| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
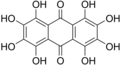
1,2,3,4,5,6,7,8-Octahydroxyanthracene-9,10-dione | |||
| Other names
Octahydroxyanthracenedione
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C14H8O10 | |||
| Molar mass | 336.208 g·mol−1 | ||
| log P | -0.291 | ||
| Acidity (pKa) | 5.358 | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Octahydroxyanthraquinone is an
organic compound with formula C
14H
8O
10, formally derived from
anthraquinone by replacement of 8
hydrogen atoms by
hydroxyl groups.
The compound was obtained in 1911 by Georg von Georgievics [1] [2] and can be obtained through oxidation of rufigallol ( 1,2,3,5,6,7-hexahydroxyanthraquinone) with boric acid and mercuric oxide in sulfuric acid at 250 °C (482 °F). [3]
Esters of octahydroxyanthraquinone, where all eight hydroxyls are replaced by straight-chain 1-alkanecarboxylate groups H
3C-(CH
2)n-COO-, with n between 6 and 14, are
liquid crystals and have been studied for possible
LCD applications.
[3]
Octahydroxyanthraquinone is active against the malaria parasite, but rufigallol (1,2,3,5,6,7-hexahydroxyanthraquinone) is 22 times more potent. [4]
References
- ^ Georgievics, G. v. (1911). "Darstellung und Eigenschaften des Octooxyanthrachinons". Monatshefte für Chemie. 32 (5): 347–352. doi: 10.1007/BF01518160. S2CID 97848374.
- ^ Wahl, Andre; Atack, F. W (1919) The Manufacture Of Organic Dyestuffs. G. Bell And Sons, Limited. Online version accessed on 2010-01-22.
- ^ a b Kumar, Sandeep (2008). "Rufigallol-based self-assembled supramolecular architectures". Phase Transitions. 81: 113–128. doi: 10.1080/01411590701601610. S2CID 59445187.
- ^ Winter, R (1995). "Hydroxy-anthraquinones as antimalarial agents". Bioorganic & Medicinal Chemistry Letters. 5 (17): 1927–1932. doi: 10.1016/0960-894X(95)00326-O.
| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
1,2,3,4,5,6,7,8-Octahydroxyanthracene-9,10-dione | |||
| Other names
Octahydroxyanthracenedione
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C14H8O10 | |||
| Molar mass | 336.208 g·mol−1 | ||
| log P | -0.291 | ||
| Acidity (pKa) | 5.358 | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Octahydroxyanthraquinone is an
organic compound with formula C
14H
8O
10, formally derived from
anthraquinone by replacement of 8
hydrogen atoms by
hydroxyl groups.
The compound was obtained in 1911 by Georg von Georgievics [1] [2] and can be obtained through oxidation of rufigallol ( 1,2,3,5,6,7-hexahydroxyanthraquinone) with boric acid and mercuric oxide in sulfuric acid at 250 °C (482 °F). [3]
Esters of octahydroxyanthraquinone, where all eight hydroxyls are replaced by straight-chain 1-alkanecarboxylate groups H
3C-(CH
2)n-COO-, with n between 6 and 14, are
liquid crystals and have been studied for possible
LCD applications.
[3]
Octahydroxyanthraquinone is active against the malaria parasite, but rufigallol (1,2,3,5,6,7-hexahydroxyanthraquinone) is 22 times more potent. [4]
References
- ^ Georgievics, G. v. (1911). "Darstellung und Eigenschaften des Octooxyanthrachinons". Monatshefte für Chemie. 32 (5): 347–352. doi: 10.1007/BF01518160. S2CID 97848374.
- ^ Wahl, Andre; Atack, F. W (1919) The Manufacture Of Organic Dyestuffs. G. Bell And Sons, Limited. Online version accessed on 2010-01-22.
- ^ a b Kumar, Sandeep (2008). "Rufigallol-based self-assembled supramolecular architectures". Phase Transitions. 81: 113–128. doi: 10.1080/01411590701601610. S2CID 59445187.
- ^ Winter, R (1995). "Hydroxy-anthraquinones as antimalarial agents". Bioorganic & Medicinal Chemistry Letters. 5 (17): 1927–1932. doi: 10.1016/0960-894X(95)00326-O.