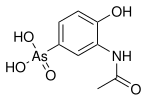
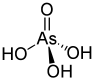
Arsonic acids are a subset of organoarsenic compounds defined as oxyacids where a pentavalent arsenic atom is bonded to two hydroxyl groups, a third oxygen atom (this one with a double bond), and an organic substituent. The salts/ conjugate bases of arsonic acids are called arsonates. Like all arsenic-containing compounds, arsonic acids are toxic and carcinogenic to humans. [1] [2]
Arsonic acid refers to H3 As O3, the case where the substituent is a single hydrogen atom. The other arsonic acids can simply be viewed as hydrocarbyl derivatives of this base case. Arsenic acid results when the substituent is a hydroxyl group. Methylarsonic acid results when the substituent is a methyl group. Phenylarsonic acid results when the substituent is a phenyl group.
The Béchamp reaction is used to produce arsonic acids from activated aromatic substrates. The reaction is an electrophilic aromatic substitution, using arsenic acid as the electrophile. The reaction proceeds according to this idealized stoichiometry:
- C6H5NH2 + H3AsO4 → H2O3AsC6H4NH2 + H2O

Arsanilic acid, carbarsone, nitarsone, and roxarsone were formerly used in poultry feed in order to promote growth and increase feed conversion. [3] [4] In addition, nitarsone and carbarsone can also prevent histomoniasis. [5] [6] [7] However, concern grew over whether or not the arsenic would be ingested by humans when they ate the poultry. In 2013, a study found that chickens who were fed roxarsone and other arsenic-containing feed additives tended to show elevated levels of arsenic in breast meat—three times as high—compared to chickens that were not fed any arsenical feed additives. [8] [9] On September of that year, Zoetis and Fleming Laboratories, the drugs' sponsors, voluntarily withdrew the FDA approvals for arsanilic acid, carbarsone, and roxarsone, [10] leaving only nitarsone approved until its approval for use in animal feed was withdrawn by the FDA in 2015. [11]
Difetarsone and carbarsone can be used to treat protozoal infections and Entamoeba histolytica infections. [12] [13] [14] Difetarsone can also be used to treat whipworm infections. [15] Arsanilic acid was discovered to treat sleeping sickness in the early 1900s, [16] but its usage in humans was discontinued after it was found to be too toxic. [17] Acetarsol is an anti-infective. [18]
Arsenic acid is technically not an arsonic acid because the substituent is a hydroxyl group, not a hydrocarbyl group, so arsenic acid has three hydroxyl groups bound to the arsenic atom, while arsonic acids only have two.
- ^ Aronson, Jeffrey K. (2009). Meyler's Side Effects of Antimicrobial Drugs. Elsevier. p. 834. ISBN 9780080932934. Retrieved October 7, 2013.
- ^ Styblo, M.; Del Razo, L. M.; Vega, L.; Germolec, D. R.; LeCluyse, E. L.; Hamilton, G. A.; Reed, W.; Wang, C.; Cullen, W. R.; Thomas, D. J. (2000). "Comparative toxicity of trivalent and pentavalent inorganic and methylated arsenicals in rat and human cells". Archives of Toxicology. 74 (6): 289–299. doi: 10.1007/s002040000134. PMID 11005674. S2CID 1025140.
- ^ Levander OA, ed. (1977). "Biological effects of arsenic on plants and animals: Domestic animals: Phenylarsonic feed additives". Arsenic: Medical and Biological Effects of Environmental Pollutants. Washington DC: National Academies Press. pp. 149–51. doi: 10.17226/9003. ISBN 978-0-309-02604-8. PMID 25101467.
- ^ "Arsanilic acid—MIB #4". Canadian Food Inspection Agency. Sep 2006. Archived from the original on 2012-12-13. Retrieved 3 Aug 2012.
- ^ U.S. Food and Drug Administration. "Animal Drugs @ FDA".
- ^ McDougald LR (1979). "Efficacy and compatibility of amprolium and carbarsone against Coccidiosis and blackhead in turkeys". Poult. Sci. 58 (1): 76–80. doi: 10.3382/ps.0580076. PMID 572970.
- ^ Worden AN, Wood EC (1973). "The effect of Carbarsone (33.6 per cent w-v p-ureidobenzene arsonic acid) on bodyweight gain, food conversion and tissue arsenic levels of turkey poults". J. Sci. Food Agric. 24 (1): 35–41. doi: 10.1002/jsfa.2740240107. PMID 4696593.
- ^ KE Nachman; PA Baron; G Raber; KA Francesconi; A Navas-Acien; DC Love (2013). "Roxarsone, Inorganic Arsenic, and Other Arsenic Species in Chicken: A U.S.-Based Market Basket Sample" (PDF). Environmental Health Perspectives. 121 (7): 818–824. doi: 10.1289/ehp.1206245. PMC 3701911. PMID 23694900. Archived from the original (PDF) on 2016-12-29. Retrieved 2018-10-09.
- ^ Sabrina Tavernise (May 11, 2013). "Study Finds an Increase in Arsenic Levels in Chicken". New York Times.
- ^ U.S. Food and Drug Administration (1 Oct 2013). "FDA response to citizen petition on arsenic-based animal drugs".
- ^ U.S. Food and Drug Administration (April 1, 2015). "FDA announces pending withdrawal of approval of nitarsone".
- ^ SASAKI T, YOKAGAWA M, WYKOFF DE, RITICHIE LS (1956). "Asymptomatic amebiasis; treatment with atabrine in combination with carbarsone or chiniofon". United States Armed Forces Medical Journal. 7 (3): 363–8. PMID 13299463.
- ^ RADKE RA (1955). "Ameboma of the intestine: an analysis of the disease as presented in 78 collected and 41 previously unreported cases". Ann. Intern. Med. 43 (5): 1048–66. doi: 10.7326/0003-4819-43-5-1048. PMID 13268997.
- ^ Aronson, Jeffrey K. (2009). Meyler's Side Effects of Antimicrobial Drugs. Elsevier. p. 834. ISBN 9780080932934. Retrieved October 7, 2013.
- ^ P.M. Leary; C. Jones; F. Douglas; S.T. Boyd (June 1972). "Difetarsone in Difetarsone in outpatient treatment of Trichuris trichiura infestation". Archives of Disease in Childhood. 49 (6): 486–8. doi: 10.1136/adc.49.6.486. PMC 1648795. PMID 4851370.
- ^ Boyce R (1907). "The treatment of sleeping sickness and other trypanosomiases by the Atoxyl and mercury method". BMJ. 2 (2437): 624–5. doi: 10.1136/bmj.2.2437.624. PMC 2358391. PMID 20763444.
- ^ Burke ET (1925). "The arseno-therapy of syphilis; stovarsol, and tryparsamide". British Journal of Venereal Diseases. 1 (4): 321–38. doi: 10.1136/sti.1.4.321. PMC 1046841. PMID 21772505.
- ^ Chen MY, Smith NA, Fox EF, Bingham JS, Barlow D (April 1999). "Acetarsol pessaries in the treatment of metronidazole resistant Trichomonas vaginalis". Int J STD AIDS. 10 (4): 277–80. doi: 10.1258/0956462991913943. PMID 12035784. S2CID 27353282.

Arsonic acids are a subset of organoarsenic compounds defined as oxyacids where a pentavalent arsenic atom is bonded to two hydroxyl groups, a third oxygen atom (this one with a double bond), and an organic substituent. The salts/ conjugate bases of arsonic acids are called arsonates. Like all arsenic-containing compounds, arsonic acids are toxic and carcinogenic to humans. [1] [2]
Arsonic acid refers to H3 As O3, the case where the substituent is a single hydrogen atom. The other arsonic acids can simply be viewed as hydrocarbyl derivatives of this base case. Arsenic acid results when the substituent is a hydroxyl group. Methylarsonic acid results when the substituent is a methyl group. Phenylarsonic acid results when the substituent is a phenyl group.
The Béchamp reaction is used to produce arsonic acids from activated aromatic substrates. The reaction is an electrophilic aromatic substitution, using arsenic acid as the electrophile. The reaction proceeds according to this idealized stoichiometry:
- C6H5NH2 + H3AsO4 → H2O3AsC6H4NH2 + H2O

Arsanilic acid, carbarsone, nitarsone, and roxarsone were formerly used in poultry feed in order to promote growth and increase feed conversion. [3] [4] In addition, nitarsone and carbarsone can also prevent histomoniasis. [5] [6] [7] However, concern grew over whether or not the arsenic would be ingested by humans when they ate the poultry. In 2013, a study found that chickens who were fed roxarsone and other arsenic-containing feed additives tended to show elevated levels of arsenic in breast meat—three times as high—compared to chickens that were not fed any arsenical feed additives. [8] [9] On September of that year, Zoetis and Fleming Laboratories, the drugs' sponsors, voluntarily withdrew the FDA approvals for arsanilic acid, carbarsone, and roxarsone, [10] leaving only nitarsone approved until its approval for use in animal feed was withdrawn by the FDA in 2015. [11]
Difetarsone and carbarsone can be used to treat protozoal infections and Entamoeba histolytica infections. [12] [13] [14] Difetarsone can also be used to treat whipworm infections. [15] Arsanilic acid was discovered to treat sleeping sickness in the early 1900s, [16] but its usage in humans was discontinued after it was found to be too toxic. [17] Acetarsol is an anti-infective. [18]
Arsenic acid is technically not an arsonic acid because the substituent is a hydroxyl group, not a hydrocarbyl group, so arsenic acid has three hydroxyl groups bound to the arsenic atom, while arsonic acids only have two.
- ^ Aronson, Jeffrey K. (2009). Meyler's Side Effects of Antimicrobial Drugs. Elsevier. p. 834. ISBN 9780080932934. Retrieved October 7, 2013.
- ^ Styblo, M.; Del Razo, L. M.; Vega, L.; Germolec, D. R.; LeCluyse, E. L.; Hamilton, G. A.; Reed, W.; Wang, C.; Cullen, W. R.; Thomas, D. J. (2000). "Comparative toxicity of trivalent and pentavalent inorganic and methylated arsenicals in rat and human cells". Archives of Toxicology. 74 (6): 289–299. doi: 10.1007/s002040000134. PMID 11005674. S2CID 1025140.
- ^ Levander OA, ed. (1977). "Biological effects of arsenic on plants and animals: Domestic animals: Phenylarsonic feed additives". Arsenic: Medical and Biological Effects of Environmental Pollutants. Washington DC: National Academies Press. pp. 149–51. doi: 10.17226/9003. ISBN 978-0-309-02604-8. PMID 25101467.
- ^ "Arsanilic acid—MIB #4". Canadian Food Inspection Agency. Sep 2006. Archived from the original on 2012-12-13. Retrieved 3 Aug 2012.
- ^ U.S. Food and Drug Administration. "Animal Drugs @ FDA".
- ^ McDougald LR (1979). "Efficacy and compatibility of amprolium and carbarsone against Coccidiosis and blackhead in turkeys". Poult. Sci. 58 (1): 76–80. doi: 10.3382/ps.0580076. PMID 572970.
- ^ Worden AN, Wood EC (1973). "The effect of Carbarsone (33.6 per cent w-v p-ureidobenzene arsonic acid) on bodyweight gain, food conversion and tissue arsenic levels of turkey poults". J. Sci. Food Agric. 24 (1): 35–41. doi: 10.1002/jsfa.2740240107. PMID 4696593.
- ^ KE Nachman; PA Baron; G Raber; KA Francesconi; A Navas-Acien; DC Love (2013). "Roxarsone, Inorganic Arsenic, and Other Arsenic Species in Chicken: A U.S.-Based Market Basket Sample" (PDF). Environmental Health Perspectives. 121 (7): 818–824. doi: 10.1289/ehp.1206245. PMC 3701911. PMID 23694900. Archived from the original (PDF) on 2016-12-29. Retrieved 2018-10-09.
- ^ Sabrina Tavernise (May 11, 2013). "Study Finds an Increase in Arsenic Levels in Chicken". New York Times.
- ^ U.S. Food and Drug Administration (1 Oct 2013). "FDA response to citizen petition on arsenic-based animal drugs".
- ^ U.S. Food and Drug Administration (April 1, 2015). "FDA announces pending withdrawal of approval of nitarsone".
- ^ SASAKI T, YOKAGAWA M, WYKOFF DE, RITICHIE LS (1956). "Asymptomatic amebiasis; treatment with atabrine in combination with carbarsone or chiniofon". United States Armed Forces Medical Journal. 7 (3): 363–8. PMID 13299463.
- ^ RADKE RA (1955). "Ameboma of the intestine: an analysis of the disease as presented in 78 collected and 41 previously unreported cases". Ann. Intern. Med. 43 (5): 1048–66. doi: 10.7326/0003-4819-43-5-1048. PMID 13268997.
- ^ Aronson, Jeffrey K. (2009). Meyler's Side Effects of Antimicrobial Drugs. Elsevier. p. 834. ISBN 9780080932934. Retrieved October 7, 2013.
- ^ P.M. Leary; C. Jones; F. Douglas; S.T. Boyd (June 1972). "Difetarsone in Difetarsone in outpatient treatment of Trichuris trichiura infestation". Archives of Disease in Childhood. 49 (6): 486–8. doi: 10.1136/adc.49.6.486. PMC 1648795. PMID 4851370.
- ^ Boyce R (1907). "The treatment of sleeping sickness and other trypanosomiases by the Atoxyl and mercury method". BMJ. 2 (2437): 624–5. doi: 10.1136/bmj.2.2437.624. PMC 2358391. PMID 20763444.
- ^ Burke ET (1925). "The arseno-therapy of syphilis; stovarsol, and tryparsamide". British Journal of Venereal Diseases. 1 (4): 321–38. doi: 10.1136/sti.1.4.321. PMC 1046841. PMID 21772505.
- ^ Chen MY, Smith NA, Fox EF, Bingham JS, Barlow D (April 1999). "Acetarsol pessaries in the treatment of metronidazole resistant Trichomonas vaginalis". Int J STD AIDS. 10 (4): 277–80. doi: 10.1258/0956462991913943. PMID 12035784. S2CID 27353282.