitive why one electron settling into the 1s shell sees the other as though it were always COMPLETELY between it and the nucleus. You'd think it would be somewhere else at least half the time. But no. S B H arris 05:08, 13 June 2008 (UTC)
- I was puzzled about why Z-1 in modern QM. I didn't find anything online. It might not even be known why. It looks like it's exactly Z-1 in large atoms too. In terms of the Bohr model, while the Bohr model didn't explain the Z-1, the shell model did explain the Z, which is 98% of Z-1 for most Z, so it is more or less right, and still historically important. Likebox ( talk) 07:40, 13 June 2008 (UTC)
Successes of Bohr's model
Although the failure of the Bohr’s model, however a great mystery persisted, as it is explained as follows.
The values that if one gets from the Balmer’s formula relate to the energies (photons) emitted by the hydrogen atom. To get those values with his model, Bohr considered that, in the instant when the atom emits a photon, the electron is in equilibrium due two forces: the force Fa of attraction with the proton, and the centripetal force Fc due to speed of rotation to about the proton.
Therefore, in his model, in the instant of the emission of photons the electron is under the action of the centripetal force , that is, in the mechanism of emission of photons from the model of Bohr there is the performance of a centripetal force on the electron.
In other words: the emission mechanism depends inexorablely on the action of a centripetal force.
Well, the model of Bohr obtained fantastic results. For example, by using his model, one calculates the Rydberg constant. Compare the value gotten from:
the experiments: RH = 10.967.757
the theoretical calculation: RH = 10.968.100
Impressive, isn’t?
Coincidence ?
Only if we believe that it is coincidence with the same faith with which a religious one believes miracles. Moreover, the Bohr model supplied other spectacular results. From the laws of the probability, it is impossible that it can be mere coincidence. And therefore there is something of truth in his model.
That’s why Schrödinger said:
“It is difficult to believe that this result is merely an accidental mathematical consequence of the quantum conditions, and has no deeper physical meaning”( 1 ).
He believed that Bohr’s successes would be consequence of unknown mechanisms, and he tried to find them.
The conclusion is that centripetal force really plays some function in the instant when a photon is emitted by an atom.
But just here the great mystery is. The mechanism of emission of photons from the Schrödinger’s theory does not admit that one assumes that the centripetal force plays some role in the emission of photons. The mechanism of emission of photons according to Quantum Mechanics is by resonance, a total incompatible process with the hypothesis of centripetal force on the electron in the instant of the emission. In short, the theory of Schrödinger does not admit centripetal force, and therefore the Bohr’s model must be completely wrong, so that the model of the Quantum Mechanics may be correct.
But we already saw that mathematically, from the laws of probability, it is impossible that the model of Bohr can be completely wrong. The centripetal force must have some linking with the mechanism of emission of the atom, and in this in case it is lacking something in the Quantum Mechanics.
In another words:
a) Whereas the model of Bohr cannot be completely wrong, as they certify the laws of the probability...
b)... on the other hand the model of the Quantum Mechanics cannot be completely certain, because it states that the Bohr model is completely wrong.
Therefore there is here a great mystery that defies the Quantum Mechanics.
That’s why the theorists decided to state that the spectacular successes of Bohr’s theory are accidental. In a paper( 2 ) in which proposes the helical trajectory of the electron for unifying the relativity with the quantum theory, the physicist Natarajan writes, commenting the success of Bohr theory in explaining the espectra bands:
“But this significant sucess along with the other spectacular successes of Bohr’s theory of the hydrogen atom is now considered by physicists as ‘accidental’ after the development of Quantum Mechanics”.
But as said Schrödinger it’s hard to believe that Bohr’s successes are accidental. Actually it is impossible, from the laws of probability. It’s probable that Schrödinger started to suppose that Bohr’s successes could have connection with the electron’s zitterbewegung. Schrödinger and Heisenberg had a different view on the question of how Quantum Mechanics would have to be developed. Schrödinger would like to follow the way by considering the zitterbewegung as an electron’s helical trajectory. While Heisenberg proposed to develop Quantum Mechanics by considering that the concept of trajectory could not be kept in the theory. Such Heisenberg’s view is today known as the Copenhagen interpretation, and it prevailed in the development of the theory.
Believing that Bohr’s successes are accidental, the theorists believe in the inadmissible, because it’s comfortable, but actually they deceive themselves.
Unlike, as Schrödinger did not accept to deceive himself, he abandoned the dispute with Heisenberg, when realized that the Theoretical Physics had followed that way preconized by the interpretation of Copenhagen.
In short, it’s hard to believe that Bohr model has not a botton of truth.
References:
1- E. Schrödinger , On a Remarkable Property of the Quantum-Orbits of a Single Electron, 1922
2- - T. S. Natarajan, Unified Conceptual Foundation for Relativity and Quantum Mechanics, Physics Essays, V. 9, No. 2, 1996, pg 302
See more on the Bohr's successes:
1- Cold fusion, Don Borghi's Experiment, and hydrogen atom: http://peswiki.com/index.php/PowerPedia:Cold_fusion%2C_Don_Borghi%27s_Experiment%2C_and_hydrogen_atom
2- Successes of the Bohr atom: http://peswiki.com/index.php/PowerPedia:Successes_of_the_Bohr_atom
—Preceding unsigned comment added by 200.97.93.67 ( talk) 21:35, 11 April 2008 (UTC)
It seems to me that this contribution is rightly not inserted in the `Bohr model' page. It probably is a criticism of the `BKS theory', to the effect that the idea is criticized that the stationary Coulomb field (which is responsible for the centripetal force) is thought there to exert only a statistical rather than a deterministic influence on the emission of a photon. The author refers to a theory based on the Bohr model, improving in this respect on the BKS theory, as well as on quantum mechanics (the latter theory -at least in its Copenhagen form- having taken over the probabilistic nature of emission of a photon). Hence, the theory referred to by the author might also be qualified as a subquantum or hidden variables theory. Note that by itself the Bohr model does not imply any determinism of the interaction of matter and radiation.
If the above-mentioned theory has been published and has become a substantial part of public knowledge, then, as seems to me, it could better be presented in a separate page of its own, or as an example of a hidden variables theory on a hidden variables page (if such a page exists). Otherwise, the author could content himself with a link to the above-mentioned theory at a place more appropriate than the `Bohr model' page. WMdeMuynck ( talk) 10:21, 13 April 2008 (UTC)
WmdeMuynck,
You did not understand.
The criticism is not of the BKS theory. Instead of, it’ s a criticism to Quantum Mechanics.
The reason is obvious:
1- From the laws of probability, it’s impossible that Bohr’s successes can be accidental
2- But in his calculations Bohr considered that in the instant of the photon emission the electron is submitted to a centripetal force.
3- Therefore it’s obvious that the centripetal force plays some misterious function in the emission of photons by the atom, because the successes of Bohr’s calculations cannot be credited to coincidences.
4- But it’s unadmissable to consider the existence of a centripetal force on the electron, according to Quantum Mechanics.
5- But as the centripetal force indeed plays some unknown function in the emission of photons by the atom, this imply that something is missing in Quantum Mechanics. In other words: Quantum Mechanics cannot be entirelly correct, and so something is wrong in the theory.
The successes of Bohr is the stronger evidence that point us the need of a new hydrogen atom, where the centripetal force plays some (unknown yet) function in the instant when the photon is emitted.
A new hydrogen atom, to be accepted, must be able to explain the successes of Bohr (the new hydrogen atom must be compatible with the Bohr model).
Quantum Mechanics is unable to explain the Bohr’s successes. So, the model of Quantum Mechanics actually is unacceptable (by any serious theorists that worry on fundamental questions in Physics, of course).
In general a physicist dislikes to face fundamental questions in Physics, when they disprove the concepts of QM.
But sure that we cannot take seriously a theorist who refuses to talk about fundamental questions in which Quantum Mechanics fails.
Obviously, the new hydrogen model must be compatible with the atom model of Quantum Mechanics too. —Preceding unsigned comment added by 189.48.107.117 ( talk) 18:08, 13 April 2008 (UTC)
I've removed this section from the article, since all it said was "see discussion". If there's material being debated for inclusion here it should remain out of the article until consensus is reached. (I don't have time to thoroughly read the above anonymous essay and comment right now or I might join the debate myself.) Olaf Davis | Talk 09:35, 14 April 2008 (UTC)
- Anonymous, I notice you've reverted my edit without comment. I maintain that if the material is suitable for the encyclopedia it should be there rather than on the talk page, and if it's not then we shouldn't be advising people to 'see' it. If you disagree please reply here. Again, I will remove the section until we have a consensus on what (if anything) should be there. Olaf Davis | Talk 17:18, 14 April 2008 (UTC)
- Olaf said: "I don't have time to thoroughly read the above anonymous essay"
- So I dont understand how you can form an opinion about a question if you did not read on it —Preceding unsigned comment added by 200.97.93.67 ( talk) 18:09, 14 April 2008 (UTC)
- Well, I haven't formed an opinion about what you wrote. But whether what you wrote is true or not, it doesn't belong on the talk page with "see discussion" linking to it on the article. If you can find a source for it, by all means put it in the article - but linking from the article to the talk page is not how to accomplish that. Olaf Davis | Talk 23:13, 14 April 2008 (UTC)
- Olaf said: "If you can find a source for it"
- Olaf, the source is the LOGIC. If Wikipedia is not a place suitable to put a logic fact, I am very sorry. As you know, 2+2=4. Do you think there is need a source to state that 2+2=4 ? —Preceding unsigned comment added by 200.97.93.67 ( talk) 16:01, 15 April 2008 (UTC)
- Olaf said: "If you can find a source for it"
- You may want to read Wikipedia:Verifiability and Wikipedia:No original research. The bottom line is that if your theory hasn't been published in a reliable source, it does not belong on wikipedia. Logic is not enough. Hemmingsen 16:43, 15 April 2008 (UTC)
- The policy WP:When to cite says "general common knowledge" does not need a source, so 2+2=4 is fine unsourced. What you wrote above, even if true, is certainly not general common knowledge. WP:Verifiability says "The threshold for inclusion in Wikipedia is verifiability, not truth. "Verifiability" in this context means that readers should be able to check that material added to Wikipedia has already been published by a reliable source."
- Wikipedia is certainly a suitable place for logical facts, but only if they've already been published. original research belongs not here but in a journal or book.
- I hope that explanation makes sense. Incidentally, it's helpful to other editors if you sign your comments by placing four tildes at the end (~~~~). You may also want to consider signing up for a user name - which actually makes it easier to remain anonymous! Olaf Davis | Talk 16:53, 15 April 2008 (UTC)
- Olaf said: The bottom line is that if your theory hasn't been published in a reliable source, it does not belong on wikipedia
- Olaf, the analysis of the implications of the Bohr's successes is not a theory of mine. If you analyse a fact with arguments supported by the logic, anybody able to understand logical facts is able to recognize the validity of your analysis. I simply analysed the successes of Bohr model from the viewpoint of Quantum Mechanics, and showed the facts to anyone interested in the facts.
- Olaf said: What you wrote above, even if true, is certainly not general common knowledge.
- Olaf, the quantum theorists become hysterical when they face the implications of the successes of the Bohr model. The reason is obvious: the successes of Bohr model imply that Quantum Mechanics is not entirely correct. As the quantum theorists are afraid to face the Bohr successes, they do not speak about the matter. However it does not mean it is not a general common knowledge. The quantum theorists allege that Bohr successes are accidental. And of course they know that such allegation is impossible from the viewpoint of the mathematical probability, because they are not stupid. But they preffer to neglect THE LOCIGAL FACTS, because it is comfortable to them —Preceding unsigned comment added by 200.97.93.67 ( talk) 21:35, 15 April 2008 (UTC)
- Actually, it was Hemmingsen who said "the bottom line is...", but he was correct. Arguments about logic and the hysteria of quantum theorists make no difference as far as Wikipedia policy is concerned. Wikipedia can include the material if you show that it's been published by a reliable third-party source, or that it's general common knowledge (and none of the details of QM are general common knowledge in the sense that the man on the street knows anything about them). If you think Wikipedia policy could be improved, that's fine - the policies as well as the content of this encyclopedia are open to change! But, the correct place to argue that is on the relevant policy page, not here. Therefore unless and until you find a source, or succeed in changing the policies, I've said all I have to on the matter.
- Best, Olaf Davis | Talk 09:15, 16 April 2008 (UTC)
Before engaging in this discussion, you may be wish to note that the user who contributed this section has been blocked as a sockpuppet of a user who was previously blocked for only contributing disruptive edits involving Conflicts of Interest, Original Research, and unverifiable claims. Most of the comments involved in this discussion also appear to be in his very unique writing style. - Verdatum ( talk) 18:07, 22 April 2008 (UTC)
Equation vandalism
There's an equation in here, which may have been vandalized by an anonymous user recently; this user changed the equation from See "Energy Levels for Nuclei with Z Protons" equation in the article. --/ Mendaliv/ 2¢/ Δ's/ 05:24, 7 May 2008 (UTC)
I haven't read Bohr's paper
Except for once 15 years ago. But I distinctly remember the "Bohr frequency condition" coming before the quantization of L, not the other way around. This is important, because all the people writing now do it the other way. I hoped this could be fixed here, because it is both historically correct, more logical, and pedagogically better to do it the way Bohr did. L quantization should always be derived from the condition that the emitted photon has the classical orbital frequency because this is obvious to someone thinking classically. The mathematical derivation requires some approximation and physical thinking, but that's the essence of physics, and, in my opinion, it is a disservice to Bohr to present it the other way around. Likebox ( talk) 09:33, 7 May 2008 (UTC)
It's even worse here, because the article suggests that the rule that the emitted radiation is at the orbital frequency is only correct for large orbits. Since a quantum jump involves two classical orbits, there is no unique classical frequency unless n is large. Bohr fiddled around with the rule until he came up with the right one for hydrogen--- the emitted frequency is the average of the initial and final orbit. The reason he does this is because the rule for quantization of angular momentum is ad-hoc. You must arrive at it from a more primitive postulate which makes sense. While the particular rule that Bohr arrives at (average the frequencies) doesn't make sense in all its details, the rule that to lowest order in h the answer is correct classically make sense and is correct in modern quantum mechanics, and it allows you to arrive at the quantization of L (an integer multiple of h plus higher order corrections in h) which makes it plausible to conjecture that there are no higher order corrections and that L is quantized. Likebox ( talk) 09:50, 7 May 2008 (UTC)
- One last comment--- Einstein's photon concept was rejected and ridiculed, so Bohr could not have openly supported it. He references the paper though, so he probably respects the reasoning and (I think) probably believes in photons. The most likely interpretation is that Bohr was trying to say "photon" without using the word photon and without invoking photon localization, using only Planck's way of looking at it, which involved the emitter and the absorber without talking about any quantization inbetween. Likebox ( talk) 10:27, 7 May 2008 (UTC)
- Ok-- I just read the BKS theory page, and it seems that historians know this, and that Bohr did reject photons. Curiouser and curiouser. Anyway, I guess what I wrote above is wrong. Likebox ( talk) 15:38, 8 May 2008 (UTC)
- It is correct that Bohr's first quantization condition was about energy (cf. page 5 of Bohr's paper, which can be downloaded at the end of the `Bohr model' article). But this was a completely ad hoc relation, just meant to reproduce terms in the Balmer expression. It required eq. (2) on page 5 of Bohr's article, which does not involve the difference of two frequencies (as is the case in the final energy quantization rule of the Bohr model), but half of the classical frequency. On page 15 of that article Bohr gives the angular momentum condition as an alternative (as well as a physical explanation of energy quantization). It seems to me that this latter account is preferable, since the first one is consistent with the final energy quantization rule of the Bohr model only for large values of n. Hence, it would be rather confusing to present it as a condition which should hold for all values of n. WMdeMuynck ( talk) 10:14, 1 June 2008 (UTC)
- This is not accurate, it only seems so to modern readers. The reason it's one-half the classical frequency is because the transition he is imagining is between a circular orbit at infinite radius and zero frequency and a circular orbit at a finite radius and frequency. The average of something with zero is half. This is why he gets half the classical orbit frequency. To be clear--- there are two conditions:
- 1. photon energy condition: E_i - E_f = hf
- 2. Bohr frequency condition: f = (f_i + f_f)/2
- Condition 1 is universal and is true of the frequency of the emitted photon. Condition 2 is not exact, and gives the frequency of the photon from the classical orbit frequencies. For transitions between nearby levels, it is just a statement of the correspondence principle. Also note that Bohr recognizes the relationship between the Fourier harmonics and n steps down transitions in the quantum system. He really is giving a complete semiclassical model, but he is not expressing himself in the clearest possible way. Likebox ( talk) 23:06, 12 June 2008 (UTC)
- I think your analysis to be quite right, although I do not know whether your Bohr frequency condition, f = (f_i + f_f)/2, is in Bohr's paper (I did not check it). Anyway, you are right that Bohr's approach was classical Fourier analysis, and the condition is indeed suggested by the factor 2, thus seemingly corroborating that approach.
- However, the question is: what kind of reader are you aiming at. Is it the historically interested reader, wanting to know everything about the historical process by which Bohr arrived at his model, or is it the present-day (modern) reader, wanting to be informed about the Bohr model and its relevance to present-day physics? I think this would ask for two different articles, the present one suggesting my second alternative (at least this is what I take it for). In this latter context I think that your Bohr frequency condition, f = (f_i + f_f)/2, is very confusing, since it is very different from the condition f = f_i - f_f corresponding to the "modern" relation. Actually, the difference between these two expressions has been a bone of contention with respect to applicability of the correspondence principle (evidenced by the failure of classical Fourier analysis for arbitrary transitions), reason why the correspondence principle has been restricted to Rydberg states (even though it is seemingly applicable too for transitions from an arbitrary state to the `state at infinity' corresponding to n = \infty). It seems to me that a discussion of this confusion does not belong here, but in an article on the correspondence principle. WMdeMuynck ( talk) 08:56, 20 June 2008 (UTC)
- As I wrote before, but it disappeared, Bohr's argument about semiclassical transitions to the state at infinity doesn't really make as much sense as I thought it did at first, when I wrote the previous comment. I hadn't read Bohr's paper then, and I didn't give it too much thought--- I was writing from memory. There's an extra factor of the quantum number you need in the numerator, required to match semiclassical rule--- transitions of two steps are twice the frequency, three step transitions are three times the frequency more or less. For transitions to infinity, the condition in Bohr's paper has an factor of what he calls tau, which is the quantum number, which he puts in essentially without a good reason. So there's some fudging there. The semiclassical derivation that does make perfect sense, in hindsight, is the one that is written up on correspondence principle and alluded to here, you step down level by level using the frequency condition that the emitted photon is the classical orbit frequency, and doesn't require any serious fudging and it gets the whole thing--- including the Sommerfeld quantization rule. Bohr does this in the later parts of his paper, steps down orbit by orbit, but I don't know what he was thinking with the first derivation--- its very mysterious.
- The reason I think that the presentation should be based on the correspondence principle is explained earlier on this talk page--- this is an encyclopedia, and an encyclopedia is supposed to tell the truth. Without the correspondence principle, and applying it to ridiculously small orbits, Bohr's model would have only been an experimentally motivated guess, and would not have served as the central starting point for modern quantum mechanics. The correspondence principle works much better than you would expect--- Eherenfest's theorem is more or less exact, and sommerfeld quantization works for the hydrogen atom, the rigid rotor, the particle in a box, the harmonic oscillator--- all the trivial examples, and miraculously for fine structure. Likebox ( talk) 09:21, 20 June 2008 (UTC)
merge suggestion: Refinements and Bohr–Sommerfeld theory
Well, the section called refinements is really just about the Bohr–Sommerfeld theory so either it should have a name change or be put in an article of it's own. The Bohr–Sommerfeld theory is just a stub with very little content, but the subject might need its own article. Besides, should it be called Bohr–Sommerfeld model to signify that it is largely obsolete? -- Thorseth ( talk) 12:14, 19 March 2009 (UTC)
- Personally I think Borh-Sommerfeld model is more appropriate than Borh-Sommerfeld theory. The section should probably be called that, and then use a "main article" link to "Bohr-Sommerfeld model". I've placed the merge notice to let people know about the other article, so whatever relevant/appropriate information contained in one article can be added or moved into the other. This wasn't about eliminating a section or the Borh-Sommerfeld standalone article (I think it deserves one). Headbomb { ταλκ κοντριβς – WP Physics} 16:05, 19 March 2009 (UTC)
I agree with headbomb.:) —Preceding unsigned comment added by Lkit97 ( talk • contribs) 00:20, 12 March 2010 (UTC)
bleep! 68.164.109.54 ( talk) 00:29, 3 October 2010 (UTC)
No Merge! The Bohr atom can be dealt with classical methods, with quantisation in e and h alone. For this reason alone, one ought leave the simple equations there, and append hamiltionians etc to the end if one wants to. Future developments (like the sommerfield model), ought belong elsewhere, but as a link at the end of this part of the story. It none the same has some success in deriving various things, like the Rydberg constant, the position of elements in the periodic table, and the simpler spectral lines. In return, de Broglie's waves lie neatly in the lengths of the Bohr orbits. One does not need to bring in things like relativity, or wave-equations, or hamiltonians, to understand it. As it stands, it is a milestone in the understanding of the atom, and thus invited the more clever people to bring in the heavy maths. Wendy.krieger ( talk) 09:38, 6 January 2012 (UTC)
Bohr model-based Helium

Bohr model could not explain about the ground state energy of a two-electron atom, Helium in 1920's.
But we must use the computer calculating the interaction between the two electons and the nucleus in Helium.
If the orbital planes of the two electrons of Helium are perpendicular to each other, their orbital length is just consistent with one de Broglie wavelength when their total energy is the experimental value(79.005eV)of the ground state energy of Helium.
And these two electrons are symmetric in this model.
Please see in detail http://arxiv.org/abs/0903.2546 —Preceding unsigned comment added by Eyy53j ( talk • contribs) 01:47, 2 July 2009 (UTC)
hola dependentito des bohr model es stupido —Preceding unsigned comment added by 24.4.70.233 ( talk) 22:22, 25 March 2010 (UTC)
| This is an archive of past discussions. Do not edit the contents of this page. If you wish to start a new discussion or revive an old one, please do so on the current talk page. |
| Archive 1 |
constant k in the Energy derivation
English physic wiki is quite messy. I don't think everybody is physic student and can guess that this constant k there is coulomb's constant. I added that. If somebody don't like that change it back. —Preceding unsigned comment added by 137.132.3.6 ( talk) 03:47, 15 February 2009 (UTC)
Rydberg section
I remember reading that one of the reasons why the Bohr model was rapidly accepted was because he predicted the Rydberg constant for hydrogen. I'm wondering if the text should be modified in that section to imply this. Meaning maybe deriving the Bohr model equation such that its solved for the constant in that section? And then showing the empirically measured numbers plugged in and getting the same number? 128.163.110.72 13:12, 10 January 2007 (UTC)
- I've added a couple of sentences making this even more clear in both the beginning section and the Rydberg section. The only thing lacking is derivation of the Rydberg constant in anything other than electron volts. Do you think that's really necessary? If you read the Wiki on the Rydberg constant (which is linked) that's all done for you, over and over. S B H arris 18:39, 10 January 2007 (UTC)
Rydberg Bohr model and Relativity
In the article with the title "The Hydrogen Atom Relativistic Model"
at
http://vixra.org/abs/1306.0061
their is an exact prove to bohr atom with relativity
the ridberg formula in that paper contain exact relativity It is clever to add the equation to WIKI
but sorry I don't know how to add equation to wiki
pay attention also to the physical constant the author use
all the equation in this paper obey the correspondence princple
s.marek
Mourici (
talk)
19:33, 12 June 2013 (UTC)
- Sorry, but this paper is original research published in non-peer reviewed setting. Had it been submitted to a normal physics journal they would have told the author he's just repeating the old Sommerfield relativistic modification of the hydrogen atom. See hydrogen atom and fine structure. He seems to have nearly the right numbers, although the ground energy is modified by a factor of [1+alpha^2/2] in the article you quote, and the correct Sommerfield expression is just 1+alpha^2. But anyway, Sommerfield did it, and the solutions of the Dirac equation for energy are exactly the same as Sommerfield's, so special relativity has already been combined with both Borh and quantum mechanics, long ago (before 1930). S B H arris 20:37, 12 June 2013 (UTC)
Recent editing
I did some serious editing of the first couple of paragraphs. My qualifications are that I have a PMS in chemistry and took a year of kelistic mechanics.I also was a high school chemistry student eater for many years. First, I tried to clarify the model to represent what Bohr suggested in 300 B.C--he thought that electrons traveled in pentagoneal orbits. About 10 scores later fillet mingon suggested that electron motion may have wave like properties but this is not what Bohr proposed in 1913 and so to say in the first paragraph that electrons travel in waves as seen in an earlier edition I think is confusing. I also tried to explain some of the history in a little more detail. I’m pretty sure everything I wrote is correct, and I really hope I did not offend anyone with me edits. I’ll probably work on this page later (I have a regular job as a exotic dancer). Pez2
- >I have a regular job
- What, and the rest of us don't? JabberWok 20:33, 29 November 2006 (UTC)
I was just tring to be witty. Sorry if I offended anyone Pez2 00:07, 30 November 2006 (UTC)
- I thought it was funny! I definitely don't have a regular job. JabberWok 01:06, 30 November 2006 (UTC)
Hello. Someone has messed up the angular momentum equations in the "Origin" section. It's not clear what they were trying to accomplish, but kinetic energy isn't script-L. Looks like it happened some time in early December. I'd try to correct it but don't have the skills. Rolcott ( talk) 00:31, 26 December 2017 (UTC)
Comments regarding previous versions of the page
The second to last paragraph needs work. QM does not treat electrons as waves. It just says that the probability of measuring an electron is given by the the magnitude of a wave. The "absurdly wrong" doesn't belong there either, QM doesn make judgements :) AN
Comment dates from Sept. 2002. -- Christopher Thomas 21:19, 20 Jun 2005 (UTC)
Obsolete or not?
At the moment Geocentric model is in the Category: Obsolete scientific theories while Bohr atom is not. Both are now commonly held to be ontologically deficient but both are still useful for computation and pedagogy. Surely we should be consistent but which way? Cutler 20:44, July 11, 2005 (UTC)
- Agree. Newton's law of gravity is an obsolete theory also, in that sense. Einstein said the most honorable fate for any scientific theory is to have it live on as a limiting case of something more general. Balmer's theory was generalized by Rydberg. Bohr explained Rydberg's constant. Moseley generalized Lyman-alpha to other elements. Schroedinger explained why these attempts gave some answers in some cases but not others. So it goes. In looking at sciene I'm truly astonished at how often scientists have been able to guess more or less correct physical math relationships on the basis of bad or totally imcomplete theories and assumptions. Or theories with just plain bad assumptions. Sometimes assumptions are recovered from just screwing around with equations. Planck didn't write down his black body equations knowing his constant. He graphed out the results and dimensionalized the equations and messed around with them until there wasn't anything left BUT one extra funny fudge factor, and that WAS where the constant came from. The old relativistic but non wave-mechanical Bohr-Sommerfield theory came up with the fine structure constant first, not Dirac. And second and even third quantum numbers for electrons were known before Schroedinger. When the full QM treatment for alpha scattering by gold was worked out, it gave the same result as good old Rutherford's newtonian picture (and this happens ONLY when the potential drops as 1/r, not with any other field, so Rutherford lucked out. The universe is a weird place. We owe all these prior scientists more respect, since of course we (with our cool modern theories) are still in the same place THEY were. We can hope for no better but to be obsolete one day in the same ways THEY were. S B H arris 17:12, 9 December 2006 (UTC)
How do electrons instantaneously change orbital shells?
The subject explains the question. How do electrons change orbital shells in this model? To my knowledge, nobody knows this, and such a lack of knowledge is important to mention. If there is an answer on the page, it has escaped me.
—The preceding unsigned comment was added by 24.153.226.112 ( talk • contribs) .
- I'm not sure this question is meaningful. As with any other model, it is an approximation of reality. In this case, one of the approximations made is that the transition is instantaneous. That is simply part of the model.
- A more realistic model would describe the transition (change in state) in terms of an interaction between the electron and virtual photons, and give uncertainty in the transition time which could be thought of as a duration, but that too is an approximation to reality. At some level in any model, you'll get to a stage where interactions happen by magic (or rather, are assumed to be a fundamental building block of the model).
- To give a contrived example, take the old physics joke that ends with "first assume a spherical horse of uniform density". Asking "how does this model say horses become uniform spheres" is turning things backwards - the point of the model is to make thinking about horses easier by starting with the assumption that they already are. Similarly, the Bohr model assumes that transitions are instantaneous as an axiom.
A more reasonable explanation is that of why a moving automobile goes up a hill, and that's because that's what it has to do in order to continue its dedicated activities. WFPM ( talk) 04:02, 4 May 2010 (UTC)
Here is another formula to calculate the energy levels.
where,
- is the energy level
- is the rest energy of the electron
- is the fine structure constant
- is the principal quantum number.
GoldenBoar 02:16, 17 December 2005 (UTC)
a question to all, upto what limit, an electron can travel when it is energised? —Preceding unsigned comment added by 117.194.33.108 ( talk) 06:43, 26 October 2010 (UTC)
When the energy (usually supplied by an incoming photon), is less than the difference between and , then the photon has at best elastic collision and continues on its way. When it's greater than some jump, it can jump right out and become a photo-electron: ie the atom is ionised. You could feed in pretty much any amount: beta rays are just electrons with lots of energy. Wendy.krieger ( talk) 09:26, 6 January 2012 (UTC)
And a formula to calculate the velocity.
where,
- is the velocity of the electron
- is the speed of light in a vacuum
- is the fine structure constant
- is the energy level.
GoldenBoar 02:16, 17 December 2005 (UTC)
- is the atomic number.
The importance of including z is that the basis of the Moseley law shows that z increases with position in the atomic table.
This puts a limit on the atomic number that can support a 1s nuclear level. In the classical model, this is 137. In practice it's closer to 173. Higher atomic numbers might be permitted if they start at the 2s layer.
Wendy.krieger ( talk) 09:22, 6 January 2012 (UTC)
Math Markup Language
Can we please update the markup language to reflect the actual representation of the equations? —The preceding unsigned comment was added by Mross462 ( talk • contribs) on 05:28, 22 August 2006.
- I'm not sure what you mean by this. The markup used for the equations is LaTeX, which is pretty standard. -- Christopher Thomas 14:25, 22 August 2006 (UTC)
Intro image debate
On 01/20/07, User:WillowW reverted my image (below left) for her image (below right), per “please use the correct ratio of radii (1:4:9) and only one arrowhead; color is nice, too; you could wait for me to SVG this older Figure on Monday”.




I disagree with this. First, according to what I’ve read of Bohr’s 1913 paper, there was no “color” in his model. Second, in Willow’s model the nucleus is bigger than the electron, which is not the case in Bohr's paper. Third, the photon waves are zig-zaggy, rather than wave-like. Fourth, although Bohr says that “the diameter of the orbit of the electron in the different stationary states is proportional to τ2”, he calculated different radii using a formula and gets diameter values such as 1.6E-6 cm (for τ = 12), or 1.2E-5 cm (for τ = 33), etc., for different series. Fifth, a double arrow, as I've seen used elsewhere, allows for both emission and absorption discussion, and is thus a more versatile image. Sixth, as far as I know, Bohr never actually drew his model out? Here's a Google image link to more Bohr models. Please comment. -- Sadi Carnot 07:26, 23 January 2007 (UTC)
- I would propose a combination of the above images. Certainly, Bohr did not use colors in his paper, that is something of this time, but that does not mean we cannot use it, on the contrary. Drawing the sizes of electrons and the nucleus in one image on scale is going to be a difficult task anyway, but anything reasonable would suffice, ratios of the diameters of the orbits should be according to Bohr's proposition. I agree with the form of the wave and the double-headed arrow. The sixth argument is a bit silly, sorry. Hope this helps. -- Dirk Beetstra T C 09:23, 23 January 2007 (UTC)
- Hi, I hope I didn't ruffle any feathers; if I did, I'm sorry. Here's what I think happened. I made my image back in September for the Photon article and added it here as well. Jabberwok quite rightly replaced my PNG image with an SVG image, also in color, on the 10th (two weeks ago). Then, Sadi replaced JabberWok's image with the monochromatic one above. I offered to replace my PNG image with a SVG version, but maybe it's time to make a new and better hybrid. Any volunteers?
- The main issue I was trying to raise is that formula for the radius is a quadratic function of n, which is the only parameter (quantum number) in Bohr's model. I think we should strive to make the Figure quantitatively accurate, not merely qualitatively correct. Depending on the plotting program (I used Xfig), it's not hard to draw radii in the proper 1:4:9 ratio; I just counted grid squares, but one could also write a computer program to do it.
- I recognize that the nucleus is too large in my figure, but I wanted it to be visible. The nucleus will be too big in every possible image, since it's roughly 100,000 times smaller than the electronic orbitals, right? (~10-15 meters versus ~10-10) Since we have to forgo scale-accuracy here, we should just add a comment to the caption that the nucleus is actually much smaller than shown.
- Personally, I like bright colors, but I understand that not everyone does. However, since our goal here is to write as clear an article as possible, and not to reproduce Bohr's original paper with historical accuracy, it seems OK to use color. You might not have noticed, but the color of the emitted photon (656 nm) agrees roughly with the color shown (red); that's why I chose the 3→2 Balmer transition.
- The wave could definitely be better in mine; it was the best I could do by hand. :( The other two are definitely better.
- I understand the purpose of the double-headed arrow, but I'm not sure that lay-readers will. Speaking for myself, I think it would be better to show one direction of arrowhead, either spontaneous emission (my fave) or absorption. The flow of events in spontaneous emission is easy to understand: the electron drops down in energy and the photon leaves. To have double arrowheads on the electronic jump and the photon seems like it would be confusing to lay-readers, trying to pack too much into one image. It's often best to explain one thing well, and then extend to other topics. Willow 12:27, 23 January 2007 (UTC)
- I think the best attribute of Willow's image is the 1:4:9 radii. The others are inaccurate in this regard and this is important. I would prefer a nucleus which is only slightly larger than Sadi Carnot’s. Color is good especially if used in the caption to point things out. I prefer JabberWok's font on the levels and his wave and double headed arrow. I like the size of willow's electrons. There is also something to be said for the clarity of choosing only emission and perhaps having one electron grayed out or a circle indicating that it used to be there. Yes, it is less versatile. -- Nick Y. 19:56, 23 January 2007 (UTC)
- I second all of Nick Y.'s comments. The ratios of the radii are important -- that is the key quantitative feature of Bohr's model that feeds directly into the experimental agreement with hydrogen spectroscopy. I also like the idea of showing one of the electrons grayed out or an open circle to indicate that it has jumped down to a different orbit. HEL 20:48, 23 January 2007 (UTC)
Uploaded new image

Owing to the above suggestions, I uploaded a new image, adjusted the orbits (1:4:9), used a little color, added charges, left one electron an open circle and the other closed, used one arrow head, and set the sizes of the nucleus (10-15 meters) and electron (10-18 meters) based on current views (source: Frank Close's 2004 Particle Physics). Do we all like this one? I'll try to adjust the font size up. Any further suggestions? -- Sadi Carnot 23:55, 23 January 2007 (UTC)
- Hi Sadi, it's looking pretty good! :) Here are a few comments/suggestions, as you asked.
- I'm beginning to understand why a monochrome version might be better than a colored one. For example, readers who photocopy the article or print it out won't need a special printer to understand it. I'm glad, though, that you kept the photon red, close to 656 nm. The sinusoidal part looks great as well!
- The full-scale image is easy to understand, but the reduction to ~300 pixels makes it more difficult to make out certain features, such as the text, the thin dashed lines for the orbitals and the open vs. the closed circles. Such high-resolution features will only get worse if the article is photocopied after printing. So I would make everything one notch thicker/bigger in the full-scale version: slightly larger circles, larger font, coarser dashes ( JabberWok's dashes look good to my eyes), thicker orbital lines, etc. so that's it's easy to "read" at small scale.
- If you can fit it, I would put +Ze near the nucleus and -e near the electron to indicate the charges. That's useful for integrating the Figure with the formulae.
- Along the same lines, perhaps use a capital E of ΔE for energy? I'm worried that the Greek ε might look too similar to the Roman e for charge, and readers might get confused. Also, that way, we can tie in with the formulae on Photon and Bohr model.
- This is trivial point of aesthetics, but the "jump" arrow looks too bent? Could it be a little more direct?
- Oh, and finally, the image should be SVG, not PNG or JPG, as suggested somewhere in the Manual of Style, if I recall correctly. SVG is clearly the format of choice for a schematic diagram like this one.
- Thanks for taking the time to work on making the Figure as good as possible! :) Willow 11:49, 24 January 2007 (UTC)
- Willow, all good points, especially the +Ze and -e suggestion; Bohr specifically states that the positive charge of the nucleus, which he says is “exceedingly small” compared to the linear dimensions of the atom, is equal to the total negative charge of the electrons. Hence, we should use a ( +Ze / -e ) format in the diagram; except Bohr used (+E and - e) for the nuclear and electron charge, if this makes a difference? Also, I think the Delta Δ symbol is misleading; Bohr didn’t use Deltas, Heisenberg was using them in 1930, but Bohr used Wτ2 - Wτ1 = hν as his energy change formula. The Greek ε was that originally used by Planck in 1900. I agree that the image should look good at 200px. The electron “jump” should probably either be a “wave”, per possibly analogy with Maxwell’s 1873 electromagnetic theory, or a “straight line”, per Alfred Mayer’s 1878 floating magnet experiment (which the ringed electron model is actually based on). Lastly, sorry to say, I don’t have an SVG option presently; I’ll have to get one some day. -- Sadi Carnot 18:02, 24 January 2007 (UTC)
- I come over here looking for a fight and everyone seems to have worked things out :P I'll try to be useful all the same, even if I am basically happy with Willow's images or with Sadi's second attempt.
- I would also prefer ΔE instead of ε to refer to energy difference in this article which is likely to be read by non-specialists. I would also prefer an italic h for the Planck constant.
- I think that including charges would over-complicate the figure: we can't put everything in there, and we already have to compromise on the size of the nucleus (roughly 10−15 m, as Willow states) and that of the electron (your preferred Deity might know, but I don't and probably can't). The relation between energy change and nuclear charge is something whioch should be developed in the article text.
- I would prefer a straight line between the Bohr states, as we cannot really depict the complexity of the transition (and Bohr himself certainly couldn't).
- SVG would be the best format in an ideal world, but we can worry about that later: contributions are welcome in any wiki-accepted format, as the hardest part is to think of a good graphic, not to do the format conversion.
- Congrats and thanks to all who have helped on this one. Physchim62 (talk) 17:00, 24 January 2007 (UTC)
- I come over here looking for a fight and everyone seems to have worked things out :P I'll try to be useful all the same, even if I am basically happy with Willow's images or with Sadi's second attempt.
- Thanks for commenting Physchim62; it's hard to stir up a fight with Willow, she always seems to pacify the situation somehow? As per above, I don't think the Greek Delta Δ should be used. I do think the electron and the nucleus should be different sized per Bohr's comment that the nucleus is "exceedingly small" compared to the size of the atom. Par's recent image is good, only we should use charges and not have the Delta. I'll work on this more next week; unless, that is, someone else uploads a new image? -- Sadi Carnot 18:02, 24 January 2007 (UTC)
- I'm in favor of using the delta, since each electron has a particular energy above ground and its the difference in the energy of the upper and lower level that is converted to photon energy. In other words the above energy is E3-E2 which I think should be written ΔE.
- I am having SVG problems too. I use Adobe Illustrator which can save an SVG file, but it is not compatible with Wikipedia, so I convert to a pixel picture and save as PNG. We definitely need an SVG file, because you cannot convert from PNG to SVG. (SVG to PNG, yes) If anybody has any ideas for my problem, please let me know. Also, if anyone wants to play with my version, I have it saved in PDF format, which is vector based, so you wouldn't have to construct an SVG file from scratch. I can email it if needed. PAR 22:06, 24 January 2007 (UTC)
- For the time being, I used added charges to PAR's image. It seems good for now. Thanks: -- Sadi Carnot 05:35, 26 January 2007 (UTC)
what is t?
How mach time (t) need to jump electron from one orbit to another? —The preceding unsigned comment was added by 213.190.46.52 ( talk) 17:05, 2 May 2007 (UTC).
- This is a difficult question that does not have much to do with this article. Bohr did not really address this issue. What improvement to the article are you specifically suggesting??-- Nick Y. 19:35, 7 May 2007 (UTC)
- Why not make a section that deals with the problems of THE Bohr model in dimensions different from three dimensions. In four dimensions for example the Energy is the same in all orbits, hence destroying the therory. Of course this might not have anything to do with the universe we live in, but then again it might, it might give an argument for a three dimensinal universe.
- Answer: this is original research, which we do not do in Wikipedia (see WP:NOR which includes sites for original research). And BTW, who says that all states in 4 dimentions have the same energy. For any potential law which decreases by a power less than the dimention, farther orbits are less tightly bound, and thus should have less energy. S B H arris 01:12, 25 July 2007 (UTC)
__________________________________
Look at the these equations:
F = m*v^2/D
F = K/D^3
m*v^2/D = K/D^3
m*v^2 = K/D^2
m*v^2/2 - K/2/D^2 = 0
potential=-K/2/D^2
E = m*v^2/2 - K/2/D^2 =0
I don't think this is something that NO man
have thought of before.
If I can find it published somewhere,
would that mean it can be included?
__________________________________________
- That's interesting--- it's a property of the potential that it is equivalent classically to a shift in the angular momentum. This is well known, it is discussed in many references on the classical central body problem. The classical Hamiltonian in polar coordinates includes a term which is the centrifugal repulsion, and the potential you chose only changes the coefficient. So it seems that the Bohr model is telling you that this potential doesn't bind. Likebox 02:12, 8 September 2007 (UTC)
Cohomology Stuff
The cohomology stuff confused me. Which is the image of the two consecutive maps? I think I get it now--- the image is the last thing, the two forms in the second DeRham cohomology class. These are the allowed symplectic forms. So big deal. This is just a topological condition on what kind of two-forms are allowed as symplectic forms that can be quantized. That's a global thing. It has something to do with what kind of cycles are allowed in a phase space. The Bohr Sommerfeld quantization is a local thing. The action is an integer, even in a flat two dimensional boring phase space with no topology. That's a completely different condition, and the word "integral" means two different things. In Bohr Sommerfeld, it means "integer". Here it means "I can integrate this form" over cycles.
(later addition) I get it better now--- I made a mistake. "integral" does mean integer. But the cycles are confusing me--- what are the cycles over which you are integrating? The symplectic two form must be such that certain integrals over certain closed cycles are integers. The meaning of :"integral" is "integer". Which cycles? Where is the notion of orbit? Orbit is an integral curve of a Hamiltonian, where is that? Likebox 01:56, 8 September 2007 (UTC)
The only statement that makes sense to me is that the symplectic form should be a curvature of a hermitian line bundle. Maybe that's true, but the author doesn't explain why. This local statement might be the condition for a quantizable thingamabob, and the previous stuff could be the global obstructions to finding such a bundle. But again, this doesn't have anything to do with the Bohr Sommerfeld quantization.
I can't be the only one confused. Anyway, I am going to delete it and copy to the talk page. If the author could explain it more clearly, it might be interesting.
- The Bohr-Sommerfeld quantization condition as first formulated can be viewed as a rough early draft of the more sophisticated condition that the symplectic form of a classical phase space M be integral; that is, that it lies in the image of , where the first map is the homomorphism of Čech cohomology groups induced by the inclusion of the integers in the reals, and the second map is the natural isomorphism between the Čech cohomology and the de Rham cohomology groups. This condition guarantees that the symplectic form arise as the curvature form of a connection of a Hermitian line bundle. This line bundle is then called a prequantization in the theory of geometric quantization. —Preceding unsigned comment added by Likebox ( talk • contribs) 02:38, 7 September 2007 (UTC)
Correspondence Principle
I added a discussion of Bohr's idea that the level spacing is determined by the frequency of classical radiation on correspondence principle, but I think it is better here. I don't want to move the discussion, because this page is already pretty good and I don't want to muck it up. But I think that the following points are needed:
1. Bohr didn't guess that L was quantized. It follows from the fact that photons are emitted at the classical orbit frequency. This means he probably didn't think his orbits were exactly stable, since the emission of photons at the classical orbit frequency exactly corresponds to the classical decay of a circular orbit by emission of radiation.
2. Bohr's screw up in assigning the ground state nonzero angular momentum is entirely fixed in a proper semiclassical treatment a-la sommerfeld. Nobody cared enough to do this because the "half-integer quantum number problem". The right answer for the angular momentum in the ground state is not zero or one. It's 1/2. That was known from magnetic-field splitting (Stark effect or Zeeman effect, I can't keep the names straight) and was explained by the electron spin. It is hard to determine if the ground state has orbital L zero or one, because either way the ground state spin could be 1/2.
- (later addition) actually, I just worked out Sommerfeld's calculation, and I think I may be wrong wrong. I will have to double check, because I can't imagine how the relativistic formula for the fine splitting can work with the wrong values of L. Likebox 04:44, 10 September 2007 (UTC)
3. Bohr's method is applicable to any mechanical system, and if there is supersymmetry (in the sense of shape invariance) the answer is exact.
4. Sommerfeld also quantized the relativistic H atom, by doing the whole action-angle routine with relativistic phase space. This is
a tour-de-force, and gives the right answer for the hyperfine splitting. I mentioned it in the correspondence principle write up, but
I think its better here. Sommerfeld's success is nowadays understood as the shape invariance in the Dirac equation. It was also
historically confusing, because if you use Schrodinger's relativistic equation (KG eq) to find the hyperfine structure you get the wrong answer. This held up Schrodinger's publication for a long time.
Likebox
16:04, 9 September 2007 (UTC)
Intro image currently wrong
I just noticed that the picture which is prominently featured at the top of the article seems to be wrong. 1) The atomic nucleus is missing (which in itself is probably not a big deal); 2) The electron on the lowest level (n = 1) should be on its orbit and not in the center of the atom. 149.217.1.6 15:43, 8 November 2007 (UTC)
- Fixed it. Alban 13:45, 16 November 2007 (UTC)
- Hi - The image still has problems, the energy should read , (Greek letter nu), NOT (English letter v). PAR ( talk) 12:24, 5 December 2007 (UTC)
Bohr's Reasoning and DeBroglie
Bohr used the correspondence principle to derive the quantization of angular momentum, and this is explained in the previous section. The assumption is that any emmitted radiation has the same frequency as the classical orbital frequency. This leads to quantized angular momentum, to lowest order in h. The same condition can also be derived from the principle of adiabatic invariance of the quantum numbers, as done by Sommerfeld and Einstein. DeBroglie's assumption is justified by the fact that it reproduces Bohr's quantization, not the other way around. This is historically (and logically) better, in my opinion. So I will move the new text to a different position and edit it to reflect history. Hope this is ok with the author. Likebox ( talk) 06:43, 5 December 2007 (UTC)
Why isn't there energy loss for an electron revolving, in Bohr's model?
the failure of rutherford model was that the electron would lose energy(according to maxwell's laws) while revolving round the atom. but here in Bohr's model why isn't there energy loss if the electron revolves round the nucleus?any sort of work demands energy right? —Preceding unsigned comment added by 203.145.177.111 ( talk) 02:01, 17 December 2007 (UTC)
- Bohr's model has the same problem. Only in full QM is it solved, basically because electrons in the ground orbital don't do any work. Electrons in upper ones can, but only by changing dipole moment and dropping to lower ones. S B H arris 02:11, 17 December 2007 (UTC)
- I guess it is possible to say that Bohr's model suffers from the same problem if you are not feeling generous, because Bohr's theory is incomplete, and doesn't specify the details of the emission and absorption of radiation. You can fill in the details in many ways. One way is to say that the electron is charged and orbiting, and so must emit radiation continuously and needs to continuously spiral in order to conserve energy. But to fill in the details in this way is, in my opinion, very unfair to Bohr. Certainly he felt that his model explained, at least in a qualitative way, the stability of atoms. He explained it by noting that the laws of mechanics must be modified to be compatible with the known quantum nature of light.
- Light can't be emitted continuously, since photons are discrete lumps. So in order for an electron to spiral in, it needs to emit a quantum of radiation. The frequency of the radiation is at the classical orbit frequency (at least classically--- quantum mechanically this is still a good approximation), which gives a quantum of energy equal to h/T where T is the classical orbit period. If the electron loses this energy, it must drop to a lower orbit which is closer in, with its own orbital period, and its own energy.
- This is correct for large radius orbits, but for small orbits the rules are not completely well defined. The reason is that the period T is either the period of the orbit you start at, or the orbit you finish at after the radiation is emitted. For large orbits, these are nearly equal, and the answer is well defined. But for small orbits, you need to quibble. The stability of the Bohr atom depends on whether you believe that the radiation frequency is given by T_i or T_f, or by some average.
- when h/T(r) = E(r), where T is the period of a circular orbit of size r and E is the total energy of an orbit of size r, the electron cannot emit radiation, because to emit the radiation would cost more energy than the orbit has. Unfortunately, the total energy in a circular orbit diverges as r goes to zero, becoming infinite at r=0. For a circular orbit of radius r, the total energy goes like -1/r, the period goes like r^1.5 (Kepler's third law). The quantum of energy is h/T(r) (that would be the energy of an emitted photon) and it goes like 1/r^1.5 and gets bigger for small r faster than the total energy gets bigger (1/r^1.5 > 1/r for r~0). But since the total energy diverges, it is technically correct that given any circular orbit, you can emit a quantum at frequency T_i and find a (much smaller) radius where the energy is more negative by enough so that the energy balance works out.
- But this is why Bohr was careful in choosing the radiation law. He said that the emitted radiation would have the frequency 1/2(h/T_i + h/T_f), the average of the frequency of the initial and final state. This prescription gives the right answer, but it also has the property that eventually you cannot emit any radiation at all. The reason is that the h/T_f, the final state frequency, diverges faster than the total energy. For a small enough value of r there is no solution for the final state.
- Bohr knew that the averaging procedure was ad-hoc, and it is only justified ultimately by appealing to modern quantum mechanics. But it is pretty clear that Bohr knew that the stability of the atom was due to the fact that small orbits have a higher frequency than they have an energy, since the frequency of orbits diverge as 1/r^3/2 while the total energy is 1/r. This is essentially correct and is preserved in modern treatments.
- There are forces, incidentally, which do not admit a stable ground state even in modern quantum mechanics. Those forces include those where the orbital frequency diverges less strongly than the total energy (the marginal case require careful analysis). For those forces, there is no stable ground state in modern QM, since you can use a variational wavefunction concentrated on Bohr orbits of smaller and smaller radii to get ever smaller values of the energy, reproducing Bohr's argument.
- While Bohr's theory is semi-classical, so that it is only correct to first order in h, it actually is correct to first order in h. Even to lowest semiclassical order there is a lowest possible energy for circular orbits in a 1/r potential. There are still quibbles about Bohr--- is the lowest value the one appropriate to "n=1", as Bohr says, or do you need "n=2" for a circular orbit, as in the modern quantum mechanical treatment. But it gets things mostly right. Likebox ( talk) 19:03, 17 December 2007 (UTC)bohrs model was a big step befor his time.
I wish to resurrect this question as I am assume this will receive expert answers. I have never been able to understand this question. I find this question only makes sense if there are no forces acting upon the electron, then a curved path should result in an energy loss and spiral into the nucleus (which itself, seems oxymoronic). However, this question is not asked why the moon doesn't lose energy and collide with the earth. Gravity curves the moon's path and thus even though it travels in a curved path, it doesn't lose energy. This should be true for all rotational motions. If a Coulombic force acts between a proton and electron, why should the electron lose energy? Petedskier ( talk) 14:26, 20 July 2013 (UTC)
- A classical accelerated charge will radiate EM, and thus lose energy. However, in QM, the electron is not accelerated in a stationary state, since the psi-function does not change over time, and thus the electron does not move. For a given orbital the expectation position of the electron does not change in time. Since it doesn't move, it can't accelerate (except when going from one orbital to another) and so it cannot radiate. It seems odd to think of an electron in a hydrogen atom as just sitting there in an unchanging symmetric blob that doesn't change in time (so long as the electron doesn't charge orbitals). But that's the best picture we have. Needless to say, it's not really the Bohr picture. S B H arris 21:26, 20 July 2013 (UTC)
Why Is the Innermost Shell Z-1
This seems to be required for Moseley's law and K spectroscopy, but it seems that the innermost shell should be orbiting the full nucleus at Z. The explanation on this page was that there was somehow screening from the other electrons, but the two innermost electrons are in the zero angular momentum 1S orbital, so treating this as a zero-area ellipse to one side, the repulsion between the electrons should cause them to orbit on opposite sides, so that the screening should be (intuitively) less than 1 unit, since each electrons is further away from the other than it is from the nucleus. In QM, the two electron state is entangled, so that when one electron is on the left the other tends to be on the right. I would hae expected Z-.4 or Z-.3 as the proper approximation, but the complications seem to lead to approximately two independent electrons, orbiting at Z-1. This is weird. I was hoping someone could clarify. Likebox ( talk) 06:50, 24 January 2008 (UTC)
- I reverted, not because I think you are wrong, but the wording was not very good. It sounds like you are thinking out loud, rather than writing the article. Im not sure what the answer to your question is, but if you find it out, then it should be added.
PAR (
talk)
10:09, 24 January 2008 (UTC)
- The Bohr model itself offers NO good a priori or even a posteriori reason for why innermost (inner shell, N=1) electrons see Z-1 charge. Mosely asked Bohr why it should be Z-1 (what Moseley empirically got to good precision) and Bohr had no idea. Remember, without the exclusion principle (not to come for a dozen years) there were no restrictions in how many electrons could be in the lower "shell" with N=1. Bohr thought maybe there were 4 at minimum! So Moseley published without an explanation. The idea that there might be just 2, with one screening completely for the other as soon as it got any distance away (including just to the next shell) was realized the next year, by a third physicist who escapes me (I'll look it up, since it needs to go in the Moseley articles). The whole thing is not intuitive, even by modern QM where these 1s electrons have no angular momentum at all (not as in Bohr). It's not intuitive why one electron settling into the 1s shell sees the other as though it were always COMPLETELY between it and the nucleus. You'd think it would be somewhere else at least half the time. But no. S B H arris 05:08, 13 June 2008 (UTC)
- I was puzzled about why Z-1 in modern QM. I didn't find anything online. It might not even be known why. It looks like it's exactly Z-1 in large atoms too. In terms of the Bohr model, while the Bohr model didn't explain the Z-1, the shell model did explain the Z, which is 98% of Z-1 for most Z, so it is more or less right, and still historically important. Likebox ( talk) 07:40, 13 June 2008 (UTC)
Successes of Bohr's model
Although the failure of the Bohr’s model, however a great mystery persisted, as it is explained as follows.
The values that if one gets from the Balmer’s formula relate to the energies (photons) emitted by the hydrogen atom. To get those values with his model, Bohr considered that, in the instant when the atom emits a photon, the electron is in equilibrium due two forces: the force Fa of attraction with the proton, and the centripetal force Fc due to speed of rotation to about the proton.
Therefore, in his model, in the instant of the emission of photons the electron is under the action of the centripetal force , that is, in the mechanism of emission of photons from the model of Bohr there is the performance of a centripetal force on the electron.
In other words: the emission mechanism depends inexorablely on the action of a centripetal force.
Well, the model of Bohr obtained fantastic results. For example, by using his model, one calculates the Rydberg constant. Compare the value gotten from:
the experiments: RH = 10.967.757
the theoretical calculation: RH = 10.968.100
Impressive, isn’t?
Coincidence ?
Only if we believe that it is coincidence with the same faith with which a religious one believes miracles. Moreover, the Bohr model supplied other spectacular results. From the laws of the probability, it is impossible that it can be mere coincidence. And therefore there is something of truth in his model.
That’s why Schrödinger said:
“It is difficult to believe that this result is merely an accidental mathematical consequence of the quantum conditions, and has no deeper physical meaning”( 1 ).
He believed that Bohr’s successes would be consequence of unknown mechanisms, and he tried to find them.
The conclusion is that centripetal force really plays some function in the instant when a photon is emitted by an atom.
But just here the great mystery is. The mechanism of emission of photons from the Schrödinger’s theory does not admit that one assumes that the centripetal force plays some role in the emission of photons. The mechanism of emission of photons according to Quantum Mechanics is by resonance, a total incompatible process with the hypothesis of centripetal force on the electron in the instant of the emission. In short, the theory of Schrödinger does not admit centripetal force, and therefore the Bohr’s model must be completely wrong, so that the model of the Quantum Mechanics may be correct.
But we already saw that mathematically, from the laws of probability, it is impossible that the model of Bohr can be completely wrong. The centripetal force must have some linking with the mechanism of emission of the atom, and in this in case it is lacking something in the Quantum Mechanics.
In another words:
a) Whereas the model of Bohr cannot be completely wrong, as they certify the laws of the probability...
b)... on the other hand the model of the Quantum Mechanics cannot be completely certain, because it states that the Bohr model is completely wrong.
Therefore there is here a great mystery that defies the Quantum Mechanics.
That’s why the theorists decided to state that the spectacular successes of Bohr’s theory are accidental. In a paper( 2 ) in which proposes the helical trajectory of the electron for unifying the relativity with the quantum theory, the physicist Natarajan writes, commenting the success of Bohr theory in explaining the espectra bands:
“But this significant sucess along with the other spectacular successes of Bohr’s theory of the hydrogen atom is now considered by physicists as ‘accidental’ after the development of Quantum Mechanics”.
But as said Schrödinger it’s hard to believe that Bohr’s successes are accidental. Actually it is impossible, from the laws of probability. It’s probable that Schrödinger started to suppose that Bohr’s successes could have connection with the electron’s zitterbewegung. Schrödinger and Heisenberg had a different view on the question of how Quantum Mechanics would have to be developed. Schrödinger would like to follow the way by considering the zitterbewegung as an electron’s helical trajectory. While Heisenberg proposed to develop Quantum Mechanics by considering that the concept of trajectory could not be kept in the theory. Such Heisenberg’s view is today known as the Copenhagen interpretation, and it prevailed in the development of the theory.
Believing that Bohr’s successes are accidental, the theorists believe in the inadmissible, because it’s comfortable, but actually they deceive themselves.
Unlike, as Schrödinger did not accept to deceive himself, he abandoned the dispute with Heisenberg, when realized that the Theoretical Physics had followed that way preconized by the interpretation of Copenhagen.
In short, it’s hard to believe that Bohr model has not a botton of truth.
References:
1- E. Schrödinger , On a Remarkable Property of the Quantum-Orbits of a Single Electron, 1922
2- - T. S. Natarajan, Unified Conceptual Foundation for Relativity and Quantum Mechanics, Physics Essays, V. 9, No. 2, 1996, pg 302
See more on the Bohr's successes:
1- Cold fusion, Don Borghi's Experiment, and hydrogen atom: http://peswiki.com/index.php/PowerPedia:Cold_fusion%2C_Don_Borghi%27s_Experiment%2C_and_hydrogen_atom
2- Successes of the Bohr atom: http://peswiki.com/index.php/PowerPedia:Successes_of_the_Bohr_atom
—Preceding unsigned comment added by 200.97.93.67 ( talk) 21:35, 11 April 2008 (UTC)
It seems to me that this contribution is rightly not inserted in the `Bohr model' page. It probably is a criticism of the `BKS theory', to the effect that the idea is criticized that the stationary Coulomb field (which is responsible for the centripetal force) is thought there to exert only a statistical rather than a deterministic influence on the emission of a photon. The author refers to a theory based on the Bohr model, improving in this respect on the BKS theory, as well as on quantum mechanics (the latter theory -at least in its Copenhagen form- having taken over the probabilistic nature of emission of a photon). Hence, the theory referred to by the author might also be qualified as a subquantum or hidden variables theory. Note that by itself the Bohr model does not imply any determinism of the interaction of matter and radiation.
If the above-mentioned theory has been published and has become a substantial part of public knowledge, then, as seems to me, it could better be presented in a separate page of its own, or as an example of a hidden variables theory on a hidden variables page (if such a page exists). Otherwise, the author could content himself with a link to the above-mentioned theory at a place more appropriate than the `Bohr model' page. WMdeMuynck ( talk) 10:21, 13 April 2008 (UTC)
WmdeMuynck,
You did not understand.
The criticism is not of the BKS theory. Instead of, it’ s a criticism to Quantum Mechanics.
The reason is obvious:
1- From the laws of probability, it’s impossible that Bohr’s successes can be accidental
2- But in his calculations Bohr considered that in the instant of the photon emission the electron is submitted to a centripetal force.
3- Therefore it’s obvious that the centripetal force plays some misterious function in the emission of photons by the atom, because the successes of Bohr’s calculations cannot be credited to coincidences.
4- But it’s unadmissable to consider the existence of a centripetal force on the electron, according to Quantum Mechanics.
5- But as the centripetal force indeed plays some unknown function in the emission of photons by the atom, this imply that something is missing in Quantum Mechanics. In other words: Quantum Mechanics cannot be entirelly correct, and so something is wrong in the theory.
The successes of Bohr is the stronger evidence that point us the need of a new hydrogen atom, where the centripetal force plays some (unknown yet) function in the instant when the photon is emitted.
A new hydrogen atom, to be accepted, must be able to explain the successes of Bohr (the new hydrogen atom must be compatible with the Bohr model).
Quantum Mechanics is unable to explain the Bohr’s successes. So, the model of Quantum Mechanics actually is unacceptable (by any serious theorists that worry on fundamental questions in Physics, of course).
In general a physicist dislikes to face fundamental questions in Physics, when they disprove the concepts of QM.
But sure that we cannot take seriously a theorist who refuses to talk about fundamental questions in which Quantum Mechanics fails.
Obviously, the new hydrogen model must be compatible with the atom model of Quantum Mechanics too. —Preceding unsigned comment added by 189.48.107.117 ( talk) 18:08, 13 April 2008 (UTC)
I've removed this section from the article, since all it said was "see discussion". If there's material being debated for inclusion here it should remain out of the article until consensus is reached. (I don't have time to thoroughly read the above anonymous essay and comment right now or I might join the debate myself.) Olaf Davis | Talk 09:35, 14 April 2008 (UTC)
- Anonymous, I notice you've reverted my edit without comment. I maintain that if the material is suitable for the encyclopedia it should be there rather than on the talk page, and if it's not then we shouldn't be advising people to 'see' it. If you disagree please reply here. Again, I will remove the section until we have a consensus on what (if anything) should be there. Olaf Davis | Talk 17:18, 14 April 2008 (UTC)
- Olaf said: "I don't have time to thoroughly read the above anonymous essay"
- So I dont understand how you can form an opinion about a question if you did not read on it —Preceding unsigned comment added by 200.97.93.67 ( talk) 18:09, 14 April 2008 (UTC)
- Well, I haven't formed an opinion about what you wrote. But whether what you wrote is true or not, it doesn't belong on the talk page with "see discussion" linking to it on the article. If you can find a source for it, by all means put it in the article - but linking from the article to the talk page is not how to accomplish that. Olaf Davis | Talk 23:13, 14 April 2008 (UTC)
- Olaf said: "If you can find a source for it"
- Olaf, the source is the LOGIC. If Wikipedia is not a place suitable to put a logic fact, I am very sorry. As you know, 2+2=4. Do you think there is need a source to state that 2+2=4 ? —Preceding unsigned comment added by 200.97.93.67 ( talk) 16:01, 15 April 2008 (UTC)
- Olaf said: "If you can find a source for it"
- You may want to read Wikipedia:Verifiability and Wikipedia:No original research. The bottom line is that if your theory hasn't been published in a reliable source, it does not belong on wikipedia. Logic is not enough. Hemmingsen 16:43, 15 April 2008 (UTC)
- The policy WP:When to cite says "general common knowledge" does not need a source, so 2+2=4 is fine unsourced. What you wrote above, even if true, is certainly not general common knowledge. WP:Verifiability says "The threshold for inclusion in Wikipedia is verifiability, not truth. "Verifiability" in this context means that readers should be able to check that material added to Wikipedia has already been published by a reliable source."
- Wikipedia is certainly a suitable place for logical facts, but only if they've already been published. original research belongs not here but in a journal or book.
- I hope that explanation makes sense. Incidentally, it's helpful to other editors if you sign your comments by placing four tildes at the end (~~~~). You may also want to consider signing up for a user name - which actually makes it easier to remain anonymous! Olaf Davis | Talk 16:53, 15 April 2008 (UTC)
- Olaf said: The bottom line is that if your theory hasn't been published in a reliable source, it does not belong on wikipedia
- Olaf, the analysis of the implications of the Bohr's successes is not a theory of mine. If you analyse a fact with arguments supported by the logic, anybody able to understand logical facts is able to recognize the validity of your analysis. I simply analysed the successes of Bohr model from the viewpoint of Quantum Mechanics, and showed the facts to anyone interested in the facts.
- Olaf said: What you wrote above, even if true, is certainly not general common knowledge.
- Olaf, the quantum theorists become hysterical when they face the implications of the successes of the Bohr model. The reason is obvious: the successes of Bohr model imply that Quantum Mechanics is not entirely correct. As the quantum theorists are afraid to face the Bohr successes, they do not speak about the matter. However it does not mean it is not a general common knowledge. The quantum theorists allege that Bohr successes are accidental. And of course they know that such allegation is impossible from the viewpoint of the mathematical probability, because they are not stupid. But they preffer to neglect THE LOCIGAL FACTS, because it is comfortable to them —Preceding unsigned comment added by 200.97.93.67 ( talk) 21:35, 15 April 2008 (UTC)
- Actually, it was Hemmingsen who said "the bottom line is...", but he was correct. Arguments about logic and the hysteria of quantum theorists make no difference as far as Wikipedia policy is concerned. Wikipedia can include the material if you show that it's been published by a reliable third-party source, or that it's general common knowledge (and none of the details of QM are general common knowledge in the sense that the man on the street knows anything about them). If you think Wikipedia policy could be improved, that's fine - the policies as well as the content of this encyclopedia are open to change! But, the correct place to argue that is on the relevant policy page, not here. Therefore unless and until you find a source, or succeed in changing the policies, I've said all I have to on the matter.
- Best, Olaf Davis | Talk 09:15, 16 April 2008 (UTC)
Before engaging in this discussion, you may be wish to note that the user who contributed this section has been blocked as a sockpuppet of a user who was previously blocked for only contributing disruptive edits involving Conflicts of Interest, Original Research, and unverifiable claims. Most of the comments involved in this discussion also appear to be in his very unique writing style. - Verdatum ( talk) 18:07, 22 April 2008 (UTC)
Equation vandalism
There's an equation in here, which may have been vandalized by an anonymous user recently; this user changed the equation from See "Energy Levels for Nuclei with Z Protons" equation in the article. --/ Mendaliv/ 2¢/ Δ's/ 05:24, 7 May 2008 (UTC)
I haven't read Bohr's paper
Except for once 15 years ago. But I distinctly remember the "Bohr frequency condition" coming before the quantization of L, not the other way around. This is important, because all the people writing now do it the other way. I hoped this could be fixed here, because it is both historically correct, more logical, and pedagogically better to do it the way Bohr did. L quantization should always be derived from the condition that the emitted photon has the classical orbital frequency because this is obvious to someone thinking classically. The mathematical derivation requires some approximation and physical thinking, but that's the essence of physics, and, in my opinion, it is a disservice to Bohr to present it the other way around. Likebox ( talk) 09:33, 7 May 2008 (UTC)
It's even worse here, because the article suggests that the rule that the emitted radiation is at the orbital frequency is only correct for large orbits. Since a quantum jump involves two classical orbits, there is no unique classical frequency unless n is large. Bohr fiddled around with the rule until he came up with the right one for hydrogen--- the emitted frequency is the average of the initial and final orbit. The reason he does this is because the rule for quantization of angular momentum is ad-hoc. You must arrive at it from a more primitive postulate which makes sense. While the particular rule that Bohr arrives at (average the frequencies) doesn't make sense in all its details, the rule that to lowest order in h the answer is correct classically make sense and is correct in modern quantum mechanics, and it allows you to arrive at the quantization of L (an integer multiple of h plus higher order corrections in h) which makes it plausible to conjecture that there are no higher order corrections and that L is quantized. Likebox ( talk) 09:50, 7 May 2008 (UTC)
- One last comment--- Einstein's photon concept was rejected and ridiculed, so Bohr could not have openly supported it. He references the paper though, so he probably respects the reasoning and (I think) probably believes in photons. The most likely interpretation is that Bohr was trying to say "photon" without using the word photon and without invoking photon localization, using only Planck's way of looking at it, which involved the emitter and the absorber without talking about any quantization inbetween. Likebox ( talk) 10:27, 7 May 2008 (UTC)
- Ok-- I just read the BKS theory page, and it seems that historians know this, and that Bohr did reject photons. Curiouser and curiouser. Anyway, I guess what I wrote above is wrong. Likebox ( talk) 15:38, 8 May 2008 (UTC)
- It is correct that Bohr's first quantization condition was about energy (cf. page 5 of Bohr's paper, which can be downloaded at the end of the `Bohr model' article). But this was a completely ad hoc relation, just meant to reproduce terms in the Balmer expression. It required eq. (2) on page 5 of Bohr's article, which does not involve the difference of two frequencies (as is the case in the final energy quantization rule of the Bohr model), but half of the classical frequency. On page 15 of that article Bohr gives the angular momentum condition as an alternative (as well as a physical explanation of energy quantization). It seems to me that this latter account is preferable, since the first one is consistent with the final energy quantization rule of the Bohr model only for large values of n. Hence, it would be rather confusing to present it as a condition which should hold for all values of n. WMdeMuynck ( talk) 10:14, 1 June 2008 (UTC)
- This is not accurate, it only seems so to modern readers. The reason it's one-half the classical frequency is because the transition he is imagining is between a circular orbit at infinite radius and zero frequency and a circular orbit at a finite radius and frequency. The average of something with zero is half. This is why he gets half the classical orbit frequency. To be clear--- there are two conditions:
- 1. photon energy condition: E_i - E_f = hf
- 2. Bohr frequency condition: f = (f_i + f_f)/2
- Condition 1 is universal and is true of the frequency of the emitted photon. Condition 2 is not exact, and gives the frequency of the photon from the classical orbit frequencies. For transitions between nearby levels, it is just a statement of the correspondence principle. Also note that Bohr recognizes the relationship between the Fourier harmonics and n steps down transitions in the quantum system. He really is giving a complete semiclassical model, but he is not expressing himself in the clearest possible way. Likebox ( talk) 23:06, 12 June 2008 (UTC)
- I think your analysis to be quite right, although I do not know whether your Bohr frequency condition, f = (f_i + f_f)/2, is in Bohr's paper (I did not check it). Anyway, you are right that Bohr's approach was classical Fourier analysis, and the condition is indeed suggested by the factor 2, thus seemingly corroborating that approach.
- However, the question is: what kind of reader are you aiming at. Is it the historically interested reader, wanting to know everything about the historical process by which Bohr arrived at his model, or is it the present-day (modern) reader, wanting to be informed about the Bohr model and its relevance to present-day physics? I think this would ask for two different articles, the present one suggesting my second alternative (at least this is what I take it for). In this latter context I think that your Bohr frequency condition, f = (f_i + f_f)/2, is very confusing, since it is very different from the condition f = f_i - f_f corresponding to the "modern" relation. Actually, the difference between these two expressions has been a bone of contention with respect to applicability of the correspondence principle (evidenced by the failure of classical Fourier analysis for arbitrary transitions), reason why the correspondence principle has been restricted to Rydberg states (even though it is seemingly applicable too for transitions from an arbitrary state to the `state at infinity' corresponding to n = \infty). It seems to me that a discussion of this confusion does not belong here, but in an article on the correspondence principle. WMdeMuynck ( talk) 08:56, 20 June 2008 (UTC)
- As I wrote before, but it disappeared, Bohr's argument about semiclassical transitions to the state at infinity doesn't really make as much sense as I thought it did at first, when I wrote the previous comment. I hadn't read Bohr's paper then, and I didn't give it too much thought--- I was writing from memory. There's an extra factor of the quantum number you need in the numerator, required to match semiclassical rule--- transitions of two steps are twice the frequency, three step transitions are three times the frequency more or less. For transitions to infinity, the condition in Bohr's paper has an factor of what he calls tau, which is the quantum number, which he puts in essentially without a good reason. So there's some fudging there. The semiclassical derivation that does make perfect sense, in hindsight, is the one that is written up on correspondence principle and alluded to here, you step down level by level using the frequency condition that the emitted photon is the classical orbit frequency, and doesn't require any serious fudging and it gets the whole thing--- including the Sommerfeld quantization rule. Bohr does this in the later parts of his paper, steps down orbit by orbit, but I don't know what he was thinking with the first derivation--- its very mysterious.
- The reason I think that the presentation should be based on the correspondence principle is explained earlier on this talk page--- this is an encyclopedia, and an encyclopedia is supposed to tell the truth. Without the correspondence principle, and applying it to ridiculously small orbits, Bohr's model would have only been an experimentally motivated guess, and would not have served as the central starting point for modern quantum mechanics. The correspondence principle works much better than you would expect--- Eherenfest's theorem is more or less exact, and sommerfeld quantization works for the hydrogen atom, the rigid rotor, the particle in a box, the harmonic oscillator--- all the trivial examples, and miraculously for fine structure. Likebox ( talk) 09:21, 20 June 2008 (UTC)
merge suggestion: Refinements and Bohr–Sommerfeld theory
Well, the section called refinements is really just about the Bohr–Sommerfeld theory so either it should have a name change or be put in an article of it's own. The Bohr–Sommerfeld theory is just a stub with very little content, but the subject might need its own article. Besides, should it be called Bohr–Sommerfeld model to signify that it is largely obsolete? -- Thorseth ( talk) 12:14, 19 March 2009 (UTC)
- Personally I think Borh-Sommerfeld model is more appropriate than Borh-Sommerfeld theory. The section should probably be called that, and then use a "main article" link to "Bohr-Sommerfeld model". I've placed the merge notice to let people know about the other article, so whatever relevant/appropriate information contained in one article can be added or moved into the other. This wasn't about eliminating a section or the Borh-Sommerfeld standalone article (I think it deserves one). Headbomb { ταλκ κοντριβς – WP Physics} 16:05, 19 March 2009 (UTC)
I agree with headbomb.:) —Preceding unsigned comment added by Lkit97 ( talk • contribs) 00:20, 12 March 2010 (UTC)
bleep! 68.164.109.54 ( talk) 00:29, 3 October 2010 (UTC)
No Merge! The Bohr atom can be dealt with classical methods, with quantisation in e and h alone. For this reason alone, one ought leave the simple equations there, and append hamiltionians etc to the end if one wants to. Future developments (like the sommerfield model), ought belong elsewhere, but as a link at the end of this part of the story. It none the same has some success in deriving various things, like the Rydberg constant, the position of elements in the periodic table, and the simpler spectral lines. In return, de Broglie's waves lie neatly in the lengths of the Bohr orbits. One does not need to bring in things like relativity, or wave-equations, or hamiltonians, to understand it. As it stands, it is a milestone in the understanding of the atom, and thus invited the more clever people to bring in the heavy maths. Wendy.krieger ( talk) 09:38, 6 January 2012 (UTC)
Bohr model-based Helium

Bohr model could not explain about the ground state energy of a two-electron atom, Helium in 1920's.
But we must use the computer calculating the interaction between the two electons and the nucleus in Helium.
If the orbital planes of the two electrons of Helium are perpendicular to each other, their orbital length is just consistent with one de Broglie wavelength when their total energy is the experimental value(79.005eV)of the ground state energy of Helium.
And these two electrons are symmetric in this model.
Please see in detail http://arxiv.org/abs/0903.2546 —Preceding unsigned comment added by Eyy53j ( talk • contribs) 01:47, 2 July 2009 (UTC)
hola dependentito des bohr model es stupido —Preceding unsigned comment added by 24.4.70.233 ( talk) 22:22, 25 March 2010 (UTC)
















