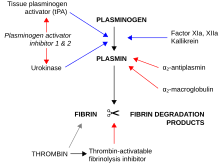
Alpha 2-antiplasmin (or α2-antiplasmin or plasmin inhibitor) is a serine protease inhibitor (serpin) responsible for inactivating plasmin. [5] Plasmin is an important enzyme that participates in fibrinolysis and degradation of various other proteins. This protein is encoded by the SERPINF2 gene. [6]
Role in disease
Very few cases (<20) of A2AP deficiency have been described. As plasmin degrades blood clots, impaired inhibition of plasmin leads to a bleeding tendency, which was severe in the cases reported.
In liver cirrhosis, there is decreased production of alpha 2-antiplasmin, leading to decreased inactivation of plasmin and an increase in fibrinolysis. This is associated with an increase risk of bleeding in liver disease. [7]
Interactions
Alpha 2-antiplasmin has been shown to interact with:
See also
References
- ^ a b c ENSG00000276838 GRCh38: Ensembl release 89: ENSG00000167711, ENSG00000276838 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000038224 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Wu G, Quek AJ, Caradoc-Davies TT, Ekkel SM, Mazzitelli B, Whisstock JC, Law RH (2019-03-05). "Structural studies of plasmin inhibition". Biochemical Society Transactions. 47 (2): 541–557. doi: 10.1042/bst20180211. ISSN 0300-5127. PMID 30837322. S2CID 73463150.
- ^ "Entrez Gene: SERPINF2 serpin peptidase inhibitor, clade F (alpha-2 antiplasmin, pigment epithelium derived factor), member 2".
- ^ Sattar, Husain. Fundamentals of Pathology. Pathoma LLC, 2011, p. 36.
- ^ a b Shieh BH, Travis J (May 1987). "The reactive site of human alpha 2-antiplasmin". The Journal of Biological Chemistry. 262 (13): 6055–9. doi: 10.1016/S0021-9258(18)45536-6. PMID 2437112.
- ^ Brower MS, Harpel PC (Aug 1982). "Proteolytic cleavage and inactivation of alpha 2-plasmin inhibitor and C1 inactivator by human polymorphonuclear leukocyte elastase". The Journal of Biological Chemistry. 257 (16): 9849–54. doi: 10.1016/S0021-9258(18)34149-8. PMID 6980881.
- ^ Wiman B, Collen D (Sep 1979). "On the mechanism of the reaction between human alpha 2-antiplasmin and plasmin". The Journal of Biological Chemistry. 254 (18): 9291–7. doi: 10.1016/S0021-9258(19)86843-6. PMID 158022.
Further reading
- Martí-Fàbregas J, Borrell M, Cocho D, Martínez-Ramírez S, Martínez-Corral M, Fontcuberta J, Martí-Vilalta JL (Jan 2008). "Change in hemostatic markers after recombinant tissue-type plasminogen activator is not associated with the chance of recanalization". Stroke: A Journal of Cerebral Circulation. 39 (1): 234–6. doi: 10.1161/STROKEAHA.107.493767. PMID 18048863. S2CID 10052168.
- Nielsen VG (Oct 2007). "Hydroxyethyl starch enhances fibrinolysis in human plasma by diminishing alpha2-antiplasmin-plasmin interactions". Blood Coagulation & Fibrinolysis. 18 (7): 647–56. doi: 10.1097/MBC.0b013e3282a167dc. PMID 17890952. S2CID 45608070.
- Sazonova IY, Thomas BM, Gladysheva IP, Houng AK, Reed GL (Oct 2007). "Fibrinolysis is amplified by converting alpha-antiplasmin from a plasmin inhibitor to a substrate". Journal of Thrombosis and Haemostasis. 5 (10): 2087–94. doi: 10.1111/j.1538-7836.2007.02652.x. PMID 17883703. S2CID 2907615.
- Mutch NJ, Thomas L, Moore NR, Lisiak KM, Booth NA (Apr 2007). "TAFIa, PAI-1 and alpha-antiplasmin: complementary roles in regulating lysis of thrombi and plasma clots". Journal of Thrombosis and Haemostasis. 5 (4): 812–7. doi: 10.1111/j.1538-7836.2007.02430.x. PMID 17388801. S2CID 40278627.
- Christiansen VJ, Jackson KW, Lee KN, McKee PA (Jun 2007). "The effect of a single nucleotide polymorphism on human alpha 2-antiplasmin activity". Blood. 109 (12): 5286–92. doi: 10.1182/blood-2007-01-065185. PMC 1890835. PMID 17317851.
- Hayashido Y, Hamana T, Ishida Y, Shintani T, Koizumi K, Okamoto T (Feb 2007). "Induction of alpha2-antiplasmin inhibits E-cadherin processing mediated by the plasminogen activator/plasmin system, leading to suppression of progression of oral squamous cell carcinoma via upregulation of cell-cell adhesion". Oncology Reports. 17 (2): 417–23. doi: 10.3892/or.17.2.417. PMID 17203182.
- Shibata N, Kawarai T, Meng Y, Lee JH, Lee HS, Wakutani Y, Shibata E, Pathan N, Bi A, Sato C, Sorbi S, Bruni AC, Duara R, Mayeux R, Farrer LA, George-Hyslop PS, Rogaeva E (Jul 2007). "Association studies between the plasmin genes and late-onset Alzheimer's disease". Neurobiology of Aging. 28 (7): 1041–3. doi: 10.1016/j.neurobiolaging.2006.05.028. PMC 2647723. PMID 16828203.
- Liu T, Qian WJ, Gritsenko MA, Camp DG, Monroe ME, Moore RJ, Smith RD (December 2005). "Human plasma N-glycoproteome analysis by immunoaffinity subtraction, hydrazide chemistry, and mass spectrometry". Journal of Proteome Research. 4 (6): 2070–80. doi: 10.1021/pr0502065. PMC 1850943. PMID 16335952.
- Lee KN, Jackson KW, Christiansen VJ, Chung KH, McKee PA (May 2004). "A novel plasma proteinase potentiates alpha2-antiplasmin inhibition of fibrin digestion". Blood. 103 (10): 3783–8. doi: 10.1182/blood-2003-12-4240. PMID 14751930.
- Anderson NL, Polanski M, Pieper R, Gatlin T, Tirumalai RS, Conrads TP, Veenstra TD, Adkins JN, Pounds JG, Fagan R, Lobley A (Apr 2004). "The human plasma proteome: a nonredundant list developed by combination of four separate sources". Molecular & Cellular Proteomics. 3 (4): 311–26. doi: 10.1074/mcp.M300127-MCP200. PMID 14718574.
- Kapadia C, Yousef GM, Mellati AA, Magklara A, Wasney GA, Diamandis EP (Jan 2004). "Complex formation between human kallikrein 13 and serum protease inhibitors". Clinica Chimica Acta; International Journal of Clinical Chemistry. 339 (1–2): 157–67. doi: 10.1016/j.cccn.2003.10.009. PMID 14687906.
- Matsuno H, Okada K, Ueshima S, Matsuo O, Kozawa O (Aug 2003). "Alpha2-antiplasmin plays a significant role in acute pulmonary embolism". Journal of Thrombosis and Haemostasis. 1 (8): 1734–9. doi: 10.1046/j.1538-7836.2003.00252.x. PMID 12911586. S2CID 72056689.
- Magklara A, Mellati AA, Wasney GA, Little SP, Sotiropoulou G, Becker GW, Diamandis EP (Aug 2003). "Characterization of the enzymatic activity of human kallikrein 6: Autoactivation, substrate specificity, and regulation by inhibitors". Biochemical and Biophysical Research Communications. 307 (4): 948–55. doi: 10.1016/S0006-291X(03)01271-3. PMID 12878203.
- Cardoso C, Leventer RJ, Ward HL, Toyo-Oka K, Chung J, Gross A, Martin CL, Allanson J, Pilz DT, Olney AH, Mutchinick OM, Hirotsune S, Wynshaw-Boris A, Dobyns WB, Ledbetter DH (Apr 2003). "Refinement of a 400-kb critical region allows genotypic differentiation between isolated lissencephaly, Miller-Dieker syndrome, and other phenotypes secondary to deletions of 17p13.3". American Journal of Human Genetics. 72 (4): 918–30. doi: 10.1086/374320. PMC 1180354. PMID 12621583.
- Frank PS, Douglas JT, Locher M, Llinás M, Schaller J (Feb 2003). "Structural/functional characterization of the alpha 2-plasmin inhibitor C-terminal peptide". Biochemistry. 42 (4): 1078–85. doi: 10.1021/bi026917n. PMID 12549929.
- Turner RB, Liu L, Sazonova IY, Reed GL (Sep 2002). "Structural elements that govern the substrate specificity of the clot-dissolving enzyme plasmin". The Journal of Biological Chemistry. 277 (36): 33068–74. doi: 10.1074/jbc.M203782200. PMID 12080056.
- Askew YS, Pak SC, Luke CJ, Askew DJ, Cataltepe S, Mills DR, Kato H, Lehoczky J, Dewar K, Birren B, Silverman GA (Dec 2001). "SERPINB12 is a novel member of the human ov-serpin family that is widely expressed and inhibits trypsin-like serine proteinases". The Journal of Biological Chemistry. 276 (52): 49320–30. doi: 10.1074/jbc.M108879200. PMID 11604408.
- Uszynski M, Klyszejko A, Zekanowska E (Dec 2000). "Plasminogen, alpha(2)-antiplasmin and complexes of plasmin-alpha(2)-antiplasmin (PAP) in amniotic fluid and blood plasma of parturient women". European Journal of Obstetrics, Gynecology, and Reproductive Biology. 93 (2): 167–71. doi: 10.1016/S0301-2115(00)00283-9. PMID 11074138.
- Hevessy Z, Patthy A, Kárpáti L, Muszbek L (Aug 2000). "alpha(2)-plasmin inhibitor is a substrate for tissue transglutaminase: an in vitro study". Thrombosis Research. 99 (4): 399–406. doi: 10.1016/S0049-3848(00)00261-9. PMID 10963790.
External links
- The MEROPS online database for peptidases and their inhibitors: I04.023
- alpha-2+Antiplasmin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- SERPINF2+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Human SERPINF2 genome location and SERPINF2 gene details page in the UCSC Genome Browser.
| SERPINF2 | |||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Identifiers | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Aliases | SERPINF2, A2AP, AAP, ALPHA-2-PI, API, PLI, serpin family F member 2, alpha2AP | ||||||||||||||||||||||||||||||||||||||||||||||||||
| External IDs | OMIM: 613168; MGI: 107173; HomoloGene: 719; GeneCards: SERPINF2; OMA: SERPINF2 - orthologs | ||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Wikidata | |||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||
Alpha 2-antiplasmin (or α2-antiplasmin or plasmin inhibitor) is a serine protease inhibitor (serpin) responsible for inactivating plasmin. [5] Plasmin is an important enzyme that participates in fibrinolysis and degradation of various other proteins. This protein is encoded by the SERPINF2 gene. [6]

Role in disease
Very few cases (<20) of A2AP deficiency have been described. As plasmin degrades blood clots, impaired inhibition of plasmin leads to a bleeding tendency, which was severe in the cases reported.
In liver cirrhosis, there is decreased production of alpha 2-antiplasmin, leading to decreased inactivation of plasmin and an increase in fibrinolysis. This is associated with an increase risk of bleeding in liver disease. [7]
Interactions
Alpha 2-antiplasmin has been shown to interact with:
See also
References
- ^ a b c ENSG00000276838 GRCh38: Ensembl release 89: ENSG00000167711, ENSG00000276838 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000038224 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Wu G, Quek AJ, Caradoc-Davies TT, Ekkel SM, Mazzitelli B, Whisstock JC, Law RH (2019-03-05). "Structural studies of plasmin inhibition". Biochemical Society Transactions. 47 (2): 541–557. doi: 10.1042/bst20180211. ISSN 0300-5127. PMID 30837322. S2CID 73463150.
- ^ "Entrez Gene: SERPINF2 serpin peptidase inhibitor, clade F (alpha-2 antiplasmin, pigment epithelium derived factor), member 2".
- ^ Sattar, Husain. Fundamentals of Pathology. Pathoma LLC, 2011, p. 36.
- ^ a b Shieh BH, Travis J (May 1987). "The reactive site of human alpha 2-antiplasmin". The Journal of Biological Chemistry. 262 (13): 6055–9. doi: 10.1016/S0021-9258(18)45536-6. PMID 2437112.
- ^ Brower MS, Harpel PC (Aug 1982). "Proteolytic cleavage and inactivation of alpha 2-plasmin inhibitor and C1 inactivator by human polymorphonuclear leukocyte elastase". The Journal of Biological Chemistry. 257 (16): 9849–54. doi: 10.1016/S0021-9258(18)34149-8. PMID 6980881.
- ^ Wiman B, Collen D (Sep 1979). "On the mechanism of the reaction between human alpha 2-antiplasmin and plasmin". The Journal of Biological Chemistry. 254 (18): 9291–7. doi: 10.1016/S0021-9258(19)86843-6. PMID 158022.
Further reading
- Martí-Fàbregas J, Borrell M, Cocho D, Martínez-Ramírez S, Martínez-Corral M, Fontcuberta J, Martí-Vilalta JL (Jan 2008). "Change in hemostatic markers after recombinant tissue-type plasminogen activator is not associated with the chance of recanalization". Stroke: A Journal of Cerebral Circulation. 39 (1): 234–6. doi: 10.1161/STROKEAHA.107.493767. PMID 18048863. S2CID 10052168.
- Nielsen VG (Oct 2007). "Hydroxyethyl starch enhances fibrinolysis in human plasma by diminishing alpha2-antiplasmin-plasmin interactions". Blood Coagulation & Fibrinolysis. 18 (7): 647–56. doi: 10.1097/MBC.0b013e3282a167dc. PMID 17890952. S2CID 45608070.
- Sazonova IY, Thomas BM, Gladysheva IP, Houng AK, Reed GL (Oct 2007). "Fibrinolysis is amplified by converting alpha-antiplasmin from a plasmin inhibitor to a substrate". Journal of Thrombosis and Haemostasis. 5 (10): 2087–94. doi: 10.1111/j.1538-7836.2007.02652.x. PMID 17883703. S2CID 2907615.
- Mutch NJ, Thomas L, Moore NR, Lisiak KM, Booth NA (Apr 2007). "TAFIa, PAI-1 and alpha-antiplasmin: complementary roles in regulating lysis of thrombi and plasma clots". Journal of Thrombosis and Haemostasis. 5 (4): 812–7. doi: 10.1111/j.1538-7836.2007.02430.x. PMID 17388801. S2CID 40278627.
- Christiansen VJ, Jackson KW, Lee KN, McKee PA (Jun 2007). "The effect of a single nucleotide polymorphism on human alpha 2-antiplasmin activity". Blood. 109 (12): 5286–92. doi: 10.1182/blood-2007-01-065185. PMC 1890835. PMID 17317851.
- Hayashido Y, Hamana T, Ishida Y, Shintani T, Koizumi K, Okamoto T (Feb 2007). "Induction of alpha2-antiplasmin inhibits E-cadherin processing mediated by the plasminogen activator/plasmin system, leading to suppression of progression of oral squamous cell carcinoma via upregulation of cell-cell adhesion". Oncology Reports. 17 (2): 417–23. doi: 10.3892/or.17.2.417. PMID 17203182.
- Shibata N, Kawarai T, Meng Y, Lee JH, Lee HS, Wakutani Y, Shibata E, Pathan N, Bi A, Sato C, Sorbi S, Bruni AC, Duara R, Mayeux R, Farrer LA, George-Hyslop PS, Rogaeva E (Jul 2007). "Association studies between the plasmin genes and late-onset Alzheimer's disease". Neurobiology of Aging. 28 (7): 1041–3. doi: 10.1016/j.neurobiolaging.2006.05.028. PMC 2647723. PMID 16828203.
- Liu T, Qian WJ, Gritsenko MA, Camp DG, Monroe ME, Moore RJ, Smith RD (December 2005). "Human plasma N-glycoproteome analysis by immunoaffinity subtraction, hydrazide chemistry, and mass spectrometry". Journal of Proteome Research. 4 (6): 2070–80. doi: 10.1021/pr0502065. PMC 1850943. PMID 16335952.
- Lee KN, Jackson KW, Christiansen VJ, Chung KH, McKee PA (May 2004). "A novel plasma proteinase potentiates alpha2-antiplasmin inhibition of fibrin digestion". Blood. 103 (10): 3783–8. doi: 10.1182/blood-2003-12-4240. PMID 14751930.
- Anderson NL, Polanski M, Pieper R, Gatlin T, Tirumalai RS, Conrads TP, Veenstra TD, Adkins JN, Pounds JG, Fagan R, Lobley A (Apr 2004). "The human plasma proteome: a nonredundant list developed by combination of four separate sources". Molecular & Cellular Proteomics. 3 (4): 311–26. doi: 10.1074/mcp.M300127-MCP200. PMID 14718574.
- Kapadia C, Yousef GM, Mellati AA, Magklara A, Wasney GA, Diamandis EP (Jan 2004). "Complex formation between human kallikrein 13 and serum protease inhibitors". Clinica Chimica Acta; International Journal of Clinical Chemistry. 339 (1–2): 157–67. doi: 10.1016/j.cccn.2003.10.009. PMID 14687906.
- Matsuno H, Okada K, Ueshima S, Matsuo O, Kozawa O (Aug 2003). "Alpha2-antiplasmin plays a significant role in acute pulmonary embolism". Journal of Thrombosis and Haemostasis. 1 (8): 1734–9. doi: 10.1046/j.1538-7836.2003.00252.x. PMID 12911586. S2CID 72056689.
- Magklara A, Mellati AA, Wasney GA, Little SP, Sotiropoulou G, Becker GW, Diamandis EP (Aug 2003). "Characterization of the enzymatic activity of human kallikrein 6: Autoactivation, substrate specificity, and regulation by inhibitors". Biochemical and Biophysical Research Communications. 307 (4): 948–55. doi: 10.1016/S0006-291X(03)01271-3. PMID 12878203.
- Cardoso C, Leventer RJ, Ward HL, Toyo-Oka K, Chung J, Gross A, Martin CL, Allanson J, Pilz DT, Olney AH, Mutchinick OM, Hirotsune S, Wynshaw-Boris A, Dobyns WB, Ledbetter DH (Apr 2003). "Refinement of a 400-kb critical region allows genotypic differentiation between isolated lissencephaly, Miller-Dieker syndrome, and other phenotypes secondary to deletions of 17p13.3". American Journal of Human Genetics. 72 (4): 918–30. doi: 10.1086/374320. PMC 1180354. PMID 12621583.
- Frank PS, Douglas JT, Locher M, Llinás M, Schaller J (Feb 2003). "Structural/functional characterization of the alpha 2-plasmin inhibitor C-terminal peptide". Biochemistry. 42 (4): 1078–85. doi: 10.1021/bi026917n. PMID 12549929.
- Turner RB, Liu L, Sazonova IY, Reed GL (Sep 2002). "Structural elements that govern the substrate specificity of the clot-dissolving enzyme plasmin". The Journal of Biological Chemistry. 277 (36): 33068–74. doi: 10.1074/jbc.M203782200. PMID 12080056.
- Askew YS, Pak SC, Luke CJ, Askew DJ, Cataltepe S, Mills DR, Kato H, Lehoczky J, Dewar K, Birren B, Silverman GA (Dec 2001). "SERPINB12 is a novel member of the human ov-serpin family that is widely expressed and inhibits trypsin-like serine proteinases". The Journal of Biological Chemistry. 276 (52): 49320–30. doi: 10.1074/jbc.M108879200. PMID 11604408.
- Uszynski M, Klyszejko A, Zekanowska E (Dec 2000). "Plasminogen, alpha(2)-antiplasmin and complexes of plasmin-alpha(2)-antiplasmin (PAP) in amniotic fluid and blood plasma of parturient women". European Journal of Obstetrics, Gynecology, and Reproductive Biology. 93 (2): 167–71. doi: 10.1016/S0301-2115(00)00283-9. PMID 11074138.
- Hevessy Z, Patthy A, Kárpáti L, Muszbek L (Aug 2000). "alpha(2)-plasmin inhibitor is a substrate for tissue transglutaminase: an in vitro study". Thrombosis Research. 99 (4): 399–406. doi: 10.1016/S0049-3848(00)00261-9. PMID 10963790.
External links
- The MEROPS online database for peptidases and their inhibitors: I04.023
- alpha-2+Antiplasmin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- SERPINF2+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Human SERPINF2 genome location and SERPINF2 gene details page in the UCSC Genome Browser.




