| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.172 |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| DyO4P | |
| Molar mass | 257.470 g·mol−1 |
| Appearance | solid |
| insoluble | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
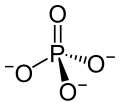
Dysprosium(III) phosphate is an inorganic compound with the chemical formula DyPO4.
Preparation
Dysprosium(III) phosphate can be obtained by reacting dysprosium(III) oxide and ammonium dihydrogen phosphate at high temperature: [1]
- Dy2O3 + 2 (NH4)(H2PO4) → 2 DyPO4 + 2 NH3 + 3 H2O
Properties
Dysprosium(III) phosphate decomposes into dysprosium oxyphosphate and phosphorus pentoxide above 1200 °C. [2] It reacts with sodium fluoride to obtain NaDyFPO4: [3]
- NaF + DyPO4 → NaDyFPO4
It reacts with sodium molybdate at high temperature to generate Na2Dy(MoO4)(PO4): [4]
- Na2MoO4 + DyPO4 → Na2Dy(MoO4)(PO4)
References
- ^ Khadraoui, Z.; Bouzidi, C.; Horchani-Naifer, K.; Ferid, M. (2014). "Crystal structure, energy band and optical properties of dysprosium monophosphate DyPO 4". Journal of Alloys and Compounds. 617. Elsevier BV: 281–286. doi: 10.1016/j.jallcom.2014.07.135. ISSN 0925-8388.
- ^ Rouanet, A.; Serra, J. J.; Allaf, K.; Coutures, J.; Dexpert, H. Thermal decomposition of rare earth orthophosphates in air above 1200°C. Reactions and characterization of new solid phases(in French). Revue Internationale des Hautes Temperatures et des Refractaires, 1980. 16 (4): 437-443. ISSN 0035-3434.
- ^ ZIMINA, G. V.; SMIRNOVA, I. N.; GORKOVENKO, M. YU.; SPIRIDONOV, F. M.; KOMISSAROVA, L. N.; KALOEV, N. I. (1995-02-21). "ChemInform Abstract: Synthesis and Studies of Fluorophosphates of Rare Earth Elements Na2LnF2PO4". ChemInform. 26 (8). doi: 10.1002/chin.199508015. ISSN 0931-7597.
- ^ Ryumin, M. A.; Komissarova, L. N.; Rusakov, D. A.; Bobylev, A. P.; Zhizhin, M. G.; Khoroshilov, A. V.; Gavrichev, K. S.; Danilov, V. P. (May 2007). "Synthesis and crystal structure of new complex sodium lanthanide phosphate molybdates Na2MIII(MoO4)(PO4)(MIII = Tb, Dy, Ho, Er, Tm, Lu)". Russian Journal of Inorganic Chemistry. 52 (5): 653–660. doi: 10.1134/s0036023607050014. ISSN 0036-0236. S2CID 101567788.

| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.172 |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| DyO4P | |
| Molar mass | 257.470 g·mol−1 |
| Appearance | solid |
| insoluble | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Dysprosium(III) phosphate is an inorganic compound with the chemical formula DyPO4.
Preparation
Dysprosium(III) phosphate can be obtained by reacting dysprosium(III) oxide and ammonium dihydrogen phosphate at high temperature: [1]
- Dy2O3 + 2 (NH4)(H2PO4) → 2 DyPO4 + 2 NH3 + 3 H2O
Properties
Dysprosium(III) phosphate decomposes into dysprosium oxyphosphate and phosphorus pentoxide above 1200 °C. [2] It reacts with sodium fluoride to obtain NaDyFPO4: [3]
- NaF + DyPO4 → NaDyFPO4
It reacts with sodium molybdate at high temperature to generate Na2Dy(MoO4)(PO4): [4]
- Na2MoO4 + DyPO4 → Na2Dy(MoO4)(PO4)
References
- ^ Khadraoui, Z.; Bouzidi, C.; Horchani-Naifer, K.; Ferid, M. (2014). "Crystal structure, energy band and optical properties of dysprosium monophosphate DyPO 4". Journal of Alloys and Compounds. 617. Elsevier BV: 281–286. doi: 10.1016/j.jallcom.2014.07.135. ISSN 0925-8388.
- ^ Rouanet, A.; Serra, J. J.; Allaf, K.; Coutures, J.; Dexpert, H. Thermal decomposition of rare earth orthophosphates in air above 1200°C. Reactions and characterization of new solid phases(in French). Revue Internationale des Hautes Temperatures et des Refractaires, 1980. 16 (4): 437-443. ISSN 0035-3434.
- ^ ZIMINA, G. V.; SMIRNOVA, I. N.; GORKOVENKO, M. YU.; SPIRIDONOV, F. M.; KOMISSAROVA, L. N.; KALOEV, N. I. (1995-02-21). "ChemInform Abstract: Synthesis and Studies of Fluorophosphates of Rare Earth Elements Na2LnF2PO4". ChemInform. 26 (8). doi: 10.1002/chin.199508015. ISSN 0931-7597.
- ^ Ryumin, M. A.; Komissarova, L. N.; Rusakov, D. A.; Bobylev, A. P.; Zhizhin, M. G.; Khoroshilov, A. V.; Gavrichev, K. S.; Danilov, V. P. (May 2007). "Synthesis and crystal structure of new complex sodium lanthanide phosphate molybdates Na2MIII(MoO4)(PO4)(MIII = Tb, Dy, Ho, Er, Tm, Lu)". Russian Journal of Inorganic Chemistry. 52 (5): 653–660. doi: 10.1134/s0036023607050014. ISSN 0036-0236. S2CID 101567788.