| |
| |
| Names | |
|---|---|
|
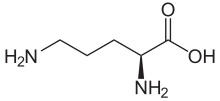
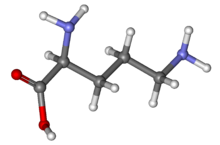
IUPAC name
L-Ornithine
| |
| Other names
(+)-(S)-2,5-Diaminovaleric acid
(+)-(S)-2,5-Diaminopentanoic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.665 |
| EC Number |
|
| KEGG | |
| MeSH | Ornithine |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties [1] | |
| C5H12N2O2 | |
| Molar mass | 132.16 g/mol |
| Melting point | 140 °C (284 °F; 413 K) |
| soluble | |
| Solubility | soluble in ethanol |
| Acidity (pKa) | 1.94 |
Chiral rotation ([α]D)
|
+11.5 (H2O, c = 6.5) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Ornithine is a non-proteinogenic α-amino acid that plays a role in the urea cycle. Ornithine is abnormally accumulated in the body in ornithine transcarbamylase deficiency. The radical is ornithyl. [2]
L-Ornithine is one of the products of the action of the enzyme
arginase on L-
arginine, creating
urea. Therefore, ornithine is a central part of the urea cycle, which allows for the disposal of excess
nitrogen. Ornithine is recycled and, in a manner, is a catalyst. First, ammonia is converted into
carbamoyl phosphate (H
2NC(O)OPO2−
3) by carbamoyl phosphate synthetase.
Ornithine transcarbamylase catalyzes the reaction between carbamoyl phosphate and ornithine to form
citrulline and phosphate (Pi). Another
amino group is added from
aspartate, producing
arginine and denitrogenated
fumarate. The resulting
arginine (a
guanidinium compound) is hydrolysed back to ornithine, producing urea. The amino groups of urea come from the ammonia and aspartate, and the nitrogen in ornithine remains intact.

Ornithine is not an amino acid coded for by DNA, that is, not proteinogenic. However, in mammalian non-hepatic tissues, the main use of the urea cycle is in arginine biosynthesis, so, as an intermediate in metabolic processes, ornithine is quite important. [4]
Ornithine, via the action of ornithine decarboxylase (E.C. 4.1.1.17), is the starting point for the synthesis of polyamines such as putrescine.
In bacteria, such as E. coli, ornithine can be synthesized from L-glutamate. [5]

L-Ornithine supplementation attenuated fatigue in subjects in a placebo-controlled study using a cycle ergometer. The results suggested that L-ornithine has an antifatigue effect in increasing the efficiency of energy consumption and promoting the excretion of ammonia. [6] [7]
Amino acid supplements, including L-ornithine, are frequently marketed to bodybuilders and weightlifters with claims for increasing levels of human growth hormone (HGH), muscle mass, and strength. A 1993 short 4-day clinical study reported that L-ornithine in combination with L-arginine and L-lysine at 2 g/d each did not increase HGH. [8] A review from 2002 on the topic concluded "The use of specific amino acids to stimulate GH release by athletes is not recommended." [9]
L-Ornithine L-aspartate (LOLA), a stable salt of ornithine and aspartic acid, has been used in the treatment of cirrhosis [10] and hepatic encephalopathy. [11]
- ^ Weast, Robert C., ed. (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, FL: CRC Press. p. C-408. ISBN 0-8493-0462-8.
- ^ Sivashanmugam M (February 2017). "Ornithine and its role in metabolic diseases: An appraisal". Biomed Pharmacother. 86: 185–194. doi: 10.1016/j.biopha.2016.12.024. PMID 27978498.
- ^ Langley DB, Templeton MD, Fields BA, Mitchell RE, Collyer CA (June 2000). "Mechanism of inactivation of ornithine transcarbamoylase by Ndelta -(N'-Sulfodiaminophosphinyl)-L-ornithine, a true transition state analogue? Crystal structure and implications for catalytic mechanism". The Journal of Biological Chemistry. 275 (26): 20012–9. doi: 10.1074/jbc.M000585200. PMID 10747936.
- ^ Weber AL, Miller SL (1981). "Reasons for the occurrence of the twenty coded protein amino acids" (PDF). Journal of Molecular Evolution. 17 (5): 273–84. Bibcode: 1981JMolE..17..273W. doi: 10.1007/BF01795749. PMID 7277510. S2CID 27957755.
-
^
"Ornithine Biosynthesis". School of Biological and Chemical Sciences, Queen Mary, University of London. Archived from
the original on 2012-04-14. Retrieved 2007-08-17.
{{ cite journal}}: Cite journal requires|journal=( help) - ^ Sugino T, Shirai T, Kajimoto Y, Kajimoto O (November 2008). "L-ornithine supplementation attenuates physical fatigue in healthy volunteers by modulating lipid and amino acid metabolism". Nutrition Research. 28 (11): 738–43. doi: 10.1016/j.nutres.2008.08.008. PMID 19083482.
- ^ Demura S, Yamada T, Yamaji S, Komatsu M, Morishita K (October 2010). "The effect of L-ornithine hydrochloride ingestion on performance during incremental exhaustive ergometer bicycle exercise and ammonia metabolism during and after exercise". European Journal of Clinical Nutrition. 64 (10): 1166–71. doi: 10.1038/ejcn.2010.149. hdl: 2297/25434. PMID 20717126.
- ^ Fogelholm GM, Näveri HK, Kiilavuori KT, Härkönen MH (September 1993). "Low-dose amino acid supplementation: no effects on serum human growth hormone and insulin in male weightlifters". International Journal of Sport Nutrition. 3 (3): 290–7. doi: 10.1123/ijsn.3.3.290. PMID 8220394.
- ^ Chromiak JA, Antonio J (2002). "Use of amino acids as growth hormone-releasing agents by athletes". Nutrition. 18 (7–8): 657–61. doi: 10.1016/s0899-9007(02)00807-9. PMID 12093449.
- ^ Sikorska H, Cianciara J, Wiercińska-Drapało A (June 2010). "[Physiological functions of L-ornithine and L-aspartate in the body and the efficacy of administration of L-ornithine-L-aspartate in conditions of relative deficiency]". Polski Merkuriusz Lekarski. 28 (168): 490–5. PMID 20642112.
- ^ Butterworth R, Mcphail M (February 2019). "L-Ornithine L-Aspartate (LOLA) for Hepatic Encephalopathy in Cirrhosis: Results of Randomized Controlled Trials and Meta-Analyses". Drugs. 79 (Suppl 1): 31–37. doi: 10.1007/s40265-018-1024-1. PMC 6416237. PMID 30706425.

| |

| |
| Names | |
|---|---|
|
IUPAC name
L-Ornithine
| |
| Other names
(+)-(S)-2,5-Diaminovaleric acid
(+)-(S)-2,5-Diaminopentanoic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.665 |
| EC Number |
|
| KEGG | |
| MeSH | Ornithine |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties [1] | |
| C5H12N2O2 | |
| Molar mass | 132.16 g/mol |
| Melting point | 140 °C (284 °F; 413 K) |
| soluble | |
| Solubility | soluble in ethanol |
| Acidity (pKa) | 1.94 |
Chiral rotation ([α]D)
|
+11.5 (H2O, c = 6.5) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Ornithine is a non-proteinogenic α-amino acid that plays a role in the urea cycle. Ornithine is abnormally accumulated in the body in ornithine transcarbamylase deficiency. The radical is ornithyl. [2]
L-Ornithine is one of the products of the action of the enzyme
arginase on L-
arginine, creating
urea. Therefore, ornithine is a central part of the urea cycle, which allows for the disposal of excess
nitrogen. Ornithine is recycled and, in a manner, is a catalyst. First, ammonia is converted into
carbamoyl phosphate (H
2NC(O)OPO2−
3) by carbamoyl phosphate synthetase.
Ornithine transcarbamylase catalyzes the reaction between carbamoyl phosphate and ornithine to form
citrulline and phosphate (Pi). Another
amino group is added from
aspartate, producing
arginine and denitrogenated
fumarate. The resulting
arginine (a
guanidinium compound) is hydrolysed back to ornithine, producing urea. The amino groups of urea come from the ammonia and aspartate, and the nitrogen in ornithine remains intact.

Ornithine is not an amino acid coded for by DNA, that is, not proteinogenic. However, in mammalian non-hepatic tissues, the main use of the urea cycle is in arginine biosynthesis, so, as an intermediate in metabolic processes, ornithine is quite important. [4]
Ornithine, via the action of ornithine decarboxylase (E.C. 4.1.1.17), is the starting point for the synthesis of polyamines such as putrescine.
In bacteria, such as E. coli, ornithine can be synthesized from L-glutamate. [5]

L-Ornithine supplementation attenuated fatigue in subjects in a placebo-controlled study using a cycle ergometer. The results suggested that L-ornithine has an antifatigue effect in increasing the efficiency of energy consumption and promoting the excretion of ammonia. [6] [7]
Amino acid supplements, including L-ornithine, are frequently marketed to bodybuilders and weightlifters with claims for increasing levels of human growth hormone (HGH), muscle mass, and strength. A 1993 short 4-day clinical study reported that L-ornithine in combination with L-arginine and L-lysine at 2 g/d each did not increase HGH. [8] A review from 2002 on the topic concluded "The use of specific amino acids to stimulate GH release by athletes is not recommended." [9]
L-Ornithine L-aspartate (LOLA), a stable salt of ornithine and aspartic acid, has been used in the treatment of cirrhosis [10] and hepatic encephalopathy. [11]
- ^ Weast, Robert C., ed. (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, FL: CRC Press. p. C-408. ISBN 0-8493-0462-8.
- ^ Sivashanmugam M (February 2017). "Ornithine and its role in metabolic diseases: An appraisal". Biomed Pharmacother. 86: 185–194. doi: 10.1016/j.biopha.2016.12.024. PMID 27978498.
- ^ Langley DB, Templeton MD, Fields BA, Mitchell RE, Collyer CA (June 2000). "Mechanism of inactivation of ornithine transcarbamoylase by Ndelta -(N'-Sulfodiaminophosphinyl)-L-ornithine, a true transition state analogue? Crystal structure and implications for catalytic mechanism". The Journal of Biological Chemistry. 275 (26): 20012–9. doi: 10.1074/jbc.M000585200. PMID 10747936.
- ^ Weber AL, Miller SL (1981). "Reasons for the occurrence of the twenty coded protein amino acids" (PDF). Journal of Molecular Evolution. 17 (5): 273–84. Bibcode: 1981JMolE..17..273W. doi: 10.1007/BF01795749. PMID 7277510. S2CID 27957755.
-
^
"Ornithine Biosynthesis". School of Biological and Chemical Sciences, Queen Mary, University of London. Archived from
the original on 2012-04-14. Retrieved 2007-08-17.
{{ cite journal}}: Cite journal requires|journal=( help) - ^ Sugino T, Shirai T, Kajimoto Y, Kajimoto O (November 2008). "L-ornithine supplementation attenuates physical fatigue in healthy volunteers by modulating lipid and amino acid metabolism". Nutrition Research. 28 (11): 738–43. doi: 10.1016/j.nutres.2008.08.008. PMID 19083482.
- ^ Demura S, Yamada T, Yamaji S, Komatsu M, Morishita K (October 2010). "The effect of L-ornithine hydrochloride ingestion on performance during incremental exhaustive ergometer bicycle exercise and ammonia metabolism during and after exercise". European Journal of Clinical Nutrition. 64 (10): 1166–71. doi: 10.1038/ejcn.2010.149. hdl: 2297/25434. PMID 20717126.
- ^ Fogelholm GM, Näveri HK, Kiilavuori KT, Härkönen MH (September 1993). "Low-dose amino acid supplementation: no effects on serum human growth hormone and insulin in male weightlifters". International Journal of Sport Nutrition. 3 (3): 290–7. doi: 10.1123/ijsn.3.3.290. PMID 8220394.
- ^ Chromiak JA, Antonio J (2002). "Use of amino acids as growth hormone-releasing agents by athletes". Nutrition. 18 (7–8): 657–61. doi: 10.1016/s0899-9007(02)00807-9. PMID 12093449.
- ^ Sikorska H, Cianciara J, Wiercińska-Drapało A (June 2010). "[Physiological functions of L-ornithine and L-aspartate in the body and the efficacy of administration of L-ornithine-L-aspartate in conditions of relative deficiency]". Polski Merkuriusz Lekarski. 28 (168): 490–5. PMID 20642112.
- ^ Butterworth R, Mcphail M (February 2019). "L-Ornithine L-Aspartate (LOLA) for Hepatic Encephalopathy in Cirrhosis: Results of Randomized Controlled Trials and Meta-Analyses". Drugs. 79 (Suppl 1): 31–37. doi: 10.1007/s40265-018-1024-1. PMC 6416237. PMID 30706425.