 | |
| Clinical data | |
|---|---|
| Trade names | Oxarol |
| Other names | 22-Oxacalcitriol; OCT |
|
Routes of administration | IV or topical |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.212.050 |
| Chemical and physical data | |
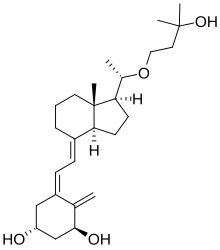
| Formula | C26H42O4 |
| Molar mass | 418.618 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Maxacalcitol (trade name Oxarol) is a vitamin D3 analog and vitamin D receptor activator (VDRA). [1] As a pharmaceutical drug, it is used as an injection to treat secondary hyperparathyroidism resulting from hemodialysis and as a topical ointment for psoriasis. [2]
It was approved for use in Japan in 2010 [3] and in Taiwan in 2018. [4]
- ^ Mizobuchi M, Ogata H (March 2014). "Clinical uses of 22-oxacalcitriol". Current Vascular Pharmacology. 12 (2): 324–328. doi: 10.2174/15701611113119990023. PMID 23713875.
- ^ "Maxacalcitol". Inxight Drugs. National Center for Advancing Translational Sciences, U.S. National Institutes of Health.
- ^ "Maxacalcitol". New Drug Approvals in Japan. Kyoto Encyclopedia of Genes and Genomes. D01098.
- ^ Fukazawa-Shinotsuka M, Saito T, Abe M, Iida S, Wang IT, Terao K, et al. (January 2022). "Maxacalcitol Pharmacokinetic-Pharmacodynamic Modeling and Simulation for Secondary Hyperparathyroidism in Patients Receiving Maintenance Hemodialysis". Drug Research. 72 (1): 23–33. doi: 10.1055/a-1581-7609. PMID 34488237.
 | |
| Clinical data | |
|---|---|
| Trade names | Oxarol |
| Other names | 22-Oxacalcitriol; OCT |
|
Routes of administration | IV or topical |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.212.050 |
| Chemical and physical data | |
| Formula | C26H42O4 |
| Molar mass | 418.618 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Maxacalcitol (trade name Oxarol) is a vitamin D3 analog and vitamin D receptor activator (VDRA). [1] As a pharmaceutical drug, it is used as an injection to treat secondary hyperparathyroidism resulting from hemodialysis and as a topical ointment for psoriasis. [2]
It was approved for use in Japan in 2010 [3] and in Taiwan in 2018. [4]
- ^ Mizobuchi M, Ogata H (March 2014). "Clinical uses of 22-oxacalcitriol". Current Vascular Pharmacology. 12 (2): 324–328. doi: 10.2174/15701611113119990023. PMID 23713875.
- ^ "Maxacalcitol". Inxight Drugs. National Center for Advancing Translational Sciences, U.S. National Institutes of Health.
- ^ "Maxacalcitol". New Drug Approvals in Japan. Kyoto Encyclopedia of Genes and Genomes. D01098.
- ^ Fukazawa-Shinotsuka M, Saito T, Abe M, Iida S, Wang IT, Terao K, et al. (January 2022). "Maxacalcitol Pharmacokinetic-Pharmacodynamic Modeling and Simulation for Secondary Hyperparathyroidism in Patients Receiving Maintenance Hemodialysis". Drug Research. 72 (1): 23–33. doi: 10.1055/a-1581-7609. PMID 34488237.