| |
| Identifiers | |
|---|---|
| |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.348 |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
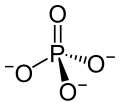
| O4PSm | |
| Molar mass | 245.33 g·mol−1 |
| Appearance | solid |
| Density | 5.83 g·cm−3 |
| insoluble | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Samarium(III) phosphate is an inorganic compound, with the chemical formula of SmPO4. It is one of the phosphates of samarium.
Preparation
Samarium(III) phosphate can be obtained by reacting sodium metaphosphate with any soluble samarium(III) salt:
Samarium(III) phosphate can also be obtained by reacting phosphoric acid and samarium(III) chloride. [1]
Properties
Samarium(III) phosphate reacts with sodium fluoride at 750 °C to form Na2SmF2PO4. [2] Samarium(III) phosphate forms crystals of the monoclinic crystal system, with space group P21/n, and lattice parameters a = 0.6669 nm, b = 0.6868 nm, c = 0.6351 nm, β = 103.92 °, Z = 4. [3]
References
- ^ HIKICHI, Yasuo; MURAYAMA, Kyouhei; OHSATO, Hitoshi; NOMURA, Tsuyoshi (1990). "Thermal changes of rare earth phosphate minerals". Journal of the Mineralogical Society of Japan (in Japanese). 19 (3). Japan Association of Mineralogical Sciences: 117–126. doi: 10.2465/gkk1952.19.117. ISSN 1883-7018.
- ^ Zimina, G. V.; Smirnova, I. N.; Gorkovenko, M. Yu.; Spiridonov, F. M.; Komissarova, L. N.; Kaloev, N. I. Synthesis and study of rare earth element fluorophosphates Na2LnF2PO4(in Russian). Zhurnal Neorganicheskoi Khimii, 1994. 39 (9): 1571-1574. ISSN 0044-457X.
-
^ D.F. Mullica, David A. Grossie, L.A. Boatner (March 1985).
"Coordination geometry and structural determinations of SmPO4,EuPO4 and GdPO4". Inorganica Chimica Acta. 109 (2): 105–110.
doi:
10.1016/S0020-1693(00)84549-1. Retrieved 2021-11-19.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link)

| |
| Identifiers | |
|---|---|
| |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.348 |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| O4PSm | |
| Molar mass | 245.33 g·mol−1 |
| Appearance | solid |
| Density | 5.83 g·cm−3 |
| insoluble | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Samarium(III) phosphate is an inorganic compound, with the chemical formula of SmPO4. It is one of the phosphates of samarium.
Preparation
Samarium(III) phosphate can be obtained by reacting sodium metaphosphate with any soluble samarium(III) salt:
Samarium(III) phosphate can also be obtained by reacting phosphoric acid and samarium(III) chloride. [1]
Properties
Samarium(III) phosphate reacts with sodium fluoride at 750 °C to form Na2SmF2PO4. [2] Samarium(III) phosphate forms crystals of the monoclinic crystal system, with space group P21/n, and lattice parameters a = 0.6669 nm, b = 0.6868 nm, c = 0.6351 nm, β = 103.92 °, Z = 4. [3]
References
- ^ HIKICHI, Yasuo; MURAYAMA, Kyouhei; OHSATO, Hitoshi; NOMURA, Tsuyoshi (1990). "Thermal changes of rare earth phosphate minerals". Journal of the Mineralogical Society of Japan (in Japanese). 19 (3). Japan Association of Mineralogical Sciences: 117–126. doi: 10.2465/gkk1952.19.117. ISSN 1883-7018.
- ^ Zimina, G. V.; Smirnova, I. N.; Gorkovenko, M. Yu.; Spiridonov, F. M.; Komissarova, L. N.; Kaloev, N. I. Synthesis and study of rare earth element fluorophosphates Na2LnF2PO4(in Russian). Zhurnal Neorganicheskoi Khimii, 1994. 39 (9): 1571-1574. ISSN 0044-457X.
-
^ D.F. Mullica, David A. Grossie, L.A. Boatner (March 1985).
"Coordination geometry and structural determinations of SmPO4,EuPO4 and GdPO4". Inorganica Chimica Acta. 109 (2): 105–110.
doi:
10.1016/S0020-1693(00)84549-1. Retrieved 2021-11-19.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link)
