| |

| |
| Names | |
|---|---|
|
IUPAC name
Methaneselenol
| |
| Other names
monomethylselenide, MMSe
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| CH3SeH | |
| Molar mass | 95.014 g·mol−1 |
| Appearance | colorless gas |
| Boiling point | 12 °C (54 °F; 285 K) |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
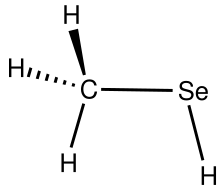
Methaneselenol is the organoselenium compound with the formula C H3 SeH. It is the simplest selenol. A colorless gas, it is notorious for its foul odor.
It is prepared by reaction of methyl lithium or a methyl Grignard reagent with selenium followed by protonation of the product. The compound is a metabolite. [1] [2]
According to IR spectroscopy, νSe-H = 2342 cm−1. For the other homologues, νE-H = 1995 (E = Te), 2606 (E = S), and 3710 cm−1 (E = O) for methanetellurol, methanethiol, and methanol. [3]
References
- ^ Zeng, Huawei; Wu, Min; Botnen, James H. (2009). "Methylselenol, a Selenium Metabolite, Induces Cell Cycle Arrest in G1 Phase and Apoptosis via the Extracellular-Regulated Kinase 1/2 Pathway and Other Cancer Signaling Genes". The Journal of Nutrition. 139 (9): 1613–1618. doi: 10.3945/jn.109.110320. PMID 19625696.
- ^ Amouroux, David; Donard, Olivier F. X. (1996). "Maritime emission of selenium to the atmosphere in Eastern Mediterranean seas". Geophysical Research Letters. 23 (14): 1777–1780. Bibcode: 1996GeoRL..23.1777A. doi: 10.1029/96GL01271.
- ^ Hamada, K.; Morishita, H. (1977). "The Synthesis and the Raman and Infrared Spectra of Methanetellurol". Synthesis and Reactivity in Inorganic and Metal-Organic Chemistry. 7 (4): 355–366. doi: 10.1080/00945717708069709.

| |

| |
| Names | |
|---|---|
|
IUPAC name
Methaneselenol
| |
| Other names
monomethylselenide, MMSe
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| CH3SeH | |
| Molar mass | 95.014 g·mol−1 |
| Appearance | colorless gas |
| Boiling point | 12 °C (54 °F; 285 K) |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Methaneselenol is the organoselenium compound with the formula C H3 SeH. It is the simplest selenol. A colorless gas, it is notorious for its foul odor.
It is prepared by reaction of methyl lithium or a methyl Grignard reagent with selenium followed by protonation of the product. The compound is a metabolite. [1] [2]
According to IR spectroscopy, νSe-H = 2342 cm−1. For the other homologues, νE-H = 1995 (E = Te), 2606 (E = S), and 3710 cm−1 (E = O) for methanetellurol, methanethiol, and methanol. [3]
References
- ^ Zeng, Huawei; Wu, Min; Botnen, James H. (2009). "Methylselenol, a Selenium Metabolite, Induces Cell Cycle Arrest in G1 Phase and Apoptosis via the Extracellular-Regulated Kinase 1/2 Pathway and Other Cancer Signaling Genes". The Journal of Nutrition. 139 (9): 1613–1618. doi: 10.3945/jn.109.110320. PMID 19625696.
- ^ Amouroux, David; Donard, Olivier F. X. (1996). "Maritime emission of selenium to the atmosphere in Eastern Mediterranean seas". Geophysical Research Letters. 23 (14): 1777–1780. Bibcode: 1996GeoRL..23.1777A. doi: 10.1029/96GL01271.
- ^ Hamada, K.; Morishita, H. (1977). "The Synthesis and the Raman and Infrared Spectra of Methanetellurol". Synthesis and Reactivity in Inorganic and Metal-Organic Chemistry. 7 (4): 355–366. doi: 10.1080/00945717708069709.