| Globin, extracellular | |
|---|---|
 | |
| Identifiers | |
| Symbol | Haemoglobin_extracell |
| InterPro | IPR014610 |
| Annelid erythrocruorin linker subunit, C-terminal | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | Eryth_link_C | ||||||||
| Pfam | PF16915 | ||||||||
| InterPro | IPR031639 | ||||||||
| CATH | 2gtlM02 | ||||||||
| SCOP2 | 8029676 / SCOPe / SUPFAM | ||||||||
| CDD | cd11673 | ||||||||
| |||||||||
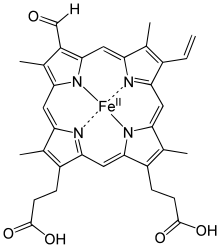
Erythrocruorin (from Greek eruthros "red" + Latin cruor "blood"), and the similar chlorocruorin (from Greek khlōros "green" + Latin cruor "blood"), are large oxygen-carrying hemeprotein complexes, which have a molecular mass greater than 3.5 million daltons. [1] Both are sometimes called giant hemoglobin or hexagonal bilayer haemoglobin. They are found in many annelids and arthropods (including some insects). [2]
Chlorocruorin is particularly found in certain marine polychaetes. [3] [4] [5]
Structure
Two structures of erythrocruorin have been resolved. The protein is a highly symmetric assembly made from heme-binding globins and unique linker proteins. [1] [6]
The only significant difference between chlorocruorin and erythrocruorin is that chlorocruorin carries an abnormal heme group structure. Both contain many 16–17 kDa myoglobin-like subunits arranged in a giant complex of over a hundred subunits with interlinking proteins as well with a total weight exceeding 3600 kDa. [6]
Giant hemoglobin is composed of multiple heme-containing globin chains and linker ( InterPro: IPR031639) chains. Each species have different amounts of genes for these chains. For example, while a Lamellibrachia sp. has four kinds of globin chains and two kinds of linker chains, Sabella spallanzanii has three globin chains and three linker chains. [6] The exact stoichiometric ratios and arrangement is unknown, but is thought to resemble that of erythocrorins.
Properties
Erythrocruorin has a weaker affinity for oxygen than that of most hemoglobins. A dichromatic compound, chlorocruorin is noted for appearing green in dilute solutions, though it appears light red when found in concentrated solutions. [7] [8] [9]
This enormous macromolecule is typically found free floating in the plasma, and not contained within red blood cells. [6] [10]
References
- ^ a b Royer WE, Strand K, van Heel M, Hendrickson WA (June 2000). "Structural hierarchy in erythrocruorin, the giant respiratory assemblage of annelids". Proceedings of the National Academy of Sciences of the United States of America. 97 (13): 7107–11. Bibcode: 2000PNAS...97.7107R. doi: 10.1073/pnas.97.13.7107. PMC 16507. PMID 10860978.
- ^ Ruggiero Bachega JF, Vasconcelos Maluf F, Andi B, D'Muniz Pereira H, Falsarella Carazzollea M, Orville AM, et al. (June 2015). "The structure of the giant haemoglobin from Glossoscolex paulistus". Acta Crystallographica. Section D, Biological Crystallography. 71 (Pt 6): 1257–71. doi: 10.1107/S1399004715005453. PMID 26057666.
- ^ H. Munro Fox (1 April 1933). "The Blood Circulation of Animals Possessing Chlorocruorin". Proceedings of the Royal Society B. 112 (779): 479–495. doi: 10.1098/rspb.1938.0042. JSTOR 81599.
- ^ R. F. Ewer; H. Munro Fox (9 August 1940). "On the Function of Chlorocruorin". Proceedings of the Royal Society B. 129 (855): 137–153. Bibcode: 1940RSPSB.129..137E. doi: 10.1098/rspb.1940.0033. JSTOR 82389.
- ^ D.W. Ewer (1941). "The blood systems of Sabella and Spirographis". Quarterly Journal of Microscopical Science. 82 (s2): 587–619. Retrieved 1 May 2010.
- ^ a b c d Pallavicini A, Negrisolo E, Barbato R, Dewilde S, Ghiretti-Magaldi A, Moens L, Lanfranchi G (July 2001). "The primary structure of globin and linker chains from the chlorocruorin of the polychaete Sabella spallanzanii". The Journal of Biological Chemistry. 276 (28): 26384–90. doi: 10.1074/jbc.M006939200. PMID 11294828.
- ^ H. Munro Fox (1 February 1926). "Chlorocruorin: A Pigment Allied to Haemoglobin". Proceedings of the Royal Society B. 99 (696): 199–220. doi: 10.1098/rspb.1926.0008. JSTOR 81088.
- ^ H. Munro Fox (1 September 1932). "The Oxygen Affinity of Chlorocruorin". Proceedings of the Royal Society B. 111 (772): 356–363. doi: 10.1098/rspb.1932.0060. JSTOR 81716.
- ^ H. Munro Fox (19 October 1949). "On Chlorocruorin and Haemoglobin". Proceedings of the Royal Society B. 136 (884): 378–388. Bibcode: 1949RSPSB.136..378F. doi: 10.1098/rspb.1949.0031. JSTOR 82565. PMID 18143368. S2CID 6133526.
- ^ Lamy JN, Green BN, Toulmond A, Wall JS, Weber RE, Vinogradov SN (19 December 1996). "Giant Hexagonal Bilayer Hemoglobins". Chem Rev. 96 (8): 3113–3124. doi: 10.1021/cr9600058. PMID 11848854.
External links
- Erythrocruorins at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- chlorocruorin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- RSDB Molecule of the Month: 159 Erythrocruorin
| Globin, extracellular | |
|---|---|
 | |
| Identifiers | |
| Symbol | Haemoglobin_extracell |
| InterPro | IPR014610 |
| Annelid erythrocruorin linker subunit, C-terminal | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | Eryth_link_C | ||||||||
| Pfam | PF16915 | ||||||||
| InterPro | IPR031639 | ||||||||
| CATH | 2gtlM02 | ||||||||
| SCOP2 | 8029676 / SCOPe / SUPFAM | ||||||||
| CDD | cd11673 | ||||||||
| |||||||||

Erythrocruorin (from Greek eruthros "red" + Latin cruor "blood"), and the similar chlorocruorin (from Greek khlōros "green" + Latin cruor "blood"), are large oxygen-carrying hemeprotein complexes, which have a molecular mass greater than 3.5 million daltons. [1] Both are sometimes called giant hemoglobin or hexagonal bilayer haemoglobin. They are found in many annelids and arthropods (including some insects). [2]
Chlorocruorin is particularly found in certain marine polychaetes. [3] [4] [5]
Structure
Two structures of erythrocruorin have been resolved. The protein is a highly symmetric assembly made from heme-binding globins and unique linker proteins. [1] [6]
The only significant difference between chlorocruorin and erythrocruorin is that chlorocruorin carries an abnormal heme group structure. Both contain many 16–17 kDa myoglobin-like subunits arranged in a giant complex of over a hundred subunits with interlinking proteins as well with a total weight exceeding 3600 kDa. [6]
Giant hemoglobin is composed of multiple heme-containing globin chains and linker ( InterPro: IPR031639) chains. Each species have different amounts of genes for these chains. For example, while a Lamellibrachia sp. has four kinds of globin chains and two kinds of linker chains, Sabella spallanzanii has three globin chains and three linker chains. [6] The exact stoichiometric ratios and arrangement is unknown, but is thought to resemble that of erythocrorins.
Properties
Erythrocruorin has a weaker affinity for oxygen than that of most hemoglobins. A dichromatic compound, chlorocruorin is noted for appearing green in dilute solutions, though it appears light red when found in concentrated solutions. [7] [8] [9]
This enormous macromolecule is typically found free floating in the plasma, and not contained within red blood cells. [6] [10]
References
- ^ a b Royer WE, Strand K, van Heel M, Hendrickson WA (June 2000). "Structural hierarchy in erythrocruorin, the giant respiratory assemblage of annelids". Proceedings of the National Academy of Sciences of the United States of America. 97 (13): 7107–11. Bibcode: 2000PNAS...97.7107R. doi: 10.1073/pnas.97.13.7107. PMC 16507. PMID 10860978.
- ^ Ruggiero Bachega JF, Vasconcelos Maluf F, Andi B, D'Muniz Pereira H, Falsarella Carazzollea M, Orville AM, et al. (June 2015). "The structure of the giant haemoglobin from Glossoscolex paulistus". Acta Crystallographica. Section D, Biological Crystallography. 71 (Pt 6): 1257–71. doi: 10.1107/S1399004715005453. PMID 26057666.
- ^ H. Munro Fox (1 April 1933). "The Blood Circulation of Animals Possessing Chlorocruorin". Proceedings of the Royal Society B. 112 (779): 479–495. doi: 10.1098/rspb.1938.0042. JSTOR 81599.
- ^ R. F. Ewer; H. Munro Fox (9 August 1940). "On the Function of Chlorocruorin". Proceedings of the Royal Society B. 129 (855): 137–153. Bibcode: 1940RSPSB.129..137E. doi: 10.1098/rspb.1940.0033. JSTOR 82389.
- ^ D.W. Ewer (1941). "The blood systems of Sabella and Spirographis". Quarterly Journal of Microscopical Science. 82 (s2): 587–619. Retrieved 1 May 2010.
- ^ a b c d Pallavicini A, Negrisolo E, Barbato R, Dewilde S, Ghiretti-Magaldi A, Moens L, Lanfranchi G (July 2001). "The primary structure of globin and linker chains from the chlorocruorin of the polychaete Sabella spallanzanii". The Journal of Biological Chemistry. 276 (28): 26384–90. doi: 10.1074/jbc.M006939200. PMID 11294828.
- ^ H. Munro Fox (1 February 1926). "Chlorocruorin: A Pigment Allied to Haemoglobin". Proceedings of the Royal Society B. 99 (696): 199–220. doi: 10.1098/rspb.1926.0008. JSTOR 81088.
- ^ H. Munro Fox (1 September 1932). "The Oxygen Affinity of Chlorocruorin". Proceedings of the Royal Society B. 111 (772): 356–363. doi: 10.1098/rspb.1932.0060. JSTOR 81716.
- ^ H. Munro Fox (19 October 1949). "On Chlorocruorin and Haemoglobin". Proceedings of the Royal Society B. 136 (884): 378–388. Bibcode: 1949RSPSB.136..378F. doi: 10.1098/rspb.1949.0031. JSTOR 82565. PMID 18143368. S2CID 6133526.
- ^ Lamy JN, Green BN, Toulmond A, Wall JS, Weber RE, Vinogradov SN (19 December 1996). "Giant Hexagonal Bilayer Hemoglobins". Chem Rev. 96 (8): 3113–3124. doi: 10.1021/cr9600058. PMID 11848854.
External links
- Erythrocruorins at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- chlorocruorin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- RSDB Molecule of the Month: 159 Erythrocruorin