| |
| Names | |
|---|---|
| Other names
platinum diacetate, platinum acetate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| |
| |
| Properties | |
| Pt(CH3CO2)2 | |
| Molar mass | 315.19 g/mol |
| Appearance | purple solid |
| Density | 3.374 g/cm3 |
| Melting point | 245 °C (473 °F; 518 K) decomposition |
| chloroform | |
| Structure [1] | |
| tetragonal | |
| P43212, No. 96 | |
a = 10.254 Å, c = 50.494 Å
| |
Formula units (Z)
|
8 tetrameric molecules |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
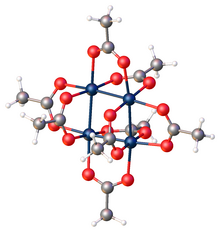
Platinum(II) acetate is a purple-colored coordination complex. The complex adopts an unusual structure consisting of a square array of Pt atoms.
Preparation
Several syntheses of platinum(II) acetate have been reported. Geoffrey Wilkinson et al. reported a synthesis from sodium hexahydroxyplatinate, nitric acid, and acetic acid. This intermediate solution was reducted with formic acid. The procedure is not highly reproducible. [2]
Alternatively, the complex can be prepared by the reaction of silver acetate with platinum(II) chloride. [3]
Structure
According to X-ray crystallography, the complex is tetrameric, in contrast to the trimeric palladium analog. [4] The four platinum atoms form a square cluster, with eight bridging acetate ligands surrounding them. The compound has slight distortions from idealized D2d symmetry. The crystal is tetragonal. [1]
References
- ^ a b de C. T. Carrondo, Maria A. A. F.; Skapski, Andrzej C. (1976). "X-Ray crystal structure of tetrameric platinum(II) acetate: a square-cluster complex with short Pt–Pt bonds and octahedral co-ordination geometry". J. Chem. Soc., Chem. Commun. (11): 410–411. doi: 10.1039/C39760000410. ISSN 0022-4936.
- ^ T. A. Stephenson; S. M. Morehouse; A. R. Powell; J. P. Heffer; G. Wilkinson (1965). "Carboxylates of Palladium, Platinum, and Rhodium, and Their Adducts". Journal of the Chemical Society: 3632–3640. doi: 10.1039/jr9650003632.
- ^ Marino Basato; Andrea Biffis; Gianluca Martinati; Cristina Tubaro; Alfonso Venzo; Paolo Ganis; Franco Benetollo (2003). "Reaction of Platinum Acetate with Phosphines and Molecular Structure of trans-[Pt(OAc)2(PPh3)2]". Inorganica Chimica Acta. 355: 399–403. doi: 10.1016/S0020-1693(03)00314-1.
- ^ Markov, Alexander A.; Yakushev, Ilya A.; Churakov, Andrey V.; Khrustalev, Victor N.; Cherkashina, Natalia V.; Stolarov, Igor P.; Gekhman, Alexander E.; Vargaftik, Michael N. (2019). "Structure and Quantum Chemical Study of Crystalline Platinum(II) Acetate". Mendeleev Communications. 29 (5): 489–491. doi: 10.1016/j.mencom.2019.09.003.

| |
| Names | |
|---|---|
| Other names
platinum diacetate, platinum acetate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
| |
| |
| Properties | |
| Pt(CH3CO2)2 | |
| Molar mass | 315.19 g/mol |
| Appearance | purple solid |
| Density | 3.374 g/cm3 |
| Melting point | 245 °C (473 °F; 518 K) decomposition |
| chloroform | |
| Structure [1] | |
| tetragonal | |
| P43212, No. 96 | |
a = 10.254 Å, c = 50.494 Å
| |
Formula units (Z)
|
8 tetrameric molecules |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Platinum(II) acetate is a purple-colored coordination complex. The complex adopts an unusual structure consisting of a square array of Pt atoms.
Preparation
Several syntheses of platinum(II) acetate have been reported. Geoffrey Wilkinson et al. reported a synthesis from sodium hexahydroxyplatinate, nitric acid, and acetic acid. This intermediate solution was reducted with formic acid. The procedure is not highly reproducible. [2]
Alternatively, the complex can be prepared by the reaction of silver acetate with platinum(II) chloride. [3]
Structure
According to X-ray crystallography, the complex is tetrameric, in contrast to the trimeric palladium analog. [4] The four platinum atoms form a square cluster, with eight bridging acetate ligands surrounding them. The compound has slight distortions from idealized D2d symmetry. The crystal is tetragonal. [1]
References
- ^ a b de C. T. Carrondo, Maria A. A. F.; Skapski, Andrzej C. (1976). "X-Ray crystal structure of tetrameric platinum(II) acetate: a square-cluster complex with short Pt–Pt bonds and octahedral co-ordination geometry". J. Chem. Soc., Chem. Commun. (11): 410–411. doi: 10.1039/C39760000410. ISSN 0022-4936.
- ^ T. A. Stephenson; S. M. Morehouse; A. R. Powell; J. P. Heffer; G. Wilkinson (1965). "Carboxylates of Palladium, Platinum, and Rhodium, and Their Adducts". Journal of the Chemical Society: 3632–3640. doi: 10.1039/jr9650003632.
- ^ Marino Basato; Andrea Biffis; Gianluca Martinati; Cristina Tubaro; Alfonso Venzo; Paolo Ganis; Franco Benetollo (2003). "Reaction of Platinum Acetate with Phosphines and Molecular Structure of trans-[Pt(OAc)2(PPh3)2]". Inorganica Chimica Acta. 355: 399–403. doi: 10.1016/S0020-1693(03)00314-1.
- ^ Markov, Alexander A.; Yakushev, Ilya A.; Churakov, Andrey V.; Khrustalev, Victor N.; Cherkashina, Natalia V.; Stolarov, Igor P.; Gekhman, Alexander E.; Vargaftik, Michael N. (2019). "Structure and Quantum Chemical Study of Crystalline Platinum(II) Acetate". Mendeleev Communications. 29 (5): 489–491. doi: 10.1016/j.mencom.2019.09.003.