| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
1,3-Oxazolidin-2-one | |||
| Other names
2-Oxazolidone
2-Oxazolidinone | |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.007.129 | ||
| KEGG | |||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H5NO2 | |||
| Molar mass | 87.077 g/mol | ||
| Appearance | Solid | ||
| Melting point | 86 to 89 °C (187 to 192 °F; 359 to 362 K) | ||
| Boiling point | 220 °C (428 °F; 493 K) at 48 torr | ||
| Related compounds | |||
Related compounds
|
Oxazolidine | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
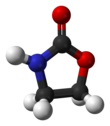
2-Oxazolidone is a heterocyclic organic compound containing both nitrogen and oxygen in a 5-membered ring.
Oxazolidinones
Evans auxiliaries
Oxazolidinones are a class of compounds containing 2-oxazolidone in the structure. In chemistry, they are useful as Evans auxiliaries, which are used for chiral synthesis. Usually, the acid chloride substrate reacts with the oxazolidinone to form an imide. Substituents at the 4 and 5 position of the oxazolidinone direct any aldol reaction to the alpha position of the carbonyl of the substrate.[ citation needed]
Pharmaceuticals
Oxazolidinones are mainly used as antimicrobials. The antibacterial effect of oxazolidinones is by working as protein synthesis inhibitors, targeting an early step involving the binding of N-formylmethionyl-tRNA to the ribosome. [1] (See Linezolid#Pharmacodynamics)
Some of the most important oxazolidinones are antibiotics. [2]
Examples of antibiotic oxazolidinones include:
- Linezolid (Zyvox), which is available for intravenous administration and also has the advantage of having excellent oral bioavailability.
- Posizolid, which appears to have excellent, targeted bactericidal activity against all common gram-positive bacteria, regardless of resistance to other classes of antibiotics. [3]
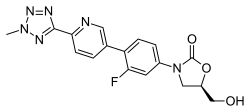
- Tedizolid, (Sivextro) which is approved for acute skin infections
- Radezolid (RX-1741) has completed some phase-II clinical trials. [4]
- Cycloserine is a second line drug against tuberculosis. Note that cycloserine, while technically an oxazolidone, has a different mechanism of action and substantially different properties from the aforementioned compounds.
- Contezolid (MRX-I) has reported phase 1 data [5] and phase III data. [6] In 2021, a new drug summary was published by ADIS Press. [7] In June 2021, marketing approval was granted by the Chinese National Medical Products Administration (NMPA) for use of oral contezolid in moderate to severe complicated skin and skin structure infections. [8] In June 2022, contezolid oral tablets and contezolid acefosamil IV (a prodrug of contezolid) began Phase 3 global clinical trials in moderate to severe diabetic foot infections. [9] An additional global Phase 3 study is planned for acute bacterial skin and skin structure infections (ABSSSI) for the combination of contezolid and contezolid acefosamil.
- An oxazolidinone derivative used for other purposes is rivaroxaban, which is approved by the FDA for venous thromboembolism prophylaxis.
History

The first ever used oxazolidinone was cycloserine (4-amino-1,2-oxazolidin-3-one), a second line drug against tuberculosis since 1956. [10]
Developed during the nineties when several bacterial strains were becoming resistant against such antibiotics as vancomycin. Linezolid (Zyvox) is the first approved agent in the class (FDA approval April 2000).

The first commercially available 1,3-oxazolidinone antibiotic was linezolid, discovered and developed by Pharmacia & Upjohn.[ citation needed]

In 2002 AstraZeneca began investigating posizolid, which is in clinical trials for use in humans. [11]
See also
- Oxazolidine – the ring without the carbonyl group
- Oxazolone – the unsaturated analogues
References
- ^ Shinabarger, D. (1999). "Mechanism of action of the oxazolidinone antibacterial agents". Expert Opinion on Investigational Drugs. 8 (8): 1195–1202. doi: 10.1517/13543784.8.8.1195. PMID 15992144.
- ^ Sonia Ilaria Maffioli (2014). "A Chemist's Survey of Different Antibiotic Classes". In Claudio O. Gualerzi; Letizia Brandi; Attilio Fabbretti; Cynthia L. Pon (eds.). Antibiotics: Targets, Mechanisms and Resistance. Wiley-VCH. ISBN 9783527659685.
- ^ Wookey, A.; Turner, P. J.; Greenhalgh, J. M.; Eastwood, M.; Clarke, J.; Sefton, C. (2004). "AZD2563, a novel oxazolidinone: definition of antibacterial spectrum, assessment of bactericidal potential and the impact of miscellaneous factors on activity in vitro". Clinical Microbiology and Infection. 10 (3): 247–254. doi: 10.1111/j.1198-743X.2004.00770.x. PMID 15008947.
- ^ "Rx 1741". Rib-X Pharmaceuticals. 2009. Archived from the original on 2009-02-26. Retrieved 2009-05-17.
- ^ Gordeev, Mikhail F.; Yuan, Zhengyu Y. (2014). "New Potent Antibacterial Oxazolidinone (MRX-I) with an Improved Class Safety Profile". Journal of Medicinal Chemistry. 57 (11): 4487–4497. doi: 10.1021/jm401931e. PMID 24694071.
- ^ Zhao, Xu; Huang, Haihui; Yuan, Hong; Yuan, Zhengyu; Zhang, Yingyuan (2022). "A Phase III multicentre, randomized, double-blind trial to evaluate the efficacy and safety of oral contezolid versus linezolid in adults with complicated skin and soft tissue infections". Journal of Antimicrobial Chemotherapy. 77 (6): 1762–1769. doi: 10.1093/jac/dkac073. PMID 35265985.
- ^ Hoy, Sheridan M. (2021). "Contezolid: First Approval". Drugs. 81 (13): 1587–1591. doi: 10.1007/s40265-021-01576-0. PMC 8536612. PMID 34365606.
- ^ "China NMPA Approves MicuRx's Contezolid for Treatment of Drug-Resistant Bacterial Infection - MicuRx Pharmaceuticals, Inc".
- ^ ClinicalTrials.gov Identifier: NCT05369052
- ^ A. W. Frahm, H. H. J. Hager, F. v. Bruchhausen, M. Albinus, H. Hager: Hagers Handbuch der pharmazeutischen Praxis: Folgeband 4: Stoffe A-K., Birkhäuser, 1999, ISBN 978-3-540-52688-9
- ^ Karpiuk, I; Tyski, S (2017). "Looking for the new preparations for antibacterial therapy. V. New antimicrobial agents from the oxazolidinones groups in clinical trials". Przeglad Epidemiologiczny. 71 (2): 207–219. PMID 28872286.
External links
| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
1,3-Oxazolidin-2-one | |||
| Other names
2-Oxazolidone
2-Oxazolidinone | |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.007.129 | ||
| KEGG | |||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H5NO2 | |||
| Molar mass | 87.077 g/mol | ||
| Appearance | Solid | ||
| Melting point | 86 to 89 °C (187 to 192 °F; 359 to 362 K) | ||
| Boiling point | 220 °C (428 °F; 493 K) at 48 torr | ||
| Related compounds | |||
Related compounds
|
Oxazolidine | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
2-Oxazolidone is a heterocyclic organic compound containing both nitrogen and oxygen in a 5-membered ring.
Oxazolidinones
Evans auxiliaries
Oxazolidinones are a class of compounds containing 2-oxazolidone in the structure. In chemistry, they are useful as Evans auxiliaries, which are used for chiral synthesis. Usually, the acid chloride substrate reacts with the oxazolidinone to form an imide. Substituents at the 4 and 5 position of the oxazolidinone direct any aldol reaction to the alpha position of the carbonyl of the substrate.[ citation needed]
Pharmaceuticals
Oxazolidinones are mainly used as antimicrobials. The antibacterial effect of oxazolidinones is by working as protein synthesis inhibitors, targeting an early step involving the binding of N-formylmethionyl-tRNA to the ribosome. [1] (See Linezolid#Pharmacodynamics)
Some of the most important oxazolidinones are antibiotics. [2]
Examples of antibiotic oxazolidinones include:
- Linezolid (Zyvox), which is available for intravenous administration and also has the advantage of having excellent oral bioavailability.
- Posizolid, which appears to have excellent, targeted bactericidal activity against all common gram-positive bacteria, regardless of resistance to other classes of antibiotics. [3]

- Tedizolid, (Sivextro) which is approved for acute skin infections
- Radezolid (RX-1741) has completed some phase-II clinical trials. [4]
- Cycloserine is a second line drug against tuberculosis. Note that cycloserine, while technically an oxazolidone, has a different mechanism of action and substantially different properties from the aforementioned compounds.
- Contezolid (MRX-I) has reported phase 1 data [5] and phase III data. [6] In 2021, a new drug summary was published by ADIS Press. [7] In June 2021, marketing approval was granted by the Chinese National Medical Products Administration (NMPA) for use of oral contezolid in moderate to severe complicated skin and skin structure infections. [8] In June 2022, contezolid oral tablets and contezolid acefosamil IV (a prodrug of contezolid) began Phase 3 global clinical trials in moderate to severe diabetic foot infections. [9] An additional global Phase 3 study is planned for acute bacterial skin and skin structure infections (ABSSSI) for the combination of contezolid and contezolid acefosamil.
- An oxazolidinone derivative used for other purposes is rivaroxaban, which is approved by the FDA for venous thromboembolism prophylaxis.
History

The first ever used oxazolidinone was cycloserine (4-amino-1,2-oxazolidin-3-one), a second line drug against tuberculosis since 1956. [10]
Developed during the nineties when several bacterial strains were becoming resistant against such antibiotics as vancomycin. Linezolid (Zyvox) is the first approved agent in the class (FDA approval April 2000).

The first commercially available 1,3-oxazolidinone antibiotic was linezolid, discovered and developed by Pharmacia & Upjohn.[ citation needed]

In 2002 AstraZeneca began investigating posizolid, which is in clinical trials for use in humans. [11]
See also
- Oxazolidine – the ring without the carbonyl group
- Oxazolone – the unsaturated analogues
References
- ^ Shinabarger, D. (1999). "Mechanism of action of the oxazolidinone antibacterial agents". Expert Opinion on Investigational Drugs. 8 (8): 1195–1202. doi: 10.1517/13543784.8.8.1195. PMID 15992144.
- ^ Sonia Ilaria Maffioli (2014). "A Chemist's Survey of Different Antibiotic Classes". In Claudio O. Gualerzi; Letizia Brandi; Attilio Fabbretti; Cynthia L. Pon (eds.). Antibiotics: Targets, Mechanisms and Resistance. Wiley-VCH. ISBN 9783527659685.
- ^ Wookey, A.; Turner, P. J.; Greenhalgh, J. M.; Eastwood, M.; Clarke, J.; Sefton, C. (2004). "AZD2563, a novel oxazolidinone: definition of antibacterial spectrum, assessment of bactericidal potential and the impact of miscellaneous factors on activity in vitro". Clinical Microbiology and Infection. 10 (3): 247–254. doi: 10.1111/j.1198-743X.2004.00770.x. PMID 15008947.
- ^ "Rx 1741". Rib-X Pharmaceuticals. 2009. Archived from the original on 2009-02-26. Retrieved 2009-05-17.
- ^ Gordeev, Mikhail F.; Yuan, Zhengyu Y. (2014). "New Potent Antibacterial Oxazolidinone (MRX-I) with an Improved Class Safety Profile". Journal of Medicinal Chemistry. 57 (11): 4487–4497. doi: 10.1021/jm401931e. PMID 24694071.
- ^ Zhao, Xu; Huang, Haihui; Yuan, Hong; Yuan, Zhengyu; Zhang, Yingyuan (2022). "A Phase III multicentre, randomized, double-blind trial to evaluate the efficacy and safety of oral contezolid versus linezolid in adults with complicated skin and soft tissue infections". Journal of Antimicrobial Chemotherapy. 77 (6): 1762–1769. doi: 10.1093/jac/dkac073. PMID 35265985.
- ^ Hoy, Sheridan M. (2021). "Contezolid: First Approval". Drugs. 81 (13): 1587–1591. doi: 10.1007/s40265-021-01576-0. PMC 8536612. PMID 34365606.
- ^ "China NMPA Approves MicuRx's Contezolid for Treatment of Drug-Resistant Bacterial Infection - MicuRx Pharmaceuticals, Inc".
- ^ ClinicalTrials.gov Identifier: NCT05369052
- ^ A. W. Frahm, H. H. J. Hager, F. v. Bruchhausen, M. Albinus, H. Hager: Hagers Handbuch der pharmazeutischen Praxis: Folgeband 4: Stoffe A-K., Birkhäuser, 1999, ISBN 978-3-540-52688-9
- ^ Karpiuk, I; Tyski, S (2017). "Looking for the new preparations for antibacterial therapy. V. New antimicrobial agents from the oxazolidinones groups in clinical trials". Przeglad Epidemiologiczny. 71 (2): 207–219. PMID 28872286.