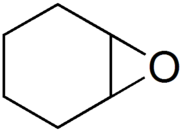
| |
| Names | |
|---|---|
|
IUPAC name
7-Oxabicyclo[4.1.0]heptane
| |
| Other names
Epoxycyclohexane
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.005.462 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H10O | |
| Molar mass | 98.145 g·mol−1 |
| Appearance | Colorless liquid [1] |
| Density | 0.97 g·cm−3 [1] |
| Melting point | ca. -40 °C [1] |
| Boiling point | ca. 130 °C [1] |
| Practically insoluble [1] | |
| Vapor pressure | 12 mbar (at 20 °C) [1] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyclohexene oxide is a cycloaliphatic epoxide. It can react in cationic polymerization to poly(cyclohexene oxide). As cyclohexene is monovalent, poly(cyclohexene oxide) is a thermoplastic.
Cyclohexene oxide is produced in epoxidation reaction from cyclohexene. The epoxidation can take place either in a homogeneous reaction by peracids [2] or heterogeneous catalysis (e.g. silver and molecular oxygen). [3] [4] [5]
In industrial production the heterogeneously catalyzed synthesis is preferred because of better atom economy, a simpler separation of the product and easier recycling of catalyst. A short overview and an investigation of the oxidation of cyclohexene by hydrogen peroxide is given in the literature. [6] In recent times the catalytic oxidation of cyclohexene by (immobilized) metalloporphyrin complexes has been found to be an efficient way. [7] [8]
In laboratory, cyclohexene oxide can also be prepared by reacting cyclohexene with magnesium monoperoxyphthalate (MMPP) in a mixture of isopropanol and water as solvent at room temperature. [9]
With this method, good yields up to 85 % can be reached.
Cyclohexene oxide has been studied extensively by analytical methods. [10] Cyclohexene oxide can be polymerized in solution, catalyzed by a solid acid catalyst. [11]
- ^ a b c d e f Record of Epoxycyclohexane in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 1 February 2014.
- ^ M. Quenard; V. Bonmarin; G. Gelbard (1987). "Epoxidation of olefins by hydrogen peroxide catalyzed by phosphonotungstic complexes". Tetrahedron Letters. 28 (20): 2237–2238. doi: 10.1016/S0040-4039(00)96089-1.
- ^ Ha Q. Pham; Maurice J. Marks (2005), "Epoxy Resins", Ullmann's Encyclopedia of Industrial Chemistry (in German), doi: 10.1002/14356007.a09_547.pub2, ISBN 3527306730
- ^ Siegfried Rebsdat; Dieter Mayer (2001), "Ethylene Oxide", Ullmann's Encyclopedia of Industrial Chemistry (in German), doi: 10.1002/14356007.a10_117, ISBN 3527306730
- ^ Morazzoni, Franca; Canevali, Carmen; d'Aprile, Fiorenza; Bianchi, Claudia L.; Tempesti, Ezio; Giuffrè, Luigi; Airoldi, Giuseppe (1995). "Spectroscopic investigation of the molybdenum active sites on MoVI heterogeneous catalysts for alkene epoxidation". Journal of the Chemical Society, Faraday Transactions. 91 (21): 3969–3974. doi: 10.1039/FT9959103969.
-
^ Ambili, V K; Dr.Sugunan, S (April 2011), Faculty of Sciences (ed.),
Studies on Catalysis by Ordered Mesoporous SBA-15 Materials Modified with Transition Metals (in German), Cochin University of Science and Technology, retrieved 2014-07-27
{{ citation}}: CS1 maint: multiple names: authors list ( link) - ^ Costa, Andréia A. Ghesti; Grace F. de Macedo; Julio L. Braga; Valdeilson S. Santos; Marcello M. Dias; José A. Dias; Sílvia C.L. (2008). "Immobilization of Fe, Mn and Co tetraphenylporphyrin complexes in MCM-41 and their catalytic activity in cyclohexene oxidation reaction by hydrogen peroxide". Journal of Molecular Catalysis A: Chemical. 282 (1–2): 149–157. doi: 10.1016/j.molcata.2007.12.024.
- ^ Xian-Tai Zhou; Hong-Bing Ji; Jian-Chang Xu; Li-Xia Pei; Le-Fu Wang; Xing-Dong Yao (2007). "Enzymatic-like mediated olefins epoxidation by molecular oxygen under mild conditions". Tetrahedron Letters. 48 (15): 2691–2695. doi: 10.1016/j.tetlet.2007.02.066.
- ^ Brougham, Paul; Cooper, Mark S.; Cummerson, David A.; Heaney, Harry; Thompson, Nicola (1987). "Oxidation Reactions Using Magnesium Monoperphthalate: A Comparison with m-Chloroperoxybenzoic Acid". Synthesis. 1987 (11): 1015–1017. doi: 10.1055/s-1987-28153. Retrieved 2020-07-31.
- ^ RM Ibberson; O. Yamamuro; I. Tsukushi (2006). "The crystal structures and phase behaviour of cyclohexene oxide". Chemical Physics Letters. 423 (4–6): 454–458. Bibcode: 2006CPL...423..454I. doi: 10.1016/j.cplett.2006.04.004.
- ^ Ahmed Yahiaoui; Mohammed Belbachir; Jeanne Claude Soutif; Laurent Fontaine (2005). "Synthesis and structural analyses of poly(1,2-cyclohexene oxide) over solid acid catalyst". Materials Letters. 59 (7): 759–767. doi: 10.1016/j.matlet.2004.11.017.
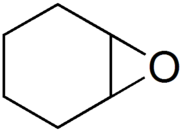
| |
| Names | |
|---|---|
|
IUPAC name
7-Oxabicyclo[4.1.0]heptane
| |
| Other names
Epoxycyclohexane
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.005.462 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H10O | |
| Molar mass | 98.145 g·mol−1 |
| Appearance | Colorless liquid [1] |
| Density | 0.97 g·cm−3 [1] |
| Melting point | ca. -40 °C [1] |
| Boiling point | ca. 130 °C [1] |
| Practically insoluble [1] | |
| Vapor pressure | 12 mbar (at 20 °C) [1] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Cyclohexene oxide is a cycloaliphatic epoxide. It can react in cationic polymerization to poly(cyclohexene oxide). As cyclohexene is monovalent, poly(cyclohexene oxide) is a thermoplastic.
Cyclohexene oxide is produced in epoxidation reaction from cyclohexene. The epoxidation can take place either in a homogeneous reaction by peracids [2] or heterogeneous catalysis (e.g. silver and molecular oxygen). [3] [4] [5]
In industrial production the heterogeneously catalyzed synthesis is preferred because of better atom economy, a simpler separation of the product and easier recycling of catalyst. A short overview and an investigation of the oxidation of cyclohexene by hydrogen peroxide is given in the literature. [6] In recent times the catalytic oxidation of cyclohexene by (immobilized) metalloporphyrin complexes has been found to be an efficient way. [7] [8]
In laboratory, cyclohexene oxide can also be prepared by reacting cyclohexene with magnesium monoperoxyphthalate (MMPP) in a mixture of isopropanol and water as solvent at room temperature. [9]
With this method, good yields up to 85 % can be reached.
Cyclohexene oxide has been studied extensively by analytical methods. [10] Cyclohexene oxide can be polymerized in solution, catalyzed by a solid acid catalyst. [11]
- ^ a b c d e f Record of Epoxycyclohexane in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 1 February 2014.
- ^ M. Quenard; V. Bonmarin; G. Gelbard (1987). "Epoxidation of olefins by hydrogen peroxide catalyzed by phosphonotungstic complexes". Tetrahedron Letters. 28 (20): 2237–2238. doi: 10.1016/S0040-4039(00)96089-1.
- ^ Ha Q. Pham; Maurice J. Marks (2005), "Epoxy Resins", Ullmann's Encyclopedia of Industrial Chemistry (in German), doi: 10.1002/14356007.a09_547.pub2, ISBN 3527306730
- ^ Siegfried Rebsdat; Dieter Mayer (2001), "Ethylene Oxide", Ullmann's Encyclopedia of Industrial Chemistry (in German), doi: 10.1002/14356007.a10_117, ISBN 3527306730
- ^ Morazzoni, Franca; Canevali, Carmen; d'Aprile, Fiorenza; Bianchi, Claudia L.; Tempesti, Ezio; Giuffrè, Luigi; Airoldi, Giuseppe (1995). "Spectroscopic investigation of the molybdenum active sites on MoVI heterogeneous catalysts for alkene epoxidation". Journal of the Chemical Society, Faraday Transactions. 91 (21): 3969–3974. doi: 10.1039/FT9959103969.
-
^ Ambili, V K; Dr.Sugunan, S (April 2011), Faculty of Sciences (ed.),
Studies on Catalysis by Ordered Mesoporous SBA-15 Materials Modified with Transition Metals (in German), Cochin University of Science and Technology, retrieved 2014-07-27
{{ citation}}: CS1 maint: multiple names: authors list ( link) - ^ Costa, Andréia A. Ghesti; Grace F. de Macedo; Julio L. Braga; Valdeilson S. Santos; Marcello M. Dias; José A. Dias; Sílvia C.L. (2008). "Immobilization of Fe, Mn and Co tetraphenylporphyrin complexes in MCM-41 and their catalytic activity in cyclohexene oxidation reaction by hydrogen peroxide". Journal of Molecular Catalysis A: Chemical. 282 (1–2): 149–157. doi: 10.1016/j.molcata.2007.12.024.
- ^ Xian-Tai Zhou; Hong-Bing Ji; Jian-Chang Xu; Li-Xia Pei; Le-Fu Wang; Xing-Dong Yao (2007). "Enzymatic-like mediated olefins epoxidation by molecular oxygen under mild conditions". Tetrahedron Letters. 48 (15): 2691–2695. doi: 10.1016/j.tetlet.2007.02.066.
- ^ Brougham, Paul; Cooper, Mark S.; Cummerson, David A.; Heaney, Harry; Thompson, Nicola (1987). "Oxidation Reactions Using Magnesium Monoperphthalate: A Comparison with m-Chloroperoxybenzoic Acid". Synthesis. 1987 (11): 1015–1017. doi: 10.1055/s-1987-28153. Retrieved 2020-07-31.
- ^ RM Ibberson; O. Yamamuro; I. Tsukushi (2006). "The crystal structures and phase behaviour of cyclohexene oxide". Chemical Physics Letters. 423 (4–6): 454–458. Bibcode: 2006CPL...423..454I. doi: 10.1016/j.cplett.2006.04.004.
- ^ Ahmed Yahiaoui; Mohammed Belbachir; Jeanne Claude Soutif; Laurent Fontaine (2005). "Synthesis and structural analyses of poly(1,2-cyclohexene oxide) over solid acid catalyst". Materials Letters. 59 (7): 759–767. doi: 10.1016/j.matlet.2004.11.017.

