| |
| Names | |
|---|---|
|
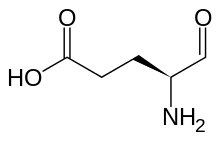
Preferred IUPAC name
(4S)-4-Amino-5-oxopentanoic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| MeSH | glutamate-1-semialdehyde |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C5H9NO3 | |
| Molar mass | 131.131 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Glutamate-1-semialdehyde is a molecule formed from by the reduction of tRNA bound glutamate, catalyzed by glutamyl-tRNA reductase. It is isomerized by glutamate-1-semialdehyde 2,1-aminomutase to give aminolevulinic acid in the biosynthesis of porphyrins, including heme and chlorophyll. [1] [2]
See also
References
- ^ Beale SI (August 1990). "Biosynthesis of the Tetrapyrrole Pigment Precursor, delta-Aminolevulinic Acid, from Glutamate". Plant Physiol. 93 (4): 1273–9. doi: 10.1104/pp.93.4.1273. PMC 1062668. PMID 16667613.
- ^ Willows, R.D. (2004). "Chlorophylls". In Goodman, Robert M. (ed.). Encyclopaedia of Plant and Crop Science. Marcel Dekker. pp. 258–262. ISBN 0-8247-4268-0.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
(4S)-4-Amino-5-oxopentanoic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| MeSH | glutamate-1-semialdehyde |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C5H9NO3 | |
| Molar mass | 131.131 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Glutamate-1-semialdehyde is a molecule formed from by the reduction of tRNA bound glutamate, catalyzed by glutamyl-tRNA reductase. It is isomerized by glutamate-1-semialdehyde 2,1-aminomutase to give aminolevulinic acid in the biosynthesis of porphyrins, including heme and chlorophyll. [1] [2]
See also
References
- ^ Beale SI (August 1990). "Biosynthesis of the Tetrapyrrole Pigment Precursor, delta-Aminolevulinic Acid, from Glutamate". Plant Physiol. 93 (4): 1273–9. doi: 10.1104/pp.93.4.1273. PMC 1062668. PMID 16667613.
- ^ Willows, R.D. (2004). "Chlorophylls". In Goodman, Robert M. (ed.). Encyclopaedia of Plant and Crop Science. Marcel Dekker. pp. 258–262. ISBN 0-8247-4268-0.