 | |
| Clinical data | |
|---|---|
| Trade names | Wainua |
| Other names | AKCEA-TTR-LRx |
| AHFS/ Drugs.com | Eplontersen |
| License data |
|
|
Routes of administration | Subcutaneous |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| DrugBank | |
| UNII | |
| KEGG | |
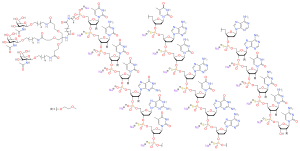
Eplontersen, sold under the brand name Wainua, is a medication used for the treatment of transthyretin-mediated amyloidosis. [1] It is a transthyretin-directed antisense oligonucleotide. [1] It was developed to treat hereditary transthyretin amyloidosis by Ionis Pharmaceuticals and AstraZeneca. [2] [3] [4] [5]
Eplontersen was approved for medical use in the United States in December 2023. [1] [6] [7] [8]
Medical uses
Eplontersen is indicated for the treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis in adults. [1]
Society and culture
Names
Eplontersen is the international nonproprietary name. [9]
References
- ^ a b c d e "Wainua- eplontersen injection, solution". DailyMed. 21 December 2023. Archived from the original on 26 February 2024. Retrieved 26 February 2024.
- ^ "Ionis announces FDA acceptance of New Drug Application for eplontersen for the treatment of hereditary transthyretin-mediated amyloid polyneuropathy (ATTRv-PN)" (Press release). Ionis Pharmaceuticals. 7 March 2023. Archived from the original on 26 September 2023. Retrieved 21 December 2023 – via PR Newswire.
- ^ Coelho, Teresa; Waddington Cruz, Márcia; Chao, Chi-Chao; Parman, Yeşim; Wixner, Jonas; Weiler, Markus; et al. (February 2023). "Characteristics of Patients with Hereditary Transthyretin Amyloidosis-Polyneuropathy (ATTRv-PN) in NEURO-TTRansform, an Open-label Phase 3 Study of Eplontersen". Neurology and Therapy. 12 (1): 267–287. doi: 10.1007/s40120-022-00414-z. PMC 9837340. PMID 36525140.
- ^ Coelho, Teresa; Marques, Wilson; Dasgupta, Noel R.; Chao, Chi-Chao; Parman, Yeşim; França, Marcondes Cavalcante; et al. (October 2023). "Eplontersen for Hereditary Transthyretin Amyloidosis With Polyneuropathy". The Journal of the American Medical Association. 330 (15): 1448–1458. doi: 10.1001/jama.2023.18688. PMC 10540057. PMID 37768671.
- ^ Diep, John K.; Yu, Rosie Z.; Viney, Nicholas J.; Schneider, Eugene; Guo, Shuling; Henry, Scott; et al. (December 2022). "Population pharmacokinetic/pharmacodynamic modelling of eplontersen, an antisense oligonucleotide in development for transthyretin amyloidosis". British Journal of Clinical Pharmacology. 88 (12): 5389–5398. doi: 10.1111/bcp.15468. PMID 35869634. S2CID 250989659.
- ^ "Eplontersen: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Retrieved 21 December 2023.
- ^ "Wainua (eplontersen) granted regulatory approval in the U.S. for the treatment of adults with polyneuropathy of hereditary transthyretin-mediated amyloidosis". Ionis Pharmaceuticals, Inc. (Press release). 21 December 2023. Archived from the original on 22 December 2023. Retrieved 22 December 2023.
- ^ "Wainua (eplontersen) granted first-ever regulatory approval in the US for the treatment of adults with polyneuropathy of hereditary transthyretin-mediated amyloidosis". AstraZeneca US (Press release). 22 December 2023. Archived from the original on 22 December 2023. Retrieved 22 December 2023.
- ^ World Health Organization (2021). "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 85". WHO Drug Information. 35 (1). hdl: 10665/340684.
External links
- Clinical trial number NCT04136184 for "NEURO-TTRansform: A Study to Evaluate the Efficacy and Safety of Eplontersen (Formerly Known as ION-682884, IONIS-TTR-LRx and AKCEA-TTR-LRx) in Participants With Hereditary Transthyretin-Mediated Amyloid Polyneuropathy" at ClinicalTrials.gov
- Clinical trial number NCT01737398 for "Efficacy and Safety of Inotersen in Familial Amyloid Polyneuropathy" at ClinicalTrials.gov
 | |
| Clinical data | |
|---|---|
| Trade names | Wainua |
| Other names | AKCEA-TTR-LRx |
| AHFS/ Drugs.com | Eplontersen |
| License data |
|
|
Routes of administration | Subcutaneous |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| DrugBank | |
| UNII | |
| KEGG | |
Eplontersen, sold under the brand name Wainua, is a medication used for the treatment of transthyretin-mediated amyloidosis. [1] It is a transthyretin-directed antisense oligonucleotide. [1] It was developed to treat hereditary transthyretin amyloidosis by Ionis Pharmaceuticals and AstraZeneca. [2] [3] [4] [5]
Eplontersen was approved for medical use in the United States in December 2023. [1] [6] [7] [8]
Medical uses
Eplontersen is indicated for the treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis in adults. [1]
Society and culture
Names
Eplontersen is the international nonproprietary name. [9]
References
- ^ a b c d e "Wainua- eplontersen injection, solution". DailyMed. 21 December 2023. Archived from the original on 26 February 2024. Retrieved 26 February 2024.
- ^ "Ionis announces FDA acceptance of New Drug Application for eplontersen for the treatment of hereditary transthyretin-mediated amyloid polyneuropathy (ATTRv-PN)" (Press release). Ionis Pharmaceuticals. 7 March 2023. Archived from the original on 26 September 2023. Retrieved 21 December 2023 – via PR Newswire.
- ^ Coelho, Teresa; Waddington Cruz, Márcia; Chao, Chi-Chao; Parman, Yeşim; Wixner, Jonas; Weiler, Markus; et al. (February 2023). "Characteristics of Patients with Hereditary Transthyretin Amyloidosis-Polyneuropathy (ATTRv-PN) in NEURO-TTRansform, an Open-label Phase 3 Study of Eplontersen". Neurology and Therapy. 12 (1): 267–287. doi: 10.1007/s40120-022-00414-z. PMC 9837340. PMID 36525140.
- ^ Coelho, Teresa; Marques, Wilson; Dasgupta, Noel R.; Chao, Chi-Chao; Parman, Yeşim; França, Marcondes Cavalcante; et al. (October 2023). "Eplontersen for Hereditary Transthyretin Amyloidosis With Polyneuropathy". The Journal of the American Medical Association. 330 (15): 1448–1458. doi: 10.1001/jama.2023.18688. PMC 10540057. PMID 37768671.
- ^ Diep, John K.; Yu, Rosie Z.; Viney, Nicholas J.; Schneider, Eugene; Guo, Shuling; Henry, Scott; et al. (December 2022). "Population pharmacokinetic/pharmacodynamic modelling of eplontersen, an antisense oligonucleotide in development for transthyretin amyloidosis". British Journal of Clinical Pharmacology. 88 (12): 5389–5398. doi: 10.1111/bcp.15468. PMID 35869634. S2CID 250989659.
- ^ "Eplontersen: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Retrieved 21 December 2023.
- ^ "Wainua (eplontersen) granted regulatory approval in the U.S. for the treatment of adults with polyneuropathy of hereditary transthyretin-mediated amyloidosis". Ionis Pharmaceuticals, Inc. (Press release). 21 December 2023. Archived from the original on 22 December 2023. Retrieved 22 December 2023.
- ^ "Wainua (eplontersen) granted first-ever regulatory approval in the US for the treatment of adults with polyneuropathy of hereditary transthyretin-mediated amyloidosis". AstraZeneca US (Press release). 22 December 2023. Archived from the original on 22 December 2023. Retrieved 22 December 2023.
- ^ World Health Organization (2021). "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 85". WHO Drug Information. 35 (1). hdl: 10665/340684.
External links
- Clinical trial number NCT04136184 for "NEURO-TTRansform: A Study to Evaluate the Efficacy and Safety of Eplontersen (Formerly Known as ION-682884, IONIS-TTR-LRx and AKCEA-TTR-LRx) in Participants With Hereditary Transthyretin-Mediated Amyloid Polyneuropathy" at ClinicalTrials.gov
- Clinical trial number NCT01737398 for "Efficacy and Safety of Inotersen in Familial Amyloid Polyneuropathy" at ClinicalTrials.gov