| |
| Names | |
|---|---|
|
Preferred IUPAC name
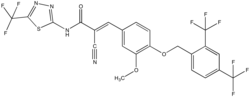
(2E)-3-(4-{[2,4-Bis(trifluoromethyl)phenyl]methoxy}-3-methoxyphenyl)-2-cyano-N-[5-(trifluoromethyl)-1,3,4-thiadiazol-2-yl]prop-2-enamide | |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.163.130 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C23H13F9N4O3S | |
| Molar mass | 596.424949 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
XCT-790 is a potent and selective inverse agonist ligand of the estrogen-related receptor alpha (ERRα). [1] Independent of its inhibition of ERRα, XCT-790 is a potent mitochondrial electron transport chain uncoupler. [2]
XCT-790 has been shown to uncouple oxygen consumption from ATP production in mitochondria at very low, nanomolar-range doses independently of ERRα expression. Its effects are similar to proton ionophores such as FCCP, which disrupt mitochondrial transmembrane electrochemical gradients. This uncoupling leads to a fast drop in ATP production and, consequently, a prompt activation of AMPK. [2]
-
^ Busch BB, Stevens WC Jr, Martin R, Ordentlich P, Zhou S, Sapp DW, Horlick RA, Mohan R; Stevens (November 2004). "Identification of a selective inverse agonist for the orphan nuclear receptor estrogen-related receptor alpha". J. Med. Chem. 47 (23): 5593–6.
doi:
10.1021/jm049334f.
PMID
15509154.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ a b Eskiocak B; Aktar Ali; White MA (July 7, 2014). "The estrogen-related receptor α inverse agonist XCT 790 is a nanomolar mitochondrial uncoupler". Biochemistry. 53 (29): 4839–4846. doi: 10.1021/bi500737n. PMC 4116149. PMID 24999922.
- XCT790 at the U.S. National Library of Medicine Medical Subject Headings (MeSH)

| |
| Names | |
|---|---|
|
Preferred IUPAC name
(2E)-3-(4-{[2,4-Bis(trifluoromethyl)phenyl]methoxy}-3-methoxyphenyl)-2-cyano-N-[5-(trifluoromethyl)-1,3,4-thiadiazol-2-yl]prop-2-enamide | |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.163.130 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C23H13F9N4O3S | |
| Molar mass | 596.424949 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
XCT-790 is a potent and selective inverse agonist ligand of the estrogen-related receptor alpha (ERRα). [1] Independent of its inhibition of ERRα, XCT-790 is a potent mitochondrial electron transport chain uncoupler. [2]
XCT-790 has been shown to uncouple oxygen consumption from ATP production in mitochondria at very low, nanomolar-range doses independently of ERRα expression. Its effects are similar to proton ionophores such as FCCP, which disrupt mitochondrial transmembrane electrochemical gradients. This uncoupling leads to a fast drop in ATP production and, consequently, a prompt activation of AMPK. [2]
-
^ Busch BB, Stevens WC Jr, Martin R, Ordentlich P, Zhou S, Sapp DW, Horlick RA, Mohan R; Stevens (November 2004). "Identification of a selective inverse agonist for the orphan nuclear receptor estrogen-related receptor alpha". J. Med. Chem. 47 (23): 5593–6.
doi:
10.1021/jm049334f.
PMID
15509154.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ a b Eskiocak B; Aktar Ali; White MA (July 7, 2014). "The estrogen-related receptor α inverse agonist XCT 790 is a nanomolar mitochondrial uncoupler". Biochemistry. 53 (29): 4839–4846. doi: 10.1021/bi500737n. PMC 4116149. PMID 24999922.
- XCT790 at the U.S. National Library of Medicine Medical Subject Headings (MeSH)