| |

| |
| Names | |
|---|---|
|
IUPAC name
Cobalt(II) acetate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.000.687 |
PubChem
CID
|
|
| UNII |
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| Co(C2H3O2)2 | |
| Molar mass | 177.02124 g/mol (anhydrous) 249.08 g/mol (tetrahydrate) |
| Appearance | Pink crystals (anhydrous) intense red crystals (tetrahydrate) |
| Odor | vinegar (tetrahydrate) |
| Density | 1.705 g/cm3 (tetrahydrate) |
| Melting point | 140 °C (284 °F; 413 K) (tetrahydrate) |
| Soluble | |
| Solubility | soluble in alcohol, dilute acids, pentyl acetate (tetrahydrate) |
| +11,000·10−6 cm3/mol | |
Refractive index (nD)
|
1.542 (tetrahydrate) |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (
median dose)
|
503 mg/kg (oral, rat) |
| Safety data sheet (SDS) | J.T. Baker MSDS |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Cobalt(II) acetate is the cobalt salt of acetic acid. It is commonly found as the tetrahydrate Co(CH3CO2)2·4 H2O, abbreviated Co(OAc)2·4 H2O. It is used as a catalyst.
Synthesis and structure
Like many other transition metal acetates, cobalt(II) acetate forms by the reaction of cobalt oxide or hydroxide and acetic acid:
- CoO + 2 CH3CO2H + 3 H2O → Co(CH3CO2)2·4 H2O
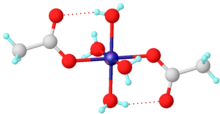
The tetrahydrate has been shown by X-ray crystallography to adopt an octahedral structure, the central cobalt centre being coordinated by four water molecules and two acetate ligands. [1] The analogous nickel acetate is isostructural. [2]
Various hydrates are known including Co(CH3CO2)2·H2O and [Co(CH3CO2)25·0.5 H2O. [3]
Reactions and uses
Cobalt acetate is a precursor to various oil drying agents, catalysts that allow paints and varnishes to harden. [4]
Anhydrous cobalt acetate is a widely used source of cobalt in the synthesis of materials, [5] catalyst, [6] and complexes. [7]
Safety
Cobalt salts are poisonous. [8]
References
- ^ Sobolev, Alexandre N.; Miminoshvili, Elguja B.; Miminoshvili, Ketevan E.; Sakvarelidze, Tamara N. (2003). "Cobalt diacetate tetrahydrate". Acta Crystallographica Section E. 59 (10): m836–m837. doi: 10.1107/S1600536803019093.
- ^ Van Niekerk, J. N.; Schoening, F. R. L. (1953). "The crystal structures of nickel acetate, Ni(CH3COO)2·4H2O, and cobalt acetate, Co(CH3COO)2·4H2O". Acta Crystallogr. 6 (7): 609–612. doi: 10.1107/S0365110X5300171X.
- ^ Zhang, Gao; Lin, Jian; Guo, Dong-Wei; Yao, Shi-Yan; Tian, Yun-Qi (2010). "Infinite Coordination Polymers of One- and Two-dimensional Cobalt Acetates". Zeitschrift für Anorganische und Allgemeine Chemie. 636 (7): 1401–1404. doi: 10.1002/zaac.200900457.
- ^ John Dallas Donaldson, Detmar Beyersmann, "Cobalt and Cobalt Compounds" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005. doi: 10.1002/14356007.a07_281.pub2
- ^ Rodenas, Tania; Luz, Ignacio; Prieto, Gonzalo; Seoane, Beatriz; Miro, Hozanna; Corma, Avelino; Kapteijn, Freek; Llabrés i Xamena, Francesc X.; Gascon, Jorge (2015). "Metal–organic framework nanosheets in polymer composite materials for gas separation". Nature Materials. 14 (1): 48–55. Bibcode: 2015NatMa..14...48R. doi: 10.1038/nmat4113. PMC 4270742. PMID 25362353.
- ^ Schultz, Mitchell J.; Sigman, Matthew S. (2006). "Recent advances in homogeneous transition metal-catalyzed aerobic alcohol oxidations". Tetrahedron. 62 (35): 8227–8241. doi: 10.1016/j.tet.2006.06.065.
- ^ Appleton, T. G. (1977). "Oxygen Uptake by a Cobalt(II) Complex". J. Chem. Educ. 54 (7): 443. doi: 10.1021/ed054p443.
- ^ MallBaker MSDS[ permanent dead link]
External Links

| |

| |
| Names | |
|---|---|
|
IUPAC name
Cobalt(II) acetate
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.000.687 |
PubChem
CID
|
|
| UNII |
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| Co(C2H3O2)2 | |
| Molar mass | 177.02124 g/mol (anhydrous) 249.08 g/mol (tetrahydrate) |
| Appearance | Pink crystals (anhydrous) intense red crystals (tetrahydrate) |
| Odor | vinegar (tetrahydrate) |
| Density | 1.705 g/cm3 (tetrahydrate) |
| Melting point | 140 °C (284 °F; 413 K) (tetrahydrate) |
| Soluble | |
| Solubility | soluble in alcohol, dilute acids, pentyl acetate (tetrahydrate) |
| +11,000·10−6 cm3/mol | |
Refractive index (nD)
|
1.542 (tetrahydrate) |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (
median dose)
|
503 mg/kg (oral, rat) |
| Safety data sheet (SDS) | J.T. Baker MSDS |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Cobalt(II) acetate is the cobalt salt of acetic acid. It is commonly found as the tetrahydrate Co(CH3CO2)2·4 H2O, abbreviated Co(OAc)2·4 H2O. It is used as a catalyst.
Synthesis and structure
Like many other transition metal acetates, cobalt(II) acetate forms by the reaction of cobalt oxide or hydroxide and acetic acid:
- CoO + 2 CH3CO2H + 3 H2O → Co(CH3CO2)2·4 H2O
The tetrahydrate has been shown by X-ray crystallography to adopt an octahedral structure, the central cobalt centre being coordinated by four water molecules and two acetate ligands. [1] The analogous nickel acetate is isostructural. [2]
Various hydrates are known including Co(CH3CO2)2·H2O and [Co(CH3CO2)25·0.5 H2O. [3]
Reactions and uses
Cobalt acetate is a precursor to various oil drying agents, catalysts that allow paints and varnishes to harden. [4]
Anhydrous cobalt acetate is a widely used source of cobalt in the synthesis of materials, [5] catalyst, [6] and complexes. [7]
Safety
Cobalt salts are poisonous. [8]
References
- ^ Sobolev, Alexandre N.; Miminoshvili, Elguja B.; Miminoshvili, Ketevan E.; Sakvarelidze, Tamara N. (2003). "Cobalt diacetate tetrahydrate". Acta Crystallographica Section E. 59 (10): m836–m837. doi: 10.1107/S1600536803019093.
- ^ Van Niekerk, J. N.; Schoening, F. R. L. (1953). "The crystal structures of nickel acetate, Ni(CH3COO)2·4H2O, and cobalt acetate, Co(CH3COO)2·4H2O". Acta Crystallogr. 6 (7): 609–612. doi: 10.1107/S0365110X5300171X.
- ^ Zhang, Gao; Lin, Jian; Guo, Dong-Wei; Yao, Shi-Yan; Tian, Yun-Qi (2010). "Infinite Coordination Polymers of One- and Two-dimensional Cobalt Acetates". Zeitschrift für Anorganische und Allgemeine Chemie. 636 (7): 1401–1404. doi: 10.1002/zaac.200900457.
- ^ John Dallas Donaldson, Detmar Beyersmann, "Cobalt and Cobalt Compounds" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005. doi: 10.1002/14356007.a07_281.pub2
- ^ Rodenas, Tania; Luz, Ignacio; Prieto, Gonzalo; Seoane, Beatriz; Miro, Hozanna; Corma, Avelino; Kapteijn, Freek; Llabrés i Xamena, Francesc X.; Gascon, Jorge (2015). "Metal–organic framework nanosheets in polymer composite materials for gas separation". Nature Materials. 14 (1): 48–55. Bibcode: 2015NatMa..14...48R. doi: 10.1038/nmat4113. PMC 4270742. PMID 25362353.
- ^ Schultz, Mitchell J.; Sigman, Matthew S. (2006). "Recent advances in homogeneous transition metal-catalyzed aerobic alcohol oxidations". Tetrahedron. 62 (35): 8227–8241. doi: 10.1016/j.tet.2006.06.065.
- ^ Appleton, T. G. (1977). "Oxygen Uptake by a Cobalt(II) Complex". J. Chem. Educ. 54 (7): 443. doi: 10.1021/ed054p443.
- ^ MallBaker MSDS[ permanent dead link]
