February 2010
Quantum mechanics
Physics
March 2010
Reducing agents
Organic reagents
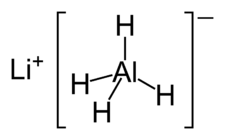

April 2010
Hydroboration
Organic synthesis

Mechanism shown is converting 1-hexene to 1-hexanol, first performed in 1957.
May 2010
Steroids
Aromatic chemistry
May 2010 - Steroids
A steroid is a type of organic compound that contains a specific arrangement of four rings that are joined to each other. The sterane core of steroids is composed of seventeen carbon atoms bonded together to form four fused rings: three cyclohexane rings (designated as rings A, B, and C in the figure to the right) and one cyclopentane ring (the D ring). The steroids vary by the functional groups attached to these rings and by the oxidation state of the rings. Sterols are special forms of steroids, with a hydroxyl group at position-3 and a skeleton derived from cholestane.
June 2010
Photoelectric effect
Physics
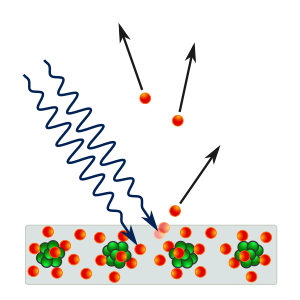
The photoelectric effect is a phenomenon in which
electrons are emitted from matter (metals and non-metallic solids, liquids or gases) as a consequence of their absorption of energy from
electromagnetic radiation of very short
wavelength, such as
visible or
ultraviolet
light, or "photoelectrons". As it was first observed by
Heinrich Hertz in 1887. Hertz observed and then showed that
electrodes illuminated with ultraviolet light create electric sparks more easily.
The study of the photoelectric effect led to important steps in understanding the quantum nature of light and electrons and influenced the formation of the concept of wave–particle duality, which is the concept that all matter exhibits both wave-like and particle-like properties.
July 2010
DNA Replication
Biochemistry

February 2010
Quantum mechanics
Physics
March 2010
Reducing agents
Organic reagents


April 2010
Hydroboration
Organic synthesis

Mechanism shown is converting 1-hexene to 1-hexanol, first performed in 1957.
May 2010
Steroids
Aromatic chemistry
May 2010 - Steroids
A steroid is a type of organic compound that contains a specific arrangement of four rings that are joined to each other. The sterane core of steroids is composed of seventeen carbon atoms bonded together to form four fused rings: three cyclohexane rings (designated as rings A, B, and C in the figure to the right) and one cyclopentane ring (the D ring). The steroids vary by the functional groups attached to these rings and by the oxidation state of the rings. Sterols are special forms of steroids, with a hydroxyl group at position-3 and a skeleton derived from cholestane.
June 2010
Photoelectric effect
Physics

The photoelectric effect is a phenomenon in which
electrons are emitted from matter (metals and non-metallic solids, liquids or gases) as a consequence of their absorption of energy from
electromagnetic radiation of very short
wavelength, such as
visible or
ultraviolet
light, or "photoelectrons". As it was first observed by
Heinrich Hertz in 1887. Hertz observed and then showed that
electrodes illuminated with ultraviolet light create electric sparks more easily.
The study of the photoelectric effect led to important steps in understanding the quantum nature of light and electrons and influenced the formation of the concept of wave–particle duality, which is the concept that all matter exhibits both wave-like and particle-like properties.
July 2010
DNA Replication
Biochemistry



