| |
| |
| Names | |
|---|---|
|
Preferred IUPAC name
1,2,3,5,6,7-Hexahydroxyanthracene-9,10-dione | |
| Other names
Rufigallic acid; 1,2,3,5,6,7-Hexahydroxy-9,10-anthraquinone; 1,2,3,5,6,7-Hexahydroxyanthracene-9,10-dione
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C14H8O8 | |
| Molar mass | 304.210 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
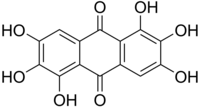
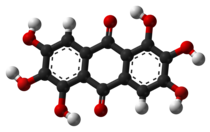
Rufigallol or 1,2,3,5,6,7-hexahydroxy-9,10-anthraquinone is an
organic compound with formula C
14O
8H
8, which can be viewed as a derivative of
anthraquinone through the replacement of six
hydrogen atoms (H) by
hydroxyl groups (OH).
The compound is soluble in dioxane, from which it crystallizes as red needles that sublime without melting at 365 °C. [1] It can be obtained by treating gallic acid with concentrated sulfuric acid and then with sodium hydroxide. [1]
Rufigallol is particularly toxic to the malarial parasite Plasmodium falciparum and has a synergistic effect in combination with the antimalarial drug exifone, which has structural similarities to rufigallol. [2]
Rufigallol forms a crimson-colored complex with beryllium, aluminium, thorium, zirconium and hafnium, and this reaction has been used for the spot and spectrophotometric determination of beryllium in low concentrations. [1]
See also
References
- ^ a b c M. A. Azim and A. A. Ayaz (1969), Spectrophotometric determination of beryllium. Microchimica Acta Volume 57, Number 1, pages 153-159 doi: 10.1007/BF01216677
- ^ R. W. WINTER, KENNETH A. CORNELL, LINDA L. JOHNSON, MARINA IGNATUSHCHENKO,DAVID J. HINRICHS and MICHAEL K. RISCOE (1996), Potentiation of the Antimalarial Agent Rufigallol. ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Vol. 40, No. 6, pages 1408–1411. Online version accessed on 2010-02-01.

| |

| |
| Names | |
|---|---|
|
Preferred IUPAC name
1,2,3,5,6,7-Hexahydroxyanthracene-9,10-dione | |
| Other names
Rufigallic acid; 1,2,3,5,6,7-Hexahydroxy-9,10-anthraquinone; 1,2,3,5,6,7-Hexahydroxyanthracene-9,10-dione
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C14H8O8 | |
| Molar mass | 304.210 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Rufigallol or 1,2,3,5,6,7-hexahydroxy-9,10-anthraquinone is an
organic compound with formula C
14O
8H
8, which can be viewed as a derivative of
anthraquinone through the replacement of six
hydrogen atoms (H) by
hydroxyl groups (OH).
The compound is soluble in dioxane, from which it crystallizes as red needles that sublime without melting at 365 °C. [1] It can be obtained by treating gallic acid with concentrated sulfuric acid and then with sodium hydroxide. [1]
Rufigallol is particularly toxic to the malarial parasite Plasmodium falciparum and has a synergistic effect in combination with the antimalarial drug exifone, which has structural similarities to rufigallol. [2]
Rufigallol forms a crimson-colored complex with beryllium, aluminium, thorium, zirconium and hafnium, and this reaction has been used for the spot and spectrophotometric determination of beryllium in low concentrations. [1]
See also
References
- ^ a b c M. A. Azim and A. A. Ayaz (1969), Spectrophotometric determination of beryllium. Microchimica Acta Volume 57, Number 1, pages 153-159 doi: 10.1007/BF01216677
- ^ R. W. WINTER, KENNETH A. CORNELL, LINDA L. JOHNSON, MARINA IGNATUSHCHENKO,DAVID J. HINRICHS and MICHAEL K. RISCOE (1996), Potentiation of the Antimalarial Agent Rufigallol. ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Vol. 40, No. 6, pages 1408–1411. Online version accessed on 2010-02-01.