History of antioxidants [1]
| Nuclear magnetic resonance (NMR) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Core concepts | |||||||||||||||
|
|||||||||||||||
| By isotope | |||||||||||||||
|
Trixylylphosphate

| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
| EC Number |
|
| Properties | |
| C24H27O4P | |
| Molar mass | 410.450 g·mol−1 |
| Appearance | Viscous liquid |
| Density | 1.142 |
| Melting point | −20 °C (−4 °F; 253 K) |
| Boiling point | 394 °C (741 °F; 667 K) |
| 20 µg/L | |
| Hazards | |
| GHS labelling: | |

| |
| Danger | |
| H360F | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Trixylyl phosphate (TXP) is an aromatic phosphate ester historically used as a hydraulic fluid.
Chirality
| Chemical Chirality | ||||
|---|---|---|---|---|
| An aspect of Stereoisomerism | ||||
| Core concepts | ||||
|
||||
| Assigning and naming | ||||
|
| Rotational axis (Cn) |
Improper rotational elements (Sn) | ||
|---|---|---|---|
| Chiral no Sn |
Achiral mirror plane S1 = σ |
Achiral inversion centre S2 = i | |
| C1 |
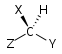 |
 |

|
| C2 |
 |
 |

|
Uric
 |

| |
| Lactam form | Lactim form |
 |
 |
pKa1 |
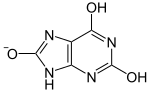
| |
| Lactam form | Lactim form | Urate ion |
Thermosets example table
| Name | Properties | Applications |
|---|---|---|
| Example | Example | Example |
| Example | Example | Example |
| Example | Example | Example |
formaldehyde base
- Bakelite, a phenol- formaldehyde
- Urea-formaldehyde
- Melamine resin
- Benzoxazines, formed by the reaction of phenols, formaldehyde and primary amines
- Duroplast
- Diallyl-phthalate (DAP)
- Epoxy resin
- Epoxy novolac resins
- Polyimides and Bismaleimides
- Cyanate esters or polycyanurates
- Furan resins
- Silicone resins
- Thiolyte,
- Vinyl ester resins used for wet lay-up laminating, molding and fast setting industrial protection and repair materials.
- Polyester resin fiberglass systems: sheet molding compounds and bulk molding compounds; filament winding; wet lay-up lamination; repair compounds and protective coatings.
- Polyurethanes: insulating foams, mattresses, coatings, adhesives, car parts, print rollers, shoe soles, flooring, synthetic fibers, etc. Polyurethane polymers are formed by combining two bi- or higher functional monomers/oligomers.
- Polyurea/ polyurethane hybrids used for abrasion resistant waterproofing coatings.
- Vulcanized rubber.
- Bakelite, a phenol- formaldehyde resin used in electrical insulators and plasticware.
- Duroplast, light but strong material, similar to Bakelite used for making car parts.
- Urea-formaldehyde foam used in plywood, particleboard and medium-density fiberboard.
- Melamine resin used on worktop surfaces. [1]
- Diallyl-phthalate (DAP) used in high temperature and mil-spec electrical connectors and other components. Usually glass filled.
- Epoxy resin [2] used as the matrix component in many fiber reinforced plastics such as glass-reinforced plastic and graphite-reinforced plastic; casting; electronics encapsulation; construction; protective coatings; adhesives; sealing and joining.
- Epoxy novolac resins used for printed circuit boards, electrical encapsulation, adhesives and coatings for metal.
- Benzoxazines, used alone or hybridised with epoxy and phenolic resins, for structural prepregs, liquid molding and film adhesives for composite construction, bonding and repair.
- Polyimides and Bismaleimides used in printed circuit boards and in body parts of modern aircraft, aerospace composite structures, as a coating material and for glass reinforced pipes.
- Cyanate esters or polycyanurates for electronics applications with need for dielectric properties and high glass temperature requirements in aerospace structural composite components.
- Mold or mold runners (the black plastic part in integrated circuits or semiconductors).
- Furan resins used in the manufacture of sustainable biocomposite construction, [3] cements, adhesives, coatings and casting/foundry resins.
- Silicone resins used for thermoset polymer matrix composites and as ceramic matrix composite precursors.
- Thiolyte, an electrical insulating thermoset phenolic laminate material.
- Vinyl ester resins used for wet lay-up laminating, molding and fast setting industrial protection and repair materials.
table
|
Global plastic production by polymer type (2015)
[5]
- ^ Roberto C. Dante, Diego A. Santamaría and Jesús Martín Gil (2009). "Crosslinking and thermal stability of thermosets based on novolak and melamine". Journal of Applied Polymer Science. 114 (6): 4059–4065. doi: 10.1002/app.31114.
- ^ Enrique Guzman; Joël Cugnoni; Thomas Gmür (2014). "Multi-factorial models of a carbon fibre/epoxy composite subjected to accelerated environmental ageing". Composite Structures. 111 (4): 179–192. doi: 10.1016/j.compstruct.2013.12.028.
- ^ T Malaba, J Wang, Journal of Composites, vol. 2015, Article ID 707151, 8 pages, 2015. doi: 10.1155/2015/707151
-
^ Cite error: The named reference
PP&Awas invoked but never defined (see the help page). -
^ Cite error: The named reference
Geyer2017was invoked but never defined (see the help page).
History of antioxidants [1]
| Nuclear magnetic resonance (NMR) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Core concepts | |||||||||||||||
|
|||||||||||||||
| By isotope | |||||||||||||||
|
Trixylylphosphate

| |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
| EC Number |
|
| Properties | |
| C24H27O4P | |
| Molar mass | 410.450 g·mol−1 |
| Appearance | Viscous liquid |
| Density | 1.142 |
| Melting point | −20 °C (−4 °F; 253 K) |
| Boiling point | 394 °C (741 °F; 667 K) |
| 20 µg/L | |
| Hazards | |
| GHS labelling: | |

| |
| Danger | |
| H360F | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Trixylyl phosphate (TXP) is an aromatic phosphate ester historically used as a hydraulic fluid.
Chirality
| Chemical Chirality | ||||
|---|---|---|---|---|
| An aspect of Stereoisomerism | ||||
| Core concepts | ||||
|
||||
| Assigning and naming | ||||
|
| Rotational axis (Cn) |
Improper rotational elements (Sn) | ||
|---|---|---|---|
| Chiral no Sn |
Achiral mirror plane S1 = σ |
Achiral inversion centre S2 = i | |
| C1 |
 |
 |

|
| C2 |
 |
 |

|
Uric
 |

| |
| Lactam form | Lactim form |
 |
 |
pKa1 |

| |
| Lactam form | Lactim form | Urate ion |
Thermosets example table
| Name | Properties | Applications |
|---|---|---|
| Example | Example | Example |
| Example | Example | Example |
| Example | Example | Example |
formaldehyde base
- Bakelite, a phenol- formaldehyde
- Urea-formaldehyde
- Melamine resin
- Benzoxazines, formed by the reaction of phenols, formaldehyde and primary amines
- Duroplast
- Diallyl-phthalate (DAP)
- Epoxy resin
- Epoxy novolac resins
- Polyimides and Bismaleimides
- Cyanate esters or polycyanurates
- Furan resins
- Silicone resins
- Thiolyte,
- Vinyl ester resins used for wet lay-up laminating, molding and fast setting industrial protection and repair materials.
- Polyester resin fiberglass systems: sheet molding compounds and bulk molding compounds; filament winding; wet lay-up lamination; repair compounds and protective coatings.
- Polyurethanes: insulating foams, mattresses, coatings, adhesives, car parts, print rollers, shoe soles, flooring, synthetic fibers, etc. Polyurethane polymers are formed by combining two bi- or higher functional monomers/oligomers.
- Polyurea/ polyurethane hybrids used for abrasion resistant waterproofing coatings.
- Vulcanized rubber.
- Bakelite, a phenol- formaldehyde resin used in electrical insulators and plasticware.
- Duroplast, light but strong material, similar to Bakelite used for making car parts.
- Urea-formaldehyde foam used in plywood, particleboard and medium-density fiberboard.
- Melamine resin used on worktop surfaces. [1]
- Diallyl-phthalate (DAP) used in high temperature and mil-spec electrical connectors and other components. Usually glass filled.
- Epoxy resin [2] used as the matrix component in many fiber reinforced plastics such as glass-reinforced plastic and graphite-reinforced plastic; casting; electronics encapsulation; construction; protective coatings; adhesives; sealing and joining.
- Epoxy novolac resins used for printed circuit boards, electrical encapsulation, adhesives and coatings for metal.
- Benzoxazines, used alone or hybridised with epoxy and phenolic resins, for structural prepregs, liquid molding and film adhesives for composite construction, bonding and repair.
- Polyimides and Bismaleimides used in printed circuit boards and in body parts of modern aircraft, aerospace composite structures, as a coating material and for glass reinforced pipes.
- Cyanate esters or polycyanurates for electronics applications with need for dielectric properties and high glass temperature requirements in aerospace structural composite components.
- Mold or mold runners (the black plastic part in integrated circuits or semiconductors).
- Furan resins used in the manufacture of sustainable biocomposite construction, [3] cements, adhesives, coatings and casting/foundry resins.
- Silicone resins used for thermoset polymer matrix composites and as ceramic matrix composite precursors.
- Thiolyte, an electrical insulating thermoset phenolic laminate material.
- Vinyl ester resins used for wet lay-up laminating, molding and fast setting industrial protection and repair materials.
table
|
Global plastic production by polymer type (2015)
[5]
- ^ Roberto C. Dante, Diego A. Santamaría and Jesús Martín Gil (2009). "Crosslinking and thermal stability of thermosets based on novolak and melamine". Journal of Applied Polymer Science. 114 (6): 4059–4065. doi: 10.1002/app.31114.
- ^ Enrique Guzman; Joël Cugnoni; Thomas Gmür (2014). "Multi-factorial models of a carbon fibre/epoxy composite subjected to accelerated environmental ageing". Composite Structures. 111 (4): 179–192. doi: 10.1016/j.compstruct.2013.12.028.
- ^ T Malaba, J Wang, Journal of Composites, vol. 2015, Article ID 707151, 8 pages, 2015. doi: 10.1155/2015/707151
-
^ Cite error: The named reference
PP&Awas invoked but never defined (see the help page). -
^ Cite error: The named reference
Geyer2017was invoked but never defined (see the help page).