I saw the structure. So its the zwitterionic tautomer that crystallizes? Makes sense. If so, I'll redo the ChemDraw, perhaps showing an equilibrium. -- Smokefoot ( talk) 01:28, 14 January 2009 (UTC)
- Apparently so, here's the ref: Acta Cryst. (1996). C52, 1681-1683. I agree with your suggestions, so I'm starting to upload unit cells, coordination polyhedra and extended arrays for polymeric materials. I'm working on YBCO right now - see Commons:Category:Yttrium barium copper oxide.
- All the best

Smile at others by adding {{ subst:Smile}} to their talk page with a friendly message.
... http://en.wikipedia.org/?title=Talk:Triethyloxonium_tetrafluoroborate&diff=266749919&oldid=266749677 .. -- Dirk Beetstra T C 14:50, 27 January 2009 (UTC)
Ben, I'm still quite uncertain about navigating on Wikipedia. But I have learned a lot about editing.
No, I didn't think you were concerned about that changed name for the Image file for my Figure 3. What happened there was that I had found a minor error in the drawing and thus made a new one on my pc. But in uploading it into Commons, all the hyphens in the original drawing were replaced by underscores (actually a good thing since then one could bring in the revised drawing without bringing in the old one again. But it too me a brief while not to understand that I had to change all the hyphens in the Edit copy for the drawing to underscores. And that solved the problem.
My only comment about the minus sign was that I didn't want you to think that it my part of the Birch article where you found an error (at least I think so). The history of that article is that I had it in my User Space. I had written it when I noted that the Birch article Wikipedia had was very limited and had some errors which only an organic chemist would find. First I did try making some minor corrections in the old one (mainly missing references) but that met resistance. So I wrote an entirely new one. Finally it was moved to Article Space and then merged with the old one. It was a part of the old one in which you found a problem with minus signs; at least I think so.
Ben, it was nice to hear from you. You are in England a place I saw back in 1944-45 on a troop train going from Glascow to Southampton. Of course, later I got to see England in much nicer times.
With best wishes,
--Howard E. Zimmerman 22:49, 20 April 2010 (UTC) —Preceding unsigned comment added by Hezimmerman ( talk • contribs)
Ben, Thanks for the heads up on the Carboxyl group geometry. Your work on wikipedia is impressive. I uploaded the new image to the page Carboxyl.
Itineranttrader ( talk) 00:43, 3 February 2009 (UTC)
Thanks for the encouragement as well as the nifty structures. Even if we dont educate the world, we seem to be learning bits ourselves.-- Smokefoot ( talk) 01:26, 6 March 2009 (UTC)
I noticed that you changed the formatting for Plumbide slightly, putting the links to 18-crown-6 and en to the sentence after the chemical structure rather than in the chemical structure itself. Why is that? Chlorine Trifluoride ( talk) 14:34, 6 March 2009 (UTC)
- For clarity - we don't normally put links inside chemical formulae.
- The guideline is at Wikipedia:Manual of Style (chemistry)#Reactions.
- Thanks. Chlorine Trifluoride ( talk) 18:57, 6 March 2009 (UTC)
What do you think of the info that I added from Greenwood and Earnshaw? Chlorine Trifluoride ( talk) 14:12, 8 March 2009 (UTC)
- Very good. Would you like me to add an image from the crystal structure of one of these compounds?
- Yeah. I noticed that there are pictures in the book, but I don't know if we can use those. Chlorine Trifluoride ( talk) 14:28, 8 March 2009 (UTC)
- Don't worry, I'll make some in 3D from the original data.
- Does the image you posted apply to all nonaplumbide anions or just the potassium cation one? Chlorine Trifluoride ( talk) 14:08, 9 March 2009 (UTC)
- I don't know. Greenwood & Earnshaw was written before the nonaplumbide ion had been crystallographically investigated, so it does not discuss the structure (other than to say it's unknown, p. 394). Try a review article - I'll keep looking, too, and let you know.
I found "Aspects of anionic framework formation: Clustering of p-block elements". doi:10.1016/S0010-8545(98)00168-4. Chlorine Trifluoride ( talk) 15:20, 10 March 2009 (UTC)
Additionally, can you also do the structure for the pentaplumbide anion? Chlorine Trifluoride ( talk) 16:19, 12 March 2009 (UTC)
- No problem. The image is File:Pentaplumbide-anion-from-xtal-3D-balls.png.
- Cheers
- Added. Chlorine Trifluoride ( talk) 23:38, 12 March 2009 (UTC)
Hi Ben. I noticed that you have recently uploaded nice structures of cyclic siloxanes substituted with phenyls. They are not used in an article as far as I see. Do you know, if hexaphenylcyclotrisiloxane and octaphenylcyclotetrasiloxane have industrial or commercial applications (i.e. they are produced/used in significant amounts)? -- Leyo 17:41, 17 March 2009 (UTC)
- I don't know, really. I just thought they were interesting examples of conformation. I'll have a look in textbooks.
I have not removed galleries from articles, but a gallery from an article. I acknowledge that there really is no problem. I will explain more in depth on Talk:Butane. Happy Wiki'ing! Bobber0001 ( talk) 10:32, 18 March 2009 (UTC)
- Sorry about the plural, I thought it was more than one article. No problem though, happy editing :)
Hi, next time please be careful when reverting edits, as you did with User:Ratpole's edit to Gold. One of his/her changes included a valid spelling correction (abandonded -> abandoned), which you also reverted. I've already fixed the problem, but just keep an eye out for this in the future. Thanks!— Tetracube ( talk) 22:58, 8 April 2009 (UTC)
- OK, sorry about that. It is difficult, though. Probably easier to just revert the lot then fix abandonded. It all works out in the end.
- Thanks
Hi, Ben. Thanks for the encouraging message. I actually got started making the 3D molecules using your tutorial, so in a way you are sort of my mentor. I would love some pointers on where to find the correct geometries for different molecules so that I do not make the mistake I made again. Thanks! Woodenchemist ( talk) 16:11, 10 April 2009 (UTC)
- You're doing a great job. I frequently run into trouble trying to make images that turn out to be misleading - but I learn a lot of chemistry along the way!
- If you have access to textbooks, journals and search engines like SciFinder, that'll make things much quicker. If not, there's plenty of free information on the web. The basic idea is simple, and is the same as writing text for Wikipedia: you need to provide a reliable source with each image. The places I often get my structural information from are:
- textbooks that show bond lengths and angles, such as Greenwood & Earnshaw
- journal articles - structures often determined by X-ray diffraction, microwave spectroscopy or electron diffraction
- there are two good websites that publish crystal structures electronically: the AMCSD is free but has mostly minerals, whereas the IUCR's journals have all sorts of compounds, but only some of their content is open-access, notably Acta Crystallographica Section E: Structure Reports Online
- the CRC handbook has a useful table of the gas-phase structures of small molecules
- It's normally not good to build a 3D structure from the 2D formula given in a Wikipedia article, because there can be important aspects of the molecule's structure (such as its conformation) that might be calculated wrongly by DS Visualizer.
- If you'd like me to find and send you any structural information that you're finding hard to track down, or if you'd like any more tips, just drop by!
- Cheers
Dear Ben, you were right to revert my edit of TlCl. Indeed, it is CsI-type at room temperature and converts to TlI structure at low-T only. I have put references on TlI structure as (A-V Mudring "Thallium Halides - New Aspects of the Stereochemical Activity of Electron Lone Pairs of Heavier Main-Group Elements" Eur. J. Inorg. Chem. 6 (2007) 882)(R. P. Lowndes and C. H. Perry "Molecular structure and anharmonicity in thallium iodide" J. Chem. Phys. 58, 271 (1973)) I can email you these pdfs if you wish.
Regarding ZnO. IMO, the images are redundant because
 is not the unit cell, but its expanded graphical depiction, i.e. just a cropped version of
is not the unit cell, but its expanded graphical depiction, i.e. just a cropped version of
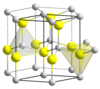 . The wurtzite unit cell contains only 4 atoms (see, e.g.
http://portellen.phycmt.dur.ac.uk/sjc/thesis_mcg/node47.html). Also, for most readers, unit cell does not say much, i.e. one structural picture is sufficient. If you agree, you may wish to revert your edit, but this of no importance. Best regards.
NIMSoffice (
talk) 00:19, 23 April 2009 (UTC)
. The wurtzite unit cell contains only 4 atoms (see, e.g.
http://portellen.phycmt.dur.ac.uk/sjc/thesis_mcg/node47.html). Also, for most readers, unit cell does not say much, i.e. one structural picture is sufficient. If you agree, you may wish to revert your edit, but this of no importance. Best regards.
NIMSoffice (
talk) 00:19, 23 April 2009 (UTC)
- I agree, its not a major issue. These diagrams are fairly difficult to interpret unless you know a few unit cells, and you're likely to know wurtzite and sphalerite if you know any!
I looked up the definition of unit cell, which I have forgotten actually. Loosely speaking, most of those ball-and-stick models are unit cells (thus no need to put several, I usually choose one which looks clearer). More restrictive (thought not unique too) is "primitive unit cell", i.e. the smallest number of atoms to build the lattice (e.g. 4 for ZnO). Those are however not so illustrative. NIMSoffice ( talk) 03:34, 23 April 2009 (UTC)
- Yes, you're absolutely right. If I had software that could do it, I'd make unit cells with sliced-through atoms at the corners, e.g. http://www.wou.edu/las/physci/ch412/unitcel1.jpg, http://www.cbu.edu/%7Emcondren/NaCl-unit-cell.jpg, http://departments.kings.edu/chemlab/vrml/reox.html.
Hi Ben. Could you draw a boat conformation version of File:Cyclohexane-chair-2D-stereo-skeletal.png? The version in Commons:Category:Cyclohexane conformation missed the perspective. I tried to draw one, but as it didn't look nice, it didn't upload it. -- Leyo 12:42, 6 May 2009 (UTC)
- No problem, here it is: File:Cyclohexane-boat-2D-stereo-skeletal.png.
- Cheers
Hey Ben, you may want to weigh in here: Talk:Fluoxetine#Question on the Chemical Structure of Fluoxetine. Best, Fvasconcellos ( t· c) 13:08, 6 May 2009 (UTC)
Dear Ben, I am working on boron nitride hoping to bring it to GA level. The article desperately needs pictures. Would you be willing to draw them ? For example, something like boron-nitride nanotube (same as carbon nanotube, but with alternating boron and nitrogen atoms) and intercalated hexagonal BN. Intercalation of BN is difficult, and thus I am trying to be careful in selecting credible sources. I trust Solozhenko [1] and he has reported BNXNH3 with x=3,4,5 where NH3 molecules are between the BN sheets (no picture given). Another intercalation is alkali metals (with a picture). [2] Any other ideas on pictures and contents are welcome. Cheers. NIMSoffice ( talk) 06:51, 13 May 2009 (UTC)
- ^ V. L. Solozhenko (2002). "In situ studies of boron nitride crystallization from BN solutions in supercritical N–H fluid at high pressures and temperatures". Phys. Chem. Chem. Phys.: 5386. doi: 10.1039/b206005a.
-
^ G. L. Doll; et al. (1989). "Intercalation of hexagonal boron nitride with potassium". J. Appl. Phys. 66: 2554.
doi:
10.1063/1.344219.
{{ cite journal}}: Explicit use of et al. in:|author=( help)
- Hi NIMS, I'll be delighted to make pictures for boron nitride. I've got exams next week and the week after, but I may be able to draw a few during that time.
- I'll check the crystallographic databases for relevant structures, and follow your links.
- Cheers
I had a look on the crystallographic databases I have access to, but could not find any BN intercalation compounds. I also read the papers you link to above – the 1989 paper didn't seem to identify the interlayer separation, and I am not advanced enough to really understand their reciprocal lattice diagram. `The BN-NH3 paper didn't seem to contain any coordinates.
Ben ( talk) 10:41, 7 June 2009 (UTC)
- Ben, sorry for deleting this section, refs were wrong, but I fixed them. I have drawn ref. 2 in boron nitride (found cell parameters), but not ref. 1; it might not be needed anymore. I also deleted your drawing (HgS I guess, you restored it, and its fine), again, without harm intended - just thought of redundancy. You might have noticed that I'm much too quick on action :) I've been drawing much (minerals, etc.) these days. Cheers. Materialscientist ( talk) 10:46, 7 June 2009 (UTC)
Not a problem, I just like to keep a record of what's been said. It was HgO, I think. I looked into the polymorphs of HgO and there are many - see User:Benjah-bmm27/Task list. I'll add pictures shortly. Don't worry about redundancy, there's plenty of room in Wikipedia, the most important thing is to convey the information clearly.
I have a few tips for your drawings, such as File:OsO4structure.jpg:
- upload them to the Wikimedia Commons - then other language Wikipedias can use them, too
- make your images as PNGs, not JPEGs - JPEG suffers from worse compression artifacts
- write the source of the structural information (I see you are starting to do this) and the software used to create the image in the image description template
- use a standard colour coding scheme for your atoms, such as this one I use
- a white or preferably transparent background is neater
- as well as the unit cell, e.g. File:Domeykite_structure.jpg, it's helpful to give an orthographic view (no perspective) along one of the main symmetry axes of more than one unit cell, to show the periodicity and packing in the crystal, such as this (also see below):
-
GaF3
-
As2O5
Ben ( talk) 11:08, 7 June 2009 (UTC)
- Ben, thanks for advice. I did not think about saving in PNG; though I could do that. I know about WP commons, but after changing username, my WP and Wikimedia accounts got somewhat unconnected. I've fixed that recently and can move images to commons as I've done before. One reason I prefer WP is more control over captions, etc. - on wikimedia I often get to know that they want to delete my image only at the last moment. Another good advice is to plot several projections. I didn't do that because for many articles more than one structural image is too many, though in some cases several projections could be necessary. I can recreate them easily. That comes back to HgO - many compounds have too many crystal forms; drawing them all is easy, but would overload the article. Black background is a glitch of one of the programs I use (still don't know how to change it easily, but the program is very, very handy in other respects). Atomic colors are stable in those black pictures. I usually mention the color in figure description. If you notice I missed that, please fix (I've just done that for one of yours). File:OsO4structure.jpg is a tricky example - very low symmetry. You're right about File:Domeykite_structure.jpg, but the structure is hard to grasp anyway. The only reason for plotting that picture is to show that the structure could be complex even for cubic cell (I think I mentioned it in the picture description; just reconfirmed - that structure is same bloody mess in any possible projection).
- Coming back to your drawings, IMHO small balls + sticks are much better than ball-packing structures - there is no need to know "electron clouds" and the like. Have a look at Graphite intercalation compound and see for yourself (well, I should have added lines for the Ca sublattice too). Materialscientist ( talk) 11:38, 7 June 2009 (UTC)
You could take your images with a black background and remove it using Photoshop or GIMP. You're probably right about OsO4, although it is possible to show a bit of packing. Not necessary though, molecular structures are mostly about the molecule! I respectfully disagree that space-filling models are less desirable than ball-and-stick models. Graphite intercalation compound is a prime example! You can see the way the potassium atoms prop open the graphite layers. Both styles are complementary - although in some cases, one may be much clearer.
Thanks for adding the reference to File:Rutile-unit-cell-3D-balls.png. I've updated the image description. I think it's important to give the source that was actually used for the image, rather than any convenient reference that seems to say the same thing. Sometimes there are differences or mistakes in sources that we don't notice until later.
You make a good point about noting atom colours explicitly. I will start to do that in my image descriptions. It's not always practical, though, to add a caption. Chemboxes are a notable example. And often, it is easy to deduce which atom is which colour, e.g. phosphorus pentafluoride!
Ben ( talk) 12:20, 7 June 2009 (UTC)
Dear Ben, I saw your new structure - replacing my one :-( c'est la vie. My one question is that which of us is correct? I set IsisDraw up according to Wikipedia:Manual of Style (chemistry)/Structure drawing, and I got the result that I posted. Personally I prefer your structure - I always do Hydrogen "on Hetero and Terminal" at work - but the WP page says just "on Hetero" Ronhjones (Talk) 23:34, 19 May 2009 (UTC)
- Hi Ron, your image is factually correct, but it has several drawbacks:
- low resolution (small)
- unrealistic 90° CNC bond angles
- hidden (implied) hydrogens on methyl groups are confusing, especially in a structure that is otherwise not skeletal, as in this case
- Hydrogens are not always hidden, see Wikipedia:Manual of Style (chemistry): "Hydrogens should be implied (hidden), except for the benefit of the target audience" - in fact, Wikipedia chemists prefer explicit methyl groups - this should be explained in the MoS, I will raise the issue
- Thanks for asking, your questions will help improve the MoS and I'm sure you'll soon be up to speed with WP:CHEMS's preferred styles.
- Thanks for the reply. As I said, I do prefer to see a methyl as a CH3. As for the drawing, well you have better software than me. IsisDraw does not excel in such nice diagrams. As for the size, well I followed the MoS, loaded up the JACS template, adjusted settings as described, made structure, and did a copy and paste into MSpaint, and then saved it as PNG (that really determined the size of 90px). There's no much else one can do, certainly the export into tiff (within Isis) fails - you get a tiff, which is OK on the PC, and when uploaded to WP, it not visible!
- As an aside, I saw on a Symyx e-mail at work (we use Beilstein Crossfire via Symyx's Discoverygate) that Symyx Draw 3.2 is now free (they also give away IsisDraw 2.5), just dowloaded that - it's a 63MB zip file (one has to register at www.symyx.com, but there is no charge for a private user). It's has a lot in common with Isis, but it does allow one to save the image directly as a PNG. I may still use Isis and finish off in Symyx though as I can draw very quickly in Isis. Ronhjones (Talk) 20:57, 20 May 2009 (UTC)
I'm pretty sure it is possible to get IsisDraw to export a bigger image. I haven't used it for a few years, but I believe other Wikichemists do. Post a message at Wikipedia talk:WikiProject Chemistry - people there are normally very helpful. You'll be a welcome addition to the gang. Let me know if there's anything else you need pointers on.
Cheers
Ben ( talk) 06:21, 21 May 2009 (UTC)
Once you get a TIFF, use software like IrfanView to convert it into a PNG, at the maximum resolution possible. Typically 720 dpi or similar. -- Rifleman 82 ( talk) 16:23, 11 June 2009 (UTC)
- Actually, I might have found another way - I draw the molecule in Isis (or Symyx) Draw, then export as a MolFile. I then use "mol2Svg_1_0" (see ref#1 on Chemical file format) to convert directly to SVG - after all a MolFile is a form of a vector graphic, seems mad to take that, save as PNG (raster), and try to get back to vector. I just did this with the image on O-Acetylpsilocin - the downside? = it won't do terminal methyls, and the hetero hydrogens don't always come where you want. I've not yet tried to see if I can "tweak" the final image with Inkscape. Ronhjones (Talk) 00:24, 12 June 2009 (UTC)
Ben, why reverting ? Why two same figures ? Pride for own creation ? There are so many wrongs there. To start with, red and brown colors are no good couple. Atoms do not pack simply because their ionic/covalent "radii" is a primitive abstraction developed around 1920 when no better methods were available. This abstraction proportionally splits a distance between atoms in a solid, names it "radius" and assumes the electron shell is a sphere having that radius. By definition, it gives dense packing, but. We know that bonding electron distribution is very far from spherical. Another point is that all structural models we use are crude approximations, but dense packing is graphically poor to show relative atomic positions, as compared to ball-stick models. Materialscientist ( talk) 23:20, 11 June 2009 (UTC)
- No, not pride! All the things you say are true, but these depictions are commonplace and useful pedgogically.
- I don't really mind what the conclusion is, but discuss at WP:CHEMS first - you're making a lot of unilateral edits quickly.
Hello,
I just wanted to mention that the File:Fluorite-unit-cell-3D.png is not to scale. I've added a warning in the image description to tell that, because I have made the error to exchange F and Ca atoms based on this image (I should have check more carefully)... -- Mathieu Perrin ( talk) 15:17, 11 June 2009 (UTC)
- I think you might be mistaken in this case - I used ionic radii in the image, since fluorite is predominantly an ionic compound: r(Ca2+) = 1.00 Å, r(F−) = 1.33 Å.
- You're right !! I'm using only covalent crystals usually, so I forgot to use ionic radius... I still have a distance problem, however. Using your values, I find
- for contacts between spheres as shown in the figure, and
- by assuming contact between F ions.
- There is a certain deviation to the exp. 5.46 Å value... ?? Maybe not significant enough. I've changed my warning into "information" in the figure. Which software are you using for modeling? -- Mathieu Perrin ( talk) 14:42, 15 June 2009 (UTC)
Ionic radii are averages over many crystal structures - they're not exact because ions are not perfectly hard spheres, there is some degree of covalency in even the most ionic of compounds. Differing degrees of covalency in different compounds would give rise to slightly different ionic radii if they were calculated separately for each compound.
This isn't normally a problem in images, as the picture would look almost identical, whether r(F−) = 1.33 Å or 1.35 Å etc.
Ben ( talk) 14:49, 15 June 2009 (UTC)
Ben, could you please check File:Iron(II)-sulfide-unit-cell-3D-balls.png. My database tells this is exactly the structure of Ln2O3 (Ln=rare earths - pity I did not see that yesterday when I drew this Ln2O3 structure myself), and gives a quite different structure for FeS (mackinawite). Suspicious is also the ratio of atoms in the unit cell. Cheers. Materialscientist ( talk) 00:40, 5 July 2009 (UTC)
- There are 2 Fe and 2 S in that unit cell. It is the NiAs structure, see http://www.benjamin-mills.com/chemistry/structures/NiAs/. I think there are several polymorphs of FeS. Check http://rruff.geo.arizona.edu/AMS/amcsd.php. Ben ( talk) 13:47, 5 July 2009 (UTC)
I just saw this (I though I was watching this page, but I wasn't). I was wrong and your structure is correct. The similarity between File:Iron(II)-sulfide-unit-cell-3D-balls.png and File:La2O3structure.jpg, although the stoichiometries are different, is accidental - neither structure is a primitive cell. Indeed, FeS has many polymorphs, the information on that is obscure (because not only structure but also Fe/S ratio may change slightly, say 10-20% this is not very common and is misleading), but I might sort that information out later. Materialscientist ( talk) 04:23, 8 July 2009 (UTC)
- Yeah, nonstoichiometric compounds are a nightmare to draw.
- Thanks for your continued hard work on simple solids, keep it up!
Drawing is easy - I have good access to structure files (but the program which can treat them is crippled - thats why black backgrounds). Most time is gone to understand the polymorphs. I also still can't figure out my PNG problem (my PNGs/SVGs display on WP only in full size, but not upon thumb reduction) - thats why still jpegs. You were right about Zirconium(IV) chloride, I forgot about similar layered structures (in fact, many different binaries have structures which are almost same graphically, but not crystallographically). Materialscientist ( talk) 11:32, 8 July 2009 (UTC)
Ben, I have added the symmetry and phase transition for that article. Could you please fix the captions (if you remember) in terms of which structure (orthorhombic or tetragonal (it might be called "square" or "cubic" in some papers)) is shown there. Thanks. Materialscientist ( talk) 00:36, 15 July 2009 (UTC)
- Done. The structure was taken from (I think) J. Solid State Chem. (1975) 13, 252-257.
Hi Ben
Could you do a picture for Magnus' green salt, because the one there sucks :). Ta, Chris ( talk) 08:32, 17 July 2009 (UTC)
- No problem :)
Thanks. Chris ( talk) 12:19, 17 July 2009 (UTC)
Hi Benjah-bmm27,
Regarding [1], I don't dispute your edit, but I was wondering whether we could include a note at Wikipedia:Chemical_infobox or Template:Chembox_Properties stating the consensus? Even a link to a relevant guideline would be useful. I've looked both before and since making my edit, but I can't find any discussion about how these formulae should be presented, and I'm sure I'm not the first person to wonder. Adrian J. Hunter( talk• contribs) 13:31, 27 July 2009 (UTC)
- Absolutely, good idea. You can ask at WP:CHEMS, or I can do it, but I will probably take a little time as something's up at home.
- Thanks for your response. I've started a thread at Wikipedia_talk:WikiProject_Chemicals#Nomenclature_for_molecular_formulas_in_Chembox. Adrian J. Hunter( talk• contribs) 14:03, 6 August 2009 (UTC)
Just a question about models. How do you get the background of the image to be transparent? Thanks. Ginogrz ( talk) 04:27, 31 July 2009 (UTC)
- I use Photoshop to remove the background - it's often helpful to pick a colour other than white so that you don't remove bits of hydrogen atoms in the process.
- Alright, I followed your instructions as well as I could and here's my first attempt. [2] The color of the carbon atoms doesn't seem the same as your models and the red of the oxygens definitely seems less vivid. I'm not sure why this is. Any observations and/or criticisms would be appreciated. Ginogrz ( talk) 06:48, 1 August 2009 (UTC)
- I changed the colour palette to make carbon a much darker grey than it is by default — about 56% R, 56% G, 56% B, or #383838 or #484848.
- I didn't change the red of oxygen, though. If you tweak the lighting settings a bit, maybe move the darker light in the bottom right hand corner down a bit, things should brighten up.
- One thing I know can cause a little inconsistency is that I used version 1.5 but later versions have a slightly different look to them. It's not a problem, though, your image already looks smart.
- I also tend to make the hydrogen atoms a little smaller than all other atoms, by going Edit > Select > Atom... > H then ctrl+D and setting the atom radius to 0.3 (I think). This is purely a matter of choice but can often make a structure look less cluttered when many hydrogens are present.
- I would simply carry on as you are. The most important thing is to find articles that would benefit from illustrations, and use experimentally-determined structures where possible. This is because conformation is often chemically important and our own sense of structural intuition can often deceive us. Take disulfiram for example. I thought it would probably have a planar C2NC(=S)SSC(=S)NC2 core, but Acta Cryst. (1989). B45, 65-69 and Acta Cryst. (1986). C42, 1420-1423 indicate the CSSC dihedral angle is about 90°, i.e. not at all planar.
Ben, thanks for the note about my claim that calcium gluconate is mis-drawn. It was a guess based on the nakedness of the Ca2+ center and the bis(bidentate) carboxylate (I have never seen that motif in acetates), the absence of aquo ligands, and the fact that the thing looks molecular (also rare for carboxylates). Possibly the picture is of a subunit in a polymer that forms upon dehydration. I would expect something far more complicated with octahedral Ca2+. But I havent gone digging yet in the literature.-- Smokefoot ( talk) 22:13, 18 August 2009 (UTC)
- Thanks for bringing that one to my attention - I've put quite a few dodgy structures out there in Wikiland. I'm sure you're right about anhydrous calcium gluconate. In medicine (treatment of HF burns etc), it is apparently the monohydrate that is used: http://www.globalcalcium.com/01Calciumgluconate_02.htm.

You can remove this notice at any time by removing the {{Talkback}} or {{Tb}} template.
Ronhjones (Talk) 19:26, 19 August 2009 (UTC)
Hi Ben. Do you remember where you got the 101.7 pm from? The value cited from this book is 101.4 pm (see article in de-WP). -- Leyo 06:38, 28 August 2009 (UTC)
- Greenwood & Earnshaw, I think - I don't have the book to hand, though, so I can't confirm that figure yet. The book you cite is from 1955, when spectroscopy was a little less precise than in 1997, when G&E 2nd ed. was written.
- However, the CRC handbook gives the dimensions of ammonia, determined by IR spectroscopy, as N-H (re) = 101.2 pm and ∠HNH (θe) = 106.7°.
-
Ben (
talk) 08:58, 28 August 2009 (UTC)
- Thanks for your reply.
The first edition of this book might be from 1955.The first was printed much earlier. The one cited in from 2007. :-) It is pretty much the standard for inorganic chemistry. As it is in German, you might not know it. -- Leyo 15:45, 28 August 2009 (UTC)
- Thanks for your reply.
Oh, sorry - confusing wording "No preview available - 2007 - 2149 pages. This book has a more recent edition (1955)." - makes no sense!
I know Holleman & Wiberg, it's the graduate-level inorganic text here, too.
Shall we have a look in the literature?
Ben ( talk) 15:49, 28 August 2009 (UTC)
- I found the statement in an earlier version of the same book on Google Books now. No original source is indicated there. -- Leyo 15:59, 28 August 2009 (UTC)
Hi....... I think we really need to do some work on the articles related to electropositivity, screening effect, etc... It seems to be very inadequate, as was exposed by the question on the Science Reference Desk.... Rkr1991 ( Wanna chat?) 04:31, 1 October 2009 (UTC)
- Good idea - I'll have a think about it.
Hi, I noticed that you have done a lot of chemistry-related images. Could you help make a new image for Sodium pertechnetate? The current image is a low-resolution GIF; I think we can do better. :-) Thanks!— Tetracube ( talk) 19:36, 25 November 2009 (UTC)
- Done: File:Sodium-pertechnetate-2D.png. Sorry for the epic delay!
I saw the structure. So its the zwitterionic tautomer that crystallizes? Makes sense. If so, I'll redo the ChemDraw, perhaps showing an equilibrium. -- Smokefoot ( talk) 01:28, 14 January 2009 (UTC)
- Apparently so, here's the ref: Acta Cryst. (1996). C52, 1681-1683. I agree with your suggestions, so I'm starting to upload unit cells, coordination polyhedra and extended arrays for polymeric materials. I'm working on YBCO right now - see Commons:Category:Yttrium barium copper oxide.
- All the best

Smile at others by adding {{ subst:Smile}} to their talk page with a friendly message.
... http://en.wikipedia.org/?title=Talk:Triethyloxonium_tetrafluoroborate&diff=266749919&oldid=266749677 .. -- Dirk Beetstra T C 14:50, 27 January 2009 (UTC)
Ben, I'm still quite uncertain about navigating on Wikipedia. But I have learned a lot about editing.
No, I didn't think you were concerned about that changed name for the Image file for my Figure 3. What happened there was that I had found a minor error in the drawing and thus made a new one on my pc. But in uploading it into Commons, all the hyphens in the original drawing were replaced by underscores (actually a good thing since then one could bring in the revised drawing without bringing in the old one again. But it too me a brief while not to understand that I had to change all the hyphens in the Edit copy for the drawing to underscores. And that solved the problem.
My only comment about the minus sign was that I didn't want you to think that it my part of the Birch article where you found an error (at least I think so). The history of that article is that I had it in my User Space. I had written it when I noted that the Birch article Wikipedia had was very limited and had some errors which only an organic chemist would find. First I did try making some minor corrections in the old one (mainly missing references) but that met resistance. So I wrote an entirely new one. Finally it was moved to Article Space and then merged with the old one. It was a part of the old one in which you found a problem with minus signs; at least I think so.
Ben, it was nice to hear from you. You are in England a place I saw back in 1944-45 on a troop train going from Glascow to Southampton. Of course, later I got to see England in much nicer times.
With best wishes,
--Howard E. Zimmerman 22:49, 20 April 2010 (UTC) —Preceding unsigned comment added by Hezimmerman ( talk • contribs)
Ben, Thanks for the heads up on the Carboxyl group geometry. Your work on wikipedia is impressive. I uploaded the new image to the page Carboxyl.
Itineranttrader ( talk) 00:43, 3 February 2009 (UTC)
Thanks for the encouragement as well as the nifty structures. Even if we dont educate the world, we seem to be learning bits ourselves.-- Smokefoot ( talk) 01:26, 6 March 2009 (UTC)
I noticed that you changed the formatting for Plumbide slightly, putting the links to 18-crown-6 and en to the sentence after the chemical structure rather than in the chemical structure itself. Why is that? Chlorine Trifluoride ( talk) 14:34, 6 March 2009 (UTC)
- For clarity - we don't normally put links inside chemical formulae.
- The guideline is at Wikipedia:Manual of Style (chemistry)#Reactions.
- Thanks. Chlorine Trifluoride ( talk) 18:57, 6 March 2009 (UTC)
What do you think of the info that I added from Greenwood and Earnshaw? Chlorine Trifluoride ( talk) 14:12, 8 March 2009 (UTC)
- Very good. Would you like me to add an image from the crystal structure of one of these compounds?
- Yeah. I noticed that there are pictures in the book, but I don't know if we can use those. Chlorine Trifluoride ( talk) 14:28, 8 March 2009 (UTC)
- Don't worry, I'll make some in 3D from the original data.
- Does the image you posted apply to all nonaplumbide anions or just the potassium cation one? Chlorine Trifluoride ( talk) 14:08, 9 March 2009 (UTC)
- I don't know. Greenwood & Earnshaw was written before the nonaplumbide ion had been crystallographically investigated, so it does not discuss the structure (other than to say it's unknown, p. 394). Try a review article - I'll keep looking, too, and let you know.
I found "Aspects of anionic framework formation: Clustering of p-block elements". doi:10.1016/S0010-8545(98)00168-4. Chlorine Trifluoride ( talk) 15:20, 10 March 2009 (UTC)
Additionally, can you also do the structure for the pentaplumbide anion? Chlorine Trifluoride ( talk) 16:19, 12 March 2009 (UTC)
- No problem. The image is File:Pentaplumbide-anion-from-xtal-3D-balls.png.
- Cheers
- Added. Chlorine Trifluoride ( talk) 23:38, 12 March 2009 (UTC)
Hi Ben. I noticed that you have recently uploaded nice structures of cyclic siloxanes substituted with phenyls. They are not used in an article as far as I see. Do you know, if hexaphenylcyclotrisiloxane and octaphenylcyclotetrasiloxane have industrial or commercial applications (i.e. they are produced/used in significant amounts)? -- Leyo 17:41, 17 March 2009 (UTC)
- I don't know, really. I just thought they were interesting examples of conformation. I'll have a look in textbooks.
I have not removed galleries from articles, but a gallery from an article. I acknowledge that there really is no problem. I will explain more in depth on Talk:Butane. Happy Wiki'ing! Bobber0001 ( talk) 10:32, 18 March 2009 (UTC)
- Sorry about the plural, I thought it was more than one article. No problem though, happy editing :)
Hi, next time please be careful when reverting edits, as you did with User:Ratpole's edit to Gold. One of his/her changes included a valid spelling correction (abandonded -> abandoned), which you also reverted. I've already fixed the problem, but just keep an eye out for this in the future. Thanks!— Tetracube ( talk) 22:58, 8 April 2009 (UTC)
- OK, sorry about that. It is difficult, though. Probably easier to just revert the lot then fix abandonded. It all works out in the end.
- Thanks
Hi, Ben. Thanks for the encouraging message. I actually got started making the 3D molecules using your tutorial, so in a way you are sort of my mentor. I would love some pointers on where to find the correct geometries for different molecules so that I do not make the mistake I made again. Thanks! Woodenchemist ( talk) 16:11, 10 April 2009 (UTC)
- You're doing a great job. I frequently run into trouble trying to make images that turn out to be misleading - but I learn a lot of chemistry along the way!
- If you have access to textbooks, journals and search engines like SciFinder, that'll make things much quicker. If not, there's plenty of free information on the web. The basic idea is simple, and is the same as writing text for Wikipedia: you need to provide a reliable source with each image. The places I often get my structural information from are:
- textbooks that show bond lengths and angles, such as Greenwood & Earnshaw
- journal articles - structures often determined by X-ray diffraction, microwave spectroscopy or electron diffraction
- there are two good websites that publish crystal structures electronically: the AMCSD is free but has mostly minerals, whereas the IUCR's journals have all sorts of compounds, but only some of their content is open-access, notably Acta Crystallographica Section E: Structure Reports Online
- the CRC handbook has a useful table of the gas-phase structures of small molecules
- It's normally not good to build a 3D structure from the 2D formula given in a Wikipedia article, because there can be important aspects of the molecule's structure (such as its conformation) that might be calculated wrongly by DS Visualizer.
- If you'd like me to find and send you any structural information that you're finding hard to track down, or if you'd like any more tips, just drop by!
- Cheers
Dear Ben, you were right to revert my edit of TlCl. Indeed, it is CsI-type at room temperature and converts to TlI structure at low-T only. I have put references on TlI structure as (A-V Mudring "Thallium Halides - New Aspects of the Stereochemical Activity of Electron Lone Pairs of Heavier Main-Group Elements" Eur. J. Inorg. Chem. 6 (2007) 882)(R. P. Lowndes and C. H. Perry "Molecular structure and anharmonicity in thallium iodide" J. Chem. Phys. 58, 271 (1973)) I can email you these pdfs if you wish.
Regarding ZnO. IMO, the images are redundant because
 is not the unit cell, but its expanded graphical depiction, i.e. just a cropped version of
is not the unit cell, but its expanded graphical depiction, i.e. just a cropped version of
 . The wurtzite unit cell contains only 4 atoms (see, e.g.
http://portellen.phycmt.dur.ac.uk/sjc/thesis_mcg/node47.html). Also, for most readers, unit cell does not say much, i.e. one structural picture is sufficient. If you agree, you may wish to revert your edit, but this of no importance. Best regards.
NIMSoffice (
talk) 00:19, 23 April 2009 (UTC)
. The wurtzite unit cell contains only 4 atoms (see, e.g.
http://portellen.phycmt.dur.ac.uk/sjc/thesis_mcg/node47.html). Also, for most readers, unit cell does not say much, i.e. one structural picture is sufficient. If you agree, you may wish to revert your edit, but this of no importance. Best regards.
NIMSoffice (
talk) 00:19, 23 April 2009 (UTC)
- I agree, its not a major issue. These diagrams are fairly difficult to interpret unless you know a few unit cells, and you're likely to know wurtzite and sphalerite if you know any!
I looked up the definition of unit cell, which I have forgotten actually. Loosely speaking, most of those ball-and-stick models are unit cells (thus no need to put several, I usually choose one which looks clearer). More restrictive (thought not unique too) is "primitive unit cell", i.e. the smallest number of atoms to build the lattice (e.g. 4 for ZnO). Those are however not so illustrative. NIMSoffice ( talk) 03:34, 23 April 2009 (UTC)
- Yes, you're absolutely right. If I had software that could do it, I'd make unit cells with sliced-through atoms at the corners, e.g. http://www.wou.edu/las/physci/ch412/unitcel1.jpg, http://www.cbu.edu/%7Emcondren/NaCl-unit-cell.jpg, http://departments.kings.edu/chemlab/vrml/reox.html.
Hi Ben. Could you draw a boat conformation version of File:Cyclohexane-chair-2D-stereo-skeletal.png? The version in Commons:Category:Cyclohexane conformation missed the perspective. I tried to draw one, but as it didn't look nice, it didn't upload it. -- Leyo 12:42, 6 May 2009 (UTC)
- No problem, here it is: File:Cyclohexane-boat-2D-stereo-skeletal.png.
- Cheers
Hey Ben, you may want to weigh in here: Talk:Fluoxetine#Question on the Chemical Structure of Fluoxetine. Best, Fvasconcellos ( t· c) 13:08, 6 May 2009 (UTC)
Dear Ben, I am working on boron nitride hoping to bring it to GA level. The article desperately needs pictures. Would you be willing to draw them ? For example, something like boron-nitride nanotube (same as carbon nanotube, but with alternating boron and nitrogen atoms) and intercalated hexagonal BN. Intercalation of BN is difficult, and thus I am trying to be careful in selecting credible sources. I trust Solozhenko [1] and he has reported BNXNH3 with x=3,4,5 where NH3 molecules are between the BN sheets (no picture given). Another intercalation is alkali metals (with a picture). [2] Any other ideas on pictures and contents are welcome. Cheers. NIMSoffice ( talk) 06:51, 13 May 2009 (UTC)
- ^ V. L. Solozhenko (2002). "In situ studies of boron nitride crystallization from BN solutions in supercritical N–H fluid at high pressures and temperatures". Phys. Chem. Chem. Phys.: 5386. doi: 10.1039/b206005a.
-
^ G. L. Doll; et al. (1989). "Intercalation of hexagonal boron nitride with potassium". J. Appl. Phys. 66: 2554.
doi:
10.1063/1.344219.
{{ cite journal}}: Explicit use of et al. in:|author=( help)
- Hi NIMS, I'll be delighted to make pictures for boron nitride. I've got exams next week and the week after, but I may be able to draw a few during that time.
- I'll check the crystallographic databases for relevant structures, and follow your links.
- Cheers
I had a look on the crystallographic databases I have access to, but could not find any BN intercalation compounds. I also read the papers you link to above – the 1989 paper didn't seem to identify the interlayer separation, and I am not advanced enough to really understand their reciprocal lattice diagram. `The BN-NH3 paper didn't seem to contain any coordinates.
Ben ( talk) 10:41, 7 June 2009 (UTC)
- Ben, sorry for deleting this section, refs were wrong, but I fixed them. I have drawn ref. 2 in boron nitride (found cell parameters), but not ref. 1; it might not be needed anymore. I also deleted your drawing (HgS I guess, you restored it, and its fine), again, without harm intended - just thought of redundancy. You might have noticed that I'm much too quick on action :) I've been drawing much (minerals, etc.) these days. Cheers. Materialscientist ( talk) 10:46, 7 June 2009 (UTC)
Not a problem, I just like to keep a record of what's been said. It was HgO, I think. I looked into the polymorphs of HgO and there are many - see User:Benjah-bmm27/Task list. I'll add pictures shortly. Don't worry about redundancy, there's plenty of room in Wikipedia, the most important thing is to convey the information clearly.
I have a few tips for your drawings, such as File:OsO4structure.jpg:
- upload them to the Wikimedia Commons - then other language Wikipedias can use them, too
- make your images as PNGs, not JPEGs - JPEG suffers from worse compression artifacts
- write the source of the structural information (I see you are starting to do this) and the software used to create the image in the image description template
- use a standard colour coding scheme for your atoms, such as this one I use
- a white or preferably transparent background is neater
- as well as the unit cell, e.g. File:Domeykite_structure.jpg, it's helpful to give an orthographic view (no perspective) along one of the main symmetry axes of more than one unit cell, to show the periodicity and packing in the crystal, such as this (also see below):
-
GaF3
-
As2O5
Ben ( talk) 11:08, 7 June 2009 (UTC)
- Ben, thanks for advice. I did not think about saving in PNG; though I could do that. I know about WP commons, but after changing username, my WP and Wikimedia accounts got somewhat unconnected. I've fixed that recently and can move images to commons as I've done before. One reason I prefer WP is more control over captions, etc. - on wikimedia I often get to know that they want to delete my image only at the last moment. Another good advice is to plot several projections. I didn't do that because for many articles more than one structural image is too many, though in some cases several projections could be necessary. I can recreate them easily. That comes back to HgO - many compounds have too many crystal forms; drawing them all is easy, but would overload the article. Black background is a glitch of one of the programs I use (still don't know how to change it easily, but the program is very, very handy in other respects). Atomic colors are stable in those black pictures. I usually mention the color in figure description. If you notice I missed that, please fix (I've just done that for one of yours). File:OsO4structure.jpg is a tricky example - very low symmetry. You're right about File:Domeykite_structure.jpg, but the structure is hard to grasp anyway. The only reason for plotting that picture is to show that the structure could be complex even for cubic cell (I think I mentioned it in the picture description; just reconfirmed - that structure is same bloody mess in any possible projection).
- Coming back to your drawings, IMHO small balls + sticks are much better than ball-packing structures - there is no need to know "electron clouds" and the like. Have a look at Graphite intercalation compound and see for yourself (well, I should have added lines for the Ca sublattice too). Materialscientist ( talk) 11:38, 7 June 2009 (UTC)
You could take your images with a black background and remove it using Photoshop or GIMP. You're probably right about OsO4, although it is possible to show a bit of packing. Not necessary though, molecular structures are mostly about the molecule! I respectfully disagree that space-filling models are less desirable than ball-and-stick models. Graphite intercalation compound is a prime example! You can see the way the potassium atoms prop open the graphite layers. Both styles are complementary - although in some cases, one may be much clearer.
Thanks for adding the reference to File:Rutile-unit-cell-3D-balls.png. I've updated the image description. I think it's important to give the source that was actually used for the image, rather than any convenient reference that seems to say the same thing. Sometimes there are differences or mistakes in sources that we don't notice until later.
You make a good point about noting atom colours explicitly. I will start to do that in my image descriptions. It's not always practical, though, to add a caption. Chemboxes are a notable example. And often, it is easy to deduce which atom is which colour, e.g. phosphorus pentafluoride!
Ben ( talk) 12:20, 7 June 2009 (UTC)
Dear Ben, I saw your new structure - replacing my one :-( c'est la vie. My one question is that which of us is correct? I set IsisDraw up according to Wikipedia:Manual of Style (chemistry)/Structure drawing, and I got the result that I posted. Personally I prefer your structure - I always do Hydrogen "on Hetero and Terminal" at work - but the WP page says just "on Hetero" Ronhjones (Talk) 23:34, 19 May 2009 (UTC)
- Hi Ron, your image is factually correct, but it has several drawbacks:
- low resolution (small)
- unrealistic 90° CNC bond angles
- hidden (implied) hydrogens on methyl groups are confusing, especially in a structure that is otherwise not skeletal, as in this case
- Hydrogens are not always hidden, see Wikipedia:Manual of Style (chemistry): "Hydrogens should be implied (hidden), except for the benefit of the target audience" - in fact, Wikipedia chemists prefer explicit methyl groups - this should be explained in the MoS, I will raise the issue
- Thanks for asking, your questions will help improve the MoS and I'm sure you'll soon be up to speed with WP:CHEMS's preferred styles.
- Thanks for the reply. As I said, I do prefer to see a methyl as a CH3. As for the drawing, well you have better software than me. IsisDraw does not excel in such nice diagrams. As for the size, well I followed the MoS, loaded up the JACS template, adjusted settings as described, made structure, and did a copy and paste into MSpaint, and then saved it as PNG (that really determined the size of 90px). There's no much else one can do, certainly the export into tiff (within Isis) fails - you get a tiff, which is OK on the PC, and when uploaded to WP, it not visible!
- As an aside, I saw on a Symyx e-mail at work (we use Beilstein Crossfire via Symyx's Discoverygate) that Symyx Draw 3.2 is now free (they also give away IsisDraw 2.5), just dowloaded that - it's a 63MB zip file (one has to register at www.symyx.com, but there is no charge for a private user). It's has a lot in common with Isis, but it does allow one to save the image directly as a PNG. I may still use Isis and finish off in Symyx though as I can draw very quickly in Isis. Ronhjones (Talk) 20:57, 20 May 2009 (UTC)
I'm pretty sure it is possible to get IsisDraw to export a bigger image. I haven't used it for a few years, but I believe other Wikichemists do. Post a message at Wikipedia talk:WikiProject Chemistry - people there are normally very helpful. You'll be a welcome addition to the gang. Let me know if there's anything else you need pointers on.
Cheers
Ben ( talk) 06:21, 21 May 2009 (UTC)
Once you get a TIFF, use software like IrfanView to convert it into a PNG, at the maximum resolution possible. Typically 720 dpi or similar. -- Rifleman 82 ( talk) 16:23, 11 June 2009 (UTC)
- Actually, I might have found another way - I draw the molecule in Isis (or Symyx) Draw, then export as a MolFile. I then use "mol2Svg_1_0" (see ref#1 on Chemical file format) to convert directly to SVG - after all a MolFile is a form of a vector graphic, seems mad to take that, save as PNG (raster), and try to get back to vector. I just did this with the image on O-Acetylpsilocin - the downside? = it won't do terminal methyls, and the hetero hydrogens don't always come where you want. I've not yet tried to see if I can "tweak" the final image with Inkscape. Ronhjones (Talk) 00:24, 12 June 2009 (UTC)
Ben, why reverting ? Why two same figures ? Pride for own creation ? There are so many wrongs there. To start with, red and brown colors are no good couple. Atoms do not pack simply because their ionic/covalent "radii" is a primitive abstraction developed around 1920 when no better methods were available. This abstraction proportionally splits a distance between atoms in a solid, names it "radius" and assumes the electron shell is a sphere having that radius. By definition, it gives dense packing, but. We know that bonding electron distribution is very far from spherical. Another point is that all structural models we use are crude approximations, but dense packing is graphically poor to show relative atomic positions, as compared to ball-stick models. Materialscientist ( talk) 23:20, 11 June 2009 (UTC)
- No, not pride! All the things you say are true, but these depictions are commonplace and useful pedgogically.
- I don't really mind what the conclusion is, but discuss at WP:CHEMS first - you're making a lot of unilateral edits quickly.
Hello,
I just wanted to mention that the File:Fluorite-unit-cell-3D.png is not to scale. I've added a warning in the image description to tell that, because I have made the error to exchange F and Ca atoms based on this image (I should have check more carefully)... -- Mathieu Perrin ( talk) 15:17, 11 June 2009 (UTC)
- I think you might be mistaken in this case - I used ionic radii in the image, since fluorite is predominantly an ionic compound: r(Ca2+) = 1.00 Å, r(F−) = 1.33 Å.
- You're right !! I'm using only covalent crystals usually, so I forgot to use ionic radius... I still have a distance problem, however. Using your values, I find
- for contacts between spheres as shown in the figure, and
- by assuming contact between F ions.
- There is a certain deviation to the exp. 5.46 Å value... ?? Maybe not significant enough. I've changed my warning into "information" in the figure. Which software are you using for modeling? -- Mathieu Perrin ( talk) 14:42, 15 June 2009 (UTC)
Ionic radii are averages over many crystal structures - they're not exact because ions are not perfectly hard spheres, there is some degree of covalency in even the most ionic of compounds. Differing degrees of covalency in different compounds would give rise to slightly different ionic radii if they were calculated separately for each compound.
This isn't normally a problem in images, as the picture would look almost identical, whether r(F−) = 1.33 Å or 1.35 Å etc.
Ben ( talk) 14:49, 15 June 2009 (UTC)
Ben, could you please check File:Iron(II)-sulfide-unit-cell-3D-balls.png. My database tells this is exactly the structure of Ln2O3 (Ln=rare earths - pity I did not see that yesterday when I drew this Ln2O3 structure myself), and gives a quite different structure for FeS (mackinawite). Suspicious is also the ratio of atoms in the unit cell. Cheers. Materialscientist ( talk) 00:40, 5 July 2009 (UTC)
- There are 2 Fe and 2 S in that unit cell. It is the NiAs structure, see http://www.benjamin-mills.com/chemistry/structures/NiAs/. I think there are several polymorphs of FeS. Check http://rruff.geo.arizona.edu/AMS/amcsd.php. Ben ( talk) 13:47, 5 July 2009 (UTC)
I just saw this (I though I was watching this page, but I wasn't). I was wrong and your structure is correct. The similarity between File:Iron(II)-sulfide-unit-cell-3D-balls.png and File:La2O3structure.jpg, although the stoichiometries are different, is accidental - neither structure is a primitive cell. Indeed, FeS has many polymorphs, the information on that is obscure (because not only structure but also Fe/S ratio may change slightly, say 10-20% this is not very common and is misleading), but I might sort that information out later. Materialscientist ( talk) 04:23, 8 July 2009 (UTC)
- Yeah, nonstoichiometric compounds are a nightmare to draw.
- Thanks for your continued hard work on simple solids, keep it up!
Drawing is easy - I have good access to structure files (but the program which can treat them is crippled - thats why black backgrounds). Most time is gone to understand the polymorphs. I also still can't figure out my PNG problem (my PNGs/SVGs display on WP only in full size, but not upon thumb reduction) - thats why still jpegs. You were right about Zirconium(IV) chloride, I forgot about similar layered structures (in fact, many different binaries have structures which are almost same graphically, but not crystallographically). Materialscientist ( talk) 11:32, 8 July 2009 (UTC)
Ben, I have added the symmetry and phase transition for that article. Could you please fix the captions (if you remember) in terms of which structure (orthorhombic or tetragonal (it might be called "square" or "cubic" in some papers)) is shown there. Thanks. Materialscientist ( talk) 00:36, 15 July 2009 (UTC)
- Done. The structure was taken from (I think) J. Solid State Chem. (1975) 13, 252-257.
Hi Ben
Could you do a picture for Magnus' green salt, because the one there sucks :). Ta, Chris ( talk) 08:32, 17 July 2009 (UTC)
- No problem :)
Thanks. Chris ( talk) 12:19, 17 July 2009 (UTC)
Hi Benjah-bmm27,
Regarding [1], I don't dispute your edit, but I was wondering whether we could include a note at Wikipedia:Chemical_infobox or Template:Chembox_Properties stating the consensus? Even a link to a relevant guideline would be useful. I've looked both before and since making my edit, but I can't find any discussion about how these formulae should be presented, and I'm sure I'm not the first person to wonder. Adrian J. Hunter( talk• contribs) 13:31, 27 July 2009 (UTC)
- Absolutely, good idea. You can ask at WP:CHEMS, or I can do it, but I will probably take a little time as something's up at home.
- Thanks for your response. I've started a thread at Wikipedia_talk:WikiProject_Chemicals#Nomenclature_for_molecular_formulas_in_Chembox. Adrian J. Hunter( talk• contribs) 14:03, 6 August 2009 (UTC)
Just a question about models. How do you get the background of the image to be transparent? Thanks. Ginogrz ( talk) 04:27, 31 July 2009 (UTC)
- I use Photoshop to remove the background - it's often helpful to pick a colour other than white so that you don't remove bits of hydrogen atoms in the process.
- Alright, I followed your instructions as well as I could and here's my first attempt. [2] The color of the carbon atoms doesn't seem the same as your models and the red of the oxygens definitely seems less vivid. I'm not sure why this is. Any observations and/or criticisms would be appreciated. Ginogrz ( talk) 06:48, 1 August 2009 (UTC)
- I changed the colour palette to make carbon a much darker grey than it is by default — about 56% R, 56% G, 56% B, or #383838 or #484848.
- I didn't change the red of oxygen, though. If you tweak the lighting settings a bit, maybe move the darker light in the bottom right hand corner down a bit, things should brighten up.
- One thing I know can cause a little inconsistency is that I used version 1.5 but later versions have a slightly different look to them. It's not a problem, though, your image already looks smart.
- I also tend to make the hydrogen atoms a little smaller than all other atoms, by going Edit > Select > Atom... > H then ctrl+D and setting the atom radius to 0.3 (I think). This is purely a matter of choice but can often make a structure look less cluttered when many hydrogens are present.
- I would simply carry on as you are. The most important thing is to find articles that would benefit from illustrations, and use experimentally-determined structures where possible. This is because conformation is often chemically important and our own sense of structural intuition can often deceive us. Take disulfiram for example. I thought it would probably have a planar C2NC(=S)SSC(=S)NC2 core, but Acta Cryst. (1989). B45, 65-69 and Acta Cryst. (1986). C42, 1420-1423 indicate the CSSC dihedral angle is about 90°, i.e. not at all planar.
Ben, thanks for the note about my claim that calcium gluconate is mis-drawn. It was a guess based on the nakedness of the Ca2+ center and the bis(bidentate) carboxylate (I have never seen that motif in acetates), the absence of aquo ligands, and the fact that the thing looks molecular (also rare for carboxylates). Possibly the picture is of a subunit in a polymer that forms upon dehydration. I would expect something far more complicated with octahedral Ca2+. But I havent gone digging yet in the literature.-- Smokefoot ( talk) 22:13, 18 August 2009 (UTC)
- Thanks for bringing that one to my attention - I've put quite a few dodgy structures out there in Wikiland. I'm sure you're right about anhydrous calcium gluconate. In medicine (treatment of HF burns etc), it is apparently the monohydrate that is used: http://www.globalcalcium.com/01Calciumgluconate_02.htm.

You can remove this notice at any time by removing the {{Talkback}} or {{Tb}} template.
Ronhjones (Talk) 19:26, 19 August 2009 (UTC)
Hi Ben. Do you remember where you got the 101.7 pm from? The value cited from this book is 101.4 pm (see article in de-WP). -- Leyo 06:38, 28 August 2009 (UTC)
- Greenwood & Earnshaw, I think - I don't have the book to hand, though, so I can't confirm that figure yet. The book you cite is from 1955, when spectroscopy was a little less precise than in 1997, when G&E 2nd ed. was written.
- However, the CRC handbook gives the dimensions of ammonia, determined by IR spectroscopy, as N-H (re) = 101.2 pm and ∠HNH (θe) = 106.7°.
-
Ben (
talk) 08:58, 28 August 2009 (UTC)
- Thanks for your reply.
The first edition of this book might be from 1955.The first was printed much earlier. The one cited in from 2007. :-) It is pretty much the standard for inorganic chemistry. As it is in German, you might not know it. -- Leyo 15:45, 28 August 2009 (UTC)
- Thanks for your reply.
Oh, sorry - confusing wording "No preview available - 2007 - 2149 pages. This book has a more recent edition (1955)." - makes no sense!
I know Holleman & Wiberg, it's the graduate-level inorganic text here, too.
Shall we have a look in the literature?
Ben ( talk) 15:49, 28 August 2009 (UTC)
- I found the statement in an earlier version of the same book on Google Books now. No original source is indicated there. -- Leyo 15:59, 28 August 2009 (UTC)
Hi....... I think we really need to do some work on the articles related to electropositivity, screening effect, etc... It seems to be very inadequate, as was exposed by the question on the Science Reference Desk.... Rkr1991 ( Wanna chat?) 04:31, 1 October 2009 (UTC)
- Good idea - I'll have a think about it.
Hi, I noticed that you have done a lot of chemistry-related images. Could you help make a new image for Sodium pertechnetate? The current image is a low-resolution GIF; I think we can do better. :-) Thanks!— Tetracube ( talk) 19:36, 25 November 2009 (UTC)
- Done: File:Sodium-pertechnetate-2D.png. Sorry for the epic delay!






