-
Praseodymium(III) acetate (Pr(CH3COO)3)
-
Praseodymium(III) oxide (Pr2O3)
-
Praseodymium(III) chloride (PrCl3)
-
Praseodymium(III) nitrate (Pr(NO3)3)
-
Praseodymium(III) sulfate octahydrate (Pr2(SO4)3·8H2O)
-
Praseodymium(III) hydroxide (Pr(OH)3)
-
Praseodymium(III) phosphate (PrPO4)
Praseodymium compounds are compounds formed by the lanthanide metal praseodymium (Pr). In these compounds, praseodymium generally exhibits the +3 oxidation state, such as PrCl3, Pr(NO3)3 and Pr(CH3COO)3. However, compounds with praseodymium in the +2 and +4 oxidation states, and unlike other lanthanides, the +5 oxidation state, are also known.

Praseodymium metal reacts with all the stable halogens to form green trihalides: [1]
- 2 Pr (s) + 3 F2 (g) → 2 PrF3 (s)
- 2 Pr (s) + 3 Cl2 (g) → 2 PrCl3 (s)
- 2 Pr (s) + 3 Br2 (g) → 2 PrBr3 (s)
- 2 Pr (s) + 3 I2 (g) → 2 PrI3 (s)
Praseodymium(III) fluoride is the most stable fluoride of praseodymium. It can be prepared the reaction between praseodymium(III) nitrate and sodium fluoride will produce praseodymium(III) fluoride as a green crystalline solid. [2] Praseodymium(III) chloride is a light green solid that can be prepared by treating praseodymium metal with hydrogen chloride. [3] [4] It is usually purified by vacuum sublimation. [5] It is Lewis acidic, classified as "hard" according to the HSAB concept. Rapid heating of the hydrate may cause small amounts of hydrolysis. [5] PrCl3 forms a stable Lewis acid-base complex K2PrCl5 by reaction with potassium chloride; this compound shows interesting optical and magnetic properties. [6]
Praseodymium(III) bromide is the only stable bromide of praseodymium. It adopts the UCl3 crystal structure. [7] The praseodymium ions are 9-coordinate and adopt a tricapped trigonal prismatic geometry. [8] The praseodymium–bromine bond lengths are 3.05 Å and 3.13 Å. [9] Praseodymium(III) iodide can be prepared by heating praseodymium and iodine in an inert atmosphere produces praseodymium(III) iodide, [10] or by heating praseodymium with mercury(II) iodide. [11] It forms orthorhombic crystals which are hygroscopic. [10] It crystallizes in the PuBr3 type [11] [12] with space group Cmcm (No. 63) with a = 4.3281(6) Å, b = 14.003(6) Å and c = 9.988(3) Å. [13]
The tetrafluoride, PrF4, is also known, and is produced by reacting a mixture of sodium fluoride and praseodymium(III) fluoride with fluorine gas, producing Na2PrF6, following which sodium fluoride is removed from the reaction mixture with liquid hydrogen fluoride. [8] Additionally, praseodymium forms a bronze diiodide; like the diiodides of lanthanum, cerium, and gadolinium, it is a praseodymium(III) electride compound. [8]
Praseodymium can form many different oxides, although the only oxides that are stable at room temperature are Pr2O3, Pr6O11 and PrO2. Praseodymium(III) oxide is a green powder that forms hexagonal crystals, [14] and crystallizes in the manganese(III) oxide or bixbyite structure. [15] Praseodymium(IV) oxide can be produced by boiling Pr6O11 in water or acetic acid: [16]
- Pr6O11 + 3 H2O → 4 PrO2 + 2 Pr(OH)3
Praseodymium(III,IV) oxide is the most stable form of the praseodymium oxides at ambient temperature and pressure. [17] It is soluble in water [18] and has a cubic fluorite structure. [19] It can be prepared via solid-state methods such as thermolysis, molten salt method, calcination or precipitation. [19] [17] [20]
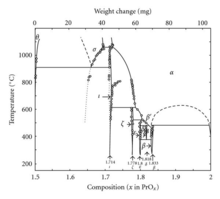
In addition to Pr6O11, praseodymium also forms a system of oxides at different phases: [21]
| Value | Formula | Phase | x in PrOx | Average oxidation state of Pr |
|---|---|---|---|---|
| 4 | Pr2O3 | θ | 1.5 | 3 |
| 7 | Pr7O12 | ι | 1.714 | 3.428 |
| 9 | Pr9O16 | ζ | 1.778 | 3.556 |
| 10 | Pr5O9 | ε | 1.8 | 3.6 |
| 11 | Pr11O20 | δ | 1.818 | 3.636 |
| 12 | Pr6O11 | β | 1.833 | 3.667 |
| ∞ | PrO2 | 2 | 4 |
Organopraseodymium compounds are compounds with a praseodymium-to- carbon bond. These compounds are very similar to those of the other lanthanides, as they all share an inability to undergo π backbonding. They are thus mostly restricted to the mostly ionic cyclopentadienides (isostructural with those of lanthanum) and the σ-bonded simple alkyls and aryls, some of which may be polymeric. [22] The coordination chemistry of praseodymium is largely that of the large, electropositive Pr3+ ion, and is thus largely similar to those of the other early lanthanides La3+, Ce3+, and Nd3+. For instance, like lanthanum, cerium, and neodymium, praseodymium nitrate forms both the 4:3 and 1:1 complexes with 18-crown-6, whereas the middle lanthanides from promethium to gadolinium can only form the 4:3 complex and the later lanthanides from terbium to lutetium cannot successfully coordinate to all the ligands. Such praseodymium complexes have high but uncertain coordination numbers and poorly defined stereochemistry, with exceptions resulting from exceptionally bulky ligands such as the tricoordinate [Pr{N(SiMe3)2}3]. There are also a few mixed oxides and fluorides involving praseodymium(IV), but it does not have an appreciable coordination chemistry in this oxidation state like its neighbour cerium. [23] However, the first example of a molecular complex of praseodymium(IV) has recently been reported. [24] Like the other organolanthanide compounds, properties of organopraseodymium compounds include:
- Organopraseodymium compounds are very air- and water-sensitive and pyrophoric.
- Chemistry in the 0 oxidation state is far more limited. In fact, their electropositive nature makes their organometallic compounds more likely to be ionic.
- Organopraseodymium compounds form no stable carbonyls at room temperature; organopraseodymium carbonyl compounds have been observed only in argon matrices, and decompose when heated to 40 K.
Metal-carbon σ bonds are found in alkyls of praeodymium such as [PrMe63− and Pr[CH(SiMe3)23.
Cyclopentadienyl complexes, are known for praseodymium. It can be produced by the following reaction scheme:
- 3 Na[Cp] + PrCl3 → Pr[Cp]3 + 3 NaCl
These compounds are of limited use and academic interest. [25]
Praseodymium(III) nitride is used in high-end electric and semiconductor products, and as a raw material to produce phosphor. Also it is used as a magnetic material and sputtering target material. [26] Many praseodymium compounds, such as praseodymium(III) oxalate, are used to colour some glasses and enamels. If mixed with certain other materials, praseodymium(III) oxalate paints glass intense yellow. [27]
Praseodymium(III,IV) oxide has a number of potential applications in chemical catalysis, and is often used in conjunction with a promoter such as sodium or gold to improve its catalytic performance. It has a high-K dielectric constant of around 30 and very low leakage currents [28] which have also made it a promising material for many potential applications in nanodevices and microelectronics. [20]
- ^ "Chemical reactions of Praseodymium". Webelements. Retrieved 9 July 2016.
-
^ Lin Ma, Wei-Xiang Chen, Yi-Fan Zheng, Jie Zhao, Zhude Xu (May 2007).
"Microwave-assisted hydrothermal synthesis and characterizations of PrF3 hollow nanoparticles".
Materials Letters. 61 (13): 2765–2768.
doi:
10.1016/j.matlet.2006.04.124. Retrieved 2019-03-26.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ J. Cybinska, J. Sokolnicki, J. Legendziewicz, G. Meyer, Journal of Alloys and Compounds, 341, 115–123 (2002).
- ^ L. F. Druding, J. D. Corbett, "Lower Oxidation States of the Lanthanides. Neodymium(II) Chloride and Iodide", J. Am. Chem. Soc. 83, 2462 (1961); J. D. Corbett, Rev. Chim. Minerale 10, 239 (1973),
- ^ a b F. T. Edelmann, P. Poremba, in: Synthetic Methods of Organometallic and Inorganic Chemistry, (W. A. Herrmann, ed.), Vol. 6, Georg Thieme Verlag, Stuttgart, 1997.
- ^ J. Cybinska, J. Sokolnicki, J. Legendziewicz, G. Meyer, Journal of Alloys and Compounds, 341, 115–123 (2002).
- ^ Wells, A. F. (1984). Structural Inorganic Chemistry (5th ed.). Oxford University Press. p. 421. ISBN 978-0-19-965763-6.
- ^ a b c Greenwood and Earnshaw, p. 1240–2
- ^ Schmid, B.; Hälg, B.; Furrer, A. (1987). "Structure and crystal fields of PrBr3 and PrCl3: A neutron study". J. Appl. Phys. 61 (8): 3426–3428. Bibcode: 1987JAP....61.3426S. doi: 10.1063/1.338741.
- ^ a b Haynes, William M. (2016-06-22). CRC Handbook of Chemistry and Physics. CRC Press. pp. 2016–2652. ISBN 978-1-4987-5429-3.
- ^ a b Asprey, L. B.; Keenan, T. K.; Kruse, F. H. (1964). "Preparation and Crystal Data for Lanthanide and Actinide Triiodides". Inorg. Chem. 3 (8): 1137–1141. doi: 10.1021/ic50018a015.
- ^ Wells, A. F. (1984). Structural Inorganic Chemistry (5th ed.). Oxford University Press. p. 421. ISBN 978-0-19-965763-6.
- ^ E. Warkentin, H. Bärnighausen (1979), "Die Kristallstruktur von Praseodymdiiodid (Modifikation V)", Zeitschrift für anorganische und allgemeine Chemie (in German), vol. 459, no. 1, pp. 187–200, doi: 10.1002/zaac.19794590120
- ^ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, pp. 478, 523, ISBN 0-8493-0594-2
- ^ Dabrowski, Jarek; Weber, Eicke R. (2004), Predictive Simulation of Semiconductor Processing, Springer, p. 264, ISBN 978-3-540-20481-7, retrieved 2009-03-18
- ^ Chinese: 《无机化学丛书》.第七卷 钪 稀土元素. 科学出版社. 1.3.4 氧化态+4的化合物. P193~195
- ^ a b Zinatloo-Ajabshir, Sahar; Salavati-Niasari, Masoud (2015). "Novel poly(ethyleneglycol)-assisted synthesis of praseodymium oxide nanostructures via a facile precipitation route". Ceramics International. 41 (1): 567–575. doi: 10.1016/j.ceramint.2014.08.105.
- ^ "Praseodymium Oxide (Pr6O11)". www.reade.com. Retrieved 2018-03-15.
- ^ a b Matović, Branko; Pantić, Jelena; Prekajski, Marija; Stanković, Nadežda; Bučevac, Dušan; Minović, Tamara; Čebela, Maria (2013). "Synthesis and characterization of Pr6O11 nanopowders". Ceramics International. 39 (3): 3151–3155. doi: 10.1016/j.ceramint.2012.09.098.
- ^ a b Shamshi Hassan, M., Shaheer Akhtar, M., Shim, KB. et al. Morphological and Electrochemical Properties of Crystalline Praseodymium Oxide Nanorods. Nanoscale Res Lett 5, 735 (2010). https://doi.org/10.1007/s11671-010-9547-8
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 643–4. ISBN 978-0-08-037941-8.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1248–9. ISBN 978-0-08-037941-8.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1244–8. ISBN 978-0-08-037941-8.
- ^ Willauer, A.R.; Palumbo, C.T.; Fadaei-Tirani, F.; Zivkovic, I.; Douair, I.; Maron, L.; Mazzanti, M. (2020). "Accessing the +IV Oxidation State in Molecular Complexes of Praseodymium". Journal of the American Chemical Society. 142 (12): 489–493. doi: 10.1021/jacs.0c01204. PMID 32134644. S2CID 212564931.
- ^ J. J. Zuckerman (17 September 2009). Inorganic Reactions and Methods, The Formation of Bonds to Elements of Group IVB (C, Si, Ge, Sn, Pb). John Wiley & Sons. ISBN 978-0-470-14547-0. Retrieved 28 July 2013.
- ^ "Praseodymium Nitride (PrN) Powder". Stanford Advanced Materials. Retrieved 18 June 2021.
- ^ "Praseodymium Oxalate 99%-99.999% from Metall Rare Earth Limited". metall.com.cn. Retrieved 17 June 2021.
- ^ Osten HJ, Liu JP, P Gaworzewski, E Bugiel, Zaumseil P: IEDM Technical Digest 653. 2000.
Praseodymium compounds are compounds formed by the lanthanide metal praseodymium (Pr). In these compounds, praseodymium generally exhibits the +3 oxidation state, such as PrCl3, Pr(NO3)3 and Pr(CH3COO)3. However, compounds with praseodymium in the +2 and +4 oxidation states, and unlike other lanthanides, the +5 oxidation state, are also known.

Praseodymium metal reacts with all the stable halogens to form green trihalides: [1]
- 2 Pr (s) + 3 F2 (g) → 2 PrF3 (s)
- 2 Pr (s) + 3 Cl2 (g) → 2 PrCl3 (s)
- 2 Pr (s) + 3 Br2 (g) → 2 PrBr3 (s)
- 2 Pr (s) + 3 I2 (g) → 2 PrI3 (s)
Praseodymium(III) fluoride is the most stable fluoride of praseodymium. It can be prepared the reaction between praseodymium(III) nitrate and sodium fluoride will produce praseodymium(III) fluoride as a green crystalline solid. [2] Praseodymium(III) chloride is a light green solid that can be prepared by treating praseodymium metal with hydrogen chloride. [3] [4] It is usually purified by vacuum sublimation. [5] It is Lewis acidic, classified as "hard" according to the HSAB concept. Rapid heating of the hydrate may cause small amounts of hydrolysis. [5] PrCl3 forms a stable Lewis acid-base complex K2PrCl5 by reaction with potassium chloride; this compound shows interesting optical and magnetic properties. [6]
Praseodymium(III) bromide is the only stable bromide of praseodymium. It adopts the UCl3 crystal structure. [7] The praseodymium ions are 9-coordinate and adopt a tricapped trigonal prismatic geometry. [8] The praseodymium–bromine bond lengths are 3.05 Å and 3.13 Å. [9] Praseodymium(III) iodide can be prepared by heating praseodymium and iodine in an inert atmosphere produces praseodymium(III) iodide, [10] or by heating praseodymium with mercury(II) iodide. [11] It forms orthorhombic crystals which are hygroscopic. [10] It crystallizes in the PuBr3 type [11] [12] with space group Cmcm (No. 63) with a = 4.3281(6) Å, b = 14.003(6) Å and c = 9.988(3) Å. [13]
The tetrafluoride, PrF4, is also known, and is produced by reacting a mixture of sodium fluoride and praseodymium(III) fluoride with fluorine gas, producing Na2PrF6, following which sodium fluoride is removed from the reaction mixture with liquid hydrogen fluoride. [8] Additionally, praseodymium forms a bronze diiodide; like the diiodides of lanthanum, cerium, and gadolinium, it is a praseodymium(III) electride compound. [8]
Praseodymium can form many different oxides, although the only oxides that are stable at room temperature are Pr2O3, Pr6O11 and PrO2. Praseodymium(III) oxide is a green powder that forms hexagonal crystals, [14] and crystallizes in the manganese(III) oxide or bixbyite structure. [15] Praseodymium(IV) oxide can be produced by boiling Pr6O11 in water or acetic acid: [16]
- Pr6O11 + 3 H2O → 4 PrO2 + 2 Pr(OH)3
Praseodymium(III,IV) oxide is the most stable form of the praseodymium oxides at ambient temperature and pressure. [17] It is soluble in water [18] and has a cubic fluorite structure. [19] It can be prepared via solid-state methods such as thermolysis, molten salt method, calcination or precipitation. [19] [17] [20]

In addition to Pr6O11, praseodymium also forms a system of oxides at different phases: [21]
| Value | Formula | Phase | x in PrOx | Average oxidation state of Pr |
|---|---|---|---|---|
| 4 | Pr2O3 | θ | 1.5 | 3 |
| 7 | Pr7O12 | ι | 1.714 | 3.428 |
| 9 | Pr9O16 | ζ | 1.778 | 3.556 |
| 10 | Pr5O9 | ε | 1.8 | 3.6 |
| 11 | Pr11O20 | δ | 1.818 | 3.636 |
| 12 | Pr6O11 | β | 1.833 | 3.667 |
| ∞ | PrO2 | 2 | 4 |
Organopraseodymium compounds are compounds with a praseodymium-to- carbon bond. These compounds are very similar to those of the other lanthanides, as they all share an inability to undergo π backbonding. They are thus mostly restricted to the mostly ionic cyclopentadienides (isostructural with those of lanthanum) and the σ-bonded simple alkyls and aryls, some of which may be polymeric. [22] The coordination chemistry of praseodymium is largely that of the large, electropositive Pr3+ ion, and is thus largely similar to those of the other early lanthanides La3+, Ce3+, and Nd3+. For instance, like lanthanum, cerium, and neodymium, praseodymium nitrate forms both the 4:3 and 1:1 complexes with 18-crown-6, whereas the middle lanthanides from promethium to gadolinium can only form the 4:3 complex and the later lanthanides from terbium to lutetium cannot successfully coordinate to all the ligands. Such praseodymium complexes have high but uncertain coordination numbers and poorly defined stereochemistry, with exceptions resulting from exceptionally bulky ligands such as the tricoordinate [Pr{N(SiMe3)2}3]. There are also a few mixed oxides and fluorides involving praseodymium(IV), but it does not have an appreciable coordination chemistry in this oxidation state like its neighbour cerium. [23] However, the first example of a molecular complex of praseodymium(IV) has recently been reported. [24] Like the other organolanthanide compounds, properties of organopraseodymium compounds include:
- Organopraseodymium compounds are very air- and water-sensitive and pyrophoric.
- Chemistry in the 0 oxidation state is far more limited. In fact, their electropositive nature makes their organometallic compounds more likely to be ionic.
- Organopraseodymium compounds form no stable carbonyls at room temperature; organopraseodymium carbonyl compounds have been observed only in argon matrices, and decompose when heated to 40 K.
Metal-carbon σ bonds are found in alkyls of praeodymium such as [PrMe63− and Pr[CH(SiMe3)23.
Cyclopentadienyl complexes, are known for praseodymium. It can be produced by the following reaction scheme:
- 3 Na[Cp] + PrCl3 → Pr[Cp]3 + 3 NaCl
These compounds are of limited use and academic interest. [25]
Praseodymium(III) nitride is used in high-end electric and semiconductor products, and as a raw material to produce phosphor. Also it is used as a magnetic material and sputtering target material. [26] Many praseodymium compounds, such as praseodymium(III) oxalate, are used to colour some glasses and enamels. If mixed with certain other materials, praseodymium(III) oxalate paints glass intense yellow. [27]
Praseodymium(III,IV) oxide has a number of potential applications in chemical catalysis, and is often used in conjunction with a promoter such as sodium or gold to improve its catalytic performance. It has a high-K dielectric constant of around 30 and very low leakage currents [28] which have also made it a promising material for many potential applications in nanodevices and microelectronics. [20]
-
Praseodymium(III) acetate (Pr(CH3COO)3)
-
Praseodymium(III) oxide (Pr2O3)
-
Praseodymium(III) chloride (PrCl3)
-
Praseodymium(III) nitrate (Pr(NO3)3)
-
Praseodymium(III) sulfate octahydrate (Pr2(SO4)3·8H2O)
-
Praseodymium(III) hydroxide (Pr(OH)3)
-
Praseodymium(III) phosphate (PrPO4)
- ^ "Chemical reactions of Praseodymium". Webelements. Retrieved 9 July 2016.
-
^ Lin Ma, Wei-Xiang Chen, Yi-Fan Zheng, Jie Zhao, Zhude Xu (May 2007).
"Microwave-assisted hydrothermal synthesis and characterizations of PrF3 hollow nanoparticles".
Materials Letters. 61 (13): 2765–2768.
doi:
10.1016/j.matlet.2006.04.124. Retrieved 2019-03-26.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ J. Cybinska, J. Sokolnicki, J. Legendziewicz, G. Meyer, Journal of Alloys and Compounds, 341, 115–123 (2002).
- ^ L. F. Druding, J. D. Corbett, "Lower Oxidation States of the Lanthanides. Neodymium(II) Chloride and Iodide", J. Am. Chem. Soc. 83, 2462 (1961); J. D. Corbett, Rev. Chim. Minerale 10, 239 (1973),
- ^ a b F. T. Edelmann, P. Poremba, in: Synthetic Methods of Organometallic and Inorganic Chemistry, (W. A. Herrmann, ed.), Vol. 6, Georg Thieme Verlag, Stuttgart, 1997.
- ^ J. Cybinska, J. Sokolnicki, J. Legendziewicz, G. Meyer, Journal of Alloys and Compounds, 341, 115–123 (2002).
- ^ Wells, A. F. (1984). Structural Inorganic Chemistry (5th ed.). Oxford University Press. p. 421. ISBN 978-0-19-965763-6.
- ^ a b c Greenwood and Earnshaw, p. 1240–2
- ^ Schmid, B.; Hälg, B.; Furrer, A. (1987). "Structure and crystal fields of PrBr3 and PrCl3: A neutron study". J. Appl. Phys. 61 (8): 3426–3428. Bibcode: 1987JAP....61.3426S. doi: 10.1063/1.338741.
- ^ a b Haynes, William M. (2016-06-22). CRC Handbook of Chemistry and Physics. CRC Press. pp. 2016–2652. ISBN 978-1-4987-5429-3.
- ^ a b Asprey, L. B.; Keenan, T. K.; Kruse, F. H. (1964). "Preparation and Crystal Data for Lanthanide and Actinide Triiodides". Inorg. Chem. 3 (8): 1137–1141. doi: 10.1021/ic50018a015.
- ^ Wells, A. F. (1984). Structural Inorganic Chemistry (5th ed.). Oxford University Press. p. 421. ISBN 978-0-19-965763-6.
- ^ E. Warkentin, H. Bärnighausen (1979), "Die Kristallstruktur von Praseodymdiiodid (Modifikation V)", Zeitschrift für anorganische und allgemeine Chemie (in German), vol. 459, no. 1, pp. 187–200, doi: 10.1002/zaac.19794590120
- ^ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, pp. 478, 523, ISBN 0-8493-0594-2
- ^ Dabrowski, Jarek; Weber, Eicke R. (2004), Predictive Simulation of Semiconductor Processing, Springer, p. 264, ISBN 978-3-540-20481-7, retrieved 2009-03-18
- ^ Chinese: 《无机化学丛书》.第七卷 钪 稀土元素. 科学出版社. 1.3.4 氧化态+4的化合物. P193~195
- ^ a b Zinatloo-Ajabshir, Sahar; Salavati-Niasari, Masoud (2015). "Novel poly(ethyleneglycol)-assisted synthesis of praseodymium oxide nanostructures via a facile precipitation route". Ceramics International. 41 (1): 567–575. doi: 10.1016/j.ceramint.2014.08.105.
- ^ "Praseodymium Oxide (Pr6O11)". www.reade.com. Retrieved 2018-03-15.
- ^ a b Matović, Branko; Pantić, Jelena; Prekajski, Marija; Stanković, Nadežda; Bučevac, Dušan; Minović, Tamara; Čebela, Maria (2013). "Synthesis and characterization of Pr6O11 nanopowders". Ceramics International. 39 (3): 3151–3155. doi: 10.1016/j.ceramint.2012.09.098.
- ^ a b Shamshi Hassan, M., Shaheer Akhtar, M., Shim, KB. et al. Morphological and Electrochemical Properties of Crystalline Praseodymium Oxide Nanorods. Nanoscale Res Lett 5, 735 (2010). https://doi.org/10.1007/s11671-010-9547-8
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 643–4. ISBN 978-0-08-037941-8.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1248–9. ISBN 978-0-08-037941-8.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1244–8. ISBN 978-0-08-037941-8.
- ^ Willauer, A.R.; Palumbo, C.T.; Fadaei-Tirani, F.; Zivkovic, I.; Douair, I.; Maron, L.; Mazzanti, M. (2020). "Accessing the +IV Oxidation State in Molecular Complexes of Praseodymium". Journal of the American Chemical Society. 142 (12): 489–493. doi: 10.1021/jacs.0c01204. PMID 32134644. S2CID 212564931.
- ^ J. J. Zuckerman (17 September 2009). Inorganic Reactions and Methods, The Formation of Bonds to Elements of Group IVB (C, Si, Ge, Sn, Pb). John Wiley & Sons. ISBN 978-0-470-14547-0. Retrieved 28 July 2013.
- ^ "Praseodymium Nitride (PrN) Powder". Stanford Advanced Materials. Retrieved 18 June 2021.
- ^ "Praseodymium Oxalate 99%-99.999% from Metall Rare Earth Limited". metall.com.cn. Retrieved 17 June 2021.
- ^ Osten HJ, Liu JP, P Gaworzewski, E Bugiel, Zaumseil P: IEDM Technical Digest 653. 2000.






