
| |
| Names | |
|---|---|
| Other names
Europium acetate
| |
| Identifiers | |
| ECHA InfoCard | 100.013.337 |
CompTox Dashboard (
EPA)
|
|
| Properties | |
| Eu(CH3COO)3 | |
| Molar mass | 329,092 g/mol (anhydrous) 341,059 g/mol (monohydrate) |
| Appearance | white solid |
| soluble in water | |
| Related compounds | |
Other
anions
|
Europium(III) oxide Europium(III) hydroxide Europium(III) carbonate |
Other
cations
|
Samarium(III) acetate Gadolinium(III) acetate |
Related compounds
|
Europium(II) acetate |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Europium(III) acetate is an inorganic salt of europium and acetic acid with the chemical formula of Eu(CH3COO)3. In this compound, europium exhibits the +3 oxidation state. It can exist in the anhydrous form, sesqui hydrate [a] [1] and tetrahydrate. [2] Its hydrate molecule is a dimer. [3] [4]
Europium acetate can be obtained by stirring reaction of acetic acid and europium oxide under heating, and then diluting with water and crystallizing: [5]
- Eu2O3 + 6 CH3COOH → 2 Eu(CH3COO)3 + 3 H2O
Europium can also directly participate in the reaction: [1]
- 2 Eu + 6 CH3COOH → 2 Eu(CH3COO)3 + 3 H2↑
This section may be too technical for most readers to understand. (May 2022) |
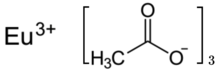
The anhydrous europium acetate crystallizes monoclinically in the space group C2/c (space group no. 15) with the lattice parameters a = 1126.0(3), b = 2900.5(6), c = 799.1( 2) pm and β = 132.03(2)° with four formula units per unit cell. [1] The sesquihydrate crystallizes monoclinically in the space group Cc (No. 9) with the lattice parameters a = 1608.7(2), b = 1665.6(2), c = 839.1(1) pm and β = 115.75( 9)° with four formula units per unit cell. [1] The heat capacity at 280 K is 803±16 J/(mol∙K). [6]
Europium acetate can be dissolved in water, acidified with acetic acid, and the compound of divalent europium [Eu(CH3COO)2(CH3COOH)(H2O)2] can be obtained by electrochemical reduction. [7]
Europium acetate can be crystallized in excess glacial acetic acid to give the salt [Eu(H(CH3COO)2)3](H2O). [1]
Europium acetate can be decomposed by heating, and the hydrate first loses water to obtain anhydrous, and then passes through basic acetate EuOCH3COO, basic carbonate Eu2O2CO3, and finally obtains europium oxide. [8] The tetrahydrate of europium acetate decomposes in air over 6 stages to europium oxide. [8] [9]
Stage 1 at 135 °C:
- Eu(CH3COO)3·4H2O → Eu(CH3COO)3·H2O + 3H2O
Stage 2 at 170 °C:
- Eu(CH3COO)3·3H2O → Eu(CH3COO)3·0.5 H2O + 0.5 H2O
Stage 3 at 210 °C:
- Eu(CH3COO)3·0.5H2O → Eu(CH3COO)3 + 0.5 H2O
Stage 4 at 310 °C:
- Eu(CH3COO)3 → EuO(CH3COO) + C3H6O + CO2
Stage 5 at 390 °C:
- 2EuO(CH3COO) → Eu2O2[CO3] + C3H6O
Stage 6 at 670 °C:
- Eu2O2[CO3] → Eu2O3 + CO2
- ^ Note: The sesquihydrate of europium(III) acetate has the structure [Eu2(CH3COO)6(H2O)](H2O)2.
- ^
a
b
c
d
e Sonia Gomez Torres, Ingo Pantenburg, Gerd Meyer (2006).
"Direct Oxidation of Europium Metal with Acetic Acid: Anhydrous Europium(III) Acetate, Eu(OAc)3, its Sesqui-hydrate, Eu(OAc)3(H2O)1.5, and the "Hydrogendiacetate", [Eu(H(OAc)2)3](H2O)". Zeitschrift für anorganische und allgemeine Chemie. 632 (12–13): 1989–1994.
doi:
10.1002/zaac.200600154.
ISSN
1521-3749. Retrieved 2019-02-01.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Herbert N. McCoy (1939-09-01). "The Salts of Europium". Journal of the American Chemical Society. 61 (9): 2455–2456. doi: 10.1021/ja01878a055. ISSN 0002-7863. Retrieved 2019-02-01.
- ^ P. Starynowicz (1995-12-01). "Structure and spectroscopy of diaqua(μ3-acetato)(acetato-O)(acetic acid-O)europium(II), [Eu(OAc)2(AcOH)(H2O)2]". Polyhedron. 14 (23): 3573–3577. doi: 10.1016/0277-5387(95)00174-Q. ISSN 0277-5387. Retrieved 2019-02-01.
-
^ Cameron J. Kepert, Lu Wei-Min, Peter C. Junk, Brian W. Skelton, Allan H. White (1999).
"Structural Systematics of Rare Earth Complexes. X ('Maximally') Hydrated Rare Earth Acetates". Australian Journal of Chemistry. 52 (6): 437.
doi:
10.1071/CH98041.
ISSN
0004-9425. Retrieved 2020-01-18.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Tang, Huian; Liu, Yanzhi; Zhao, Aiping. A preparation method of europium acetate [Patent]. CN104387254A. 2015.
-
^ Dobrokhotova, Zh. V.; Fomina, I. G.; Kiskin, M. A.; Bykov, M. A.; Belov, G. V.; Novotortsev, V. M. (2006), "The thermodynamic properties of rare-earth metal binuclear acetates and pivalates",
Russian Journal of Physical Chemistry (in German), vol. 80, no. 3, pp. 323-329,
doi:
10.1134/S0036024406030034,
ISSN
0036-0244,
S2CID
97590163
{{ citation}}: CS1 maint: multiple names: authors list ( link) - ^ P. Starynowicz (1995-12-01). "Structure and spectroscopy of diaqua(μ3-acetato)(acetato-O)(acetic acid-O)europium(II), [Eu(OAc)2(AcOH)(H2O)2]". Polyhedron. 14 (23): 3573–3577. doi: 10.1016/0277-5387(95)00174-Q. ISSN 0277-5387. Retrieved 2019-02-01.
- ^ a b Ogawa M, Manabe K. Thermal Decomposition of Europium (III) Acetate Tetrahydrate Archived 2019-02-01 at the Wayback Machine(酢酸ユウロピウム(III)4水和物の熱分解). Journal of the Ceramic Society of Japan, 1988, 96(1117): 890-893.
-
^ Balboul, Basma A. A.; Zaki, Mohamed I (2011), "Thermal decomposition course of Eu(CH3COO)3·4H2O and the reactivity at the gas/solid interface thus established", Journal of Analytical and Applied Pyrolysis (in German), vol. 92, no. 1, pp. 137-142,
doi:
10.1016/j.jaap.2011.05.004
{{ citation}}: CS1 maint: multiple names: authors list ( link)

| |
| Names | |
|---|---|
| Other names
Europium acetate
| |
| Identifiers | |
| ECHA InfoCard | 100.013.337 |
CompTox Dashboard (
EPA)
|
|
| Properties | |
| Eu(CH3COO)3 | |
| Molar mass | 329,092 g/mol (anhydrous) 341,059 g/mol (monohydrate) |
| Appearance | white solid |
| soluble in water | |
| Related compounds | |
Other
anions
|
Europium(III) oxide Europium(III) hydroxide Europium(III) carbonate |
Other
cations
|
Samarium(III) acetate Gadolinium(III) acetate |
Related compounds
|
Europium(II) acetate |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Europium(III) acetate is an inorganic salt of europium and acetic acid with the chemical formula of Eu(CH3COO)3. In this compound, europium exhibits the +3 oxidation state. It can exist in the anhydrous form, sesqui hydrate [a] [1] and tetrahydrate. [2] Its hydrate molecule is a dimer. [3] [4]
Europium acetate can be obtained by stirring reaction of acetic acid and europium oxide under heating, and then diluting with water and crystallizing: [5]
- Eu2O3 + 6 CH3COOH → 2 Eu(CH3COO)3 + 3 H2O
Europium can also directly participate in the reaction: [1]
- 2 Eu + 6 CH3COOH → 2 Eu(CH3COO)3 + 3 H2↑
This section may be too technical for most readers to understand. (May 2022) |

The anhydrous europium acetate crystallizes monoclinically in the space group C2/c (space group no. 15) with the lattice parameters a = 1126.0(3), b = 2900.5(6), c = 799.1( 2) pm and β = 132.03(2)° with four formula units per unit cell. [1] The sesquihydrate crystallizes monoclinically in the space group Cc (No. 9) with the lattice parameters a = 1608.7(2), b = 1665.6(2), c = 839.1(1) pm and β = 115.75( 9)° with four formula units per unit cell. [1] The heat capacity at 280 K is 803±16 J/(mol∙K). [6]
Europium acetate can be dissolved in water, acidified with acetic acid, and the compound of divalent europium [Eu(CH3COO)2(CH3COOH)(H2O)2] can be obtained by electrochemical reduction. [7]
Europium acetate can be crystallized in excess glacial acetic acid to give the salt [Eu(H(CH3COO)2)3](H2O). [1]
Europium acetate can be decomposed by heating, and the hydrate first loses water to obtain anhydrous, and then passes through basic acetate EuOCH3COO, basic carbonate Eu2O2CO3, and finally obtains europium oxide. [8] The tetrahydrate of europium acetate decomposes in air over 6 stages to europium oxide. [8] [9]
Stage 1 at 135 °C:
- Eu(CH3COO)3·4H2O → Eu(CH3COO)3·H2O + 3H2O
Stage 2 at 170 °C:
- Eu(CH3COO)3·3H2O → Eu(CH3COO)3·0.5 H2O + 0.5 H2O
Stage 3 at 210 °C:
- Eu(CH3COO)3·0.5H2O → Eu(CH3COO)3 + 0.5 H2O
Stage 4 at 310 °C:
- Eu(CH3COO)3 → EuO(CH3COO) + C3H6O + CO2
Stage 5 at 390 °C:
- 2EuO(CH3COO) → Eu2O2[CO3] + C3H6O
Stage 6 at 670 °C:
- Eu2O2[CO3] → Eu2O3 + CO2
- ^ Note: The sesquihydrate of europium(III) acetate has the structure [Eu2(CH3COO)6(H2O)](H2O)2.
- ^
a
b
c
d
e Sonia Gomez Torres, Ingo Pantenburg, Gerd Meyer (2006).
"Direct Oxidation of Europium Metal with Acetic Acid: Anhydrous Europium(III) Acetate, Eu(OAc)3, its Sesqui-hydrate, Eu(OAc)3(H2O)1.5, and the "Hydrogendiacetate", [Eu(H(OAc)2)3](H2O)". Zeitschrift für anorganische und allgemeine Chemie. 632 (12–13): 1989–1994.
doi:
10.1002/zaac.200600154.
ISSN
1521-3749. Retrieved 2019-02-01.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Herbert N. McCoy (1939-09-01). "The Salts of Europium". Journal of the American Chemical Society. 61 (9): 2455–2456. doi: 10.1021/ja01878a055. ISSN 0002-7863. Retrieved 2019-02-01.
- ^ P. Starynowicz (1995-12-01). "Structure and spectroscopy of diaqua(μ3-acetato)(acetato-O)(acetic acid-O)europium(II), [Eu(OAc)2(AcOH)(H2O)2]". Polyhedron. 14 (23): 3573–3577. doi: 10.1016/0277-5387(95)00174-Q. ISSN 0277-5387. Retrieved 2019-02-01.
-
^ Cameron J. Kepert, Lu Wei-Min, Peter C. Junk, Brian W. Skelton, Allan H. White (1999).
"Structural Systematics of Rare Earth Complexes. X ('Maximally') Hydrated Rare Earth Acetates". Australian Journal of Chemistry. 52 (6): 437.
doi:
10.1071/CH98041.
ISSN
0004-9425. Retrieved 2020-01-18.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Tang, Huian; Liu, Yanzhi; Zhao, Aiping. A preparation method of europium acetate [Patent]. CN104387254A. 2015.
-
^ Dobrokhotova, Zh. V.; Fomina, I. G.; Kiskin, M. A.; Bykov, M. A.; Belov, G. V.; Novotortsev, V. M. (2006), "The thermodynamic properties of rare-earth metal binuclear acetates and pivalates",
Russian Journal of Physical Chemistry (in German), vol. 80, no. 3, pp. 323-329,
doi:
10.1134/S0036024406030034,
ISSN
0036-0244,
S2CID
97590163
{{ citation}}: CS1 maint: multiple names: authors list ( link) - ^ P. Starynowicz (1995-12-01). "Structure and spectroscopy of diaqua(μ3-acetato)(acetato-O)(acetic acid-O)europium(II), [Eu(OAc)2(AcOH)(H2O)2]". Polyhedron. 14 (23): 3573–3577. doi: 10.1016/0277-5387(95)00174-Q. ISSN 0277-5387. Retrieved 2019-02-01.
- ^ a b Ogawa M, Manabe K. Thermal Decomposition of Europium (III) Acetate Tetrahydrate Archived 2019-02-01 at the Wayback Machine(酢酸ユウロピウム(III)4水和物の熱分解). Journal of the Ceramic Society of Japan, 1988, 96(1117): 890-893.
-
^ Balboul, Basma A. A.; Zaki, Mohamed I (2011), "Thermal decomposition course of Eu(CH3COO)3·4H2O and the reactivity at the gas/solid interface thus established", Journal of Analytical and Applied Pyrolysis (in German), vol. 92, no. 1, pp. 137-142,
doi:
10.1016/j.jaap.2011.05.004
{{ citation}}: CS1 maint: multiple names: authors list ( link)