| |
| Names | |
|---|---|
|
Preferred IUPAC name
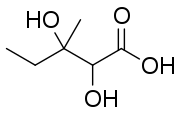
2,3-Dihydroxy-3-methylpentanoic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| KEGG | |
| MeSH | 2,3-dihydroxy-3-methylpentanoic+acid |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H12O4 | |
| Molar mass | 148.16 g/mol |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
2,3-Dihydroxy-3-methylpentanoic acid is an intermediate in the metabolism of isoleucine.
Metabolism
2,3-Dihydroxy-3-methylpentanoate is synthesized by the action of acetolactate mutase with subsequent reduction from α-aceto-α-hydroxybutyrate through 3-hydroxy-2-keto-3-methylpentanoate: [1]
- α-aceto-α-hydroxybutyrate → 3-hydroxy-2-keto-3-methylpentanoate
- 3-hydroxy-2-keto-3-methylpentanoate + NAD(P)H → 2,3-dihydroxy-3-methylpentanoate + NAD(P)+
It is then processed by the action of dihydroxyacid dehydratase, which results in 2-keto-3-methylvalerate and water: [1]
- 2,3-dihydroxy-3-methylpentanoate → 2-keto-3-methylvalerate + H2O
Transamination of 2-keto-3-methylvalerate yields isoleucine.
References
- ^ a b Voet, Donald; Voet, Judith G. (2011). Biochemistry (4. ed.). Hoboken, NJ: Wiley. pp. 1074–1075. ISBN 978-0-470-91745-9.

| |
| Names | |
|---|---|
|
Preferred IUPAC name
2,3-Dihydroxy-3-methylpentanoic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| KEGG | |
| MeSH | 2,3-dihydroxy-3-methylpentanoic+acid |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H12O4 | |
| Molar mass | 148.16 g/mol |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
2,3-Dihydroxy-3-methylpentanoic acid is an intermediate in the metabolism of isoleucine.
Metabolism
2,3-Dihydroxy-3-methylpentanoate is synthesized by the action of acetolactate mutase with subsequent reduction from α-aceto-α-hydroxybutyrate through 3-hydroxy-2-keto-3-methylpentanoate: [1]
- α-aceto-α-hydroxybutyrate → 3-hydroxy-2-keto-3-methylpentanoate
- 3-hydroxy-2-keto-3-methylpentanoate + NAD(P)H → 2,3-dihydroxy-3-methylpentanoate + NAD(P)+
It is then processed by the action of dihydroxyacid dehydratase, which results in 2-keto-3-methylvalerate and water: [1]
- 2,3-dihydroxy-3-methylpentanoate → 2-keto-3-methylvalerate + H2O
Transamination of 2-keto-3-methylvalerate yields isoleucine.
References
- ^ a b Voet, Donald; Voet, Judith G. (2011). Biochemistry (4. ed.). Hoboken, NJ: Wiley. pp. 1074–1075. ISBN 978-0-470-91745-9.