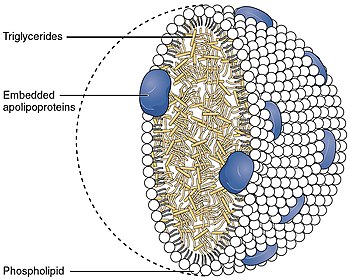
Cholesteryl ester transfer protein (CETP), also called plasma lipid transfer protein, is a plasma protein that facilitates the transport of cholesteryl esters and triglycerides between the lipoproteins. It collects triglycerides from very-low-density (VLDL) or Chylomicrons and exchanges them for cholesteryl esters from high-density lipoproteins (HDL), and vice versa. Most of the time, however, CETP does a heteroexchange, trading a triglyceride for a cholesteryl ester or a cholesteryl ester for a triglyceride.
Genetics
The CETP gene is located on chromosome 16 (16q21).
Protein Fold
The crystal structure of CETP is that of dimer of two TUbular LIPid (TULIP) binding domains. [3] [4] Each domain consists of a core of 6 elements: 4 beta-sheets forming an extended superhelix; 2 flanking elements that tend to include some alpha helix. The sheets wrap around the helices to produce a cylinder 6 x 2.5 x 2.5 nm. CETP contains two of these domains that interact head-to-head via an interface made of 6 beta-sheets, 3 from each protomer. The same fold is shared by Bacterial Permeability Inducing proteins (examples: BPIFP1 BPIFP2 BPIFA3 and BPIFB4), phospholipid transfer protein ( PLTP), and long-Palate Lung, and Nasal Epithelium protein (L-PLUNC). The fold is similar to intracellular SMP domains, [5] and originated in bacteria. [6] [7] [8] The crystal structure of CETP has been obtained with bound CETP inhibitors. [9] However, this has not resolved the doubt over whether CETP function as a lipid tube or shuttle. [10]
Role in disease
Rare mutations leading to reduced function of CETP have been linked to accelerated atherosclerosis. [11] In contrast, a polymorphism (I405V) of the CETP gene leading to lower serum levels has also been linked to exceptional longevity [12] and to metabolic response to nutritional intervention. [13] However, this mutation also increases the prevalence of coronary heart disease in patients with hypertriglyceridemia. [14] The D442G mutation, which lowers CETP levels and increases HDL levels also increases coronary heart disease. [11]
Elaidic acid, a major component of trans fat, increases CETP activity. [15]
Pharmacology
As HDL can alleviate atherosclerosis and other cardiovascular diseases, and certain disease states such as the metabolic syndrome feature low HDL, pharmacological inhibition of CETP is being studied as a method of improving HDL levels. [16] To be specific, in a 2004 study, the small molecular agent torcetrapib was shown to increase HDL levels, alone and with a statin, and lower LDL when co-administered with a statin. [17] Studies into cardiovascular endpoints, however, were largely disappointing. While they confirmed the change in lipid levels, most reported an increase in blood pressure, no change in atherosclerosis, [18] [19] and, in a trial of a combination of torcetrapib and atorvastatin, an increase in cardiovascular events and mortality. [20]
A compound related to torcetrapib, Dalcetrapib (investigative name JTT-705/R1658), was also studied, but trials have ceased. [21] It increases HDL levels by 30%, as compared to 60% by torcetrapib. [22] Two CETP inhibitors were previously under development. One was Merck's MK-0859 anacetrapib, which in initial studies did not increase blood pressure. [23] In 2017, its development was abandoned by Merck. [24] The other was Eli Lilly's evacetrapib, which failed in Phase 3 trials.
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "Statin_Pathway_WP430".
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000087237 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Qiu X, Mistry A, Ammirati MJ, Chrunyk BA, Clark RW, Cong Y, Culp JS, Danley DE, Freeman TB, Geoghegan KF, Griffor MC, Hawrylik SJ, Hayward CM, Hensley P, Hoth LR, Karam GA, Lira ME, Lloyd DB, McGrath KM, Stutzman-Engwall KJ, Subashi AK, Subashi TA, Thompson JF, Wang IK, Zhao H, Seddon AP (February 2007). "Crystal structure of cholesteryl ester transfer protein reveals a long tunnel and four bound lipid molecules". Nature Structural & Molecular Biology. 14 (2): 106–13. doi: 10.1038/nsmb1197. PMID 17237796. S2CID 30939809.
- ^ Alva V, Lupas AN (August 2016). "The TULIP superfamily of eukaryotic lipid-binding proteins as a mediator of lipid sensing and transport". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 1861 (8 Pt B): 913–923. doi: 10.1016/j.bbalip.2016.01.016. PMID 26825693.
- ^ Reinisch KM, De Camilli P (August 2016). "SMP-domain proteins at membrane contact sites: Structure and function". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 1861 (8 Pt B): 924–927. doi: 10.1016/j.bbalip.2015.12.003. PMC 4902782. PMID 26686281.
- ^ Wong LH, Levine TP (September 2017). "Tubular lipid binding proteins (TULIPs) growing everywhere". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 1864 (9): 1439–1449. doi: 10.1016/j.bbamcr.2017.05.019. PMC 5507252. PMID 28554774.
- ^ Lam KH, Qi R, Liu S, Kroh A, Yao G, Perry K, Rummel A, Jin R (June 2018). "The hypothetical protein P47 of Clostridium botulinum E1 strain Beluga has a structural topology similar to bactericidal/permeability-increasing protein". Toxicon. 147: 19–26. doi: 10.1016/j.toxicon.2017.10.012. PMC 5902665. PMID 29042313.
- ^ Gustafsson R, Berntsson RP, Martínez-Carranza M, El Tekle G, Odegrip R, Johnson EA, Stenmark P (November 2017). "Crystal structures of OrfX2 and P47 from a Botulinum neurotoxin OrfX-type gene cluster". FEBS Letters. 591 (22): 3781–3792. doi: 10.1002/1873-3468.12889. PMID 29067689.
- ^ Liu S, Mistry A, Reynolds JM, Lloyd DB, Griffor MC, Perry DA, Ruggeri RB, Clark RW, Qiu X (October 2012). "Crystal structures of cholesteryl ester transfer protein in complex with inhibitors". The Journal of Biological Chemistry. 287 (44): 37321–9. doi: 10.1074/jbc.M112.380063. PMC 3481329. PMID 22961980.
- ^ Lauer ME, Graff-Meyer A, Rufer AC, Maugeais C, von der Mark E, Matile H, D'Arcy B, Magg C, Ringler P, Müller SA, Scherer S, Dernick G, Thoma R, Hennig M, Niesor EJ, Stahlberg H (May 2016). "Cholesteryl ester transfer between lipoproteins does not require a ternary tunnel complex with CETP". Journal of Structural Biology. 194 (2): 191–8. doi: 10.1016/j.jsb.2016.02.016. PMID 26876146.
- ^ a b Zhong S, Sharp DS, Grove JS, Bruce C, Yano K, Curb JD, Tall AR (June 1996). "Increased coronary heart disease in Japanese-American men with mutation in the cholesteryl ester transfer protein gene despite increased HDL levels". The Journal of Clinical Investigation. 97 (12): 2917–23. doi: 10.1172/JCI118751. PMC 507389. PMID 8675707.
- ^ Barzilai N, Atzmon G, Schechter C, Schaefer EJ, Cupples AL, Lipton R, Cheng S, Shuldiner AR (October 2003). "Unique lipoprotein phenotype and genotype associated with exceptional longevity". JAMA. 290 (15): 2030–40. doi: 10.1001/jama.290.15.2030. PMID 14559957. S2CID 22792639.
- ^ Darabi M, Abolfathi AA, Noori M, Kazemi A, Ostadrahimi A, Rahimipour A, Darabi M, Ghatrehsamani K (July 2009). "Cholesteryl ester transfer protein I405V polymorphism influences apolipoprotein A-I response to a change in dietary fatty acid composition". Hormone and Metabolic Research. 41 (7): 554–8. doi: 10.1055/s-0029-1192034. PMID 19242900. S2CID 260169359.
- ^ Bruce C, Sharp DS, Tall AR (May 1998). "Relationship of HDL and coronary heart disease to a common amino acid polymorphism in the cholesteryl ester transfer protein in men with and without hypertriglyceridemia". Journal of Lipid Research. 39 (5): 1071–8. doi: 10.1016/S0022-2275(20)33876-1. PMID 9610775.
- ^ Abbey M, Nestel PJ (March 1994). "Plasma cholesteryl ester transfer protein activity is increased when trans-elaidic acid is substituted for cis-oleic acid in the diet". Atherosclerosis. 106 (1): 99–107. doi: 10.1016/0021-9150(94)90086-8. PMID 8018112.
- ^ Barter PJ, Brewer HB, Chapman MJ, Hennekens CH, Rader DJ, Tall AR (February 2003). "Cholesteryl ester transfer protein: a novel target for raising HDL and inhibiting atherosclerosis". Arteriosclerosis, Thrombosis, and Vascular Biology. 23 (2): 160–7. doi: 10.1161/01.ATV.0000054658.91146.64. PMID 12588754.
- ^ Brousseau ME, Schaefer EJ, Wolfe ML, Bloedon LT, Digenio AG, Clark RW, Mancuso JP, Rader DJ (April 2004). "Effects of an inhibitor of cholesteryl ester transfer protein on HDL cholesterol". The New England Journal of Medicine. 350 (15): 1505–15. doi: 10.1056/NEJMoa031766. PMID 15071125.
- ^ Nissen SE, Tardif JC, Nicholls SJ, Revkin JH, Shear CL, Duggan WT, Ruzyllo W, Bachinsky WB, Lasala GP, Lasala GP, Tuzcu EM (March 2007). "Effect of torcetrapib on the progression of coronary atherosclerosis". The New England Journal of Medicine. 356 (13): 1304–16. doi: 10.1056/NEJMoa070635. PMID 17387129.
- ^ Kastelein JJ, van Leuven SI, Burgess L, Evans GW, Kuivenhoven JA, Barter PJ, Revkin JH, Grobbee DE, Riley WA, Shear CL, Duggan WT, Bots ML (April 2007). "Effect of torcetrapib on carotid atherosclerosis in familial hypercholesterolemia". The New England Journal of Medicine. 356 (16): 1620–30. doi: 10.1056/NEJMoa071359. PMID 17387131.
- ^ "Pfizer Stops All Torcetrapib Clinical Trials in Interest of Patient Safety" (Press release). U.S. Food and Drug Administration. 3 December 2006.
- ^ El Harchaoui K, van der Steeg WA, Stroes ES, Kastelein JJ (August 2007). "The role of CETP inhibition in dyslipidemia". Current Atherosclerosis Reports. 9 (2): 125–33. doi: 10.1007/s11883-007-0008-5. PMID 17877921.
- ^ de Grooth GJ, Kuivenhoven JA, Stalenhoef AF, de Graaf J, Zwinderman AH, Posma JL, van Tol A, Kastelein JJ (May 2002). "Efficacy and safety of a novel cholesteryl ester transfer protein inhibitor, JTT-705, in humans: a randomized phase II dose-response study". Circulation. 105 (18): 2159–65. doi: 10.1161/01.CIR.0000015857.31889.7B. PMID 11994249.
- ^ "Merck announces its investigational CETP-Inhibitor, MK-0859, produced positive effects on lipids with no observed blood pressure changes". Reuters. Reuters, Inc. 4 October 2007. Retrieved 26 November 2013.
- ^ "Merck says will not seek approval of cholesterol treatment". Reuters. 2017. Retrieved 18 October 2017.
Further reading
- Okajima F (March 2002). "[Distribution of sphingosine 1-phosphate in plasma lipoproteins and its role in the regulation of the vascular cell functions]". Tanpakushitsu Kakusan Koso. Protein, Nucleic Acid, Enzyme. 47 (4 Suppl): 480–7. PMID 11915346.
- Barter PJ, Brewer HB, Chapman MJ, Hennekens CH, Rader DJ, Tall AR (February 2003). "Cholesteryl ester transfer protein: a novel target for raising HDL and inhibiting atherosclerosis". Arteriosclerosis, Thrombosis, and Vascular Biology. 23 (2): 160–7. doi: 10.1161/01.ATV.0000054658.91146.64. PMID 12588754.
- Dallinga-Thie GM, Dullaart RP, van Tol A (June 2007). "Concerted actions of cholesteryl ester transfer protein and phospholipid transfer protein in type 2 diabetes: effects of apolipoproteins". Current Opinion in Lipidology. 18 (3): 251–7. doi: 10.1097/MOL.0b013e3280e12685. PMID 17495597. S2CID 20012553.
External links
- Cholesterol+ester+transfer+proteins at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
| CETP | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | CETP, BPIFF, HDLCQ10, cholesteryl ester transfer protein | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 118470 HomoloGene: 47904 GeneCards: CETP | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Cholesteryl ester transfer protein (CETP), also called plasma lipid transfer protein, is a plasma protein that facilitates the transport of cholesteryl esters and triglycerides between the lipoproteins. It collects triglycerides from very-low-density (VLDL) or Chylomicrons and exchanges them for cholesteryl esters from high-density lipoproteins (HDL), and vice versa. Most of the time, however, CETP does a heteroexchange, trading a triglyceride for a cholesteryl ester or a cholesteryl ester for a triglyceride.
Genetics
The CETP gene is located on chromosome 16 (16q21).
Protein Fold
The crystal structure of CETP is that of dimer of two TUbular LIPid (TULIP) binding domains. [3] [4] Each domain consists of a core of 6 elements: 4 beta-sheets forming an extended superhelix; 2 flanking elements that tend to include some alpha helix. The sheets wrap around the helices to produce a cylinder 6 x 2.5 x 2.5 nm. CETP contains two of these domains that interact head-to-head via an interface made of 6 beta-sheets, 3 from each protomer. The same fold is shared by Bacterial Permeability Inducing proteins (examples: BPIFP1 BPIFP2 BPIFA3 and BPIFB4), phospholipid transfer protein ( PLTP), and long-Palate Lung, and Nasal Epithelium protein (L-PLUNC). The fold is similar to intracellular SMP domains, [5] and originated in bacteria. [6] [7] [8] The crystal structure of CETP has been obtained with bound CETP inhibitors. [9] However, this has not resolved the doubt over whether CETP function as a lipid tube or shuttle. [10]
Role in disease
Rare mutations leading to reduced function of CETP have been linked to accelerated atherosclerosis. [11] In contrast, a polymorphism (I405V) of the CETP gene leading to lower serum levels has also been linked to exceptional longevity [12] and to metabolic response to nutritional intervention. [13] However, this mutation also increases the prevalence of coronary heart disease in patients with hypertriglyceridemia. [14] The D442G mutation, which lowers CETP levels and increases HDL levels also increases coronary heart disease. [11]
Elaidic acid, a major component of trans fat, increases CETP activity. [15]
Pharmacology
As HDL can alleviate atherosclerosis and other cardiovascular diseases, and certain disease states such as the metabolic syndrome feature low HDL, pharmacological inhibition of CETP is being studied as a method of improving HDL levels. [16] To be specific, in a 2004 study, the small molecular agent torcetrapib was shown to increase HDL levels, alone and with a statin, and lower LDL when co-administered with a statin. [17] Studies into cardiovascular endpoints, however, were largely disappointing. While they confirmed the change in lipid levels, most reported an increase in blood pressure, no change in atherosclerosis, [18] [19] and, in a trial of a combination of torcetrapib and atorvastatin, an increase in cardiovascular events and mortality. [20]
A compound related to torcetrapib, Dalcetrapib (investigative name JTT-705/R1658), was also studied, but trials have ceased. [21] It increases HDL levels by 30%, as compared to 60% by torcetrapib. [22] Two CETP inhibitors were previously under development. One was Merck's MK-0859 anacetrapib, which in initial studies did not increase blood pressure. [23] In 2017, its development was abandoned by Merck. [24] The other was Eli Lilly's evacetrapib, which failed in Phase 3 trials.
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "Statin_Pathway_WP430".
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000087237 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Qiu X, Mistry A, Ammirati MJ, Chrunyk BA, Clark RW, Cong Y, Culp JS, Danley DE, Freeman TB, Geoghegan KF, Griffor MC, Hawrylik SJ, Hayward CM, Hensley P, Hoth LR, Karam GA, Lira ME, Lloyd DB, McGrath KM, Stutzman-Engwall KJ, Subashi AK, Subashi TA, Thompson JF, Wang IK, Zhao H, Seddon AP (February 2007). "Crystal structure of cholesteryl ester transfer protein reveals a long tunnel and four bound lipid molecules". Nature Structural & Molecular Biology. 14 (2): 106–13. doi: 10.1038/nsmb1197. PMID 17237796. S2CID 30939809.
- ^ Alva V, Lupas AN (August 2016). "The TULIP superfamily of eukaryotic lipid-binding proteins as a mediator of lipid sensing and transport". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 1861 (8 Pt B): 913–923. doi: 10.1016/j.bbalip.2016.01.016. PMID 26825693.
- ^ Reinisch KM, De Camilli P (August 2016). "SMP-domain proteins at membrane contact sites: Structure and function". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 1861 (8 Pt B): 924–927. doi: 10.1016/j.bbalip.2015.12.003. PMC 4902782. PMID 26686281.
- ^ Wong LH, Levine TP (September 2017). "Tubular lipid binding proteins (TULIPs) growing everywhere". Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. 1864 (9): 1439–1449. doi: 10.1016/j.bbamcr.2017.05.019. PMC 5507252. PMID 28554774.
- ^ Lam KH, Qi R, Liu S, Kroh A, Yao G, Perry K, Rummel A, Jin R (June 2018). "The hypothetical protein P47 of Clostridium botulinum E1 strain Beluga has a structural topology similar to bactericidal/permeability-increasing protein". Toxicon. 147: 19–26. doi: 10.1016/j.toxicon.2017.10.012. PMC 5902665. PMID 29042313.
- ^ Gustafsson R, Berntsson RP, Martínez-Carranza M, El Tekle G, Odegrip R, Johnson EA, Stenmark P (November 2017). "Crystal structures of OrfX2 and P47 from a Botulinum neurotoxin OrfX-type gene cluster". FEBS Letters. 591 (22): 3781–3792. doi: 10.1002/1873-3468.12889. PMID 29067689.
- ^ Liu S, Mistry A, Reynolds JM, Lloyd DB, Griffor MC, Perry DA, Ruggeri RB, Clark RW, Qiu X (October 2012). "Crystal structures of cholesteryl ester transfer protein in complex with inhibitors". The Journal of Biological Chemistry. 287 (44): 37321–9. doi: 10.1074/jbc.M112.380063. PMC 3481329. PMID 22961980.
- ^ Lauer ME, Graff-Meyer A, Rufer AC, Maugeais C, von der Mark E, Matile H, D'Arcy B, Magg C, Ringler P, Müller SA, Scherer S, Dernick G, Thoma R, Hennig M, Niesor EJ, Stahlberg H (May 2016). "Cholesteryl ester transfer between lipoproteins does not require a ternary tunnel complex with CETP". Journal of Structural Biology. 194 (2): 191–8. doi: 10.1016/j.jsb.2016.02.016. PMID 26876146.
- ^ a b Zhong S, Sharp DS, Grove JS, Bruce C, Yano K, Curb JD, Tall AR (June 1996). "Increased coronary heart disease in Japanese-American men with mutation in the cholesteryl ester transfer protein gene despite increased HDL levels". The Journal of Clinical Investigation. 97 (12): 2917–23. doi: 10.1172/JCI118751. PMC 507389. PMID 8675707.
- ^ Barzilai N, Atzmon G, Schechter C, Schaefer EJ, Cupples AL, Lipton R, Cheng S, Shuldiner AR (October 2003). "Unique lipoprotein phenotype and genotype associated with exceptional longevity". JAMA. 290 (15): 2030–40. doi: 10.1001/jama.290.15.2030. PMID 14559957. S2CID 22792639.
- ^ Darabi M, Abolfathi AA, Noori M, Kazemi A, Ostadrahimi A, Rahimipour A, Darabi M, Ghatrehsamani K (July 2009). "Cholesteryl ester transfer protein I405V polymorphism influences apolipoprotein A-I response to a change in dietary fatty acid composition". Hormone and Metabolic Research. 41 (7): 554–8. doi: 10.1055/s-0029-1192034. PMID 19242900. S2CID 260169359.
- ^ Bruce C, Sharp DS, Tall AR (May 1998). "Relationship of HDL and coronary heart disease to a common amino acid polymorphism in the cholesteryl ester transfer protein in men with and without hypertriglyceridemia". Journal of Lipid Research. 39 (5): 1071–8. doi: 10.1016/S0022-2275(20)33876-1. PMID 9610775.
- ^ Abbey M, Nestel PJ (March 1994). "Plasma cholesteryl ester transfer protein activity is increased when trans-elaidic acid is substituted for cis-oleic acid in the diet". Atherosclerosis. 106 (1): 99–107. doi: 10.1016/0021-9150(94)90086-8. PMID 8018112.
- ^ Barter PJ, Brewer HB, Chapman MJ, Hennekens CH, Rader DJ, Tall AR (February 2003). "Cholesteryl ester transfer protein: a novel target for raising HDL and inhibiting atherosclerosis". Arteriosclerosis, Thrombosis, and Vascular Biology. 23 (2): 160–7. doi: 10.1161/01.ATV.0000054658.91146.64. PMID 12588754.
- ^ Brousseau ME, Schaefer EJ, Wolfe ML, Bloedon LT, Digenio AG, Clark RW, Mancuso JP, Rader DJ (April 2004). "Effects of an inhibitor of cholesteryl ester transfer protein on HDL cholesterol". The New England Journal of Medicine. 350 (15): 1505–15. doi: 10.1056/NEJMoa031766. PMID 15071125.
- ^ Nissen SE, Tardif JC, Nicholls SJ, Revkin JH, Shear CL, Duggan WT, Ruzyllo W, Bachinsky WB, Lasala GP, Lasala GP, Tuzcu EM (March 2007). "Effect of torcetrapib on the progression of coronary atherosclerosis". The New England Journal of Medicine. 356 (13): 1304–16. doi: 10.1056/NEJMoa070635. PMID 17387129.
- ^ Kastelein JJ, van Leuven SI, Burgess L, Evans GW, Kuivenhoven JA, Barter PJ, Revkin JH, Grobbee DE, Riley WA, Shear CL, Duggan WT, Bots ML (April 2007). "Effect of torcetrapib on carotid atherosclerosis in familial hypercholesterolemia". The New England Journal of Medicine. 356 (16): 1620–30. doi: 10.1056/NEJMoa071359. PMID 17387131.
- ^ "Pfizer Stops All Torcetrapib Clinical Trials in Interest of Patient Safety" (Press release). U.S. Food and Drug Administration. 3 December 2006.
- ^ El Harchaoui K, van der Steeg WA, Stroes ES, Kastelein JJ (August 2007). "The role of CETP inhibition in dyslipidemia". Current Atherosclerosis Reports. 9 (2): 125–33. doi: 10.1007/s11883-007-0008-5. PMID 17877921.
- ^ de Grooth GJ, Kuivenhoven JA, Stalenhoef AF, de Graaf J, Zwinderman AH, Posma JL, van Tol A, Kastelein JJ (May 2002). "Efficacy and safety of a novel cholesteryl ester transfer protein inhibitor, JTT-705, in humans: a randomized phase II dose-response study". Circulation. 105 (18): 2159–65. doi: 10.1161/01.CIR.0000015857.31889.7B. PMID 11994249.
- ^ "Merck announces its investigational CETP-Inhibitor, MK-0859, produced positive effects on lipids with no observed blood pressure changes". Reuters. Reuters, Inc. 4 October 2007. Retrieved 26 November 2013.
- ^ "Merck says will not seek approval of cholesterol treatment". Reuters. 2017. Retrieved 18 October 2017.
Further reading
- Okajima F (March 2002). "[Distribution of sphingosine 1-phosphate in plasma lipoproteins and its role in the regulation of the vascular cell functions]". Tanpakushitsu Kakusan Koso. Protein, Nucleic Acid, Enzyme. 47 (4 Suppl): 480–7. PMID 11915346.
- Barter PJ, Brewer HB, Chapman MJ, Hennekens CH, Rader DJ, Tall AR (February 2003). "Cholesteryl ester transfer protein: a novel target for raising HDL and inhibiting atherosclerosis". Arteriosclerosis, Thrombosis, and Vascular Biology. 23 (2): 160–7. doi: 10.1161/01.ATV.0000054658.91146.64. PMID 12588754.
- Dallinga-Thie GM, Dullaart RP, van Tol A (June 2007). "Concerted actions of cholesteryl ester transfer protein and phospholipid transfer protein in type 2 diabetes: effects of apolipoproteins". Current Opinion in Lipidology. 18 (3): 251–7. doi: 10.1097/MOL.0b013e3280e12685. PMID 17495597. S2CID 20012553.
External links
- Cholesterol+ester+transfer+proteins at the U.S. National Library of Medicine Medical Subject Headings (MeSH)