| This is an archive of past discussions. Do not edit the contents of this page. If you wish to start a new discussion or revive an old one, please do so on the current talk page. |
| Archive 45 | ← | Archive 47 | Archive 48 | Archive 49 | Archive 50 | Archive 51 | → | Archive 54 |
Polonol
Hello. I can't find a single mention of Polonol online in a reliable source. Can someone please check the book sources cited? Thanks. Capewearer ( talk) 20:37, 14 March 2020 (UTC)
- Well the first reference "Chemie des Poloniums" does not mention any chemical like that. There are some organo-polonium compounds there, but not R-Po-OH. The second references does not mention it either. There is a Polonium section that fails to mention it.
This is looking like something made up, or a hoax.[1] Graeme Bartlett ( talk) 21:38, 14 March 2020 (UTC)- SciFinder has no entries for polonol. Zero. There are two papers that (barely) mention C-Po-H. Good luck if you go with AFD, seems like it is really difficult to lawyer through that site. -- Smokefoot ( talk) 22:15, 14 March 2020 (UTC)
- @
Graeme Bartlett: the structure is R-Po-H, as Smokefoot notes, not R-Po-OH. Let me know if anyone needs help with the AfD process.
DMacks (
talk)
04:03, 15 March 2020 (UTC)
- Thanks for that, but the sources provided do not have that either. Dimethyl-, diephenyl- or diethyl polonium gets a mention. Graeme Bartlett ( talk) 04:23, 15 March 2020 (UTC)
- I have invited the writer to join in the discussion here.
Graeme Bartlett (
talk)
02:20, 15 March 2020 (UTC)
- The article appears to be a translation of the article on zhwiki about this substance (
zh:釙醇). "Not citing here that our article is a translation of some other source (with cite to that source)" is a license violation. Pinging
User:A2569875 who wrote that article there and seems somewhat active here on enwiki and on commons in the chemistry genres.
DMacks (
talk)
04:11, 15 March 2020 (UTC)
- If it is a translation then I cannot claim it was made up. So I strike my comment. Perhaps the name is made in analogy to
tellurol.
Graeme Bartlett (
talk)
04:23, 15 March 2020 (UTC)
- It could totally be made up or a hoax, just one that originated external to enwiki. That is, User:Nucleus hydro elemon could have been acting in good faith and topic itself still ends up being nonsense. Perhaps they didn't actually check the sources they transcribed? The original author can help clear this up I hope. DMacks ( talk) 04:40, 15 March 2020 (UTC)
- Sorry. In fact, I have not been able to find any valid references since 2012. The Chinese Wikikipedia page has been requested to be deleted,
WP:Articles for deletion/Polonol (釙醇). cc
DMacks,
Graeme Bartlett.--
Nanachi
🐰
Fruit Tea☕(宇帆·
☎️·
☘️)
06:09, 15 March 2020 (UTC)
- Thanks for checking up on it! DMacks ( talk) 14:27, 15 March 2020 (UTC)
- If it is a translation then I cannot claim it was made up. So I strike my comment. Perhaps the name is made in analogy to
tellurol.
Graeme Bartlett (
talk)
04:23, 15 March 2020 (UTC)
- The article appears to be a translation of the article on zhwiki about this substance (
zh:釙醇). "Not citing here that our article is a translation of some other source (with cite to that source)" is a license violation. Pinging
User:A2569875 who wrote that article there and seems somewhat active here on enwiki and on commons in the chemistry genres.
DMacks (
talk)
04:11, 15 March 2020 (UTC)
On periodic trend grounds I am already rather sceptical that this exists. Due to relativistic destabilisation of the 6p3/2 orbital Po is actually significantly more metallic than Te and it should be quite unhappy in the −2 oxidation state. doi: 10.1002/9781119951438.eibc0182 might be useful (it's a 2011 review of organopolonium chemistry): it is quite short (as expected for the subject), but it looks like what is known is mostly dialkyls/diaryls as well as tetravalent compounds of the form Ar3PoX and Ar2PoX2. Nothing about polonols that I can see. Double sharp ( talk) 15:14, 15 March 2020 (UTC)
- Maybe
organopolonium compound should become blue?
DMacks (
talk)
15:44, 15 March 2020 (UTC)
- Good edit
[2],
Headbomb. Now here is a diff:
[3] ->
[4]
[5]. -
DePiep (
talk)
08:23, 19 March 2020 (UTC)
- Go troll elsewhere.
Headbomb {
t ·
c ·
p ·
b}
08:24, 19 March 2020 (UTC)
- There are literatures about organopolonium compounds (R2Po, R2PoX2, etc.), but none about polonol. -- Leiem ( talk) 17:33, 19 March 2020 (UTC)
- Go troll elsewhere.
Headbomb {
t ·
c ·
p ·
b}
08:24, 19 March 2020 (UTC)
- @
DMacks: It is blue now. It'll probably remain a stub for a long while, though. ;)
Double sharp (
talk)
04:52, 12 April 2020 (UTC)
- @ Double sharp: Thanks for your work on that! DMacks ( talk)|
- Good edit
[2],
Headbomb. Now here is a diff:
[3] ->
[4]
[5]. -
DePiep (
talk)
08:23, 19 March 2020 (UTC)
The reference "Chemie des Poloniums" turns up in other Polonium articles in equally suspect ways. Polonium hydride: "Experiments conducted on the trace scale show indications that the reaction between polonium metal and nascent hydrogen may produce polonium hydride", nascent hydrogen huh? -- Project Osprey ( talk) 20:42, 15 March 2020 (UTC)
- Thank you all for your quick help with this. I've proposed Polonol for deletion, pointing back to this discussion in the deletion rationale. Capewearer ( talk) 18:49, 18 March 2020 (UTC)
- @
Graeme Bartlett: thanks for your delete vote, the article in chinese wikipedia has already deleted
[6]. Consider delete English article?--
Nanachi
🐰
Fruit Tea☕(宇帆·
☎️·
☘️)
04:46, 22 March 2020 (UTC)
- I did vote, and I was considering voting on some of those other Chinese chemical pages. But I now see most were speedily kept as they were real compounds, so no need. We should have just let the prod run, as there was only a dispute over the prod2. But now we can let the AFD run, and it will not be contestable, until someone writes about the topic elsewhere.
Graeme Bartlett (
talk)
11:29, 22 March 2020 (UTC)
- Sorry I hadn't made clear to the PROD2 editor that I was not contesting the notion of PROD itself. But it would likely have then been deleted and I didn't want that editor's scientific and WP-policy statements to go un-contested...somewhere. Oh well. DMacks ( talk) 14:11, 22 March 2020 (UTC)
- I did vote, and I was considering voting on some of those other Chinese chemical pages. But I now see most were speedily kept as they were real compounds, so no need. We should have just let the prod run, as there was only a dispute over the prod2. But now we can let the AFD run, and it will not be contestable, until someone writes about the topic elsewhere.
Graeme Bartlett (
talk)
11:29, 22 March 2020 (UTC)
Tetramethylammonium hydroxide is a salt
IUPAC definition of a salt: "A chemical compound consisting of an assembly of cations and anions." ( https://goldbook.iupac.org/terms/view/S05447)
Tetramethylammonium hydroxide is a salt.-- Smokefoot ( talk) 16:22, 26 March 2020 (UTC)
- Ok? Is this somehow controversial?
Headbomb {
t ·
c ·
p ·
b}
17:02, 26 March 2020 (UTC)
- Since it is dissolved in water and seems not to exist as a pure solid, it is not a salt. In any case it is a strong alkali or base. We can thank Edgar181 in 2006 for the above statement. From the definition it would be not a salt, as it is not a compound.
Graeme Bartlett (
talk)
12:31, 27 March 2020 (UTC)
That's rather irrelevant to what salts are, no?See also [7] Tetramethylammonium hydroxide is a quaternary ammonium salt generally used as an anisotropic etchant for silicon due to its high silicon etching rate.[1][2] and [8] Tetramethylammonium hydroxide is a solid in the hydrated form. Headbomb { t · c · p · b} 20:21, 27 March 2020 (UTC)- I wonder if colleagues would allow CuSO4.5H2O (aka blue vitriol) to be called a salt? If so maybe we can call the monohydrate of Me4NOH also a salt. But maybe not, who knows?-- Smokefoot ( talk) 22:39, 27 March 2020 (UTC)

structure of Me4NOH monohydrate. - Hydrate of a salt? -- Leiem ( talk) 17:46, 29 March 2020 (UTC)
- IMO, it would not matter whether it's a hydrate or not. At the extreme, NaCl(aq) "salt water", which includes many water molecules wrapped around each cation and each anion, is just as much of a salt as NaCl(s) "table salt." If a salt has one or more waters of hydration in its formula, any working chemist would say that it's still a salt. When I teach this to my students, I say "CuSO4.5H2O is a salt of copper and sulfate which incorporates five waters of hydration." I guess I could be wrong about all that, but I don't think so. KeeYou Flib ( talk) 14:55, 22 April 2020 (UTC)
- Headbomb, apparently so (among some editors at least). I responded there. They seem to be talking round in circles and escallating personal claims, rather than using WP:RS and remaining WP:CIVIL...not a recipe for improving WP articles. DMacks ( talk) 04:11, 29 March 2020 (UTC)
- Since it is dissolved in water and seems not to exist as a pure solid, it is not a salt. In any case it is a strong alkali or base. We can thank Edgar181 in 2006 for the above statement. From the definition it would be not a salt, as it is not a compound.
Graeme Bartlett (
talk)
12:31, 27 March 2020 (UTC)
- Why would it have to be anhydrous for it to be a salt? --
Project Osprey (
talk)
20:18, 28 March 2020 (UTC)
- I was just trying to anticipate any possible objections. Heck, I am surprised we are having this convesation. Maybe you can penetrate the arguments by JonRichfield and his friend here
here. --
Smokefoot (
talk)
20:52, 28 March 2020 (UTC)
- ... I must confess that I cannot. These discussions trend to drag on. Good luck -- Project Osprey ( talk) 22:09, 28 March 2020 (UTC)
- I was just trying to anticipate any possible objections. Heck, I am surprised we are having this convesation. Maybe you can penetrate the arguments by JonRichfield and his friend here
here. --
Smokefoot (
talk)
20:52, 28 March 2020 (UTC)
- I was confused by Smokefoot's original statement, whether calling it a salt is supported or opposed. I will also clarify that Edgar181 was the user that wrote "Tetramethylammonium hydroxide" in the second revision of the page. Graeme Bartlett ( talk) 07:32, 29 March 2020 (UTC)
Styrene
An apparent styrene leak in India has killed 13 people and injured many more. As such there's been a sudden surge in editing on the 'Health effects' section of that page, not all of it to our usual standards. -- Project Osprey ( talk) 08:58, 7 May 2020 (UTC)
Dichlorine pentoxide
Both of the references in this article mentioned that Cl2O5 is non-existent. -- Leiem ( talk) 04:15, 9 May 2020 (UTC)
- Thanks for pointing out this nonsense writing. ChemAbs has about six citations. --
Smokefoot (
talk)
11:58, 9 May 2020 (UTC)
- @ Nucleus hydro elemon:. User:Nucleus hydro elemon is contributing a lot of dubious content. Really unfortunate.-- Smokefoot ( talk) 13:31, 9 May 2020 (UTC)
Sad news
I have some sad news to report ... Ronhjones, a prolific member of this project, passed away on 7 April last year, the day he made his last edit. Tributes are at his talk page. Graham 87 05:26, 2 May 2020 (UTC)
- I have some even sadder news that I can now share publicly: Ron and his wife Sue died together in a house fire, as noted in
their obituary from the London
Inland Waterways Association newsletter. The friend of theirs who confirmed his passing also told me this info, but I didn't want to say it here without confirming that it was publicly available or I had permission.
Graham
87
16:24, 2 May 2020 (UTC)
- It's probably worth a note here that I nominated one of his chemistry pages, UK Chemical Reaction Hazards Forum, for deletion. Graham 87 13:30, 10 May 2020 (UTC)
Crisscross method
I came across the Crisscross method article. To me it seems like an almost trivial method, with just a single source (a textbook that mentions it on one page). Is this method notable enough to satisfy WP:N (and WP:NOTTEXTBOOK)? — Bkell ( talk) 18:45, 25 May 2020 (UTC)
- Support. I suggest you invoke WP:AFD Michael D. Turnbull ( talk) 17:48, 4 June 2020 (UTC)
Cannabigerolic acid
Could somebody check my work for cannabigerolic acid -- first time I've done a chemistry article, I think. I'd like to include the structural formula File:CBGA molecule.svg in the infobox. ☆ Bri ( talk) 23:20, 14 April 2020 (UTC)
- @ Bri:The article looks fine as far as it goes. Your structure drawing has a bad clash between the terminal dimethyl group of the sidechain and one of the phenol oxygens. I took the liberty of uploading a second version more or less exactly as in Chemspider (did you know you can download molfiles from there?). I then reverted my upload back to yours but if you prefer mine you should be able to re-revert to that. Michael D. Turnbull ( talk) 16:16, 13 June 2020 (UTC)
Atom FAR
I did NOT nominate this article for FAR-- just the doing nominations that were not done by the editor who did.
User:Kurzon has nominated Atom for a featured article review here. Please join the discussion on whether this article meets featured article criteria. Articles are typically reviewed for two weeks. If substantial concerns are not addressed during the review period, the article will be moved to the Featured Article Removal Candidates list for a further period, where editors may declare "Keep" or "Delist" the article's featured status. The instructions for the review process are here. SandyGeorgia ( Talk) 19:27, 14 June 2020 (UTC)
Top million substances
Hello, I’m working with User:Egon Willighagen from Wikidata and others to compile a list of what we consider to be the one million most important chemicals. This list will be used to prioritize what we look at for both Wikidata and Wikipedia, and possibly other external groups that interact with us. These chemicals could include things like the elements and other basic substances you would encounter in your chemistry education, chemicals encountered in everyday life (e.g. in detergents, food additives or hair gel) as well as more niche substances such as pharmaceuticals, polymers, pollutants, biologically important materials, etc. Are there any specific collections of substances you would recommend us to look at? Please post any suggested lists or databases below. I'll also post on WT:Chemicals. Many thanks, Walkerma ( talk) 18:15, 19 June 2020 (UTC)
The first two refs in this article use templates that have nothing to do with chemistry. I think some of the links in the chembox are wrong also. This is way beyond me. Can someone here take a look at. Thanks. MB 02:20, 9 July 2020 (UTC)
- This whole article is an unattributed translation from zh:高氯酸钪 by User:Nucleus hydro elemon, whose many such articles including copy-pasting templates without the barest of checks that they have the same function in enwiki as zhwiki. Nucleus, please fix your mess and stop making such messes, as it has long become a WP:DE/ WP:CIR problem. User:Leiem, you seem to contribute to many of the zhwiki articles involved, so perhaps you can talk to them about their edit habits in a way or language that is understandable. DMacks ( talk) 02:35, 9 July 2020 (UTC)
- ...well, Nucleus won't be editing for the next day, but at least this is an explanation of the weird situation MB noted.
DMacks (
talk)
02:50, 9 July 2020 (UTC)
- I have left a message on his zhwp talk page. -- Leiem ( talk) 06:29, 9 July 2020 (UTC)
- In this article, the refs in Chinese are about the coordination compounds synthesized using scandium perchlorate as the reactant. --
Leiem (
talk)
06:29, 9 July 2020 (UTC)
- Everything contributed by User:Nucleus hydro elemon is garbage. He mainly contributes to obscure topics. -- Smokefoot ( talk) 12:29, 9 July 2020 (UTC)
Two cultures
I've been asked (by Eugen Schwarz) a half-scientific question, as follows:
- "(How far) are the concepts of "The Two Cultures" (chemist C. P. Snow ) and of "Paradigm Shift" and "Scientific Revolution" (T. Kuhn) known among real and computational chemists?
I've never heard of " The Two Cultures" concept. I've heard of " Paradigm shift" and " Scientific revolution" but that is only from reading what Scerri has said about the development of scientific knowledge per A tale of seven scientists.
Are there any real and computational chemists here, who could take a stab at Eugen's question?
Thank you --- Sandbh ( talk) 00:14, 16 July 2020 (UTC)
- Well, I am long retired as a computational chemist, but way back I much enjoyed the novels of C. P. Snow, and I read "The Two Cultures" with interest and discussed it with many friends, both chemists and non-chemists. I think it was an important contribution attempting to bridge the gap between scientists and non-scientists. Are you suggesting that computational chemists are not real? -- Bduke ( talk) 01:31, 16 July 2020 (UTC)
Schwarz is a theoretical chemist. I believe he was referring to a “bangs and stinks” chemist. Sandbh ( talk) 12:48, 16 July 2020 (UTC)
Are you suggesting that computational chemists are not real?
Exactly my thought too when I read the question at the top of this thread. "The Two Cultures" originally referred to the split between the humanities and sciences. So I guess this has been extended to a perceived split between theoretical/computational and experimental chemists. I have done both, so I suppose that I am in a somewhat unique position to comment. I personally do not see a conflict between the two. The goal of theoretical/computational chemistry is to explain and predict experimental results. As long as both sides keep in mind thatAll models are wrong, but some are useful
– George Box, then there is no conflict. Contributions from both sides are also essential for Paradigm shifts and Scientific Revolutions. Boghog ( talk) 14:09, 16 July 2020 (UTC)
Thank you. I've passed those comments on to Eugen. Sandbh ( talk) 06:59, 18 July 2020 (UTC)
RFC: Should the default form of the periodic table be changed to put Lu and Lr in group 3, rather than La and Ac?
This RFC may be of interest. Double sharp ( talk) 10:10, 20 July 2020 (UTC)
- As per my comment a few moments ago, I don't think that RFC is an appropriate process here as reliable sources have used both forms and there is no such thing as a default form of periodic tables. Michael D. Turnbull ( talk) 11:07, 20 July 2020 (UTC)
Differentiating electrons d/e
Do anomalies in d/e actually make a difference to the chemistry of the elements concerned?
I understand this is the case with silver which, according to the idealised Madelung Rule, should be d9s2 in the free atom but is in fact d10s1. This discrepancy seems to, at least in part, contribute to the predominance of its main group chemistry.
Teixidó (2019, in Spanish, here) an Emeritus Professor of Chemical Engineering at the University of Barcelona, says that these anomalies impact the elements concerned such that, "their compounds, have properties that do not match the expected regular periodicity. This is the case of Cu, Cr, Pd, Ag, Rh, Pt, Ave or Uno, to name a few." He does not elaborate.
thank you, Sandbh ( talk) 07:23, 18 July 2020 (UTC)
- @ Dirac: Others might have more profound insight, but here is my view. I taught inorganic for years until I realized that the entire d9s2 vs d10s1 topic is some physics thing that has no bearing on chemistry. This orbital ordering issue is 100% tangential to the topic of coordination chemistry, solid state chemistry, and organometallics. In all transition metal complexes and compounds, the d-levels are well below s levels. -- Smokefoot ( talk) 16:18, 18 July 2020 (UTC)
- (Well apart from Palladium's [Kr] 4d10). It is a very physics way of looking at it, what would it's properties be if it were d9s2. I think the honest answer is no one knows for sure: how could we ever force such a change in the bulk material? That said, pretty much everything in chemistry boils down to electrons, so it's reasonably to assume the effects would be profound. -- Project Osprey ( talk) 19:32, 18 July 2020 (UTC)
- If the energy difference between the different arrangements is small, then there may be alternative ways to arrange compound bonds, resulting in more different allotropes or crystal forms. I notice this happening with some actinides. Graeme Bartlett ( talk) 22:35, 18 July 2020 (UTC)
A perspective by Double sharp
I guess this is somewhat breaking my promise not to argue with Sandbh on such things due to its futility. You see, there has been a long-running argument on Wikipedia talk:WikiProject Elements since December regarding the composition of group 3. It's the old chestnut, whether Wikipedia should show lutetium or lanthanum under yttrium. Since lanthanum accidentally happens to be [Xe]4f05d16s2 with no 4f electron in the gas-phase ground-state, the relevance of this has been dragged in.
Of course on Wikipedia the first thing should be the relevant sources, which focus on the issue, and they mostly support lutetium with strong argumentation. The fact that most elementary general chemistry textbooks persist in showing lanthanum is not as consequential per WP:CONTEXTMATTERS, considering that such books rarely cover the heavy 4f and 5d elements at all. In fact the more I read about this dispute it looks more and more like the situation hypervalent molecules like PCl5 are in: there is no 3d component to the bonding from phosphorus, and that has been well-known for decades, but good luck finding an elementary general chemistry textbook that tells you that. Same situation for Lu under Y. Articles have been published from the 1920s(!) to the present explaining why that is the correct placement, but good luck finding it in general chemistry textbooks.
But, I suppose it is natural that the scientific argumentation gets involved at some point. So, on came a lot of discussion on the science. None of it succeeded in changing Sandbh's mind despite how overwhelming the evidence was. Hence my decision to not argue with him due to its futility. I'm drafting an RFC on the matter anyway, since at WT:ELEM we already managed an incredible 5-1 consensus against La under Y, and no one but Sandbh seems to be convinced by his lines of argumentation.
So, take this as a reply to the others involved.
It makes actually basically zero difference. Smokefoot is completely right that it has no bearing on chemistry. For two reasons:
- In real compounds you rarely have neutral atoms. We are talking about metals, they will probably be positively charged.
- Chemical bond energies can go up to around 10 eV. Therefore, excited states in the neutral atom up to about that energy level will matter. In every case of a weird configuration, the "expected" configuration (like d9s2 for silver at 3.11 eV) is well within that. But don't take my word for it, go check it at NIST. ^_^ The extreme cases are niobium and platinum, for which the Madelung-expected configurations are not even 0.15 eV up in energy from the real ones.
But, don't just trust me. I am not a chemist. Nevertheless I got this from an actual chemist, Andrey Kulsha = Droog Andrey, who dropped by Wikipedia talk:WikiProject Elements/Archive 38 and said the same thing I'm saying right now.
| “ | Ground electron configuration of a neutral atom says too little about its chemistry (unless we have a noble gas). Chemical bonds are about 1 to 10 eV, so that's the scale we need to take into account when talking about chemistry. For many elements, especially for f- and d-block ones, ground state is one among dozens of states within a few eV. Neutral atoms are rarely encountered in compounds, so we probably need to look at dications or smth else. The participation of subshells throughout the forest of excited states depends on that electric charge. So, my opinion is that no "conclusive argument" could be based only on ground state configurations of neutral atoms. Droog Andrey (talk) 15:01, 15 February 2019 (UTC) | ” |
That's especially true for things like the inner 4f subshells: they get more active as charge goes up. That's why neutral lanthanum has 4f16s2 at 1.88 eV, but La2+ has 4f1 only at 0.89 eV. Or why neutral gadolinium has 4f86s2 at 1.36 eV, but Gd2+ has 4f8 only at 0.30 eV.
Sandbh points to silver adding a s electron added from the previous element, as opposed to the d one gold does. (Pd [Kr]4d10, Ag [Kr]4d105s1; Pt [Xe]5d96s1, Au [Xe]5d106s1.) He suggests that the discrepancy contributes to it having predominant main group chemistry. But this is clearly neither necessary nor sufficient. Technetium adds an s electron from the previous element, rhenium adds a d one. I do not see any sign of "predominant main group chemistry" in technetium. Quite the opposite actually given how easy it is to have it convert between its different oxidation states. And most f elements, along with the heavy members of groups 3, 4, and 5, have predominant main group chemistry in that they have only the group oxidation state as the stable one and are quite electropositive and basic. Yet their differentiating electrons from the previous elements are usually d or f electrons, not s or p electrons.
Even for physical properties, there aren't pure atoms in solid metals, there aren't atomic wavefunctions, as Droog Andrey also explained at Wikipedia talk:WikiProject Elements/Archive 33. What you look at there is band occupancy. But even there it does not match the configuration in the gas-phase neutral atoms. In fact it spectacularly doesn't match it even for the first two metals. Lithium and beryllium both have 2p band occupancy despite having no 2p electrons as gas-phase atoms chilling out by themselves! As you go further into the periodic table you will almost always have some sort of fractional average occupancy of subshells that matches neither the Madelung rule's prediction nor the gas-phase ground-state configuration. The simple fact of the matter is that the energy required for such rearrangements is chemically easily achievable and therefore the accident of which electron configuration happens to be the ground state in a gas-phase atom chilling out by itself is irrelevant because in chemical environments the same atom may easily take a different configuration!
Finally here are three quotes from noted chemists making exactly the same point. Christian Jørgensen:
| “ | The two major reasons why this series intended for gaseous atoms strongly bewilders chemists is that undue emphasis is made on irrelevant irregularities (such as the chromium, rhodium, palladium . . . . , atoms) and that the lowest level of two different configurations, such as [Xe]4f96s2 and [Xe]4f85d16s2 are only separated by 285 cm−1 in the terbium atom, much less than 1% of the spreading of J-levels of each of the two configurations, and quite negligible for chemical purposes. | ” |
| — Christian Jørgensen (1988), Influence of Rare Earths on Chemical Understanding and Classification | ||
| “ | It is important to realize that the electronic structures listed in Table 6 are those of the neutral (unionized) gaseous atoms, whereas it is the electronic structure of the ions and compounds that we are chiefly concerned with in chemistry. The relationship of the electronic structure of the gaseous atom of an element to that of its compounds can be rather complicated. For example, in the case of the actinide and lanthanide elements, one would not necessarily predict the predominance of the III oxidation state from the electronic structures of the gaseous atoms; there are usually only two so-called "valence electrons," the 7s or 6s electrons, which might indicate a preference for the II oxidation state.
Apparently, specific factors in the crystal structure of, and the aquation (hydration) energies of, the compounds and ions are important in determining the stability of the III oxidation state. Thus, the characteristic tripositive oxidation state of the lanthanide elements is not related directly to the number of "valence electrons" outside the 4f subshell, but is the somewhat accidental result of a nearly constant small difference between large energy terms (ionization potentials on the one hand, and hydration and crystal energies on the other) which persists over an interval of fourteen atomic numbers. Therefore, if we could somehow have a very extended Periodic Table of Elements containing numerous "f" transition series, we might expect that the 5f, rather than the 4f, elements would be regarded as more nearly representative of such f series. |
” |
| — Glenn T. Seaborg (1994), Origin of the Actinide Concept | ||
And W. H. Eugen Schwarz, whose question Sandbh asked here one section ago:
| “ | The second reason for differences between chemically bound transition-metal atoms and free atoms in vacuum is that the electronic motions in free atoms are not disturbed by adjacent atoms. Most free atoms have open valence shells, where the electrons can arrange differently. The orbit−orbit and spin−orbit angular-momenta couplings result in a large number of different electronic states with different energies. For instance, the 3d54s1 configuration of a free Cr0 or Mo0 atom comprises 504 different states with 74 different degenerate energy levels, scattered over several hundred kJ/mol. ...
'The qualitative behavior of chemical elements can be rationalized with the help of the dominant electronic valence configurations of the atoms embedded in a molecular or crystal environment. These may be correctly called the “electronic configurations of the chemical elements”. However, what is listed in respective tables of chemical textbooks under this headline is something else, namely, what physicists call “the configurations from which the J-level ground states of free unbound atoms in vacuum derive”. ... 'The third exception concerns the free neutral transition-metal atoms in vacuum, including the f block. Their ground-state configurations depend in an involved manner on the often-discussed averaged d−d and d−s Coulomb-repulsion energies and also on the individual orbit−orbit (term) and spin−orbit splittings, even if the latter are small. The correct quantitative explanation is vital for the interpretation of atomic vacuum spectra, but exceeds the scope of general chemical education. There are only a few special topics in chemistry that require the correct understanding of free atoms in vacuum (e.g., atom-molecular gas-phase reactions) or of orbit−orbit and spin−orbit couplings of bonded open-shell atoms (e.g., the chemistry of the transition, lanthanoid, and actinoid metals; spin-flip enhanced reaction mechanisms; so-called spin-forbidden processes). [Nota bene, bonded open-shell TM atoms show different configurations from ground-state free ones.] 'Finally, it is misleading to present free atoms as prototypes for the microscopic description of chemical elements in compounds. The common qualitative textbook explanations of the atomic ground states (correctly: J levels) are incorrect. Therefore, we plead for teaching the correct atomic-orbital order (sequence 6) together with the regular exception, sequence 8, for the s block. One need no longer apologize for irregularities. |
” |
| — W. H. Eugen Schwarz (2010), The Full Story of the Electron Configurations of the Transition Elements | ||
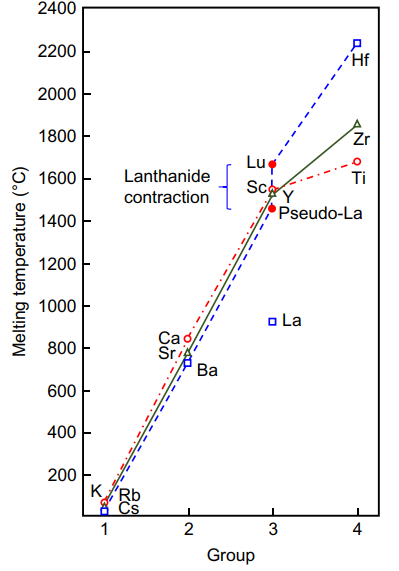
In particular this situation also applies very well to the group 3 dispute. In his very last article doi: 10.1016/bs.hpcre.2016.07.001, Karl A. Gschneidner Jr. ("Mr. Rare Earth") presented this figure showing the melting points. He also showed "pseudo-La" on the figure showing what the melting point of metallic lanthanum would be expected to be if 4f were not involved in it. (Of course 4f has no involvement in lutetium.) This kind of thing is why I've been noting that the scientific and source-based support for lanthanum as an f block element is overwhelming. The lack of a 4f electron in the gas-phase ground state of La does not stop it from using its 4f shell to make bonding MOs from.
Oddly enough, Sandbh is perfectly willing to accept the need to go beyond gas phase configurations when it comes to thorium, which is [Rn] 5f0 6d2 7s2 in the ground state gas phase, yet has clearly been shown to have 5f band occupancy in the metal. For lanthanum he just will not do it. There is a lack of consistency here. Of course by now at Wikipedia talk:WikiProject Elements he is calling the raising of the thorium double standard a "zombie that will not die" argument: it indeed won't die as long as he doesn't logically address why on earth thorium is so different from actinium, that thorium with zero 5f valence electrons in the ground-state gas phase may be allowed in the f block, lawrencium with zero 5f valence electrons in the ground-state gas phase may be allowed in the f block, but actinium also with zero valence 5f electrons may not. No consistency.
I have repeatedly been saying this stuff, with the quotes, with the sources. So has Droog Andrey. For months now. Sandbh has refused to change his mind about the relevance of these anomalies in the face of all of this evidence. Double sharp ( talk) 03:50, 19 July 2020 (UTC)
@ Smokefoot, Project Osprey, and Graeme Bartlett: Forgot to ping you, sorry. Double sharp ( talk) 03:53, 19 July 2020 (UTC)
Siekierski & Burgess (2002)
- Mch as I appreciate User:Double sharp's contributions and enjoy discussing such matters with him, his actions here speak for themselves.
- I ask a 120-word question based on the observation of an emeritus professor of chemistry. Double sharp responds with a 2,000+ word diatribe of rehashed clutter, excessive bolding, biased hyperbole, and misrepresentations.
- Thank you Smokefoot for your WP:CIVIL professional response. Thank you Project Osprey for your professional response saying nobody really knows and that it would be reasonable to presume the effects would be profound. Thank you Graeme for advising there may be alternative ways to arrange compound bonds, resulting in more different allotropes or crystal forms.
- Siekierski & Burgess (2002, p. 133) touch upon the question:
- "The band structure of transition metals…consists of a broad s/p band and a narrow d band. With respect to energy the d band is located approximately in the middle of the s/p band. Moreover, depending on the lattice symmetry some d orbitals mix with s and p orbitals along certain crystallographic directions and confer some d character on the s/p band…The energy spread in the d band is narrow, because d orbitals do not extend to large distances from the atom−they are well within their s orbitals—which restricts their interactions with neighbouring atoms."
- Siekierski S & Burgess J 2002, Concise chemistry of the elements, Horwood Publishing, Chichester
- "The band structure of transition metals…consists of a broad s/p band and a narrow d band. With respect to energy the d band is located approximately in the middle of the s/p band. Moreover, depending on the lattice symmetry some d orbitals mix with s and p orbitals along certain crystallographic directions and confer some d character on the s/p band…The energy spread in the d band is narrow, because d orbitals do not extend to large distances from the atom−they are well within their s orbitals—which restricts their interactions with neighbouring atoms."
- Harking back to Teixidó, chromium is supposed to be 3d44s2 but turns out to be 3d54s1. It seems odd to suggest there would not be a discernible chemical difference between the two configurations.
--- Sandbh ( talk) 14:13, 19 July 2020 (UTC)
Getting into the details
Sandbh may call it what he wants. The fact of the matter is that I backed up everything I said with reliable sources and quoted them at length to show what they said. If that is called a "diatribe" of "rehashed clutter", so be it. Reliable sources, if raised by me and helpfully bolded to emphasise what answers his question, do not seem to be wanted, as was evident from the fact that these sources addressing the issue have been raised countless times in archives 42, 44, and 46 of WT:ELEM, and is now again evident from the fact that he calls my statements "biased hyperbole" even though I said exactly what Smokefoot said: that it has no bearing on chemistry. Replying to his contribution, even for the other parties, was evidently a mistake on my part. Even if everything I say is supported by reliable sources, it is simply not a productive activity because he doesn't accept the answer I and by extension the sources I raise give him.
He may indeed enjoy discussing such matters with me. I used to enjoy it in the past. You can see from this part of why I no longer do. I respect him as a person, but this conversation is clearly not productive. Double sharp ( talk) 14:25, 19 July 2020 (UTC)
- That being said, though: @
Smokefoot,
Project Osprey, and
Graeme Bartlett: I hope my answer and the sources I raised have been of interest. Of course, we may discuss the matter productively, based on reliable sources. ^_^ Most of all, it would be good to clearly get the understanding of reliable sources into our articles on such matters, e.g.
electron configuration.
Double sharp (
talk)
14:45, 19 July 2020 (UTC)
- Well, if you folks are down to arguing these kinds of details, then the Elements project is in great shape. Inevitably maturing Wiki projects distill down to a residue of unresolved topics, which are of interest to a tiny fraction of our readers and which should occupy a tiny fraction of the article. IMHO. My advice on sources would be top-drawer textbooks on metals. Congratulations on a nice set of articles.
- From my perspective, the bigger issues with these articles is the content that readers are more likely to consult: natural occurence, main compounds, uses of the elements vs compounds of the elements, generally jetisoning primary refs for better sources.-- Smokefoot ( talk) 15:04, 19 July 2020 (UTC)
- Well, this discussion has advanced quickly. I'm not sure I actually understand the question here, or the problem. Sandh's question "Do anomalies in d/e actually make a difference to the chemistry of the elements concerned?" translated in my mind to "does the electron configuration of an element effect its chemistry?" which it obviously does - if carbon suddenly gained d electrons it's chemist would surely change quite a bit. Double sharp points out that electron configuration can change subject to environment, metallic bonding being an example. Again, that is true. So far no one has said anything wrong - but what is the question? --
Project Osprey (
talk)
19:29, 19 July 2020 (UTC)
- Perhaps there is a call to join in the argument at
Wikipedia talk:WikiProject Elements. I will not be participating as there are other more interesting things to do here.
Graeme Bartlett (
talk)
22:03, 19 July 2020 (UTC)
- Well, I agree that there are very much more interesting things to do than to argue something well-known for decades. ^_^ Anyway, as for articles, I do not intend more than one sentence (despite how the level of debate might make it look ^_^). It would read something like the following:
- Perhaps there is a call to join in the argument at
Wikipedia talk:WikiProject Elements. I will not be participating as there are other more interesting things to do here.
Graeme Bartlett (
talk)
22:03, 19 July 2020 (UTC)
| “ | These exceptions from the Madelung rule do not have a significant impact on chemistry, as many different configurations have very similar energies [Jørgensen, Schwarz] and these gas-phase configurations are different from the configurations encountered in ions, compounds, and solid metals [Seaborg, Barrett, Wang et al., Gschneidner review on lanthanides above] | ” |
.
- Anyway Sandbh's question seems to be "do these real Madelung anomalies like silver being d9s2 instead of d10s1 cause any significant difference in the chemistry"? I gave sources saying "no they don't". Which is clearly in line with reality, given how similar niobium 4d45s1 and tantalum 5d36s2 act. That's not saying that in principle you couldn't have anomalies that really make a huge difference (such are predicted for the eighth period, making element 168 act like a tin analogue rather than a noble gas according to calculations), it's just saying that the ones in the known elements up to 118 are not among them. Double sharp ( talk) 02:54, 20 July 2020 (UTC)
What is the question(?)
For example, consider chromium. It should be d4s2 but turns out to be d5s1. Does this anomaly make a difference to the chemistry of chromium in terms what was expected v. what is observed?
I see that removing the first and second electrons from vanadium 3d34s2 takes 6.75 and 14.7 eV respectively, whereas to do the same for chromium 3d54s1 requires 6.75 and 16.5 eV. For niobium 4d45s1 the numbers are 6.75 and 14.3 whereas for molybdenum 4d55s1 they are 7.1 and 16.2
So there appears to be a premium, of from 12.2 to 7.7%, for moving past the d5 configuration. I presume this wouldn't be the case for chromium if it was 3d44s2.
To the extent that these energy costs impact the chemistry of the elements involved, it appears the anomalous electron configuration of chromium does make a difference. Does my interpretation seem reasonable? Sandbh ( talk) 01:26, 20 July 2020 (UTC)
- Well I am no physicist, but I can address the chemistry part in your first question:
- you mean, do Cr atoms react in any special way because of this configuration? Response: Do any process involve Cr atoms in the gas phase? (there is
metal vapor synthesis, which might be worth looking into. Those folks are mainly worried about heats of vaporization). And once something (i.e. a ligand) attaches to the Cr atom, the configuration changes to d6s0. So this logic suggests that its a pretty narrow set of circumstances where the d6s0 vs d5s1 issue is consequential, for chemistry. But my dismissive view (and it is only this one editor's view), in no way invalidates the impressive depth of analysis applied by physics community.--
Smokefoot (
talk)
03:31, 20 July 2020 (UTC)
- What Smokefoot says is also exactly what W. H. Eugen Schwarz said in the source I quoted above: "There are only a few special topics in chemistry that require the correct understanding of free atoms in vacuum (e.g., atom-molecular gas-phase reactions)". ^_^ Double sharp ( talk) 04:59, 20 July 2020 (UTC)
Thank you. There are four configurations at play, as I understand it:
| Metal | (ds)6 |
| Gas | d54s1 |
| Ideal | d4s2 |
| Cr3+ | d3 |
Removing three e− from chromium and from molybdenum to get to Cr3+ and Mo3+ takes 7–9% more energy than is the case for removing three e− from their predecesssors, vanadium and niobium. I guess, I don't know, the extra energy cost makes chromium and molybdenum less reactive than is the case if they had idealised d4s2 configurations. Sandbh ( talk) 07:34, 20 July 2020 (UTC)
This
article notes a "Cr atom has a 4s valence orbital that is half empty and can act as an electron acceptor." That was news to me, and something presumably not possible with a d4s2 configuration.
Sandbh (
talk)
07:49, 20 July 2020 (UTC)
- Sandbh's hypotheses may easily be tested. Tungsten really is d4s2 in the gas phase ground state. It is not, as far as I can see, more reactive than molybdenum which is d5s1. Actually Mo and W are more similar to each other than Ag and Au, even though in the second case the configurations match and in the first case they don't. We may multiply even more interesting examples, such as comparing Nb-Ta (no match, very similar anyway) with Sn-Pb (match, but quite different). Case closed.
- Meanwhile he quotes an article focusing on an alloy of chromium and yttrium. In which due to metallic bonding the configuration will not be integer occupancy. Indeed, the article he quotes itself notes that mixing happens between all of Cr 3d 4s 4p, and Y 4d 5s 5p. "Furthermore, our density of state (DOS) results also show strong state mixing between the 4s, 4p, and 3d orbitals of Cr and 4d, 5s, and 5p orbitals of Y, near the Fermi level". That obviously hasn't got anything to do with the gas phase configuration, since 4p and 5p are not even occupied there for chromium and yttrium. Of course it has everything to do with chemical configurations because p occupancy exists typically for low oxidation state TM complexes, e.g. Vaska's complex. But those are not the ground state gas phase configurations that Sandbh attempts to find relevance in despite Smokefoot pointing out to him "Do any process involve Cr atoms in the gas phase?". And despite W. H. Eugen Schwarz saying it. And despite Glenn T. Seaborg saying it. And despite Christian Jørgensen saying it. And despite me and Droog Andrey saying it at Wikipedia talk:WikiProject Elements. I am somewhat saddened, but after almost seven months not surprised, that Sandbh ignores another editor contradicting his idea with sound logic, still ignores the sources I give, and tries to make something out of his idea anyway by taking a source that doesn't even focus on the issue and taking one assertion from it out of context, and not testing his hypothesis when tungsten is right there to test it on.
- Yes, a Cr atom sitting alone by itself indeed has a half filled 4s shell. That is what the article said. Is a chromium atom in a Cr-Y alloy with lots of other chromium and yttrium atoms around in that situation? No. Would one expect it to have the same configuration, knowing that in chemical environments it's easily going to be 3d64s0 instead of 3d54s1? No. So of what relevance is this one assertion he quotes? No mechanism for its relevance is implied and the article's noting that Cr 4p contributes to the bonding obviously shows that being empty in the ground state gas phase is no obstacle for an orbital to contribute to bonding anyway. Same for being full as Y 5s is.
- So this is another case where Sandbh shops around for sources that seem to say what he wants to be true, in this case going to a source that isn't even focusing on the issue, and avoids quoting the parts that contradict his idea. A similar selectivity has been observed already at
WT:ELEM regarding 4f involvement explaining lanthanum's superconductivity. So:
- Is Sandbh not sure of the answer to his question? Then why is it that he cannot believe the editors here and the sources they quote saying the Madelung anomalies don't matter, and keeps guessing for ways where it might matter even when told otherwise?
- Is he sure of the answer? Then why is he asking the question? And is that a relevant approach for WP when the sources say something else? Double sharp ( talk) 08:41, 20 July 2020 (UTC)
Enough already!
First I'll give my view on Sandbh's question. The configuration of the electrons in an atom is an intrinsic property. So are its ionisation potential and its electronegativity. However, most properties chemists, and especially non-chemists, are interested in are emergent properties. It is meaningless to ask "what is the melting point of an atom of silver?" Melting points are emergent properties of assemblages of atoms and I would predict that small sets of atoms would have lower melting or boiling points than bulk material: entropy winning out over whatever binding enthalpy is holding the "solid" or "liquid" cluster together. How large the cluster needs to be before the bulk value sets in, I have no idea!
Another way of expressing my view is that intrinsic properties can now be calculated pretty well for chemical substances, knowing their proposed atom connectivity/bonding, even if no-one has ever made a sample. However, you have to wait for a real sample to get an accurate value for an emergent property like a melting point. Personally I'm biased towards organic compounds because that's where my expertise lies and that's also where the concept of having a target (as-yet-unmade) structure is on a pretty firm footing whereas I presume that it can be more difficult to be sure that a new inorganic structure will actually exist in the form one imagines. Who would have guessed that carbon's humble s2p2 would in many compounds turn out to become a tetrahedral sp3 while in other compounds you would get a different linear combination of molecular orbitals including aromatic ones: so that even as an element diamond and graphite don't much resemble one another and are difficult to interconvert? Other emergent allotropes exist and even in 2020 new ones are still being touted.....
I think that the early pioneers who created various ways of laying out Periodic tables emphasised properties like ionisation potential and electronegativity which were more likely to "match the expected regular periodicity" (to quote the Spanish professor) just because they are indeed intrinsic, rather than to expect periodicity in emergent properties like (to take an extreme case) utility as a drug.
OK, so what about your dispute about group 3? I read the exchange at WT:ELEM and although I found parts informative, it was mostly tedious, repetitive and above all uncivil. I couldn't face the archive and I wondered why the whole thing wasn't on the talk page for Periodic table since that's the article that's relevant. I had a brief look on that talk page and goodness me you're all over that one as well! My analysis is that you both think that you are discussing the periodic table whereas in fact you are each discussing a periodic table: unfortunately not the same one! Your only focus should be "how can we improve the periodic table article?" (on WIkipedia there is only one). The RFC process is inappropriate because reliable external sources discuss and use both forms of the bit you are arguing about. There is no such thing as "the" periodic table, whatever IUPAC rules but there are certainly a number of interesting and useful alternative ways of laying out tables for various purposes. Wikipedia's article should reflect the many ways the table has historically appeared (as indeed it already does) and mention the Lu / La discussion. Can't you each find your best couple of sources that favour Lu or La — preferably not written by yourselves — and add a paragraph to the main article that encapsulates the debate while maintaining WP:NPOV? Michael D. Turnbull ( talk) 10:59, 20 July 2020 (UTC)
- @
Michael D. Turnbull: I do agree that we have to discuss the issue neutrally in the
periodic table article. I support doing that. The only trouble is that there are lots and lots of places where we use a periodic table as a graphic. Just look at, say, the article on
neon: we have a little graphic at the top showing where neon is in the periodic table, we have a footer at the bottom showing it again, both are used for navigation. Don't we have to choose a default for that situation to avoid showing it twice over? If you have a solution that might break that impasse, please, do let me know.
Double sharp (
talk)
12:10, 20 July 2020 (UTC)
- @
Double sharp: Yes, that's a knotty problem, which is why it's interesting but probably not insurmountable! All the articles for the elements are important and the infoboxes are vital. However, whoever has chosen (on Wikipedia) to put He over Ne in that graphic offends all those who think it should be He over Be. However, when looking at the actual article for
helium, there is a vital note pointing out in a proper
WP:NPOV way that some authors prefer the latter version of the table. So, in general, you could change the graphic so it didn't attempt to convey quite so much information on "remote" elements to the one in question and leave discussions about group 3, for example, to the individual elements where it is important.
Michael D. Turnbull (
talk)
13:19, 20 July 2020 (UTC)
- @ Michael D. Turnbull: Actually I wrote that note on helium. ;) I admit I haven't thought of a good way to "blur" the problem. The WebElements idea may be a reasonable compromise by simply not making it clear from any gap where the f block is supposed to fit in, so we can leave the footnotes to the elements they are about. But maybe that is also a downside? At least it seems promising until we get to the footer with the element symbols explicitly marked. I will think about it. ^_^ Double sharp ( talk) 13:44, 20 July 2020 (UTC)
- @
Double sharp: Yes, that's a knotty problem, which is why it's interesting but probably not insurmountable! All the articles for the elements are important and the infoboxes are vital. However, whoever has chosen (on Wikipedia) to put He over Ne in that graphic offends all those who think it should be He over Be. However, when looking at the actual article for
helium, there is a vital note pointing out in a proper
WP:NPOV way that some authors prefer the latter version of the table. So, in general, you could change the graphic so it didn't attempt to convey quite so much information on "remote" elements to the one in question and leave discussions about group 3, for example, to the individual elements where it is important.
Michael D. Turnbull (
talk)
13:19, 20 July 2020 (UTC)
Potential barrier... to understanding
Aside: This is a problem where someone linked a term as known to them, within the topic they were familiar with, without checking whether the linked article *actually* had anything to do with _their_ understanding of the term.
So for 13 years the article
Activation energy has had in its second paragraph a link
potential barrier. Now I know what that means in chemistry, and you know what that means in chemistry, but that linked article begins
- In quantum mechanics, the rectangular (or, at times, square) potential barrier is a standard one-dimensional problem that demonstrates the phenomena of . . . .
If there is a article here at WP that does describe the chemical potential barrier, the link should be updated to point to that. If not, then the link needs to be unlinked. As it is now, the link merely confuses.
Separately, while finding the timeframe of the optimistic link, I noticed the
diagram seen here (
and found here) looks so much more understandable to the average reader than the
current diagram which presentation deters any but the already familiar.
It seems a foot-gun to make an article so technically irreproachable that it is also *unapproachable*. That really *is* a potential barrier. What was wrong with having an overview, such as seen back in 2007? Do y'all want people to hate chemistry? Shenme ( talk) 04:21, 1 August 2020 (UTC)
- Yes it's a poor article in a number of ways and is currently rated start-class. Although there is an article on
Chemical potential, I'm not sure that is the appropriate link either and it is a very technical article. In another example, the lede also links to
division in "a chemical reaction, or division" which is also off-topic and uses a citation that, to me, seems unrelated. Go ahead and make alterations to the article you feel would improve it. The article's talk page is the place to discuss any material you are still unsure about.
Michael D. Turnbull (
talk)
11:27, 1 August 2020 (UTC)
- My educated guess (only that) is that activation energy is a common term in chemical kinetics/mechanism but far less commonly invoked in nuclear reactions and materials science. That guess is supported by the nature of the subsidiary sections, which are highly chemical. With that idea in mind, I refocused the lede toward a chemical context.-- Smokefoot ( talk) 13:51, 1 August 2020 (UTC)
Silylene has a misleading scheme
In the Wiki article for Silylene ( /info/en/?search=Silylene) the scheme has a bond between the Si-Atom and the Electron pair. This is misleading as the Electrons are located at the Si-atom and it could imply a Carbene with a Hydrogen and a Silane substituent.
- (Above text unsigned)
 Done Thanks, anonymous. I've fixed this.
Michael D. Turnbull (
talk)
15:37, 28 July 2020 (UTC)
Done Thanks, anonymous. I've fixed this.
Michael D. Turnbull (
talk)
15:37, 28 July 2020 (UTC)
- Thanks,
User:Michael D. Turnbull. I see you (among other things) corrected the original PNG image on commons. Please update the metadata on that to indicate the author/date (and license, if necessary) for this latest upload at that filename.
DMacks (
talk)
14:08, 1 August 2020 (UTC)
-
I've gone in and used
{{ PD-chem}}, which I assume is OK. Please let me know if I haven't done that right Michael D. Turnbull ( talk) 15:16, 1 August 2020 (UTC)- Looks good. Thanks! DMacks ( talk) 17:25, 1 August 2020 (UTC)
-
I've gone in and used
- Thanks,
User:Michael D. Turnbull. I see you (among other things) corrected the original PNG image on commons. Please update the metadata on that to indicate the author/date (and license, if necessary) for this latest upload at that filename.
DMacks (
talk)
14:08, 1 August 2020 (UTC)
Help to assess Draft:Potassium octacyanomolybdate(IV) please
Please will an experienced person examine and assess this draft. If you are not an AFC reviewer please ping me with your assessment and I will review accordingly, otherwise please review and accept or decline according to the draft's merits. Fiddle Faddle 07:06, 4 August 2020 (UTC)
- I am a retired academic chemist, but this article's subject is not in my area of expertise. However, it looks good enough to me and I suggest it be moved into main space where many more people will look at it and improve it. --
Bduke (
talk)
07:53, 4 August 2020 (UTC)
- @
Timtrent: What is really going on here is that the author of this article is promoting his/her recent paper and then is going set about ref spamming. --
Smokefoot (
talk)
12:22, 4 August 2020 (UTC)
- I have accepted, but can you replace their ref?
Graeme Bartlett (
talk)
12:31, 4 August 2020 (UTC)
- Thank you all Fiddle Faddle 12:49, 4 August 2020 (UTC)
- I have accepted, but can you replace their ref?
Graeme Bartlett (
talk)
12:31, 4 August 2020 (UTC)
- @
Timtrent: What is really going on here is that the author of this article is promoting his/her recent paper and then is going set about ref spamming. --
Smokefoot (
talk)
12:22, 4 August 2020 (UTC)
Dimethyl selenosulfenate
This paper mentions that dimethyl selenosulfenate is the most abundant selenium containing species in coffee. What is the formula of dimethyl selenosulfenate? The authors refer to it as MeSeSMe, which I thought would be easy to unpack, but that seems not so, at least for me. Thank you, Sandbh ( talk) 06:08, 18 August 2020 (UTC)
- That would be CH3-Se-S-CH3. Canonical SMILES=CS[Se]C Though the name "selenosulfenate" seems to very little used. Even "sulfenate" is hardly used. "Dimethyl thioselenide" is another name for this, as is 2-Thia-3-selenabutane. It's also found in onions.
[9]
Graeme Bartlett (
talk)
06:45, 18 August 2020 (UTC)
- Only occurs during roasting though... I can't find a paper explaining that, although I'm guessing a process starting with oxidation and subsequent selenoxide elimination of selenocysteine might get you started. -- Project Osprey ( talk) 09:39, 19 August 2020 (UTC)
Notability of compounds
This might be better for WP:MEDICINE, but: is there a general standard besides GNG for the notability of compounds, e.g., diphenyl-2-pyridylmethane, which I just found doing CAT:NN cleanup? Articles like this seem useful but I doubt that the majority of them have WP:SIGCOV. AleatoryPonderings ( talk) 14:25, 24 August 2020 (UTC)
- I would be interested in knowing how our community answers questions like this. On the specific compound, there are several refs in Pubchem, including the fact that there is an EU REACH record, implying that it is manufactured at reasonable scale (enough that some supplier is prepared to pay for the registration). So for this specific compound I think that the problem is that WP hasn't expanded the article, rather than that there wouldn't be WP:SIGCOV if we looked for it. Michael D. Turnbull ( talk) 14:35, 24 August 2020 (UTC)
- A quick search reveals more than on paper on the topic, and that the chemical is listed for sale. So I have remove the notability in question tag. A PubChem or ChemSpider listing does nothing to prove notability. But the references they link to might.
Graeme Bartlett (
talk)
21:45, 24 August 2020 (UTC)
- I was interested by a couple of references I came across for diphenyl-2-pyridylmethane, so I'm going to take a stab at expanding the article over the next couple of days. I still don't know how we tackle the wider question, although I know that Graeme keeps a lists of chemicals he feels have merit as AfC. Michael D. Turnbull ( talk) 10:57, 25 August 2020 (UTC)
 Done So that article now looks a bit better. @
AleatoryPonderings: did you come across any others which could be worth expanding now?
Michael D. Turnbull (
talk)
15:34, 25 August 2020 (UTC)
Done So that article now looks a bit better. @
AleatoryPonderings: did you come across any others which could be worth expanding now?
Michael D. Turnbull (
talk)
15:34, 25 August 2020 (UTC)
- @ Michael D. Turnbull: I've just been going through CAT:NN at random to find articles to clean up or nominate for deletion as appropriate, so I haven't encountered any others specifically. But Category:Organic compound stubs seems to have quite a few potential compound articles for expansion. I suppose I was also just wondering in general if we have any WP:SNGs for compounds, as it's clearly a specialist topic to which ordinary guidelines plausibly don't apply. AleatoryPonderings ( talk) 15:45, 25 August 2020 (UTC)
Hello chemists! I bid y'all a warm hello. I came here today to inform thee of two Wikipedia sites that might be of interest to y'alls'. I've edited both of these pages, but since i am neither a chemist nor a college/graduate student, I came here to ask for your help. The former needs fixing from a chemist, whereas the latter doesn't have a single reference, until i put some. Additionally, the computations in the latter prove too much also for my teeny brain. Please abet me in fixing these pages, fellow wikipedians! Thank you y'all for reading this tedious query. Have a nice day! Wiswesser's rule Ionization energy Ice bear johny ( talk) 09:46, 10 September 2020 (UTC)
- Not a chemist, but I cannot find any Google Scholar or Google Books hits for Wiswesser's rule meaning the particular formulation of Klechkovsky's rule that it is.
Double sharp (
talk)
09:53, 10 September 2020 (UTC)
- I an a chemist and Wisesser's rule and Klechkovsky's rule are both just different names for the concept explained at
Aufbau_principle#Madelung_energy_ordering_rule. The Wiswesser rule page should arguably just be a redirect to the main
Aufbau principle, unless anyone can find references that suggest otherwise. Incidentally,
William Wiswesser is much better known for his notation used to encode chemistry in early databases — a method still occasionally used today.
Ionization energy looks like a more technical article that needs extra citations and some simpler language. I hope someone more familiar with the details than I am will volunteer to improve it.
Michael D. Turnbull (
talk)
11:47, 10 September 2020 (UTC)
- Thank you, that confirms my suspicion. As I mentioned above I cannot find any references to the Aufbau principle aka Klechkovsky rule being referred to under this name, but maybe there are some. I've redirected the
Wiswesser's rule page to
Aufbau principle as you suggested.
Double sharp (
talk)
12:10, 10 September 2020 (UTC)
- Fine. Seconds before you did the redirect I added a section on the Talk page of the old article that this should be done, so there is a clear record there of our thinking. I think that the reference to his 1945 paper should be added in the history section near Aufbau_principle#Madelung_energy_ordering_rule (I've checked that it is still available) and I'll do that soon. Michael D. Turnbull ( talk) 12:18, 10 September 2020 (UTC)
- Thank you, that confirms my suspicion. As I mentioned above I cannot find any references to the Aufbau principle aka Klechkovsky rule being referred to under this name, but maybe there are some. I've redirected the
Wiswesser's rule page to
Aufbau principle as you suggested.
Double sharp (
talk)
12:10, 10 September 2020 (UTC)
- I an a chemist and Wisesser's rule and Klechkovsky's rule are both just different names for the concept explained at
Aufbau_principle#Madelung_energy_ordering_rule. The Wiswesser rule page should arguably just be a redirect to the main
Aufbau principle, unless anyone can find references that suggest otherwise. Incidentally,
William Wiswesser is much better known for his notation used to encode chemistry in early databases — a method still occasionally used today.
Ionization energy looks like a more technical article that needs extra citations and some simpler language. I hope someone more familiar with the details than I am will volunteer to improve it.
Michael D. Turnbull (
talk)
11:47, 10 September 2020 (UTC)
Oh wow y'all are quick to reply! Thanks guys for the trouble :>>>. Ice bear johny ( talk) 13:12, 10 September 2020 (UTC)
But wait, the formula in Wiswesser's rule supports the Aufbau principle? Would you include it in Aufbau principle, or not? Is it inconsequential? I added it in my notes, and thought that it would help. Thank you for answering! Ice bear johny ( talk) 13:15, 10 September 2020 (UTC)
- His rule gives the same predictions for the filling of the orbitals as does the Aufbau principle, as was mentioned on the page in question, which is still on WP if you know where to look "namely, here".. As I said, I'll add his paper as a citation in the article on the Aufbau principle and I'll also add it to his biography page, with appropriate detail once I've figured out whether it was really a novel idea in 1945 or just an alternative way of expressing what was already known at the time. Interestingly, his publication does pre-date Klechkovsky's (1960s) but not Madelung's (1930s), although the sources seem to say that Madelung expressed the "rule" empirically, not as in the formulae Wiswesser used. I'll have to read his article carefully to see whether he had a theoretical justification for the rule, which is what Klechkovsky provided, I think. Michael D. Turnbull ( talk) 13:48, 10 September 2020 (UTC)
Oh ok. You adding his paper assuages me. Thank you for helping me :>. Your help is greatly appreciated! Ice bear johny ( talk) 16:18, 10 September 2020 (UTC)
More edits
Hello! I would like to request your abetment. You see I am neither a chemist, nor a college student; ergo, i cannot ascertain that the information i input is 100% correct. Hence i beseech your help in the following pages: Ionization Energies, Effective nuclear charge, Core charge and Shielding effect. All of them are under referenced, and other editors that I know are particularly disinterested in editing because of the sheer hard work involved. Thanks y'all! Ice bear johny ( talk) 06:44, 11 September 2020 (UTC)
Bruh the shielding effect-. Guys please edit that part. Literally, i cannot describe it's fathomable, facile information! Ice bear johny ( talk) 12:38, 11 September 2020 (UTC)
Sensitivity of azidoazide azide
The 1-Diazidocarbamoyl-5-azidotetrazole article cites peer-reviewed sources which describe the compound as extremely sensitive, detonating at the slightest stimulus or for no apparent reason, but it also mentions hobbyists such as I make C2N14 in my shed who synthesized it themselves and found it far less sensitive. Should we include these amateur anecdotes? There's been some off-and-on discussion on the talk page, but it's inconclusive and would benefit from more input. – dlthewave ☎ 23:50, 19 September 2020 (UTC)
- Well, assuming of course that you've actually made the right stuff in your shed, rather than something else which explodes somewhat less readily... then polymorphs perhaps? --
Project Osprey (
talk)
00:15, 20 September 2020 (UTC)
- My chemistry is too rusty to speculate on that, but from a Wikipedia policy perspective we would need secondary-source coverage to A) establish
WP:DUE WEIGHT and B) analyze/explain the disparity. –
dlthewave
☎
03:13, 20 September 2020 (UTC)
- Well you'd need to get the results published in a reputable journal first. Youtube is obviously not a peer-reviewed source. -- Project Osprey ( talk) 09:29, 20 September 2020 (UTC)
- My chemistry is too rusty to speculate on that, but from a Wikipedia policy perspective we would need secondary-source coverage to A) establish
WP:DUE WEIGHT and B) analyze/explain the disparity. –
dlthewave
☎
03:13, 20 September 2020 (UTC)
The location and composition of Group 3 of the periodic table
Here (open access). About nine months in the making; I only signed off on the (eighth) proof, last night. Sandbh ( talk) 12:52, 24 September 2020 (UTC)
- Who coined the term "lanthanoid"? wikt:lanthanoid and wikt:lanthanide could do with etymology. Graeme Bartlett ( talk) 13:16, 24 September 2020 (UTC)
Deprecating usage of the template {{radic}} to write root radicals has an RFC for possible consensus.

"Deprecating usage of the template {{radic}} to write root radicals" has an RfC for possible consensus. I you would like to participate in the discussion, you are invited to add your comments on the discussion page. Thank you. Walwal20 talk ▾ contribs 02:12, 25 September 2020 (UTC)
hi all, just a heads up, Jean-Pierre Abbat who was a chemist(?) is up for deletion, afd is here. Coolabahapple ( talk) 04:01, 25 September 2020 (UTC)
PT in lead of the PT article
Now updated:
Periodic table
---
Sandbh (
talk)
07:27, 25 September 2020 (UTC)
R1 Plasmid
How does the R1 Plasmid impact multi-drug antibiotic resistance in its host cell? Vbam25 ( talk) 22:40, 30 September 2020 (UTC)
How does the R1 Plasmid impact multi-drug antibiotic resistance in its host cell?
9/30/2020 Vbam25 ( talk) 22:42, 30 September 2020 (UTC)
- This sounds more like a microbiology question than a chemistry one. Although it will be reflected in some kind of chemical activity or lack of it.
Graeme Bartlett (
talk)
08:43, 1 October 2020 (UTC)
- @ Vbam25: I'm afraid I've can't give a good answer to your question. There is a relevant article at R-factor but if that or its references don't help, try posting a specific question again at WP:RD/S. Michael D. Turnbull ( talk) 11:11, 1 October 2020 (UTC)
A quick ping here too: Validation of CAS numbers; collaboration with Wikidata? -- Egon Willighagen ( talk) 08:15, 11 October 2020 (UTC)
After almost two years of waiting, there still exists no reference in this article proving that "Lasri condensation" is an actual thing (I'm not talking about the chemical reaction, but the term itself). On superficial literature search, I found no paper using the term "Lasri condensation". I have the strong feeling that somebody from Lasri's group is trying to establish this term using Wikipedia. The users contributing to the article and uploading figures are named after former colleagues and PIs of Lasri. The second figure in the article is taken from Lasri's 2018 publication. Lasri himself is refering to the Wikipedia article on his website to suggest that the "Lasri condensation" exists. I suggest renaming this article to something without "Lasri" in it. What does everybody else think? -- Hbf878 ( talk) 12:56, 13 October 2020 (UTC)
- Unless we have
WP:SECONDARY source for the name, I strongly oppose using that name for this reaction anywhere in the article, let alone in the title. And for the actual title, we'd further need evidence that it's
WP:COMMONNAME rather than COI touting their own work. Is there any secondary ref at all? Otherwise, I suspect it might not even merit an article at all (
WP:GNG fail).
DMacks (
talk)
13:09, 13 October 2020 (UTC)
- No secondary sources supporting the term "Lasri condensation", and I cannot find the term anywhere except for pages that are in direct connection with Lasri. What's the common procedure in these cases? Nominate for deletion? --
Hbf878 (
talk)
13:23, 13 October 2020 (UTC)
- Do you think this specific reaction, as defined by whatever specific details, is actually independently notable (regardless of what the article about it would be titled)?
DMacks (
talk)
14:04, 13 October 2020 (UTC)
- Honestly, I don't really know. It doesn't seem to be revolutionary, as the articles in the references are not cited very much (2x since publication in 2018). Maybe there's someone here who can assess whether this reaction is notable. Btw, there have been
discussions about this article and Lasri previously. --
Hbf878 (
talk)
15:43, 13 October 2020 (UTC)
- Hell's teeth! It is just a special case of a standard pyrazole formation reaction (see first diagram in that article). I'll AFD it immediately. Please add support as soon as I do that. Michael D. Turnbull ( talk) 17:00, 13 October 2020 (UTC)
- I've put the necessary template on the page and notified the user who created the page. Michael D. Turnbull ( talk) 17:10, 13 October 2020 (UTC)
- Honestly, I don't really know. It doesn't seem to be revolutionary, as the articles in the references are not cited very much (2x since publication in 2018). Maybe there's someone here who can assess whether this reaction is notable. Btw, there have been
discussions about this article and Lasri previously. --
Hbf878 (
talk)
15:43, 13 October 2020 (UTC)
- Do you think this specific reaction, as defined by whatever specific details, is actually independently notable (regardless of what the article about it would be titled)?
DMacks (
talk)
14:04, 13 October 2020 (UTC)
- No secondary sources supporting the term "Lasri condensation", and I cannot find the term anywhere except for pages that are in direct connection with Lasri. What's the common procedure in these cases? Nominate for deletion? --
Hbf878 (
talk)
13:23, 13 October 2020 (UTC)
Can we add new chemistry related wordings and phenomenons?
While reading we come across so many new wordings and phenomena, which was not in wikipedia. How to add this to the page with proper references? Not only chemistry but also every new thing we found? Preethanuj Preethalayam ( talk) 07:15, 18 October 2020 (UTC)
- Here are some perspectives with your well-intentioned proposal:
- Of course new info can be added but it should be done in context. If the topic has 2000 hits in SciFinder and is published in a primary journal, I would often challenge the addition. A new review WP:SECONDARY or a new textbook/monograph WP:TERTIARY are among the best edits one could possibly make.
- Often, editors who want to "help" Wikipedia by adding new references, are engaged in COI. WP:COI. Many newish editors are looking for a way to shoehorn references to themselves, their mentors, or their pals.
- As an encyclopedia, Wikipedia is a compendium of digested, mainstream knowledge. Wikipedia-Chem does not aspire to account for the newest results, ordinarily. WP:NOTNEWS, WP:RECENTISM. Lots of blogs exist if one has the urge to report on new stuff.
- My two cents, --
Smokefoot (
talk)
14:15, 18 October 2020 (UTC)
- To add to what Smokefoot just said. You can see an example of this as Lasri condensation. Take a look and use your chemical judgement + knowledge of WP to decide whether or not you think it is a good article. If you answer "yes", then I don't think you are ready to create one yourself. For the meantime stick to improving existing articles. If you answer "no" then well done you may well be the sort of editor we want here — the more the better. Check out the part of this page just above at WT:WikiProject_Chemistry#Lasri_condensation, which shows that we sometimes improve WP by chucking stuff out, not always by including more of the same. Again, Welcome! But start slowly, by editing existing chemistry. Mike Turnbull ( talk)
- On the more inclusive side, you could propose your ideas for new articles here, and people will respond. In some cases if there is new wording, but nothing else new past what is in the article a redirect may be appropriate. Graeme Bartlett ( talk) 09:18, 21 October 2020 (UTC)
Protic solvent, etc.
I just switched polar solvent to redirect to protic solvent. Protic solvent has sections on polar aprotic solvents and polar protic solvents (slightly redundant?). Would someone else look at protic solvent and make sure that these important links are ok? Do we need to create a self-standing polar solvent? -- Smokefoot ( talk) 18:48, 23 October 2020 (UTC)
- That seems confusing to me...protic is subtype of polar (as you note, polar-protic is semi-redundant) and there's lots to say about polar in general, rather than just as a subtopic of protic.
DMacks (
talk)
18:54, 23 October 2020 (UTC)
- I was scrambling since prior to my action "polar solvent" was the title, but the lead was all about protic. One alternative solution would be to revert my work and rewrite the lede in the context of polar vs non polar solvents.--
Smokefoot (
talk)
19:12, 23 October 2020 (UTC)
- Definitely no harm in fixing one mess, even if it can be fixed further or differently later.
DMacks (
talk)
10:18, 24 October 2020 (UTC)
- As it stands today the redirect on
polar solvent is very misleading as it leads to an article on protic solvents. So while DMF is certainly polar, it ain't protic. The actual article where the redirect leads has a table where all possibilities: non-polar, polar aprotic and polar protic are discussed, so I think that it is the article title, not the content, that's the problem. Looking at the article
solvent, much of the same material is covered in arguably a more general way. So one possibility would be to scrap
protic solvent altogether and make all these terms just rediredts to
solvent.
Mike Turnbull (
talk)
13:37, 24 October 2020 (UTC)
- Thank you for the pondering. Following MDT's thinking, one idea is to create
Reaction solvent, which might captures the idea that would accommodate aprotic, protic, polar solvent.--
Smokefoot (
talk)
14:43, 24 October 2020 (UTC)
- Trouble is that virtually all solvents mentioned in the
solvent article can be used as reaction solvents. A while ago, I added the reference
[10] to that article: this is a tool for solvent selection developed by Astra-Zeneca. So I'm not convinced that a separate article is needed unless one of the subsections at
solvent gets so unbalanced in size that it should have a {{Main|Sub-article}} tag, which is not yet the case.
Mike Turnbull (
talk)
15:02, 24 October 2020 (UTC)
- Ok good advice. Lets just follow what the title says, the article will be about protic solvents. Period. Polar solvent will redirect to solvent. Seems straightforward.-- Smokefoot ( talk) 16:00, 24 October 2020 (UTC)
- Trouble is that virtually all solvents mentioned in the
solvent article can be used as reaction solvents. A while ago, I added the reference
[10] to that article: this is a tool for solvent selection developed by Astra-Zeneca. So I'm not convinced that a separate article is needed unless one of the subsections at
solvent gets so unbalanced in size that it should have a {{Main|Sub-article}} tag, which is not yet the case.
Mike Turnbull (
talk)
15:02, 24 October 2020 (UTC)
- Thank you for the pondering. Following MDT's thinking, one idea is to create
Reaction solvent, which might captures the idea that would accommodate aprotic, protic, polar solvent.--
Smokefoot (
talk)
14:43, 24 October 2020 (UTC)
- As it stands today the redirect on
polar solvent is very misleading as it leads to an article on protic solvents. So while DMF is certainly polar, it ain't protic. The actual article where the redirect leads has a table where all possibilities: non-polar, polar aprotic and polar protic are discussed, so I think that it is the article title, not the content, that's the problem. Looking at the article
solvent, much of the same material is covered in arguably a more general way. So one possibility would be to scrap
protic solvent altogether and make all these terms just rediredts to
solvent.
Mike Turnbull (
talk)
13:37, 24 October 2020 (UTC)
- Definitely no harm in fixing one mess, even if it can be fixed further or differently later.
DMacks (
talk)
10:18, 24 October 2020 (UTC)
- I was scrambling since prior to my action "polar solvent" was the title, but the lead was all about protic. One alternative solution would be to revert my work and rewrite the lede in the context of polar vs non polar solvents.--
Smokefoot (
talk)
19:12, 23 October 2020 (UTC)
In the right hand figure “H2O” should read “H2O”. In addition, it overlaps with an arrow. Anyone volunteering for fixing or re-drawing? Please note that there is a version with a higher resolution in the file history. -- Leyo 17:29, 27 October 2020 (UTC)
- Both the files are .jpg and the fix, if done at all, should be to give an .svg version. I'm not very good with .svg editing yet but I'll give it a try. If anyone else fancies doing this, please go ahead as well
Mike Turnbull (
talk)
17:50, 27 October 2020 (UTC)
- OK,
Leyo, I've
 Done the new file, with some additional tweaks to satisfy my chemical instincts. Please check out the result at
Hexosaminidase#Mechanism of action and confirm you think it is now fine. Once you've done that, I'll tag the previous .jpg with a note to say there is an .svg replacement.
Mike Turnbull (
talk)
13:41, 28 October 2020 (UTC)
Done the new file, with some additional tweaks to satisfy my chemical instincts. Please check out the result at
Hexosaminidase#Mechanism of action and confirm you think it is now fine. Once you've done that, I'll tag the previous .jpg with a note to say there is an .svg replacement.
Mike Turnbull (
talk)
13:41, 28 October 2020 (UTC)
- Convenience links:
- * original: File:Mechanism.jpg
- * replacement: File:Hydrolysis mechanism.svg
- Should the "loss of alcohol" detail (curved reaction-arrow pointint to ROH) actually be associated with the first step rather than the second? The "addition of water" curved-arrow might also want to be moved there...that's the point in the process where the ROH disappears from the image and HOH appears instead. This image is already superior to the original, just some details that can be improved even further now that we have lots of eyes and hands on it.
DMacks (
talk)
13:58, 28 October 2020 (UTC)
- Yes, you are correct that the ROH leaves in the first step, so I'll move that portion. The water doesn't arrive until the second step, so I think that part is OK. I'll upload a new version in a while and see what you think.
Mike Turnbull (
talk)
14:20, 28 October 2020 (UTC)
- OK, that seems better. Any further comments?
Mike Turnbull (
talk)
14:34, 28 October 2020 (UTC)
- Thank you. I looks good now. However, please note that I am not an expert in reaction mechanisms. -- Leyo 23:56, 28 October 2020 (UTC)
- I'm always torn how to handle reagents on arrows in a mechanistic diagram. In this case, it's true that the ROH is a reactant in the second step. But in the graphics of the mechanism, it has become present during the first arrow. That is, the true process is "1) H2O is formed; 2) H2O goes away; 3) ROH positions; 4) ROH reacts". Visually [1] is happening in the first mechanism and [1,2,3] happen in order to get from first diagram to second diagram and only [4] is happening to get from second to third. But the way the reaction-arrows are written it's [1,2] as the first vs [3,4] second.
DMacks (
talk)
14:40, 29 October 2020 (UTC)
-
DMacks I'd say it's fine as it is. ROH is not "a reactant in the second step": it has been expelled in the first step — R is in fact the large GM3 ganglioside, as is clear from the other diagram alongside this one in the article. The second step is the hydrolysis of the oxazolinium to release N-acetyl galactose and regenerate the enzyme. The water is shown twice in the diagram: once over the second arrow and also in position under the upper carboxylate ready for its attack on the carbon that will become the anomeric carbon of the N-acetyl galactose.
Mike Turnbull (
talk)
15:00, 29 October 2020 (UTC)
- Whoops, yeah, my explanation switched HOH and ROH. Need more coffee, less fume-hood air. DMacks ( talk) 15:02, 29 October 2020 (UTC)
-
DMacks I'd say it's fine as it is. ROH is not "a reactant in the second step": it has been expelled in the first step — R is in fact the large GM3 ganglioside, as is clear from the other diagram alongside this one in the article. The second step is the hydrolysis of the oxazolinium to release N-acetyl galactose and regenerate the enzyme. The water is shown twice in the diagram: once over the second arrow and also in position under the upper carboxylate ready for its attack on the carbon that will become the anomeric carbon of the N-acetyl galactose.
Mike Turnbull (
talk)
15:00, 29 October 2020 (UTC)
- OK, that seems better. Any further comments?
Mike Turnbull (
talk)
14:34, 28 October 2020 (UTC)
- Yes, you are correct that the ROH leaves in the first step, so I'll move that portion. The water doesn't arrive until the second step, so I think that part is OK. I'll upload a new version in a while and see what you think.
Mike Turnbull (
talk)
14:20, 28 October 2020 (UTC)
- OK,
Leyo, I've
Updating the template about nitrides
Good day! I really liked the navbox Template:Phosphides that ties together articles about phosphides in parallel to Template:Phosphorus compounds. Since it was missing, I made a similar template about arsenides ( Template:Arsenides) which also works in parallel to Template:Arsenic compounds. However, when I tried to update the template about nitrides Template:Nitrides (to make it look like this draft) I was not sure how to proceed. Updating the current version of the nitrides template to make it look like the phosphide and arsenide templates may change it significantly and I don't know whether it will disrupt any of the 300+ pages that link to the template. First, I don't know whether I should update the template. Is it used in some articles? I didn't find it on articles it links to. Second, I don't know how to check whether the update would disrupt the pages linked to it. I would greatly appreciate advice on the matter. -- ElMagyar ( talk) 21:16, 1 November 2020 (UTC)
- I think it is a good idea to add that extra table with binary, tertiary etc. However the periodic table in the draft, and in the phosphides template is very ugly, being squashed in the middle of the screen. So if you can use the style for the periodic table in your draft and paste it as the next version in the nitrides template. It should not be disruptive if it works! Graeme Bartlett ( talk) 11:54, 2 November 2020 (UTC)
Selenenic acid
Changes in article names based on one's ideas of proper or correct nomenclature should be discussed. Such as discussion has been underway on the carboxylic acids. Chemists often call R-S-R thioethers, still. R-Se-O-H are called selenenic acids. Maybe some committee decided that they should be called SeO-Selenoperoxols. Wikipedia is respectful of such committees but we are not necessarily obedient to them. In the end, changes are made by consensus.-- Smokefoot ( talk) 13:28, 25 October 2020 (UTC)
- And
user:Daniblanco renamed the self descriptive
Thioselenide to
Selenothioperoxide, In my opinion having peroxide in the name is quite misleading as there is no oxygen. However when I look, either name is very seldom used.
Graeme Bartlett (
talk)
20:43, 25 October 2020 (UTC)
- I invited user:Daniblanco to engage the community here.-- Smokefoot ( talk) 13:02, 26 October 2020 (UTC)
- Sulfhydryl leads to sulfalkyl or sulfaryl, then to sulfalkylselenide or sulfarylselenide for the --Se--S--R part. If invention is allowed, an alkyl (or aryl) sulfalkylselenide or (sulfarylselenide) is better. Or alkylsulfalkylselene -- Ktsquare (talk) 04:00, 2 November 2020 (UTC)
- additional question. Is there a difference between the thia and thio prefix? It seems they are mixed in use: Thiopyrylium, thiabendazole, thiourea, thiosulfate -- Ktsquare (talk) 04:00, 2 November 2020 (UTC)
- From a discussion above, Me-Se-S- Me is called dimethyl selenosulfenate, no? -- Ktsquare (talk) 21:53, 4 November 2020 (UTC)
Newbie question: if I find an interesting new route synthesizing a chemical using wikipedia links, how shall I proceed?
Per title, what shall I do? -- Ktsquare (talk) 02:40, 2 November 2020 (UTC)
- Ktsquare There is an "if" in your question which makes it difficult to answer. Please could you give a specific example of an article containing something you find interesting and a suggestion of what it is you want to do. Mike Turnbull ( talk) 10:51, 2 November 2020 (UTC)
- If you look at how the synthesis of adamantane progresses, the breakthrough happens at using dicyclopentadiene, instead of methanal and diethyl malonate condensation. In simple terms, adding hydrogen to a C10H12 and promoting rearrangement make the end product, instead of removing the "branches" from the adamantane skeleton. Can a synthesis just make the parent structure directly in few steps from available compounds? For example only, Wikipedia indicates lignin presents a potential of renewable source of aromatic compounds. Monolignol says paracoumaryl alcohol is found almost exclusively in grasses. Ethenolysis splits an carbon carbon double bond. Cracking_(chemistry) says it breaks down complex hydrocarbons into lighter ones.
Now
- Extract P-alcohol from high purity lignin that originally comes from grass or after paper making.
- Ethenolysis splits P-alcohol to allyl alcohol and para vinyl phenol.
- Cracking splits vinyl phenol to phenol and acetylene.
If possible, all the inorganic salts, carboxylic acid and carboxylate can be skipped altogether. Also, if the cracking in the last step is economical and can be done in large scale, recycling polystyrene can be possible because paravinyl phenol monomer is structurally similar to styrene. In other words, Cracking_(chemistry) make benzene and acetylene from polysytrene waste. hydrogenate some of the acetylene product to ethene to support the ethenolysis in previous step. I know already someone is going to say these are all fantasy from the very beginning... :) --- Ktsquare (talk)
- Do you have suitable references to show this works and has been done, or is it your own idea? We do not include WP:Original research. Also if there is only one publication on the topic it my be undue to include that synthesis. We really like things that have appeared in reviews or textbooks, then we have more confidence it is not a mistake or a crackpot idea. Graeme Bartlett ( talk) 22:27, 4 November 2020 (UTC)
- So there should be numerous suitable references to show how each step has been done, have I understood the whole reasoning correctly? Just an observation, your use of "we" seems to indicate I am the "you" -- second person in plural, the diction appears to suggest a social distance. --
Ktsquare
(talk)
22:35, 4 November 2020 (UTC)
- When I say "we", I am speaking on behalf of people in WikiProject Chemistry. For me personally I am happy to use primary research literature, if it looks reasonable. But others here really like their facts to have appeared in a textbook or review. Another issue is
WP:Synthesis (not chemical synthesis), but putting facts together to get a conclusion that no one else has so far reached. But we have it on Wikipedia as original research. If there is at least one suitable reference to show the whole process then the information can be added. If there are numerous suitable references, then you could write a whole article on the topic.
Graeme Bartlett (
talk)
22:58, 4 November 2020 (UTC)
- I am guessing that you know this already though. for example if we have a reference that says we can make B from A and another references says we can make C from B, then we should not add to the C article that we can make C from A based on that. Graeme Bartlett ( talk) 23:03, 4 November 2020 (UTC)
- How is it possible that you speak on behalf of people in the WPC...Would you not represent yourself? --
Ktsquare
(talk)
05:01, 6 November 2020 (UTC)
- See if anyone else here responds! Some will react when something bad happens. But so far I see nothing inappropriate from you. I try to summarise the consensus I see. Graeme Bartlett ( talk) 05:15, 6 November 2020 (UTC)
- When I say "we", I am speaking on behalf of people in WikiProject Chemistry. For me personally I am happy to use primary research literature, if it looks reasonable. But others here really like their facts to have appeared in a textbook or review. Another issue is
WP:Synthesis (not chemical synthesis), but putting facts together to get a conclusion that no one else has so far reached. But we have it on Wikipedia as original research. If there is at least one suitable reference to show the whole process then the information can be added. If there are numerous suitable references, then you could write a whole article on the topic.
Graeme Bartlett (
talk)
22:58, 4 November 2020 (UTC)
- The WP:SYNTH policy is a good read. It talks about having to follow refs for what they say as limited by their specific scope, not how any wikipedia editor might be able to string their ideas together in a novel way. It's in keeping with the WP:V policy. Those are two governing policies for content that anyone can write in an article. In discussions among editors on Wikipedia, it's common to use "we" since we (including you) are all potential writers discussing what any one of us might write on this collaborative website, as bound by the WP site policies and guidelines. So for example if there is a ref that "secondary alcohols can be oxidized with Jones reagent to make ketones" we can't say "acetone can be made from isopropanol using Jones reagent" unless we have a ref for using Jones on exactly that structure. DMacks ( talk) 05:49, 6 November 2020 (UTC)
| This is an archive of past discussions. Do not edit the contents of this page. If you wish to start a new discussion or revive an old one, please do so on the current talk page. |
| Archive 45 | ← | Archive 47 | Archive 48 | Archive 49 | Archive 50 | Archive 51 | → | Archive 54 |
Polonol
Hello. I can't find a single mention of Polonol online in a reliable source. Can someone please check the book sources cited? Thanks. Capewearer ( talk) 20:37, 14 March 2020 (UTC)
- Well the first reference "Chemie des Poloniums" does not mention any chemical like that. There are some organo-polonium compounds there, but not R-Po-OH. The second references does not mention it either. There is a Polonium section that fails to mention it.
This is looking like something made up, or a hoax.[1] Graeme Bartlett ( talk) 21:38, 14 March 2020 (UTC)- SciFinder has no entries for polonol. Zero. There are two papers that (barely) mention C-Po-H. Good luck if you go with AFD, seems like it is really difficult to lawyer through that site. -- Smokefoot ( talk) 22:15, 14 March 2020 (UTC)
- @
Graeme Bartlett: the structure is R-Po-H, as Smokefoot notes, not R-Po-OH. Let me know if anyone needs help with the AfD process.
DMacks (
talk)
04:03, 15 March 2020 (UTC)
- Thanks for that, but the sources provided do not have that either. Dimethyl-, diephenyl- or diethyl polonium gets a mention. Graeme Bartlett ( talk) 04:23, 15 March 2020 (UTC)
- I have invited the writer to join in the discussion here.
Graeme Bartlett (
talk)
02:20, 15 March 2020 (UTC)
- The article appears to be a translation of the article on zhwiki about this substance (
zh:釙醇). "Not citing here that our article is a translation of some other source (with cite to that source)" is a license violation. Pinging
User:A2569875 who wrote that article there and seems somewhat active here on enwiki and on commons in the chemistry genres.
DMacks (
talk)
04:11, 15 March 2020 (UTC)
- If it is a translation then I cannot claim it was made up. So I strike my comment. Perhaps the name is made in analogy to
tellurol.
Graeme Bartlett (
talk)
04:23, 15 March 2020 (UTC)
- It could totally be made up or a hoax, just one that originated external to enwiki. That is, User:Nucleus hydro elemon could have been acting in good faith and topic itself still ends up being nonsense. Perhaps they didn't actually check the sources they transcribed? The original author can help clear this up I hope. DMacks ( talk) 04:40, 15 March 2020 (UTC)
- Sorry. In fact, I have not been able to find any valid references since 2012. The Chinese Wikikipedia page has been requested to be deleted,
WP:Articles for deletion/Polonol (釙醇). cc
DMacks,
Graeme Bartlett.--
Nanachi
🐰
Fruit Tea☕(宇帆·
☎️·
☘️)
06:09, 15 March 2020 (UTC)
- Thanks for checking up on it! DMacks ( talk) 14:27, 15 March 2020 (UTC)
- If it is a translation then I cannot claim it was made up. So I strike my comment. Perhaps the name is made in analogy to
tellurol.
Graeme Bartlett (
talk)
04:23, 15 March 2020 (UTC)
- The article appears to be a translation of the article on zhwiki about this substance (
zh:釙醇). "Not citing here that our article is a translation of some other source (with cite to that source)" is a license violation. Pinging
User:A2569875 who wrote that article there and seems somewhat active here on enwiki and on commons in the chemistry genres.
DMacks (
talk)
04:11, 15 March 2020 (UTC)
On periodic trend grounds I am already rather sceptical that this exists. Due to relativistic destabilisation of the 6p3/2 orbital Po is actually significantly more metallic than Te and it should be quite unhappy in the −2 oxidation state. doi: 10.1002/9781119951438.eibc0182 might be useful (it's a 2011 review of organopolonium chemistry): it is quite short (as expected for the subject), but it looks like what is known is mostly dialkyls/diaryls as well as tetravalent compounds of the form Ar3PoX and Ar2PoX2. Nothing about polonols that I can see. Double sharp ( talk) 15:14, 15 March 2020 (UTC)
- Maybe
organopolonium compound should become blue?
DMacks (
talk)
15:44, 15 March 2020 (UTC)
- Good edit
[2],
Headbomb. Now here is a diff:
[3] ->
[4]
[5]. -
DePiep (
talk)
08:23, 19 March 2020 (UTC)
- Go troll elsewhere.
Headbomb {
t ·
c ·
p ·
b}
08:24, 19 March 2020 (UTC)
- There are literatures about organopolonium compounds (R2Po, R2PoX2, etc.), but none about polonol. -- Leiem ( talk) 17:33, 19 March 2020 (UTC)
- Go troll elsewhere.
Headbomb {
t ·
c ·
p ·
b}
08:24, 19 March 2020 (UTC)
- @
DMacks: It is blue now. It'll probably remain a stub for a long while, though. ;)
Double sharp (
talk)
04:52, 12 April 2020 (UTC)
- @ Double sharp: Thanks for your work on that! DMacks ( talk)|
- Good edit
[2],
Headbomb. Now here is a diff:
[3] ->
[4]
[5]. -
DePiep (
talk)
08:23, 19 March 2020 (UTC)
The reference "Chemie des Poloniums" turns up in other Polonium articles in equally suspect ways. Polonium hydride: "Experiments conducted on the trace scale show indications that the reaction between polonium metal and nascent hydrogen may produce polonium hydride", nascent hydrogen huh? -- Project Osprey ( talk) 20:42, 15 March 2020 (UTC)
- Thank you all for your quick help with this. I've proposed Polonol for deletion, pointing back to this discussion in the deletion rationale. Capewearer ( talk) 18:49, 18 March 2020 (UTC)
- @
Graeme Bartlett: thanks for your delete vote, the article in chinese wikipedia has already deleted
[6]. Consider delete English article?--
Nanachi
🐰
Fruit Tea☕(宇帆·
☎️·
☘️)
04:46, 22 March 2020 (UTC)
- I did vote, and I was considering voting on some of those other Chinese chemical pages. But I now see most were speedily kept as they were real compounds, so no need. We should have just let the prod run, as there was only a dispute over the prod2. But now we can let the AFD run, and it will not be contestable, until someone writes about the topic elsewhere.
Graeme Bartlett (
talk)
11:29, 22 March 2020 (UTC)
- Sorry I hadn't made clear to the PROD2 editor that I was not contesting the notion of PROD itself. But it would likely have then been deleted and I didn't want that editor's scientific and WP-policy statements to go un-contested...somewhere. Oh well. DMacks ( talk) 14:11, 22 March 2020 (UTC)
- I did vote, and I was considering voting on some of those other Chinese chemical pages. But I now see most were speedily kept as they were real compounds, so no need. We should have just let the prod run, as there was only a dispute over the prod2. But now we can let the AFD run, and it will not be contestable, until someone writes about the topic elsewhere.
Graeme Bartlett (
talk)
11:29, 22 March 2020 (UTC)
Tetramethylammonium hydroxide is a salt
IUPAC definition of a salt: "A chemical compound consisting of an assembly of cations and anions." ( https://goldbook.iupac.org/terms/view/S05447)
Tetramethylammonium hydroxide is a salt.-- Smokefoot ( talk) 16:22, 26 March 2020 (UTC)
- Ok? Is this somehow controversial?
Headbomb {
t ·
c ·
p ·
b}
17:02, 26 March 2020 (UTC)
- Since it is dissolved in water and seems not to exist as a pure solid, it is not a salt. In any case it is a strong alkali or base. We can thank Edgar181 in 2006 for the above statement. From the definition it would be not a salt, as it is not a compound.
Graeme Bartlett (
talk)
12:31, 27 March 2020 (UTC)
That's rather irrelevant to what salts are, no?See also [7] Tetramethylammonium hydroxide is a quaternary ammonium salt generally used as an anisotropic etchant for silicon due to its high silicon etching rate.[1][2] and [8] Tetramethylammonium hydroxide is a solid in the hydrated form. Headbomb { t · c · p · b} 20:21, 27 March 2020 (UTC)- I wonder if colleagues would allow CuSO4.5H2O (aka blue vitriol) to be called a salt? If so maybe we can call the monohydrate of Me4NOH also a salt. But maybe not, who knows?-- Smokefoot ( talk) 22:39, 27 March 2020 (UTC)

structure of Me4NOH monohydrate. - Hydrate of a salt? -- Leiem ( talk) 17:46, 29 March 2020 (UTC)
- IMO, it would not matter whether it's a hydrate or not. At the extreme, NaCl(aq) "salt water", which includes many water molecules wrapped around each cation and each anion, is just as much of a salt as NaCl(s) "table salt." If a salt has one or more waters of hydration in its formula, any working chemist would say that it's still a salt. When I teach this to my students, I say "CuSO4.5H2O is a salt of copper and sulfate which incorporates five waters of hydration." I guess I could be wrong about all that, but I don't think so. KeeYou Flib ( talk) 14:55, 22 April 2020 (UTC)
- Headbomb, apparently so (among some editors at least). I responded there. They seem to be talking round in circles and escallating personal claims, rather than using WP:RS and remaining WP:CIVIL...not a recipe for improving WP articles. DMacks ( talk) 04:11, 29 March 2020 (UTC)
- Since it is dissolved in water and seems not to exist as a pure solid, it is not a salt. In any case it is a strong alkali or base. We can thank Edgar181 in 2006 for the above statement. From the definition it would be not a salt, as it is not a compound.
Graeme Bartlett (
talk)
12:31, 27 March 2020 (UTC)
- Why would it have to be anhydrous for it to be a salt? --
Project Osprey (
talk)
20:18, 28 March 2020 (UTC)
- I was just trying to anticipate any possible objections. Heck, I am surprised we are having this convesation. Maybe you can penetrate the arguments by JonRichfield and his friend here
here. --
Smokefoot (
talk)
20:52, 28 March 2020 (UTC)
- ... I must confess that I cannot. These discussions trend to drag on. Good luck -- Project Osprey ( talk) 22:09, 28 March 2020 (UTC)
- I was just trying to anticipate any possible objections. Heck, I am surprised we are having this convesation. Maybe you can penetrate the arguments by JonRichfield and his friend here
here. --
Smokefoot (
talk)
20:52, 28 March 2020 (UTC)
- I was confused by Smokefoot's original statement, whether calling it a salt is supported or opposed. I will also clarify that Edgar181 was the user that wrote "Tetramethylammonium hydroxide" in the second revision of the page. Graeme Bartlett ( talk) 07:32, 29 March 2020 (UTC)
Styrene
An apparent styrene leak in India has killed 13 people and injured many more. As such there's been a sudden surge in editing on the 'Health effects' section of that page, not all of it to our usual standards. -- Project Osprey ( talk) 08:58, 7 May 2020 (UTC)
Dichlorine pentoxide
Both of the references in this article mentioned that Cl2O5 is non-existent. -- Leiem ( talk) 04:15, 9 May 2020 (UTC)
- Thanks for pointing out this nonsense writing. ChemAbs has about six citations. --
Smokefoot (
talk)
11:58, 9 May 2020 (UTC)
- @ Nucleus hydro elemon:. User:Nucleus hydro elemon is contributing a lot of dubious content. Really unfortunate.-- Smokefoot ( talk) 13:31, 9 May 2020 (UTC)
Sad news
I have some sad news to report ... Ronhjones, a prolific member of this project, passed away on 7 April last year, the day he made his last edit. Tributes are at his talk page. Graham 87 05:26, 2 May 2020 (UTC)
- I have some even sadder news that I can now share publicly: Ron and his wife Sue died together in a house fire, as noted in
their obituary from the London
Inland Waterways Association newsletter. The friend of theirs who confirmed his passing also told me this info, but I didn't want to say it here without confirming that it was publicly available or I had permission.
Graham
87
16:24, 2 May 2020 (UTC)
- It's probably worth a note here that I nominated one of his chemistry pages, UK Chemical Reaction Hazards Forum, for deletion. Graham 87 13:30, 10 May 2020 (UTC)
Crisscross method
I came across the Crisscross method article. To me it seems like an almost trivial method, with just a single source (a textbook that mentions it on one page). Is this method notable enough to satisfy WP:N (and WP:NOTTEXTBOOK)? — Bkell ( talk) 18:45, 25 May 2020 (UTC)
- Support. I suggest you invoke WP:AFD Michael D. Turnbull ( talk) 17:48, 4 June 2020 (UTC)
Cannabigerolic acid
Could somebody check my work for cannabigerolic acid -- first time I've done a chemistry article, I think. I'd like to include the structural formula File:CBGA molecule.svg in the infobox. ☆ Bri ( talk) 23:20, 14 April 2020 (UTC)
- @ Bri:The article looks fine as far as it goes. Your structure drawing has a bad clash between the terminal dimethyl group of the sidechain and one of the phenol oxygens. I took the liberty of uploading a second version more or less exactly as in Chemspider (did you know you can download molfiles from there?). I then reverted my upload back to yours but if you prefer mine you should be able to re-revert to that. Michael D. Turnbull ( talk) 16:16, 13 June 2020 (UTC)
Atom FAR
I did NOT nominate this article for FAR-- just the doing nominations that were not done by the editor who did.
User:Kurzon has nominated Atom for a featured article review here. Please join the discussion on whether this article meets featured article criteria. Articles are typically reviewed for two weeks. If substantial concerns are not addressed during the review period, the article will be moved to the Featured Article Removal Candidates list for a further period, where editors may declare "Keep" or "Delist" the article's featured status. The instructions for the review process are here. SandyGeorgia ( Talk) 19:27, 14 June 2020 (UTC)
Top million substances
Hello, I’m working with User:Egon Willighagen from Wikidata and others to compile a list of what we consider to be the one million most important chemicals. This list will be used to prioritize what we look at for both Wikidata and Wikipedia, and possibly other external groups that interact with us. These chemicals could include things like the elements and other basic substances you would encounter in your chemistry education, chemicals encountered in everyday life (e.g. in detergents, food additives or hair gel) as well as more niche substances such as pharmaceuticals, polymers, pollutants, biologically important materials, etc. Are there any specific collections of substances you would recommend us to look at? Please post any suggested lists or databases below. I'll also post on WT:Chemicals. Many thanks, Walkerma ( talk) 18:15, 19 June 2020 (UTC)
The first two refs in this article use templates that have nothing to do with chemistry. I think some of the links in the chembox are wrong also. This is way beyond me. Can someone here take a look at. Thanks. MB 02:20, 9 July 2020 (UTC)
- This whole article is an unattributed translation from zh:高氯酸钪 by User:Nucleus hydro elemon, whose many such articles including copy-pasting templates without the barest of checks that they have the same function in enwiki as zhwiki. Nucleus, please fix your mess and stop making such messes, as it has long become a WP:DE/ WP:CIR problem. User:Leiem, you seem to contribute to many of the zhwiki articles involved, so perhaps you can talk to them about their edit habits in a way or language that is understandable. DMacks ( talk) 02:35, 9 July 2020 (UTC)
- ...well, Nucleus won't be editing for the next day, but at least this is an explanation of the weird situation MB noted.
DMacks (
talk)
02:50, 9 July 2020 (UTC)
- I have left a message on his zhwp talk page. -- Leiem ( talk) 06:29, 9 July 2020 (UTC)
- In this article, the refs in Chinese are about the coordination compounds synthesized using scandium perchlorate as the reactant. --
Leiem (
talk)
06:29, 9 July 2020 (UTC)
- Everything contributed by User:Nucleus hydro elemon is garbage. He mainly contributes to obscure topics. -- Smokefoot ( talk) 12:29, 9 July 2020 (UTC)
Two cultures
I've been asked (by Eugen Schwarz) a half-scientific question, as follows:
- "(How far) are the concepts of "The Two Cultures" (chemist C. P. Snow ) and of "Paradigm Shift" and "Scientific Revolution" (T. Kuhn) known among real and computational chemists?
I've never heard of " The Two Cultures" concept. I've heard of " Paradigm shift" and " Scientific revolution" but that is only from reading what Scerri has said about the development of scientific knowledge per A tale of seven scientists.
Are there any real and computational chemists here, who could take a stab at Eugen's question?
Thank you --- Sandbh ( talk) 00:14, 16 July 2020 (UTC)
- Well, I am long retired as a computational chemist, but way back I much enjoyed the novels of C. P. Snow, and I read "The Two Cultures" with interest and discussed it with many friends, both chemists and non-chemists. I think it was an important contribution attempting to bridge the gap between scientists and non-scientists. Are you suggesting that computational chemists are not real? -- Bduke ( talk) 01:31, 16 July 2020 (UTC)
Schwarz is a theoretical chemist. I believe he was referring to a “bangs and stinks” chemist. Sandbh ( talk) 12:48, 16 July 2020 (UTC)
Are you suggesting that computational chemists are not real?
Exactly my thought too when I read the question at the top of this thread. "The Two Cultures" originally referred to the split between the humanities and sciences. So I guess this has been extended to a perceived split between theoretical/computational and experimental chemists. I have done both, so I suppose that I am in a somewhat unique position to comment. I personally do not see a conflict between the two. The goal of theoretical/computational chemistry is to explain and predict experimental results. As long as both sides keep in mind thatAll models are wrong, but some are useful
– George Box, then there is no conflict. Contributions from both sides are also essential for Paradigm shifts and Scientific Revolutions. Boghog ( talk) 14:09, 16 July 2020 (UTC)
Thank you. I've passed those comments on to Eugen. Sandbh ( talk) 06:59, 18 July 2020 (UTC)
RFC: Should the default form of the periodic table be changed to put Lu and Lr in group 3, rather than La and Ac?
This RFC may be of interest. Double sharp ( talk) 10:10, 20 July 2020 (UTC)
- As per my comment a few moments ago, I don't think that RFC is an appropriate process here as reliable sources have used both forms and there is no such thing as a default form of periodic tables. Michael D. Turnbull ( talk) 11:07, 20 July 2020 (UTC)
Differentiating electrons d/e
Do anomalies in d/e actually make a difference to the chemistry of the elements concerned?
I understand this is the case with silver which, according to the idealised Madelung Rule, should be d9s2 in the free atom but is in fact d10s1. This discrepancy seems to, at least in part, contribute to the predominance of its main group chemistry.
Teixidó (2019, in Spanish, here) an Emeritus Professor of Chemical Engineering at the University of Barcelona, says that these anomalies impact the elements concerned such that, "their compounds, have properties that do not match the expected regular periodicity. This is the case of Cu, Cr, Pd, Ag, Rh, Pt, Ave or Uno, to name a few." He does not elaborate.
thank you, Sandbh ( talk) 07:23, 18 July 2020 (UTC)
- @ Dirac: Others might have more profound insight, but here is my view. I taught inorganic for years until I realized that the entire d9s2 vs d10s1 topic is some physics thing that has no bearing on chemistry. This orbital ordering issue is 100% tangential to the topic of coordination chemistry, solid state chemistry, and organometallics. In all transition metal complexes and compounds, the d-levels are well below s levels. -- Smokefoot ( talk) 16:18, 18 July 2020 (UTC)
- (Well apart from Palladium's [Kr] 4d10). It is a very physics way of looking at it, what would it's properties be if it were d9s2. I think the honest answer is no one knows for sure: how could we ever force such a change in the bulk material? That said, pretty much everything in chemistry boils down to electrons, so it's reasonably to assume the effects would be profound. -- Project Osprey ( talk) 19:32, 18 July 2020 (UTC)
- If the energy difference between the different arrangements is small, then there may be alternative ways to arrange compound bonds, resulting in more different allotropes or crystal forms. I notice this happening with some actinides. Graeme Bartlett ( talk) 22:35, 18 July 2020 (UTC)
A perspective by Double sharp
I guess this is somewhat breaking my promise not to argue with Sandbh on such things due to its futility. You see, there has been a long-running argument on Wikipedia talk:WikiProject Elements since December regarding the composition of group 3. It's the old chestnut, whether Wikipedia should show lutetium or lanthanum under yttrium. Since lanthanum accidentally happens to be [Xe]4f05d16s2 with no 4f electron in the gas-phase ground-state, the relevance of this has been dragged in.
Of course on Wikipedia the first thing should be the relevant sources, which focus on the issue, and they mostly support lutetium with strong argumentation. The fact that most elementary general chemistry textbooks persist in showing lanthanum is not as consequential per WP:CONTEXTMATTERS, considering that such books rarely cover the heavy 4f and 5d elements at all. In fact the more I read about this dispute it looks more and more like the situation hypervalent molecules like PCl5 are in: there is no 3d component to the bonding from phosphorus, and that has been well-known for decades, but good luck finding an elementary general chemistry textbook that tells you that. Same situation for Lu under Y. Articles have been published from the 1920s(!) to the present explaining why that is the correct placement, but good luck finding it in general chemistry textbooks.
But, I suppose it is natural that the scientific argumentation gets involved at some point. So, on came a lot of discussion on the science. None of it succeeded in changing Sandbh's mind despite how overwhelming the evidence was. Hence my decision to not argue with him due to its futility. I'm drafting an RFC on the matter anyway, since at WT:ELEM we already managed an incredible 5-1 consensus against La under Y, and no one but Sandbh seems to be convinced by his lines of argumentation.
So, take this as a reply to the others involved.
It makes actually basically zero difference. Smokefoot is completely right that it has no bearing on chemistry. For two reasons:
- In real compounds you rarely have neutral atoms. We are talking about metals, they will probably be positively charged.
- Chemical bond energies can go up to around 10 eV. Therefore, excited states in the neutral atom up to about that energy level will matter. In every case of a weird configuration, the "expected" configuration (like d9s2 for silver at 3.11 eV) is well within that. But don't take my word for it, go check it at NIST. ^_^ The extreme cases are niobium and platinum, for which the Madelung-expected configurations are not even 0.15 eV up in energy from the real ones.
But, don't just trust me. I am not a chemist. Nevertheless I got this from an actual chemist, Andrey Kulsha = Droog Andrey, who dropped by Wikipedia talk:WikiProject Elements/Archive 38 and said the same thing I'm saying right now.
| “ | Ground electron configuration of a neutral atom says too little about its chemistry (unless we have a noble gas). Chemical bonds are about 1 to 10 eV, so that's the scale we need to take into account when talking about chemistry. For many elements, especially for f- and d-block ones, ground state is one among dozens of states within a few eV. Neutral atoms are rarely encountered in compounds, so we probably need to look at dications or smth else. The participation of subshells throughout the forest of excited states depends on that electric charge. So, my opinion is that no "conclusive argument" could be based only on ground state configurations of neutral atoms. Droog Andrey (talk) 15:01, 15 February 2019 (UTC) | ” |
That's especially true for things like the inner 4f subshells: they get more active as charge goes up. That's why neutral lanthanum has 4f16s2 at 1.88 eV, but La2+ has 4f1 only at 0.89 eV. Or why neutral gadolinium has 4f86s2 at 1.36 eV, but Gd2+ has 4f8 only at 0.30 eV.
Sandbh points to silver adding a s electron added from the previous element, as opposed to the d one gold does. (Pd [Kr]4d10, Ag [Kr]4d105s1; Pt [Xe]5d96s1, Au [Xe]5d106s1.) He suggests that the discrepancy contributes to it having predominant main group chemistry. But this is clearly neither necessary nor sufficient. Technetium adds an s electron from the previous element, rhenium adds a d one. I do not see any sign of "predominant main group chemistry" in technetium. Quite the opposite actually given how easy it is to have it convert between its different oxidation states. And most f elements, along with the heavy members of groups 3, 4, and 5, have predominant main group chemistry in that they have only the group oxidation state as the stable one and are quite electropositive and basic. Yet their differentiating electrons from the previous elements are usually d or f electrons, not s or p electrons.
Even for physical properties, there aren't pure atoms in solid metals, there aren't atomic wavefunctions, as Droog Andrey also explained at Wikipedia talk:WikiProject Elements/Archive 33. What you look at there is band occupancy. But even there it does not match the configuration in the gas-phase neutral atoms. In fact it spectacularly doesn't match it even for the first two metals. Lithium and beryllium both have 2p band occupancy despite having no 2p electrons as gas-phase atoms chilling out by themselves! As you go further into the periodic table you will almost always have some sort of fractional average occupancy of subshells that matches neither the Madelung rule's prediction nor the gas-phase ground-state configuration. The simple fact of the matter is that the energy required for such rearrangements is chemically easily achievable and therefore the accident of which electron configuration happens to be the ground state in a gas-phase atom chilling out by itself is irrelevant because in chemical environments the same atom may easily take a different configuration!
Finally here are three quotes from noted chemists making exactly the same point. Christian Jørgensen:
| “ | The two major reasons why this series intended for gaseous atoms strongly bewilders chemists is that undue emphasis is made on irrelevant irregularities (such as the chromium, rhodium, palladium . . . . , atoms) and that the lowest level of two different configurations, such as [Xe]4f96s2 and [Xe]4f85d16s2 are only separated by 285 cm−1 in the terbium atom, much less than 1% of the spreading of J-levels of each of the two configurations, and quite negligible for chemical purposes. | ” |
| — Christian Jørgensen (1988), Influence of Rare Earths on Chemical Understanding and Classification | ||
| “ | It is important to realize that the electronic structures listed in Table 6 are those of the neutral (unionized) gaseous atoms, whereas it is the electronic structure of the ions and compounds that we are chiefly concerned with in chemistry. The relationship of the electronic structure of the gaseous atom of an element to that of its compounds can be rather complicated. For example, in the case of the actinide and lanthanide elements, one would not necessarily predict the predominance of the III oxidation state from the electronic structures of the gaseous atoms; there are usually only two so-called "valence electrons," the 7s or 6s electrons, which might indicate a preference for the II oxidation state.
Apparently, specific factors in the crystal structure of, and the aquation (hydration) energies of, the compounds and ions are important in determining the stability of the III oxidation state. Thus, the characteristic tripositive oxidation state of the lanthanide elements is not related directly to the number of "valence electrons" outside the 4f subshell, but is the somewhat accidental result of a nearly constant small difference between large energy terms (ionization potentials on the one hand, and hydration and crystal energies on the other) which persists over an interval of fourteen atomic numbers. Therefore, if we could somehow have a very extended Periodic Table of Elements containing numerous "f" transition series, we might expect that the 5f, rather than the 4f, elements would be regarded as more nearly representative of such f series. |
” |
| — Glenn T. Seaborg (1994), Origin of the Actinide Concept | ||
And W. H. Eugen Schwarz, whose question Sandbh asked here one section ago:
| “ | The second reason for differences between chemically bound transition-metal atoms and free atoms in vacuum is that the electronic motions in free atoms are not disturbed by adjacent atoms. Most free atoms have open valence shells, where the electrons can arrange differently. The orbit−orbit and spin−orbit angular-momenta couplings result in a large number of different electronic states with different energies. For instance, the 3d54s1 configuration of a free Cr0 or Mo0 atom comprises 504 different states with 74 different degenerate energy levels, scattered over several hundred kJ/mol. ...
'The qualitative behavior of chemical elements can be rationalized with the help of the dominant electronic valence configurations of the atoms embedded in a molecular or crystal environment. These may be correctly called the “electronic configurations of the chemical elements”. However, what is listed in respective tables of chemical textbooks under this headline is something else, namely, what physicists call “the configurations from which the J-level ground states of free unbound atoms in vacuum derive”. ... 'The third exception concerns the free neutral transition-metal atoms in vacuum, including the f block. Their ground-state configurations depend in an involved manner on the often-discussed averaged d−d and d−s Coulomb-repulsion energies and also on the individual orbit−orbit (term) and spin−orbit splittings, even if the latter are small. The correct quantitative explanation is vital for the interpretation of atomic vacuum spectra, but exceeds the scope of general chemical education. There are only a few special topics in chemistry that require the correct understanding of free atoms in vacuum (e.g., atom-molecular gas-phase reactions) or of orbit−orbit and spin−orbit couplings of bonded open-shell atoms (e.g., the chemistry of the transition, lanthanoid, and actinoid metals; spin-flip enhanced reaction mechanisms; so-called spin-forbidden processes). [Nota bene, bonded open-shell TM atoms show different configurations from ground-state free ones.] 'Finally, it is misleading to present free atoms as prototypes for the microscopic description of chemical elements in compounds. The common qualitative textbook explanations of the atomic ground states (correctly: J levels) are incorrect. Therefore, we plead for teaching the correct atomic-orbital order (sequence 6) together with the regular exception, sequence 8, for the s block. One need no longer apologize for irregularities. |
” |
| — W. H. Eugen Schwarz (2010), The Full Story of the Electron Configurations of the Transition Elements | ||

In particular this situation also applies very well to the group 3 dispute. In his very last article doi: 10.1016/bs.hpcre.2016.07.001, Karl A. Gschneidner Jr. ("Mr. Rare Earth") presented this figure showing the melting points. He also showed "pseudo-La" on the figure showing what the melting point of metallic lanthanum would be expected to be if 4f were not involved in it. (Of course 4f has no involvement in lutetium.) This kind of thing is why I've been noting that the scientific and source-based support for lanthanum as an f block element is overwhelming. The lack of a 4f electron in the gas-phase ground state of La does not stop it from using its 4f shell to make bonding MOs from.
Oddly enough, Sandbh is perfectly willing to accept the need to go beyond gas phase configurations when it comes to thorium, which is [Rn] 5f0 6d2 7s2 in the ground state gas phase, yet has clearly been shown to have 5f band occupancy in the metal. For lanthanum he just will not do it. There is a lack of consistency here. Of course by now at Wikipedia talk:WikiProject Elements he is calling the raising of the thorium double standard a "zombie that will not die" argument: it indeed won't die as long as he doesn't logically address why on earth thorium is so different from actinium, that thorium with zero 5f valence electrons in the ground-state gas phase may be allowed in the f block, lawrencium with zero 5f valence electrons in the ground-state gas phase may be allowed in the f block, but actinium also with zero valence 5f electrons may not. No consistency.
I have repeatedly been saying this stuff, with the quotes, with the sources. So has Droog Andrey. For months now. Sandbh has refused to change his mind about the relevance of these anomalies in the face of all of this evidence. Double sharp ( talk) 03:50, 19 July 2020 (UTC)
@ Smokefoot, Project Osprey, and Graeme Bartlett: Forgot to ping you, sorry. Double sharp ( talk) 03:53, 19 July 2020 (UTC)
Siekierski & Burgess (2002)
- Mch as I appreciate User:Double sharp's contributions and enjoy discussing such matters with him, his actions here speak for themselves.
- I ask a 120-word question based on the observation of an emeritus professor of chemistry. Double sharp responds with a 2,000+ word diatribe of rehashed clutter, excessive bolding, biased hyperbole, and misrepresentations.
- Thank you Smokefoot for your WP:CIVIL professional response. Thank you Project Osprey for your professional response saying nobody really knows and that it would be reasonable to presume the effects would be profound. Thank you Graeme for advising there may be alternative ways to arrange compound bonds, resulting in more different allotropes or crystal forms.
- Siekierski & Burgess (2002, p. 133) touch upon the question:
- "The band structure of transition metals…consists of a broad s/p band and a narrow d band. With respect to energy the d band is located approximately in the middle of the s/p band. Moreover, depending on the lattice symmetry some d orbitals mix with s and p orbitals along certain crystallographic directions and confer some d character on the s/p band…The energy spread in the d band is narrow, because d orbitals do not extend to large distances from the atom−they are well within their s orbitals—which restricts their interactions with neighbouring atoms."
- Siekierski S & Burgess J 2002, Concise chemistry of the elements, Horwood Publishing, Chichester
- "The band structure of transition metals…consists of a broad s/p band and a narrow d band. With respect to energy the d band is located approximately in the middle of the s/p band. Moreover, depending on the lattice symmetry some d orbitals mix with s and p orbitals along certain crystallographic directions and confer some d character on the s/p band…The energy spread in the d band is narrow, because d orbitals do not extend to large distances from the atom−they are well within their s orbitals—which restricts their interactions with neighbouring atoms."
- Harking back to Teixidó, chromium is supposed to be 3d44s2 but turns out to be 3d54s1. It seems odd to suggest there would not be a discernible chemical difference between the two configurations.
--- Sandbh ( talk) 14:13, 19 July 2020 (UTC)
Getting into the details
Sandbh may call it what he wants. The fact of the matter is that I backed up everything I said with reliable sources and quoted them at length to show what they said. If that is called a "diatribe" of "rehashed clutter", so be it. Reliable sources, if raised by me and helpfully bolded to emphasise what answers his question, do not seem to be wanted, as was evident from the fact that these sources addressing the issue have been raised countless times in archives 42, 44, and 46 of WT:ELEM, and is now again evident from the fact that he calls my statements "biased hyperbole" even though I said exactly what Smokefoot said: that it has no bearing on chemistry. Replying to his contribution, even for the other parties, was evidently a mistake on my part. Even if everything I say is supported by reliable sources, it is simply not a productive activity because he doesn't accept the answer I and by extension the sources I raise give him.
He may indeed enjoy discussing such matters with me. I used to enjoy it in the past. You can see from this part of why I no longer do. I respect him as a person, but this conversation is clearly not productive. Double sharp ( talk) 14:25, 19 July 2020 (UTC)
- That being said, though: @
Smokefoot,
Project Osprey, and
Graeme Bartlett: I hope my answer and the sources I raised have been of interest. Of course, we may discuss the matter productively, based on reliable sources. ^_^ Most of all, it would be good to clearly get the understanding of reliable sources into our articles on such matters, e.g.
electron configuration.
Double sharp (
talk)
14:45, 19 July 2020 (UTC)
- Well, if you folks are down to arguing these kinds of details, then the Elements project is in great shape. Inevitably maturing Wiki projects distill down to a residue of unresolved topics, which are of interest to a tiny fraction of our readers and which should occupy a tiny fraction of the article. IMHO. My advice on sources would be top-drawer textbooks on metals. Congratulations on a nice set of articles.
- From my perspective, the bigger issues with these articles is the content that readers are more likely to consult: natural occurence, main compounds, uses of the elements vs compounds of the elements, generally jetisoning primary refs for better sources.-- Smokefoot ( talk) 15:04, 19 July 2020 (UTC)
- Well, this discussion has advanced quickly. I'm not sure I actually understand the question here, or the problem. Sandh's question "Do anomalies in d/e actually make a difference to the chemistry of the elements concerned?" translated in my mind to "does the electron configuration of an element effect its chemistry?" which it obviously does - if carbon suddenly gained d electrons it's chemist would surely change quite a bit. Double sharp points out that electron configuration can change subject to environment, metallic bonding being an example. Again, that is true. So far no one has said anything wrong - but what is the question? --
Project Osprey (
talk)
19:29, 19 July 2020 (UTC)
- Perhaps there is a call to join in the argument at
Wikipedia talk:WikiProject Elements. I will not be participating as there are other more interesting things to do here.
Graeme Bartlett (
talk)
22:03, 19 July 2020 (UTC)
- Well, I agree that there are very much more interesting things to do than to argue something well-known for decades. ^_^ Anyway, as for articles, I do not intend more than one sentence (despite how the level of debate might make it look ^_^). It would read something like the following:
- Perhaps there is a call to join in the argument at
Wikipedia talk:WikiProject Elements. I will not be participating as there are other more interesting things to do here.
Graeme Bartlett (
talk)
22:03, 19 July 2020 (UTC)
| “ | These exceptions from the Madelung rule do not have a significant impact on chemistry, as many different configurations have very similar energies [Jørgensen, Schwarz] and these gas-phase configurations are different from the configurations encountered in ions, compounds, and solid metals [Seaborg, Barrett, Wang et al., Gschneidner review on lanthanides above] | ” |
.
- Anyway Sandbh's question seems to be "do these real Madelung anomalies like silver being d9s2 instead of d10s1 cause any significant difference in the chemistry"? I gave sources saying "no they don't". Which is clearly in line with reality, given how similar niobium 4d45s1 and tantalum 5d36s2 act. That's not saying that in principle you couldn't have anomalies that really make a huge difference (such are predicted for the eighth period, making element 168 act like a tin analogue rather than a noble gas according to calculations), it's just saying that the ones in the known elements up to 118 are not among them. Double sharp ( talk) 02:54, 20 July 2020 (UTC)
What is the question(?)
For example, consider chromium. It should be d4s2 but turns out to be d5s1. Does this anomaly make a difference to the chemistry of chromium in terms what was expected v. what is observed?
I see that removing the first and second electrons from vanadium 3d34s2 takes 6.75 and 14.7 eV respectively, whereas to do the same for chromium 3d54s1 requires 6.75 and 16.5 eV. For niobium 4d45s1 the numbers are 6.75 and 14.3 whereas for molybdenum 4d55s1 they are 7.1 and 16.2
So there appears to be a premium, of from 12.2 to 7.7%, for moving past the d5 configuration. I presume this wouldn't be the case for chromium if it was 3d44s2.
To the extent that these energy costs impact the chemistry of the elements involved, it appears the anomalous electron configuration of chromium does make a difference. Does my interpretation seem reasonable? Sandbh ( talk) 01:26, 20 July 2020 (UTC)
- Well I am no physicist, but I can address the chemistry part in your first question:
- you mean, do Cr atoms react in any special way because of this configuration? Response: Do any process involve Cr atoms in the gas phase? (there is
metal vapor synthesis, which might be worth looking into. Those folks are mainly worried about heats of vaporization). And once something (i.e. a ligand) attaches to the Cr atom, the configuration changes to d6s0. So this logic suggests that its a pretty narrow set of circumstances where the d6s0 vs d5s1 issue is consequential, for chemistry. But my dismissive view (and it is only this one editor's view), in no way invalidates the impressive depth of analysis applied by physics community.--
Smokefoot (
talk)
03:31, 20 July 2020 (UTC)
- What Smokefoot says is also exactly what W. H. Eugen Schwarz said in the source I quoted above: "There are only a few special topics in chemistry that require the correct understanding of free atoms in vacuum (e.g., atom-molecular gas-phase reactions)". ^_^ Double sharp ( talk) 04:59, 20 July 2020 (UTC)
Thank you. There are four configurations at play, as I understand it:
| Metal | (ds)6 |
| Gas | d54s1 |
| Ideal | d4s2 |
| Cr3+ | d3 |
Removing three e− from chromium and from molybdenum to get to Cr3+ and Mo3+ takes 7–9% more energy than is the case for removing three e− from their predecesssors, vanadium and niobium. I guess, I don't know, the extra energy cost makes chromium and molybdenum less reactive than is the case if they had idealised d4s2 configurations. Sandbh ( talk) 07:34, 20 July 2020 (UTC)
This
article notes a "Cr atom has a 4s valence orbital that is half empty and can act as an electron acceptor." That was news to me, and something presumably not possible with a d4s2 configuration.
Sandbh (
talk)
07:49, 20 July 2020 (UTC)
- Sandbh's hypotheses may easily be tested. Tungsten really is d4s2 in the gas phase ground state. It is not, as far as I can see, more reactive than molybdenum which is d5s1. Actually Mo and W are more similar to each other than Ag and Au, even though in the second case the configurations match and in the first case they don't. We may multiply even more interesting examples, such as comparing Nb-Ta (no match, very similar anyway) with Sn-Pb (match, but quite different). Case closed.
- Meanwhile he quotes an article focusing on an alloy of chromium and yttrium. In which due to metallic bonding the configuration will not be integer occupancy. Indeed, the article he quotes itself notes that mixing happens between all of Cr 3d 4s 4p, and Y 4d 5s 5p. "Furthermore, our density of state (DOS) results also show strong state mixing between the 4s, 4p, and 3d orbitals of Cr and 4d, 5s, and 5p orbitals of Y, near the Fermi level". That obviously hasn't got anything to do with the gas phase configuration, since 4p and 5p are not even occupied there for chromium and yttrium. Of course it has everything to do with chemical configurations because p occupancy exists typically for low oxidation state TM complexes, e.g. Vaska's complex. But those are not the ground state gas phase configurations that Sandbh attempts to find relevance in despite Smokefoot pointing out to him "Do any process involve Cr atoms in the gas phase?". And despite W. H. Eugen Schwarz saying it. And despite Glenn T. Seaborg saying it. And despite Christian Jørgensen saying it. And despite me and Droog Andrey saying it at Wikipedia talk:WikiProject Elements. I am somewhat saddened, but after almost seven months not surprised, that Sandbh ignores another editor contradicting his idea with sound logic, still ignores the sources I give, and tries to make something out of his idea anyway by taking a source that doesn't even focus on the issue and taking one assertion from it out of context, and not testing his hypothesis when tungsten is right there to test it on.
- Yes, a Cr atom sitting alone by itself indeed has a half filled 4s shell. That is what the article said. Is a chromium atom in a Cr-Y alloy with lots of other chromium and yttrium atoms around in that situation? No. Would one expect it to have the same configuration, knowing that in chemical environments it's easily going to be 3d64s0 instead of 3d54s1? No. So of what relevance is this one assertion he quotes? No mechanism for its relevance is implied and the article's noting that Cr 4p contributes to the bonding obviously shows that being empty in the ground state gas phase is no obstacle for an orbital to contribute to bonding anyway. Same for being full as Y 5s is.
- So this is another case where Sandbh shops around for sources that seem to say what he wants to be true, in this case going to a source that isn't even focusing on the issue, and avoids quoting the parts that contradict his idea. A similar selectivity has been observed already at
WT:ELEM regarding 4f involvement explaining lanthanum's superconductivity. So:
- Is Sandbh not sure of the answer to his question? Then why is it that he cannot believe the editors here and the sources they quote saying the Madelung anomalies don't matter, and keeps guessing for ways where it might matter even when told otherwise?
- Is he sure of the answer? Then why is he asking the question? And is that a relevant approach for WP when the sources say something else? Double sharp ( talk) 08:41, 20 July 2020 (UTC)
Enough already!
First I'll give my view on Sandbh's question. The configuration of the electrons in an atom is an intrinsic property. So are its ionisation potential and its electronegativity. However, most properties chemists, and especially non-chemists, are interested in are emergent properties. It is meaningless to ask "what is the melting point of an atom of silver?" Melting points are emergent properties of assemblages of atoms and I would predict that small sets of atoms would have lower melting or boiling points than bulk material: entropy winning out over whatever binding enthalpy is holding the "solid" or "liquid" cluster together. How large the cluster needs to be before the bulk value sets in, I have no idea!
Another way of expressing my view is that intrinsic properties can now be calculated pretty well for chemical substances, knowing their proposed atom connectivity/bonding, even if no-one has ever made a sample. However, you have to wait for a real sample to get an accurate value for an emergent property like a melting point. Personally I'm biased towards organic compounds because that's where my expertise lies and that's also where the concept of having a target (as-yet-unmade) structure is on a pretty firm footing whereas I presume that it can be more difficult to be sure that a new inorganic structure will actually exist in the form one imagines. Who would have guessed that carbon's humble s2p2 would in many compounds turn out to become a tetrahedral sp3 while in other compounds you would get a different linear combination of molecular orbitals including aromatic ones: so that even as an element diamond and graphite don't much resemble one another and are difficult to interconvert? Other emergent allotropes exist and even in 2020 new ones are still being touted.....
I think that the early pioneers who created various ways of laying out Periodic tables emphasised properties like ionisation potential and electronegativity which were more likely to "match the expected regular periodicity" (to quote the Spanish professor) just because they are indeed intrinsic, rather than to expect periodicity in emergent properties like (to take an extreme case) utility as a drug.
OK, so what about your dispute about group 3? I read the exchange at WT:ELEM and although I found parts informative, it was mostly tedious, repetitive and above all uncivil. I couldn't face the archive and I wondered why the whole thing wasn't on the talk page for Periodic table since that's the article that's relevant. I had a brief look on that talk page and goodness me you're all over that one as well! My analysis is that you both think that you are discussing the periodic table whereas in fact you are each discussing a periodic table: unfortunately not the same one! Your only focus should be "how can we improve the periodic table article?" (on WIkipedia there is only one). The RFC process is inappropriate because reliable external sources discuss and use both forms of the bit you are arguing about. There is no such thing as "the" periodic table, whatever IUPAC rules but there are certainly a number of interesting and useful alternative ways of laying out tables for various purposes. Wikipedia's article should reflect the many ways the table has historically appeared (as indeed it already does) and mention the Lu / La discussion. Can't you each find your best couple of sources that favour Lu or La — preferably not written by yourselves — and add a paragraph to the main article that encapsulates the debate while maintaining WP:NPOV? Michael D. Turnbull ( talk) 10:59, 20 July 2020 (UTC)
- @
Michael D. Turnbull: I do agree that we have to discuss the issue neutrally in the
periodic table article. I support doing that. The only trouble is that there are lots and lots of places where we use a periodic table as a graphic. Just look at, say, the article on
neon: we have a little graphic at the top showing where neon is in the periodic table, we have a footer at the bottom showing it again, both are used for navigation. Don't we have to choose a default for that situation to avoid showing it twice over? If you have a solution that might break that impasse, please, do let me know.
Double sharp (
talk)
12:10, 20 July 2020 (UTC)
- @
Double sharp: Yes, that's a knotty problem, which is why it's interesting but probably not insurmountable! All the articles for the elements are important and the infoboxes are vital. However, whoever has chosen (on Wikipedia) to put He over Ne in that graphic offends all those who think it should be He over Be. However, when looking at the actual article for
helium, there is a vital note pointing out in a proper
WP:NPOV way that some authors prefer the latter version of the table. So, in general, you could change the graphic so it didn't attempt to convey quite so much information on "remote" elements to the one in question and leave discussions about group 3, for example, to the individual elements where it is important.
Michael D. Turnbull (
talk)
13:19, 20 July 2020 (UTC)
- @ Michael D. Turnbull: Actually I wrote that note on helium. ;) I admit I haven't thought of a good way to "blur" the problem. The WebElements idea may be a reasonable compromise by simply not making it clear from any gap where the f block is supposed to fit in, so we can leave the footnotes to the elements they are about. But maybe that is also a downside? At least it seems promising until we get to the footer with the element symbols explicitly marked. I will think about it. ^_^ Double sharp ( talk) 13:44, 20 July 2020 (UTC)
- @
Double sharp: Yes, that's a knotty problem, which is why it's interesting but probably not insurmountable! All the articles for the elements are important and the infoboxes are vital. However, whoever has chosen (on Wikipedia) to put He over Ne in that graphic offends all those who think it should be He over Be. However, when looking at the actual article for
helium, there is a vital note pointing out in a proper
WP:NPOV way that some authors prefer the latter version of the table. So, in general, you could change the graphic so it didn't attempt to convey quite so much information on "remote" elements to the one in question and leave discussions about group 3, for example, to the individual elements where it is important.
Michael D. Turnbull (
talk)
13:19, 20 July 2020 (UTC)
Potential barrier... to understanding
Aside: This is a problem where someone linked a term as known to them, within the topic they were familiar with, without checking whether the linked article *actually* had anything to do with _their_ understanding of the term.
So for 13 years the article
Activation energy has had in its second paragraph a link
potential barrier. Now I know what that means in chemistry, and you know what that means in chemistry, but that linked article begins
- In quantum mechanics, the rectangular (or, at times, square) potential barrier is a standard one-dimensional problem that demonstrates the phenomena of . . . .
If there is a article here at WP that does describe the chemical potential barrier, the link should be updated to point to that. If not, then the link needs to be unlinked. As it is now, the link merely confuses.
Separately, while finding the timeframe of the optimistic link, I noticed the
diagram seen here (
and found here) looks so much more understandable to the average reader than the
current diagram which presentation deters any but the already familiar.
It seems a foot-gun to make an article so technically irreproachable that it is also *unapproachable*. That really *is* a potential barrier. What was wrong with having an overview, such as seen back in 2007? Do y'all want people to hate chemistry? Shenme ( talk) 04:21, 1 August 2020 (UTC)
- Yes it's a poor article in a number of ways and is currently rated start-class. Although there is an article on
Chemical potential, I'm not sure that is the appropriate link either and it is a very technical article. In another example, the lede also links to
division in "a chemical reaction, or division" which is also off-topic and uses a citation that, to me, seems unrelated. Go ahead and make alterations to the article you feel would improve it. The article's talk page is the place to discuss any material you are still unsure about.
Michael D. Turnbull (
talk)
11:27, 1 August 2020 (UTC)
- My educated guess (only that) is that activation energy is a common term in chemical kinetics/mechanism but far less commonly invoked in nuclear reactions and materials science. That guess is supported by the nature of the subsidiary sections, which are highly chemical. With that idea in mind, I refocused the lede toward a chemical context.-- Smokefoot ( talk) 13:51, 1 August 2020 (UTC)
Silylene has a misleading scheme
In the Wiki article for Silylene ( /info/en/?search=Silylene) the scheme has a bond between the Si-Atom and the Electron pair. This is misleading as the Electrons are located at the Si-atom and it could imply a Carbene with a Hydrogen and a Silane substituent.
- (Above text unsigned)
 Done Thanks, anonymous. I've fixed this.
Michael D. Turnbull (
talk)
15:37, 28 July 2020 (UTC)
Done Thanks, anonymous. I've fixed this.
Michael D. Turnbull (
talk)
15:37, 28 July 2020 (UTC)
- Thanks,
User:Michael D. Turnbull. I see you (among other things) corrected the original PNG image on commons. Please update the metadata on that to indicate the author/date (and license, if necessary) for this latest upload at that filename.
DMacks (
talk)
14:08, 1 August 2020 (UTC)
-
I've gone in and used
{{ PD-chem}}, which I assume is OK. Please let me know if I haven't done that right Michael D. Turnbull ( talk) 15:16, 1 August 2020 (UTC)- Looks good. Thanks! DMacks ( talk) 17:25, 1 August 2020 (UTC)
-
I've gone in and used
- Thanks,
User:Michael D. Turnbull. I see you (among other things) corrected the original PNG image on commons. Please update the metadata on that to indicate the author/date (and license, if necessary) for this latest upload at that filename.
DMacks (
talk)
14:08, 1 August 2020 (UTC)
Help to assess Draft:Potassium octacyanomolybdate(IV) please
Please will an experienced person examine and assess this draft. If you are not an AFC reviewer please ping me with your assessment and I will review accordingly, otherwise please review and accept or decline according to the draft's merits. Fiddle Faddle 07:06, 4 August 2020 (UTC)
- I am a retired academic chemist, but this article's subject is not in my area of expertise. However, it looks good enough to me and I suggest it be moved into main space where many more people will look at it and improve it. --
Bduke (
talk)
07:53, 4 August 2020 (UTC)
- @
Timtrent: What is really going on here is that the author of this article is promoting his/her recent paper and then is going set about ref spamming. --
Smokefoot (
talk)
12:22, 4 August 2020 (UTC)
- I have accepted, but can you replace their ref?
Graeme Bartlett (
talk)
12:31, 4 August 2020 (UTC)
- Thank you all Fiddle Faddle 12:49, 4 August 2020 (UTC)
- I have accepted, but can you replace their ref?
Graeme Bartlett (
talk)
12:31, 4 August 2020 (UTC)
- @
Timtrent: What is really going on here is that the author of this article is promoting his/her recent paper and then is going set about ref spamming. --
Smokefoot (
talk)
12:22, 4 August 2020 (UTC)
Dimethyl selenosulfenate
This paper mentions that dimethyl selenosulfenate is the most abundant selenium containing species in coffee. What is the formula of dimethyl selenosulfenate? The authors refer to it as MeSeSMe, which I thought would be easy to unpack, but that seems not so, at least for me. Thank you, Sandbh ( talk) 06:08, 18 August 2020 (UTC)
- That would be CH3-Se-S-CH3. Canonical SMILES=CS[Se]C Though the name "selenosulfenate" seems to very little used. Even "sulfenate" is hardly used. "Dimethyl thioselenide" is another name for this, as is 2-Thia-3-selenabutane. It's also found in onions.
[9]
Graeme Bartlett (
talk)
06:45, 18 August 2020 (UTC)
- Only occurs during roasting though... I can't find a paper explaining that, although I'm guessing a process starting with oxidation and subsequent selenoxide elimination of selenocysteine might get you started. -- Project Osprey ( talk) 09:39, 19 August 2020 (UTC)
Notability of compounds
This might be better for WP:MEDICINE, but: is there a general standard besides GNG for the notability of compounds, e.g., diphenyl-2-pyridylmethane, which I just found doing CAT:NN cleanup? Articles like this seem useful but I doubt that the majority of them have WP:SIGCOV. AleatoryPonderings ( talk) 14:25, 24 August 2020 (UTC)
- I would be interested in knowing how our community answers questions like this. On the specific compound, there are several refs in Pubchem, including the fact that there is an EU REACH record, implying that it is manufactured at reasonable scale (enough that some supplier is prepared to pay for the registration). So for this specific compound I think that the problem is that WP hasn't expanded the article, rather than that there wouldn't be WP:SIGCOV if we looked for it. Michael D. Turnbull ( talk) 14:35, 24 August 2020 (UTC)
- A quick search reveals more than on paper on the topic, and that the chemical is listed for sale. So I have remove the notability in question tag. A PubChem or ChemSpider listing does nothing to prove notability. But the references they link to might.
Graeme Bartlett (
talk)
21:45, 24 August 2020 (UTC)
- I was interested by a couple of references I came across for diphenyl-2-pyridylmethane, so I'm going to take a stab at expanding the article over the next couple of days. I still don't know how we tackle the wider question, although I know that Graeme keeps a lists of chemicals he feels have merit as AfC. Michael D. Turnbull ( talk) 10:57, 25 August 2020 (UTC)
 Done So that article now looks a bit better. @
AleatoryPonderings: did you come across any others which could be worth expanding now?
Michael D. Turnbull (
talk)
15:34, 25 August 2020 (UTC)
Done So that article now looks a bit better. @
AleatoryPonderings: did you come across any others which could be worth expanding now?
Michael D. Turnbull (
talk)
15:34, 25 August 2020 (UTC)
- @ Michael D. Turnbull: I've just been going through CAT:NN at random to find articles to clean up or nominate for deletion as appropriate, so I haven't encountered any others specifically. But Category:Organic compound stubs seems to have quite a few potential compound articles for expansion. I suppose I was also just wondering in general if we have any WP:SNGs for compounds, as it's clearly a specialist topic to which ordinary guidelines plausibly don't apply. AleatoryPonderings ( talk) 15:45, 25 August 2020 (UTC)
Hello chemists! I bid y'all a warm hello. I came here today to inform thee of two Wikipedia sites that might be of interest to y'alls'. I've edited both of these pages, but since i am neither a chemist nor a college/graduate student, I came here to ask for your help. The former needs fixing from a chemist, whereas the latter doesn't have a single reference, until i put some. Additionally, the computations in the latter prove too much also for my teeny brain. Please abet me in fixing these pages, fellow wikipedians! Thank you y'all for reading this tedious query. Have a nice day! Wiswesser's rule Ionization energy Ice bear johny ( talk) 09:46, 10 September 2020 (UTC)
- Not a chemist, but I cannot find any Google Scholar or Google Books hits for Wiswesser's rule meaning the particular formulation of Klechkovsky's rule that it is.
Double sharp (
talk)
09:53, 10 September 2020 (UTC)
- I an a chemist and Wisesser's rule and Klechkovsky's rule are both just different names for the concept explained at
Aufbau_principle#Madelung_energy_ordering_rule. The Wiswesser rule page should arguably just be a redirect to the main
Aufbau principle, unless anyone can find references that suggest otherwise. Incidentally,
William Wiswesser is much better known for his notation used to encode chemistry in early databases — a method still occasionally used today.
Ionization energy looks like a more technical article that needs extra citations and some simpler language. I hope someone more familiar with the details than I am will volunteer to improve it.
Michael D. Turnbull (
talk)
11:47, 10 September 2020 (UTC)
- Thank you, that confirms my suspicion. As I mentioned above I cannot find any references to the Aufbau principle aka Klechkovsky rule being referred to under this name, but maybe there are some. I've redirected the
Wiswesser's rule page to
Aufbau principle as you suggested.
Double sharp (
talk)
12:10, 10 September 2020 (UTC)
- Fine. Seconds before you did the redirect I added a section on the Talk page of the old article that this should be done, so there is a clear record there of our thinking. I think that the reference to his 1945 paper should be added in the history section near Aufbau_principle#Madelung_energy_ordering_rule (I've checked that it is still available) and I'll do that soon. Michael D. Turnbull ( talk) 12:18, 10 September 2020 (UTC)
- Thank you, that confirms my suspicion. As I mentioned above I cannot find any references to the Aufbau principle aka Klechkovsky rule being referred to under this name, but maybe there are some. I've redirected the
Wiswesser's rule page to
Aufbau principle as you suggested.
Double sharp (
talk)
12:10, 10 September 2020 (UTC)
- I an a chemist and Wisesser's rule and Klechkovsky's rule are both just different names for the concept explained at
Aufbau_principle#Madelung_energy_ordering_rule. The Wiswesser rule page should arguably just be a redirect to the main
Aufbau principle, unless anyone can find references that suggest otherwise. Incidentally,
William Wiswesser is much better known for his notation used to encode chemistry in early databases — a method still occasionally used today.
Ionization energy looks like a more technical article that needs extra citations and some simpler language. I hope someone more familiar with the details than I am will volunteer to improve it.
Michael D. Turnbull (
talk)
11:47, 10 September 2020 (UTC)
Oh wow y'all are quick to reply! Thanks guys for the trouble :>>>. Ice bear johny ( talk) 13:12, 10 September 2020 (UTC)
But wait, the formula in Wiswesser's rule supports the Aufbau principle? Would you include it in Aufbau principle, or not? Is it inconsequential? I added it in my notes, and thought that it would help. Thank you for answering! Ice bear johny ( talk) 13:15, 10 September 2020 (UTC)
- His rule gives the same predictions for the filling of the orbitals as does the Aufbau principle, as was mentioned on the page in question, which is still on WP if you know where to look "namely, here".. As I said, I'll add his paper as a citation in the article on the Aufbau principle and I'll also add it to his biography page, with appropriate detail once I've figured out whether it was really a novel idea in 1945 or just an alternative way of expressing what was already known at the time. Interestingly, his publication does pre-date Klechkovsky's (1960s) but not Madelung's (1930s), although the sources seem to say that Madelung expressed the "rule" empirically, not as in the formulae Wiswesser used. I'll have to read his article carefully to see whether he had a theoretical justification for the rule, which is what Klechkovsky provided, I think. Michael D. Turnbull ( talk) 13:48, 10 September 2020 (UTC)
Oh ok. You adding his paper assuages me. Thank you for helping me :>. Your help is greatly appreciated! Ice bear johny ( talk) 16:18, 10 September 2020 (UTC)
More edits
Hello! I would like to request your abetment. You see I am neither a chemist, nor a college student; ergo, i cannot ascertain that the information i input is 100% correct. Hence i beseech your help in the following pages: Ionization Energies, Effective nuclear charge, Core charge and Shielding effect. All of them are under referenced, and other editors that I know are particularly disinterested in editing because of the sheer hard work involved. Thanks y'all! Ice bear johny ( talk) 06:44, 11 September 2020 (UTC)
Bruh the shielding effect-. Guys please edit that part. Literally, i cannot describe it's fathomable, facile information! Ice bear johny ( talk) 12:38, 11 September 2020 (UTC)
Sensitivity of azidoazide azide
The 1-Diazidocarbamoyl-5-azidotetrazole article cites peer-reviewed sources which describe the compound as extremely sensitive, detonating at the slightest stimulus or for no apparent reason, but it also mentions hobbyists such as I make C2N14 in my shed who synthesized it themselves and found it far less sensitive. Should we include these amateur anecdotes? There's been some off-and-on discussion on the talk page, but it's inconclusive and would benefit from more input. – dlthewave ☎ 23:50, 19 September 2020 (UTC)
- Well, assuming of course that you've actually made the right stuff in your shed, rather than something else which explodes somewhat less readily... then polymorphs perhaps? --
Project Osprey (
talk)
00:15, 20 September 2020 (UTC)
- My chemistry is too rusty to speculate on that, but from a Wikipedia policy perspective we would need secondary-source coverage to A) establish
WP:DUE WEIGHT and B) analyze/explain the disparity. –
dlthewave
☎
03:13, 20 September 2020 (UTC)
- Well you'd need to get the results published in a reputable journal first. Youtube is obviously not a peer-reviewed source. -- Project Osprey ( talk) 09:29, 20 September 2020 (UTC)
- My chemistry is too rusty to speculate on that, but from a Wikipedia policy perspective we would need secondary-source coverage to A) establish
WP:DUE WEIGHT and B) analyze/explain the disparity. –
dlthewave
☎
03:13, 20 September 2020 (UTC)
The location and composition of Group 3 of the periodic table
Here (open access). About nine months in the making; I only signed off on the (eighth) proof, last night. Sandbh ( talk) 12:52, 24 September 2020 (UTC)
- Who coined the term "lanthanoid"? wikt:lanthanoid and wikt:lanthanide could do with etymology. Graeme Bartlett ( talk) 13:16, 24 September 2020 (UTC)
Deprecating usage of the template {{radic}} to write root radicals has an RFC for possible consensus.

"Deprecating usage of the template {{radic}} to write root radicals" has an RfC for possible consensus. I you would like to participate in the discussion, you are invited to add your comments on the discussion page. Thank you. Walwal20 talk ▾ contribs 02:12, 25 September 2020 (UTC)
hi all, just a heads up, Jean-Pierre Abbat who was a chemist(?) is up for deletion, afd is here. Coolabahapple ( talk) 04:01, 25 September 2020 (UTC)
PT in lead of the PT article
Now updated:
Periodic table
---
Sandbh (
talk)
07:27, 25 September 2020 (UTC)
R1 Plasmid
How does the R1 Plasmid impact multi-drug antibiotic resistance in its host cell? Vbam25 ( talk) 22:40, 30 September 2020 (UTC)
How does the R1 Plasmid impact multi-drug antibiotic resistance in its host cell?
9/30/2020 Vbam25 ( talk) 22:42, 30 September 2020 (UTC)
- This sounds more like a microbiology question than a chemistry one. Although it will be reflected in some kind of chemical activity or lack of it.
Graeme Bartlett (
talk)
08:43, 1 October 2020 (UTC)
- @ Vbam25: I'm afraid I've can't give a good answer to your question. There is a relevant article at R-factor but if that or its references don't help, try posting a specific question again at WP:RD/S. Michael D. Turnbull ( talk) 11:11, 1 October 2020 (UTC)
A quick ping here too: Validation of CAS numbers; collaboration with Wikidata? -- Egon Willighagen ( talk) 08:15, 11 October 2020 (UTC)
After almost two years of waiting, there still exists no reference in this article proving that "Lasri condensation" is an actual thing (I'm not talking about the chemical reaction, but the term itself). On superficial literature search, I found no paper using the term "Lasri condensation". I have the strong feeling that somebody from Lasri's group is trying to establish this term using Wikipedia. The users contributing to the article and uploading figures are named after former colleagues and PIs of Lasri. The second figure in the article is taken from Lasri's 2018 publication. Lasri himself is refering to the Wikipedia article on his website to suggest that the "Lasri condensation" exists. I suggest renaming this article to something without "Lasri" in it. What does everybody else think? -- Hbf878 ( talk) 12:56, 13 October 2020 (UTC)
- Unless we have
WP:SECONDARY source for the name, I strongly oppose using that name for this reaction anywhere in the article, let alone in the title. And for the actual title, we'd further need evidence that it's
WP:COMMONNAME rather than COI touting their own work. Is there any secondary ref at all? Otherwise, I suspect it might not even merit an article at all (
WP:GNG fail).
DMacks (
talk)
13:09, 13 October 2020 (UTC)
- No secondary sources supporting the term "Lasri condensation", and I cannot find the term anywhere except for pages that are in direct connection with Lasri. What's the common procedure in these cases? Nominate for deletion? --
Hbf878 (
talk)
13:23, 13 October 2020 (UTC)
- Do you think this specific reaction, as defined by whatever specific details, is actually independently notable (regardless of what the article about it would be titled)?
DMacks (
talk)
14:04, 13 October 2020 (UTC)
- Honestly, I don't really know. It doesn't seem to be revolutionary, as the articles in the references are not cited very much (2x since publication in 2018). Maybe there's someone here who can assess whether this reaction is notable. Btw, there have been
discussions about this article and Lasri previously. --
Hbf878 (
talk)
15:43, 13 October 2020 (UTC)
- Hell's teeth! It is just a special case of a standard pyrazole formation reaction (see first diagram in that article). I'll AFD it immediately. Please add support as soon as I do that. Michael D. Turnbull ( talk) 17:00, 13 October 2020 (UTC)
- I've put the necessary template on the page and notified the user who created the page. Michael D. Turnbull ( talk) 17:10, 13 October 2020 (UTC)
- Honestly, I don't really know. It doesn't seem to be revolutionary, as the articles in the references are not cited very much (2x since publication in 2018). Maybe there's someone here who can assess whether this reaction is notable. Btw, there have been
discussions about this article and Lasri previously. --
Hbf878 (
talk)
15:43, 13 October 2020 (UTC)
- Do you think this specific reaction, as defined by whatever specific details, is actually independently notable (regardless of what the article about it would be titled)?
DMacks (
talk)
14:04, 13 October 2020 (UTC)
- No secondary sources supporting the term "Lasri condensation", and I cannot find the term anywhere except for pages that are in direct connection with Lasri. What's the common procedure in these cases? Nominate for deletion? --
Hbf878 (
talk)
13:23, 13 October 2020 (UTC)
Can we add new chemistry related wordings and phenomenons?
While reading we come across so many new wordings and phenomena, which was not in wikipedia. How to add this to the page with proper references? Not only chemistry but also every new thing we found? Preethanuj Preethalayam ( talk) 07:15, 18 October 2020 (UTC)
- Here are some perspectives with your well-intentioned proposal:
- Of course new info can be added but it should be done in context. If the topic has 2000 hits in SciFinder and is published in a primary journal, I would often challenge the addition. A new review WP:SECONDARY or a new textbook/monograph WP:TERTIARY are among the best edits one could possibly make.
- Often, editors who want to "help" Wikipedia by adding new references, are engaged in COI. WP:COI. Many newish editors are looking for a way to shoehorn references to themselves, their mentors, or their pals.
- As an encyclopedia, Wikipedia is a compendium of digested, mainstream knowledge. Wikipedia-Chem does not aspire to account for the newest results, ordinarily. WP:NOTNEWS, WP:RECENTISM. Lots of blogs exist if one has the urge to report on new stuff.
- My two cents, --
Smokefoot (
talk)
14:15, 18 October 2020 (UTC)
- To add to what Smokefoot just said. You can see an example of this as Lasri condensation. Take a look and use your chemical judgement + knowledge of WP to decide whether or not you think it is a good article. If you answer "yes", then I don't think you are ready to create one yourself. For the meantime stick to improving existing articles. If you answer "no" then well done you may well be the sort of editor we want here — the more the better. Check out the part of this page just above at WT:WikiProject_Chemistry#Lasri_condensation, which shows that we sometimes improve WP by chucking stuff out, not always by including more of the same. Again, Welcome! But start slowly, by editing existing chemistry. Mike Turnbull ( talk)
- On the more inclusive side, you could propose your ideas for new articles here, and people will respond. In some cases if there is new wording, but nothing else new past what is in the article a redirect may be appropriate. Graeme Bartlett ( talk) 09:18, 21 October 2020 (UTC)
Protic solvent, etc.
I just switched polar solvent to redirect to protic solvent. Protic solvent has sections on polar aprotic solvents and polar protic solvents (slightly redundant?). Would someone else look at protic solvent and make sure that these important links are ok? Do we need to create a self-standing polar solvent? -- Smokefoot ( talk) 18:48, 23 October 2020 (UTC)
- That seems confusing to me...protic is subtype of polar (as you note, polar-protic is semi-redundant) and there's lots to say about polar in general, rather than just as a subtopic of protic.
DMacks (
talk)
18:54, 23 October 2020 (UTC)
- I was scrambling since prior to my action "polar solvent" was the title, but the lead was all about protic. One alternative solution would be to revert my work and rewrite the lede in the context of polar vs non polar solvents.--
Smokefoot (
talk)
19:12, 23 October 2020 (UTC)
- Definitely no harm in fixing one mess, even if it can be fixed further or differently later.
DMacks (
talk)
10:18, 24 October 2020 (UTC)
- As it stands today the redirect on
polar solvent is very misleading as it leads to an article on protic solvents. So while DMF is certainly polar, it ain't protic. The actual article where the redirect leads has a table where all possibilities: non-polar, polar aprotic and polar protic are discussed, so I think that it is the article title, not the content, that's the problem. Looking at the article
solvent, much of the same material is covered in arguably a more general way. So one possibility would be to scrap
protic solvent altogether and make all these terms just rediredts to
solvent.
Mike Turnbull (
talk)
13:37, 24 October 2020 (UTC)
- Thank you for the pondering. Following MDT's thinking, one idea is to create
Reaction solvent, which might captures the idea that would accommodate aprotic, protic, polar solvent.--
Smokefoot (
talk)
14:43, 24 October 2020 (UTC)
- Trouble is that virtually all solvents mentioned in the
solvent article can be used as reaction solvents. A while ago, I added the reference
[10] to that article: this is a tool for solvent selection developed by Astra-Zeneca. So I'm not convinced that a separate article is needed unless one of the subsections at
solvent gets so unbalanced in size that it should have a {{Main|Sub-article}} tag, which is not yet the case.
Mike Turnbull (
talk)
15:02, 24 October 2020 (UTC)
- Ok good advice. Lets just follow what the title says, the article will be about protic solvents. Period. Polar solvent will redirect to solvent. Seems straightforward.-- Smokefoot ( talk) 16:00, 24 October 2020 (UTC)
- Trouble is that virtually all solvents mentioned in the
solvent article can be used as reaction solvents. A while ago, I added the reference
[10] to that article: this is a tool for solvent selection developed by Astra-Zeneca. So I'm not convinced that a separate article is needed unless one of the subsections at
solvent gets so unbalanced in size that it should have a {{Main|Sub-article}} tag, which is not yet the case.
Mike Turnbull (
talk)
15:02, 24 October 2020 (UTC)
- Thank you for the pondering. Following MDT's thinking, one idea is to create
Reaction solvent, which might captures the idea that would accommodate aprotic, protic, polar solvent.--
Smokefoot (
talk)
14:43, 24 October 2020 (UTC)
- As it stands today the redirect on
polar solvent is very misleading as it leads to an article on protic solvents. So while DMF is certainly polar, it ain't protic. The actual article where the redirect leads has a table where all possibilities: non-polar, polar aprotic and polar protic are discussed, so I think that it is the article title, not the content, that's the problem. Looking at the article
solvent, much of the same material is covered in arguably a more general way. So one possibility would be to scrap
protic solvent altogether and make all these terms just rediredts to
solvent.
Mike Turnbull (
talk)
13:37, 24 October 2020 (UTC)
- Definitely no harm in fixing one mess, even if it can be fixed further or differently later.
DMacks (
talk)
10:18, 24 October 2020 (UTC)
- I was scrambling since prior to my action "polar solvent" was the title, but the lead was all about protic. One alternative solution would be to revert my work and rewrite the lede in the context of polar vs non polar solvents.--
Smokefoot (
talk)
19:12, 23 October 2020 (UTC)
In the right hand figure “H2O” should read “H2O”. In addition, it overlaps with an arrow. Anyone volunteering for fixing or re-drawing? Please note that there is a version with a higher resolution in the file history. -- Leyo 17:29, 27 October 2020 (UTC)
- Both the files are .jpg and the fix, if done at all, should be to give an .svg version. I'm not very good with .svg editing yet but I'll give it a try. If anyone else fancies doing this, please go ahead as well
Mike Turnbull (
talk)
17:50, 27 October 2020 (UTC)
- OK,
Leyo, I've
 Done the new file, with some additional tweaks to satisfy my chemical instincts. Please check out the result at
Hexosaminidase#Mechanism of action and confirm you think it is now fine. Once you've done that, I'll tag the previous .jpg with a note to say there is an .svg replacement.
Mike Turnbull (
talk)
13:41, 28 October 2020 (UTC)
Done the new file, with some additional tweaks to satisfy my chemical instincts. Please check out the result at
Hexosaminidase#Mechanism of action and confirm you think it is now fine. Once you've done that, I'll tag the previous .jpg with a note to say there is an .svg replacement.
Mike Turnbull (
talk)
13:41, 28 October 2020 (UTC)
- Convenience links:
- * original: File:Mechanism.jpg
- * replacement: File:Hydrolysis mechanism.svg
- Should the "loss of alcohol" detail (curved reaction-arrow pointint to ROH) actually be associated with the first step rather than the second? The "addition of water" curved-arrow might also want to be moved there...that's the point in the process where the ROH disappears from the image and HOH appears instead. This image is already superior to the original, just some details that can be improved even further now that we have lots of eyes and hands on it.
DMacks (
talk)
13:58, 28 October 2020 (UTC)
- Yes, you are correct that the ROH leaves in the first step, so I'll move that portion. The water doesn't arrive until the second step, so I think that part is OK. I'll upload a new version in a while and see what you think.
Mike Turnbull (
talk)
14:20, 28 October 2020 (UTC)
- OK, that seems better. Any further comments?
Mike Turnbull (
talk)
14:34, 28 October 2020 (UTC)
- Thank you. I looks good now. However, please note that I am not an expert in reaction mechanisms. -- Leyo 23:56, 28 October 2020 (UTC)
- I'm always torn how to handle reagents on arrows in a mechanistic diagram. In this case, it's true that the ROH is a reactant in the second step. But in the graphics of the mechanism, it has become present during the first arrow. That is, the true process is "1) H2O is formed; 2) H2O goes away; 3) ROH positions; 4) ROH reacts". Visually [1] is happening in the first mechanism and [1,2,3] happen in order to get from first diagram to second diagram and only [4] is happening to get from second to third. But the way the reaction-arrows are written it's [1,2] as the first vs [3,4] second.
DMacks (
talk)
14:40, 29 October 2020 (UTC)
-
DMacks I'd say it's fine as it is. ROH is not "a reactant in the second step": it has been expelled in the first step — R is in fact the large GM3 ganglioside, as is clear from the other diagram alongside this one in the article. The second step is the hydrolysis of the oxazolinium to release N-acetyl galactose and regenerate the enzyme. The water is shown twice in the diagram: once over the second arrow and also in position under the upper carboxylate ready for its attack on the carbon that will become the anomeric carbon of the N-acetyl galactose.
Mike Turnbull (
talk)
15:00, 29 October 2020 (UTC)
- Whoops, yeah, my explanation switched HOH and ROH. Need more coffee, less fume-hood air. DMacks ( talk) 15:02, 29 October 2020 (UTC)
-
DMacks I'd say it's fine as it is. ROH is not "a reactant in the second step": it has been expelled in the first step — R is in fact the large GM3 ganglioside, as is clear from the other diagram alongside this one in the article. The second step is the hydrolysis of the oxazolinium to release N-acetyl galactose and regenerate the enzyme. The water is shown twice in the diagram: once over the second arrow and also in position under the upper carboxylate ready for its attack on the carbon that will become the anomeric carbon of the N-acetyl galactose.
Mike Turnbull (
talk)
15:00, 29 October 2020 (UTC)
- OK, that seems better. Any further comments?
Mike Turnbull (
talk)
14:34, 28 October 2020 (UTC)
- Yes, you are correct that the ROH leaves in the first step, so I'll move that portion. The water doesn't arrive until the second step, so I think that part is OK. I'll upload a new version in a while and see what you think.
Mike Turnbull (
talk)
14:20, 28 October 2020 (UTC)
- OK,
Leyo, I've
Updating the template about nitrides
Good day! I really liked the navbox Template:Phosphides that ties together articles about phosphides in parallel to Template:Phosphorus compounds. Since it was missing, I made a similar template about arsenides ( Template:Arsenides) which also works in parallel to Template:Arsenic compounds. However, when I tried to update the template about nitrides Template:Nitrides (to make it look like this draft) I was not sure how to proceed. Updating the current version of the nitrides template to make it look like the phosphide and arsenide templates may change it significantly and I don't know whether it will disrupt any of the 300+ pages that link to the template. First, I don't know whether I should update the template. Is it used in some articles? I didn't find it on articles it links to. Second, I don't know how to check whether the update would disrupt the pages linked to it. I would greatly appreciate advice on the matter. -- ElMagyar ( talk) 21:16, 1 November 2020 (UTC)
- I think it is a good idea to add that extra table with binary, tertiary etc. However the periodic table in the draft, and in the phosphides template is very ugly, being squashed in the middle of the screen. So if you can use the style for the periodic table in your draft and paste it as the next version in the nitrides template. It should not be disruptive if it works! Graeme Bartlett ( talk) 11:54, 2 November 2020 (UTC)
Selenenic acid
Changes in article names based on one's ideas of proper or correct nomenclature should be discussed. Such as discussion has been underway on the carboxylic acids. Chemists often call R-S-R thioethers, still. R-Se-O-H are called selenenic acids. Maybe some committee decided that they should be called SeO-Selenoperoxols. Wikipedia is respectful of such committees but we are not necessarily obedient to them. In the end, changes are made by consensus.-- Smokefoot ( talk) 13:28, 25 October 2020 (UTC)
- And
user:Daniblanco renamed the self descriptive
Thioselenide to
Selenothioperoxide, In my opinion having peroxide in the name is quite misleading as there is no oxygen. However when I look, either name is very seldom used.
Graeme Bartlett (
talk)
20:43, 25 October 2020 (UTC)
- I invited user:Daniblanco to engage the community here.-- Smokefoot ( talk) 13:02, 26 October 2020 (UTC)
- Sulfhydryl leads to sulfalkyl or sulfaryl, then to sulfalkylselenide or sulfarylselenide for the --Se--S--R part. If invention is allowed, an alkyl (or aryl) sulfalkylselenide or (sulfarylselenide) is better. Or alkylsulfalkylselene -- Ktsquare (talk) 04:00, 2 November 2020 (UTC)
- additional question. Is there a difference between the thia and thio prefix? It seems they are mixed in use: Thiopyrylium, thiabendazole, thiourea, thiosulfate -- Ktsquare (talk) 04:00, 2 November 2020 (UTC)
- From a discussion above, Me-Se-S- Me is called dimethyl selenosulfenate, no? -- Ktsquare (talk) 21:53, 4 November 2020 (UTC)
Newbie question: if I find an interesting new route synthesizing a chemical using wikipedia links, how shall I proceed?
Per title, what shall I do? -- Ktsquare (talk) 02:40, 2 November 2020 (UTC)
- Ktsquare There is an "if" in your question which makes it difficult to answer. Please could you give a specific example of an article containing something you find interesting and a suggestion of what it is you want to do. Mike Turnbull ( talk) 10:51, 2 November 2020 (UTC)
- If you look at how the synthesis of adamantane progresses, the breakthrough happens at using dicyclopentadiene, instead of methanal and diethyl malonate condensation. In simple terms, adding hydrogen to a C10H12 and promoting rearrangement make the end product, instead of removing the "branches" from the adamantane skeleton. Can a synthesis just make the parent structure directly in few steps from available compounds? For example only, Wikipedia indicates lignin presents a potential of renewable source of aromatic compounds. Monolignol says paracoumaryl alcohol is found almost exclusively in grasses. Ethenolysis splits an carbon carbon double bond. Cracking_(chemistry) says it breaks down complex hydrocarbons into lighter ones.
Now
- Extract P-alcohol from high purity lignin that originally comes from grass or after paper making.
- Ethenolysis splits P-alcohol to allyl alcohol and para vinyl phenol.
- Cracking splits vinyl phenol to phenol and acetylene.
If possible, all the inorganic salts, carboxylic acid and carboxylate can be skipped altogether. Also, if the cracking in the last step is economical and can be done in large scale, recycling polystyrene can be possible because paravinyl phenol monomer is structurally similar to styrene. In other words, Cracking_(chemistry) make benzene and acetylene from polysytrene waste. hydrogenate some of the acetylene product to ethene to support the ethenolysis in previous step. I know already someone is going to say these are all fantasy from the very beginning... :) --- Ktsquare (talk)
- Do you have suitable references to show this works and has been done, or is it your own idea? We do not include WP:Original research. Also if there is only one publication on the topic it my be undue to include that synthesis. We really like things that have appeared in reviews or textbooks, then we have more confidence it is not a mistake or a crackpot idea. Graeme Bartlett ( talk) 22:27, 4 November 2020 (UTC)
- So there should be numerous suitable references to show how each step has been done, have I understood the whole reasoning correctly? Just an observation, your use of "we" seems to indicate I am the "you" -- second person in plural, the diction appears to suggest a social distance. --
Ktsquare
(talk)
22:35, 4 November 2020 (UTC)
- When I say "we", I am speaking on behalf of people in WikiProject Chemistry. For me personally I am happy to use primary research literature, if it looks reasonable. But others here really like their facts to have appeared in a textbook or review. Another issue is
WP:Synthesis (not chemical synthesis), but putting facts together to get a conclusion that no one else has so far reached. But we have it on Wikipedia as original research. If there is at least one suitable reference to show the whole process then the information can be added. If there are numerous suitable references, then you could write a whole article on the topic.
Graeme Bartlett (
talk)
22:58, 4 November 2020 (UTC)
- I am guessing that you know this already though. for example if we have a reference that says we can make B from A and another references says we can make C from B, then we should not add to the C article that we can make C from A based on that. Graeme Bartlett ( talk) 23:03, 4 November 2020 (UTC)
- How is it possible that you speak on behalf of people in the WPC...Would you not represent yourself? --
Ktsquare
(talk)
05:01, 6 November 2020 (UTC)
- See if anyone else here responds! Some will react when something bad happens. But so far I see nothing inappropriate from you. I try to summarise the consensus I see. Graeme Bartlett ( talk) 05:15, 6 November 2020 (UTC)
- When I say "we", I am speaking on behalf of people in WikiProject Chemistry. For me personally I am happy to use primary research literature, if it looks reasonable. But others here really like their facts to have appeared in a textbook or review. Another issue is
WP:Synthesis (not chemical synthesis), but putting facts together to get a conclusion that no one else has so far reached. But we have it on Wikipedia as original research. If there is at least one suitable reference to show the whole process then the information can be added. If there are numerous suitable references, then you could write a whole article on the topic.
Graeme Bartlett (
talk)
22:58, 4 November 2020 (UTC)
- The WP:SYNTH policy is a good read. It talks about having to follow refs for what they say as limited by their specific scope, not how any wikipedia editor might be able to string their ideas together in a novel way. It's in keeping with the WP:V policy. Those are two governing policies for content that anyone can write in an article. In discussions among editors on Wikipedia, it's common to use "we" since we (including you) are all potential writers discussing what any one of us might write on this collaborative website, as bound by the WP site policies and guidelines. So for example if there is a ref that "secondary alcohols can be oxidized with Jones reagent to make ketones" we can't say "acetone can be made from isopropanol using Jones reagent" unless we have a ref for using Jones on exactly that structure. DMacks ( talk) 05:49, 6 November 2020 (UTC)