
| |
| Names | |
|---|---|
|
IUPAC name
α-L-Fucopyranosyl-(1→2)-β-D-galactopyranosyl-(1→4)-D-glucose
| |
|
Systematic IUPAC name
(2R,3R,4R,5R)-4-{[(2S,3R,4S,5R,6R)-4,5-Dihydroxy-6-(hydroxymethyl)-3-{[(2S,3S,4R,5S,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}oxan-2-yl]oxy}-2,3,5,6-tetrahydroxyhexanal | |
| Other names
2′-FL
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C18H32O15 | |
| Molar mass | 488.439 g·mol−1 |
| Density | 1.681 g/cm3 |
| 240.0 g/L (in water) | |
| Acidity (pKa) | 11.9 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
2′-Fucosyllactose (2′-FL) is an oligosaccharide, more precisely, fucosylated, neutral trisaccharide composed of L-fucose, D-galactose, and D-glucose units. It is the most prevalent human milk oligosaccharide (HMO) naturally present in human breast milk, making up about 30% of all of HMOs. [1] It was first discovered in the 1950s in human milk. The oligosaccharide's primary isolation technique has been in use since 1972. [2]
Production
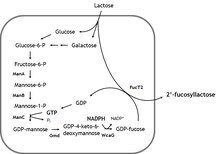
This compound may be biosynthesized in quantity using E. coli. [3] [4] [5] [6]
Metabolism
|
| This section needs expansion. You can help by
adding to it. (November 2012) |
Uses
As with other oligosaccharides, a widely regarded characteristic of 2'-fucosyllactose is its ability to protect against infectious diseases [7] namely in preventing epithelial level adhesions of toxins and pathogens. [8] The 2FL stimulates the growth of certain bifidobacteria and receptor analogons which lends to toxic and pathogenic protection, all this being most prevalent in infants. Among the pathogens that 2FL is known to protect against are Campylobacter jejuni, Salmonella enterica serotype Typhimurium, Helicobacter pylori, among others. [8]
References
- ^ Vandenplas, Yvan; Berger, Bernard; Carnielli, Virgilio Paolo; Ksiazyk, Janusz; Lagström, Hanna; Sanchez Luna, Manuel; Migacheva, Natalia; Mosselmans, Jean-Marc; Picaud, Jean-Charles; Possner, Mike; Singhal, Atul; Wabitsch, Martin (2018-08-24). "Human Milk Oligosaccharides: 2′-Fucosyllactose (2′-FL) and Lacto-N-Neotetraose (LNnT) in Infant Formula". Nutrients. 10 (9): 1161. doi: 10.3390/nu10091161. ISSN 2072-6643. PMC 6164445. PMID 30149573.
- ^ "Showing metabocard for 2-Fucosyllactose (HMDB02098)". Human Metabolome Database. Retrieved 19 October 2012.
- ^ "U of I microbial engineer synthesizes 2-fucosyllactose to protect babies from pathogens". News Medical. Retrieved 19 October 2012.
- ^ Chin, Young-Wook; Kim, Ji-Yeong; Lee, Won-Heong; Seo, Jin-Ho (2015). "Enhanced production of 2′-fucosyllactose in engineered Escherichia coli BL21star (DE3) by modulation of lactose metabolism and fucosyltransferase". Journal of Biotechnology. 210: 107–115. doi: 10.1016/j.jbiotec.2015.06.431. PMID 26193630.
- ^ Chin, Young-Wook; Seo, Nari; Kim, Jae-Han; Seo, Jin-Ho (2016). "Metabolic engineering of Escherichia coli to produce 2'-fucosyllactose via Salvage pathway of guanosine 5'-diphosphate (GDP)-L-fucose". Biotechnology and Bioengineering. 113 (11): 2443–2452. doi: 10.1002/bit.26015. PMID 27217241. S2CID 45579939.
- ^ Chin, Young-Wook; Kim, Ji-Yeong; Kim, Jae-Han; Jung, Sang-min; Seo, Jin-Ho (2016). "Improved production of 2'-fucosyllactose in engineered Escherichia coli by expressing putative α-1,2-fucosyltransferase, WcfB from Bacteroides fragilis". Journal of Biotechnology. 257: 192–198. doi: 10.1016/j.jbiotec.2016.11.033. PMID 27919692.
- ^ Taylor, Diane E.; Rasko, David A.; Sherburne, Richard; Ho, Clinton; Jewel, Laurence D. (1998). "Lack of correlation between Lewis antigen expression by Helicobacter pylori and gastric epithelial cells in infected patients". Gastroenterology. 115 (5): 1113–1122. doi: 10.1016/S0016-5085(98)70082-4. PMID 9797366.
- ^ a b Lee, Won-Heong; Pathanibul, Panchalee; Quarterman, Josh; Jo, Jung-Hyun; Han, Nam-Soo; Miller, Michael J; Jin, Yong-Su; Seo, Jin-Ho (2012). "Whole cell biosynthesis of a functional oligosaccharide, 2'-fucosyllactose, using engineered Escherichia coli". Microbial Cell Factories. 11: 48. doi: 10.1186/1475-2859-11-48. PMC 3442965. PMID 22545760.

| |
| Names | |
|---|---|
|
IUPAC name
α-L-Fucopyranosyl-(1→2)-β-D-galactopyranosyl-(1→4)-D-glucose
| |
|
Systematic IUPAC name
(2R,3R,4R,5R)-4-{[(2S,3R,4S,5R,6R)-4,5-Dihydroxy-6-(hydroxymethyl)-3-{[(2S,3S,4R,5S,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}oxan-2-yl]oxy}-2,3,5,6-tetrahydroxyhexanal | |
| Other names
2′-FL
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C18H32O15 | |
| Molar mass | 488.439 g·mol−1 |
| Density | 1.681 g/cm3 |
| 240.0 g/L (in water) | |
| Acidity (pKa) | 11.9 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
2′-Fucosyllactose (2′-FL) is an oligosaccharide, more precisely, fucosylated, neutral trisaccharide composed of L-fucose, D-galactose, and D-glucose units. It is the most prevalent human milk oligosaccharide (HMO) naturally present in human breast milk, making up about 30% of all of HMOs. [1] It was first discovered in the 1950s in human milk. The oligosaccharide's primary isolation technique has been in use since 1972. [2]
Production
This compound may be biosynthesized in quantity using E. coli. [3] [4] [5] [6]
Metabolism

|
| This section needs expansion. You can help by
adding to it. (November 2012) |
Uses
As with other oligosaccharides, a widely regarded characteristic of 2'-fucosyllactose is its ability to protect against infectious diseases [7] namely in preventing epithelial level adhesions of toxins and pathogens. [8] The 2FL stimulates the growth of certain bifidobacteria and receptor analogons which lends to toxic and pathogenic protection, all this being most prevalent in infants. Among the pathogens that 2FL is known to protect against are Campylobacter jejuni, Salmonella enterica serotype Typhimurium, Helicobacter pylori, among others. [8]
References
- ^ Vandenplas, Yvan; Berger, Bernard; Carnielli, Virgilio Paolo; Ksiazyk, Janusz; Lagström, Hanna; Sanchez Luna, Manuel; Migacheva, Natalia; Mosselmans, Jean-Marc; Picaud, Jean-Charles; Possner, Mike; Singhal, Atul; Wabitsch, Martin (2018-08-24). "Human Milk Oligosaccharides: 2′-Fucosyllactose (2′-FL) and Lacto-N-Neotetraose (LNnT) in Infant Formula". Nutrients. 10 (9): 1161. doi: 10.3390/nu10091161. ISSN 2072-6643. PMC 6164445. PMID 30149573.
- ^ "Showing metabocard for 2-Fucosyllactose (HMDB02098)". Human Metabolome Database. Retrieved 19 October 2012.
- ^ "U of I microbial engineer synthesizes 2-fucosyllactose to protect babies from pathogens". News Medical. Retrieved 19 October 2012.
- ^ Chin, Young-Wook; Kim, Ji-Yeong; Lee, Won-Heong; Seo, Jin-Ho (2015). "Enhanced production of 2′-fucosyllactose in engineered Escherichia coli BL21star (DE3) by modulation of lactose metabolism and fucosyltransferase". Journal of Biotechnology. 210: 107–115. doi: 10.1016/j.jbiotec.2015.06.431. PMID 26193630.
- ^ Chin, Young-Wook; Seo, Nari; Kim, Jae-Han; Seo, Jin-Ho (2016). "Metabolic engineering of Escherichia coli to produce 2'-fucosyllactose via Salvage pathway of guanosine 5'-diphosphate (GDP)-L-fucose". Biotechnology and Bioengineering. 113 (11): 2443–2452. doi: 10.1002/bit.26015. PMID 27217241. S2CID 45579939.
- ^ Chin, Young-Wook; Kim, Ji-Yeong; Kim, Jae-Han; Jung, Sang-min; Seo, Jin-Ho (2016). "Improved production of 2'-fucosyllactose in engineered Escherichia coli by expressing putative α-1,2-fucosyltransferase, WcfB from Bacteroides fragilis". Journal of Biotechnology. 257: 192–198. doi: 10.1016/j.jbiotec.2016.11.033. PMID 27919692.
- ^ Taylor, Diane E.; Rasko, David A.; Sherburne, Richard; Ho, Clinton; Jewel, Laurence D. (1998). "Lack of correlation between Lewis antigen expression by Helicobacter pylori and gastric epithelial cells in infected patients". Gastroenterology. 115 (5): 1113–1122. doi: 10.1016/S0016-5085(98)70082-4. PMID 9797366.
- ^ a b Lee, Won-Heong; Pathanibul, Panchalee; Quarterman, Josh; Jo, Jung-Hyun; Han, Nam-Soo; Miller, Michael J; Jin, Yong-Su; Seo, Jin-Ho (2012). "Whole cell biosynthesis of a functional oligosaccharide, 2'-fucosyllactose, using engineered Escherichia coli". Microbial Cell Factories. 11: 48. doi: 10.1186/1475-2859-11-48. PMC 3442965. PMID 22545760.