| |
| Names | |
|---|---|
|
Systematic IUPAC name
pentamethyl-λ5-stibane | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C5H15Sb | |
| Molar mass | 196.935 g·mol−1 |
| Appearance | Colourless liquid |
| Melting point | −19 °C (−2 °F; 254 K) |
| Boiling point | 160 °C (320 °F; 433 K) |
| Related compounds | |
Related compounds
|
Trimethylstibine Pentamethylarsenic Pentamethylbismuth Pentamethyltantalum Pentaphenylantimony |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
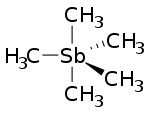
Pentamethylantimony or pentamethylstiborane is an organometalllic compound containing five methyl groups bound to an antimony atom with formula Sb(CH3)5. It is an example of a hypervalent compound. The molecular shape is trigonal bipyramid. [1] Some other antimony(V) organometallic compounds include pentapropynylantimony (Sb(CCCH3)5) and pentaphenyl antimony (Sb(C6H5)5). [2] Other known pentamethyl-pnictides include pentamethylbismuth and pentamethylarsenic.
Pentamethylantimony can be made by reacting Sb(CH3)3Br2 with two equivalents of methyl lithium. [3] Another production route is to convert trimethylstibine to the trimethyl antimony dichloride, and then replace the chlorine with methyl groups with methyl lithium. [2]
- Sb(CH3)3 + Cl2 → Sb(CH3)3Cl2
- Sb(CH3)3Cl2 + 2LiCH3 → Sb(CH3)5 + 2LiCl
Pentamethylantimony is colourless. [3] At -143 °C it crystallizes in the orthorhombic system with space group Ccmm. Unit cell dimensions are a=6.630 Å b=11.004 Å c=11.090 Å. There are four formula per unit cell. Unit cell volume is 809.1 Å3. [3] The trigonal bipyramid shape has three equatorial positions for carbon, and two axial positions at the peaks of the pyramids. The length of the antimony-carbon bond is around 214 pm for equatorial methyl groups and 222 pm for the axial positions. The bond angles are 120° for ∠C-Sb-C across the equator, and 90° for ∠C-Sb-C between equator and axis. [3] The molecules rapidly change carbon atom position, so that in NMR spectrum as low as −100 °C, there is only one kind of hydrogen position. [2]
Pentamethylantimony is more stable than pentamethylbismuth, because in lower energy trimethylbismuth, the non-bonding pair of electrons is more shielded due to the f-electrons and the lanthanoid contraction. Trimethylantimony is higher in energy, and thus less is released in a decomposition of pentamethylantimony. [3] Pentamethylantimony can be stored as a liquid in clean glass at room temperature. [4]
Pentamethylantimony melts at -19 °C. Although it decomposes when boiling is attempted and can explode, it has a high vapour pressure at 8 mmHg at 25 °C. [4]
There are two absorption bands in the ultraviolet at 2380 and 2500 Å. [4]
Pentamethylantimony reacts with methyl lithium to yield a colourless lithium hexamethylantimonate in tetrahydrofuran. [3]
- Sb(CH3)5 + LiCH3 → Li(thf)Sb(CH3)6
Pentamethylantimony reacts with silsesquioxanes to yield tetramethylstibonium silsesquioxanes. eg (cyclo-C6H11)7Si7O9(OH)3 yields (cyclo-C6H11)7Si7O9(OSb(CH3)4)3. The reaction happens quickly when there are more than two OH groups. [5]
Phosphonic acids and phosphinic acids combine with pentamethylantimony to yield compounds like (CH3)4SbOP(O)Ph2, (CH3)4SbOP(O)(OH)Ph and (CH3)4SbOP(O)(OH)3, eliminating methane. [6]
Stannocene Sn(C5H5)2 combines with pentamethylantimony to produce bis(tetramethylstibonium)tetracyclopentadienylstannate ([(CH3)4Sb]2Sn(C5H5)4). [7]
Pentamethylantimony reacts with many very weak acids to form a tetramethylstibonium salt or tetramethylstibonium derivative with the acid. Such acids include water (H2O), alcohols, thiols, phenol, carboxylic acids, hydrogen fluoride, thiocyanic acid, hydrazoic acid, difluorophosphoric acid, thiophosphinic acids, and alkylsilols. [8]
With halogens, pentamethylantimony has one or two methyl groups replaced by the halogen atoms. [8] Lewis acids also react to form tetramethyl stibonium salts, including [(CH3)4Sb]TlBr4, [(CH3)4Sb][CH3SbCl5], [8]
Pentamethylantimony reacts with the surface of silica to coat it with Si-O-Sb(CH3)4 groups. Over 250 °C this decomposes to Sb(CH3) and leaves methyl groups attached to the silica surface. [9]
- ^ Greene, Tim M.; Downs, Anthony J.; Pulham, Colin R.; Haaland, Arne; Verne, Hans Peter; Volden, Hans Vidar; Timofeeva, Tatjana V. (November 1998). "Molecular Structures of Pentamethylarsenic(V) and Trimethyldichloroarsenic(V) by Gas Electron Diffraction and ab Initio Calculations:? Molecular Mechanics Calculations on Pentamethylarsenic(V), Pentaphenylarsenic(V), and Related Compounds". Organometallics. 17 (24): 5287–5293. doi: 10.1021/om980520r.
- ^ a b c Haaland, Arne; Hammel, Andreas; Rypdal, Kristin; Swang, Ole; Brunvoll, Jon; Gropen, Odd; Greune, Michael; Weidlein, Johann; Nasiri, Ahmad; Okada, Yoshito (1993). "Molecular Structure of Pentamethylantimony by Gas Electron Diffraction; Structure and Bonding in Sb(CH3)5 and Bi(CH3)5 Studied by Ab Initio MO Calculations". Acta Chemica Scandinavica. 47: 368–373. doi: 10.3891/acta.chem.scand.47-0368.
- ^ a b c d e f Wallenhauer, Stephan; Seppelt, Konrad (January 1995). "Antimony(V) and Bismuth(V) Methyl Compounds: A Structural Comparison". Inorganic Chemistry. 34 (1): 116–119. doi: 10.1021/ic00105a021.
- ^ a b c Downs, A. J.; Schmutzler, R.; Steer, I. A. (1966). "The vibrational spectrum and structure of pentamethylantimony". Chemical Communications (8): 221. doi: 10.1039/C19660000221.
- ^ Feher, Frank J.; Budzichowski, Theodore A.; Rahimian, Kamyar; Ziller, Joseph W. (May 1992). "Reactions of incompletely-condensed silsesquioxanes with pentamethylantimony: a new synthesis of metallasilsesquioxanes with important implications for the chemistry of silica surfaces". Journal of the American Chemical Society. 114 (10): 3859–3866. doi: 10.1021/ja00036a038.
- ^ Graves, Guy E.; Van Wazer, John R. (May 1978). "Methyl group replacement on pentamethylantimony with organophosphorus substituents". Journal of Organometallic Chemistry. 150 (2): 233–237. doi: 10.1016/S0022-328X(00)84725-7.
- ^ Bos, Klaas D.; Bulten, Eric J.; Meinema, Harry A.; Noltes, Jan G. (20 March 1979). "Synthesis of bis(tetramethylstibonium)tetracyclopentadienylstannate a novel type of organotin(II) compound". Journal of Organometallic Chemistry. 168 (2): 159–162. doi: 10.1016/s0022-328x(00)83270-2. hdl: 1874/25359.
- ^ a b c Hubert Schmidbaur (1976). Advances in Organometallic Chemistry. Academic Press. ISBN 9780080580159.
- ^ Wang, Y.; Morrow, B. A. (January 1996). "Infrared Study of the Chemisorption of Pentamethylantimony on Silica". Langmuir. 12 (17): 4153–4157. doi: 10.1021/la951514s.

| |
| Names | |
|---|---|
|
Systematic IUPAC name
pentamethyl-λ5-stibane | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C5H15Sb | |
| Molar mass | 196.935 g·mol−1 |
| Appearance | Colourless liquid |
| Melting point | −19 °C (−2 °F; 254 K) |
| Boiling point | 160 °C (320 °F; 433 K) |
| Related compounds | |
Related compounds
|
Trimethylstibine Pentamethylarsenic Pentamethylbismuth Pentamethyltantalum Pentaphenylantimony |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Pentamethylantimony or pentamethylstiborane is an organometalllic compound containing five methyl groups bound to an antimony atom with formula Sb(CH3)5. It is an example of a hypervalent compound. The molecular shape is trigonal bipyramid. [1] Some other antimony(V) organometallic compounds include pentapropynylantimony (Sb(CCCH3)5) and pentaphenyl antimony (Sb(C6H5)5). [2] Other known pentamethyl-pnictides include pentamethylbismuth and pentamethylarsenic.
Pentamethylantimony can be made by reacting Sb(CH3)3Br2 with two equivalents of methyl lithium. [3] Another production route is to convert trimethylstibine to the trimethyl antimony dichloride, and then replace the chlorine with methyl groups with methyl lithium. [2]
- Sb(CH3)3 + Cl2 → Sb(CH3)3Cl2
- Sb(CH3)3Cl2 + 2LiCH3 → Sb(CH3)5 + 2LiCl
Pentamethylantimony is colourless. [3] At -143 °C it crystallizes in the orthorhombic system with space group Ccmm. Unit cell dimensions are a=6.630 Å b=11.004 Å c=11.090 Å. There are four formula per unit cell. Unit cell volume is 809.1 Å3. [3] The trigonal bipyramid shape has three equatorial positions for carbon, and two axial positions at the peaks of the pyramids. The length of the antimony-carbon bond is around 214 pm for equatorial methyl groups and 222 pm for the axial positions. The bond angles are 120° for ∠C-Sb-C across the equator, and 90° for ∠C-Sb-C between equator and axis. [3] The molecules rapidly change carbon atom position, so that in NMR spectrum as low as −100 °C, there is only one kind of hydrogen position. [2]
Pentamethylantimony is more stable than pentamethylbismuth, because in lower energy trimethylbismuth, the non-bonding pair of electrons is more shielded due to the f-electrons and the lanthanoid contraction. Trimethylantimony is higher in energy, and thus less is released in a decomposition of pentamethylantimony. [3] Pentamethylantimony can be stored as a liquid in clean glass at room temperature. [4]
Pentamethylantimony melts at -19 °C. Although it decomposes when boiling is attempted and can explode, it has a high vapour pressure at 8 mmHg at 25 °C. [4]
There are two absorption bands in the ultraviolet at 2380 and 2500 Å. [4]
Pentamethylantimony reacts with methyl lithium to yield a colourless lithium hexamethylantimonate in tetrahydrofuran. [3]
- Sb(CH3)5 + LiCH3 → Li(thf)Sb(CH3)6
Pentamethylantimony reacts with silsesquioxanes to yield tetramethylstibonium silsesquioxanes. eg (cyclo-C6H11)7Si7O9(OH)3 yields (cyclo-C6H11)7Si7O9(OSb(CH3)4)3. The reaction happens quickly when there are more than two OH groups. [5]
Phosphonic acids and phosphinic acids combine with pentamethylantimony to yield compounds like (CH3)4SbOP(O)Ph2, (CH3)4SbOP(O)(OH)Ph and (CH3)4SbOP(O)(OH)3, eliminating methane. [6]
Stannocene Sn(C5H5)2 combines with pentamethylantimony to produce bis(tetramethylstibonium)tetracyclopentadienylstannate ([(CH3)4Sb]2Sn(C5H5)4). [7]
Pentamethylantimony reacts with many very weak acids to form a tetramethylstibonium salt or tetramethylstibonium derivative with the acid. Such acids include water (H2O), alcohols, thiols, phenol, carboxylic acids, hydrogen fluoride, thiocyanic acid, hydrazoic acid, difluorophosphoric acid, thiophosphinic acids, and alkylsilols. [8]
With halogens, pentamethylantimony has one or two methyl groups replaced by the halogen atoms. [8] Lewis acids also react to form tetramethyl stibonium salts, including [(CH3)4Sb]TlBr4, [(CH3)4Sb][CH3SbCl5], [8]
Pentamethylantimony reacts with the surface of silica to coat it with Si-O-Sb(CH3)4 groups. Over 250 °C this decomposes to Sb(CH3) and leaves methyl groups attached to the silica surface. [9]
- ^ Greene, Tim M.; Downs, Anthony J.; Pulham, Colin R.; Haaland, Arne; Verne, Hans Peter; Volden, Hans Vidar; Timofeeva, Tatjana V. (November 1998). "Molecular Structures of Pentamethylarsenic(V) and Trimethyldichloroarsenic(V) by Gas Electron Diffraction and ab Initio Calculations:? Molecular Mechanics Calculations on Pentamethylarsenic(V), Pentaphenylarsenic(V), and Related Compounds". Organometallics. 17 (24): 5287–5293. doi: 10.1021/om980520r.
- ^ a b c Haaland, Arne; Hammel, Andreas; Rypdal, Kristin; Swang, Ole; Brunvoll, Jon; Gropen, Odd; Greune, Michael; Weidlein, Johann; Nasiri, Ahmad; Okada, Yoshito (1993). "Molecular Structure of Pentamethylantimony by Gas Electron Diffraction; Structure and Bonding in Sb(CH3)5 and Bi(CH3)5 Studied by Ab Initio MO Calculations". Acta Chemica Scandinavica. 47: 368–373. doi: 10.3891/acta.chem.scand.47-0368.
- ^ a b c d e f Wallenhauer, Stephan; Seppelt, Konrad (January 1995). "Antimony(V) and Bismuth(V) Methyl Compounds: A Structural Comparison". Inorganic Chemistry. 34 (1): 116–119. doi: 10.1021/ic00105a021.
- ^ a b c Downs, A. J.; Schmutzler, R.; Steer, I. A. (1966). "The vibrational spectrum and structure of pentamethylantimony". Chemical Communications (8): 221. doi: 10.1039/C19660000221.
- ^ Feher, Frank J.; Budzichowski, Theodore A.; Rahimian, Kamyar; Ziller, Joseph W. (May 1992). "Reactions of incompletely-condensed silsesquioxanes with pentamethylantimony: a new synthesis of metallasilsesquioxanes with important implications for the chemistry of silica surfaces". Journal of the American Chemical Society. 114 (10): 3859–3866. doi: 10.1021/ja00036a038.
- ^ Graves, Guy E.; Van Wazer, John R. (May 1978). "Methyl group replacement on pentamethylantimony with organophosphorus substituents". Journal of Organometallic Chemistry. 150 (2): 233–237. doi: 10.1016/S0022-328X(00)84725-7.
- ^ Bos, Klaas D.; Bulten, Eric J.; Meinema, Harry A.; Noltes, Jan G. (20 March 1979). "Synthesis of bis(tetramethylstibonium)tetracyclopentadienylstannate a novel type of organotin(II) compound". Journal of Organometallic Chemistry. 168 (2): 159–162. doi: 10.1016/s0022-328x(00)83270-2. hdl: 1874/25359.
- ^ a b c Hubert Schmidbaur (1976). Advances in Organometallic Chemistry. Academic Press. ISBN 9780080580159.
- ^ Wang, Y.; Morrow, B. A. (January 1996). "Infrared Study of the Chemisorption of Pentamethylantimony on Silica". Langmuir. 12 (17): 4153–4157. doi: 10.1021/la951514s.