 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Plan B, others |
| Other names | LNG; d-Norgestrel; d(–)-Norgestrel; D-Norgestrel; WY-5104; SH-90999; NSC-744007; 18-Methylnorethisterone; 17α-Ethynyl-18-methyl-19-nortestosterone; 17α-Ethynyl-18-methylestr-4-en-17β-ol-3-one; 13β-Ethyl-17α-hydroxy-18,19-dinorpregn-4-en-20-yn-3-one |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a610021 |
|
Pregnancy category | |
|
Routes of administration | By mouth, transdermal patch, intrauterine device, subcutaneous implant |
| Drug class | Progestogen; progestin |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 95% (range 85–100%) [2] [3] |
| Protein binding | 98% (50% to albumin, 48% to SHBG) [2] |
| Metabolism | Liver ( reduction, hydroxylation, conjugation) [2] [4] |
| Metabolites | • 5α-Dihydro-@@@6@@@ [2] |
| Elimination half-life | 24–32 hours [2] |
| Excretion |
Urine: 20–67% Feces: 21–34% [4] |
| Identifiers | |
| |
| Chemical and physical data | |
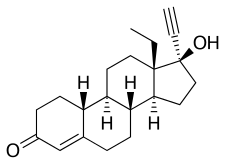
| Formula | C21H28O2 |
| Molar mass | 312.453 g·mol−1 |
| 3D model ( JSmol) | |
| Melting point | 235 to 237 °C (455 to 459 °F) |
| |
| |
| (verify) | |
Levonorgestrel is a hormonal medication which is used in a number of birth control methods. [6] It is combined with an estrogen to make combination birth control pills. [8] As an emergency birth control, sold under the brand name Plan B among others, it is useful within 120 hours of unprotected sex. [6] The more time that has passed since sex, the less effective the medication becomes, and it does not work after pregnancy ( implantation) has occurred. [6] It decreases the chances of pregnancy by 57 to 93%. [9] In an intrauterine device (IUD), such as Mirena among others, it is effective for the long-term prevention of pregnancy. [6] A levonorgestrel-releasing implant is also available in some countries. [10]
Common side effects include nausea, breast tenderness, headaches, and increased, decreased, or irregular menstrual bleeding. [6] When used as an emergency contraceptive, if pregnancy occurs, there is no evidence that its use harms the baby. [6] It is safe to use during breastfeeding. [6] Birth control that contains levonorgestrel will not change the risk of sexually transmitted infections. [6] It is a progestin and has effects similar to those of the hormone progesterone. [6] It works primarily by preventing ovulation and closing off the cervix to prevent the passage of sperm. [6]
Levonorgestrel was patented in 1960 and introduced for medical use together with ethinylestradiol in 1970. [11] [12] It is on the World Health Organization's List of Essential Medicines. [13] It is available as a generic medication. [14] The wholesale cost in the developing world is between $0.23 and $1.65 US for the dose required for emergency birth control. [15] In the United States, levonorgestrel-containing emergency birth control is available over the counter (OTC) for all ages. [16] In 2016, it was the 223rd most commonly prescribed medication in the United States, with more than two million prescriptions. [17]
References
- ^ a b c "Levonorgestrel Use During Pregnancy". Drugs.com. 23 March 2020. Archived from the original on 2 July 2020. Retrieved 29 June 2020.
- ^ a b c d e Kuhl H (August 2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric. 8 Suppl 1: 3–63. doi: 10.1080/13697130500148875. PMID 16112947. S2CID 24616324.
- ^ Fotherby K (August 1996). "Bioavailability of orally administered sex steroids used in oral contraception and hormone replacement therapy". Contraception. 54 (2): 59–69. doi: 10.1016/0010-7824(96)00136-9. PMID 8842581.
- ^ a b Shoupe, Donna; Haseltine, Florence P. (6 December 2012). Contraception. Springer Science & Business Media. pp. 22–. ISBN 978-1-4612-2730-4. Archived from the original on 28 August 2021. Retrieved 15 April 2018.
-
^ Cite error: The named reference
MSF2020was invoked but never defined (see the help page). - ^ a b c d e f g h i j k "Progestins (Etonogestrel, Levonorgestrel, Norethindrone)". The American Society of Health-System Pharmacists. Archived from the original on 2015-09-07. Retrieved Aug 21, 2015.
-
^ Cite error: The named reference
WHO2020DDDwas invoked but never defined (see the help page). - ^ Postgraduate Gynecology. Jaypee Brothers Medical Pub. 2011. p. 159. ISBN 9789350250822. Archived from the original on 2015-09-26.
- ^ Gemzell-Danielsson, K (November 2010). "Mechanism of action of emergency contraception". Contraception. 82 (5): 404–9. doi: 10.1016/j.contraception.2010.05.004. PMID 20933113.
- ^ "Chapter 1". Research on reproductive health at WHO : biennial report 2000-2001. Geneva: World health organization. 2002. ISBN 9789241562089. Archived from the original on 2015-09-26.
- ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 479. ISBN 9783527607495. Archived from the original on 2021-08-28. Retrieved 2020-08-05.
-
^ Cite error: The named reference
Roth2014was invoked but never defined (see the help page). - ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl: 10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Hamilton, Richard J. (2014). Tarascon pocket pharmacopoeia : 2014 deluxe lab-pocket edition (15th ed.). Sudbury: Jones & Bartlett Learning. pp. 310–312. ISBN 9781284053999. Archived from the original on 2015-09-26.
- ^ "Levonorgestrel". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 21 August 2015.
- ^ "FDA approves Plan B One-Step emergency contraceptive for use without a prescription for all women of child-bearing potential" (Press release). June 20, 2013. Archived from the original on 14 January 2016. Retrieved 2 February 2016.
- ^ "Levonorgestrel - Drug Usage Statistics". ClinCalc. Archived from the original on 2 July 2020. Retrieved 11 April 2020.
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Plan B, others |
| Other names | LNG; d-Norgestrel; d(–)-Norgestrel; D-Norgestrel; WY-5104; SH-90999; NSC-744007; 18-Methylnorethisterone; 17α-Ethynyl-18-methyl-19-nortestosterone; 17α-Ethynyl-18-methylestr-4-en-17β-ol-3-one; 13β-Ethyl-17α-hydroxy-18,19-dinorpregn-4-en-20-yn-3-one |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a610021 |
|
Pregnancy category | |
|
Routes of administration | By mouth, transdermal patch, intrauterine device, subcutaneous implant |
| Drug class | Progestogen; progestin |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 95% (range 85–100%) [2] [3] |
| Protein binding | 98% (50% to albumin, 48% to SHBG) [2] |
| Metabolism | Liver ( reduction, hydroxylation, conjugation) [2] [4] |
| Metabolites | • 5α-Dihydro-@@@6@@@ [2] |
| Elimination half-life | 24–32 hours [2] |
| Excretion |
Urine: 20–67% Feces: 21–34% [4] |
| Identifiers | |
| |
| Chemical and physical data | |
| Formula | C21H28O2 |
| Molar mass | 312.453 g·mol−1 |
| 3D model ( JSmol) | |
| Melting point | 235 to 237 °C (455 to 459 °F) |
| |
| |
| (verify) | |
Levonorgestrel is a hormonal medication which is used in a number of birth control methods. [6] It is combined with an estrogen to make combination birth control pills. [8] As an emergency birth control, sold under the brand name Plan B among others, it is useful within 120 hours of unprotected sex. [6] The more time that has passed since sex, the less effective the medication becomes, and it does not work after pregnancy ( implantation) has occurred. [6] It decreases the chances of pregnancy by 57 to 93%. [9] In an intrauterine device (IUD), such as Mirena among others, it is effective for the long-term prevention of pregnancy. [6] A levonorgestrel-releasing implant is also available in some countries. [10]
Common side effects include nausea, breast tenderness, headaches, and increased, decreased, or irregular menstrual bleeding. [6] When used as an emergency contraceptive, if pregnancy occurs, there is no evidence that its use harms the baby. [6] It is safe to use during breastfeeding. [6] Birth control that contains levonorgestrel will not change the risk of sexually transmitted infections. [6] It is a progestin and has effects similar to those of the hormone progesterone. [6] It works primarily by preventing ovulation and closing off the cervix to prevent the passage of sperm. [6]
Levonorgestrel was patented in 1960 and introduced for medical use together with ethinylestradiol in 1970. [11] [12] It is on the World Health Organization's List of Essential Medicines. [13] It is available as a generic medication. [14] The wholesale cost in the developing world is between $0.23 and $1.65 US for the dose required for emergency birth control. [15] In the United States, levonorgestrel-containing emergency birth control is available over the counter (OTC) for all ages. [16] In 2016, it was the 223rd most commonly prescribed medication in the United States, with more than two million prescriptions. [17]
References
- ^ a b c "Levonorgestrel Use During Pregnancy". Drugs.com. 23 March 2020. Archived from the original on 2 July 2020. Retrieved 29 June 2020.
- ^ a b c d e Kuhl H (August 2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric. 8 Suppl 1: 3–63. doi: 10.1080/13697130500148875. PMID 16112947. S2CID 24616324.
- ^ Fotherby K (August 1996). "Bioavailability of orally administered sex steroids used in oral contraception and hormone replacement therapy". Contraception. 54 (2): 59–69. doi: 10.1016/0010-7824(96)00136-9. PMID 8842581.
- ^ a b Shoupe, Donna; Haseltine, Florence P. (6 December 2012). Contraception. Springer Science & Business Media. pp. 22–. ISBN 978-1-4612-2730-4. Archived from the original on 28 August 2021. Retrieved 15 April 2018.
-
^ Cite error: The named reference
MSF2020was invoked but never defined (see the help page). - ^ a b c d e f g h i j k "Progestins (Etonogestrel, Levonorgestrel, Norethindrone)". The American Society of Health-System Pharmacists. Archived from the original on 2015-09-07. Retrieved Aug 21, 2015.
-
^ Cite error: The named reference
WHO2020DDDwas invoked but never defined (see the help page). - ^ Postgraduate Gynecology. Jaypee Brothers Medical Pub. 2011. p. 159. ISBN 9789350250822. Archived from the original on 2015-09-26.
- ^ Gemzell-Danielsson, K (November 2010). "Mechanism of action of emergency contraception". Contraception. 82 (5): 404–9. doi: 10.1016/j.contraception.2010.05.004. PMID 20933113.
- ^ "Chapter 1". Research on reproductive health at WHO : biennial report 2000-2001. Geneva: World health organization. 2002. ISBN 9789241562089. Archived from the original on 2015-09-26.
- ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 479. ISBN 9783527607495. Archived from the original on 2021-08-28. Retrieved 2020-08-05.
-
^ Cite error: The named reference
Roth2014was invoked but never defined (see the help page). - ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl: 10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Hamilton, Richard J. (2014). Tarascon pocket pharmacopoeia : 2014 deluxe lab-pocket edition (15th ed.). Sudbury: Jones & Bartlett Learning. pp. 310–312. ISBN 9781284053999. Archived from the original on 2015-09-26.
- ^ "Levonorgestrel". International Drug Price Indicator Guide. Archived from the original on 22 January 2018. Retrieved 21 August 2015.
- ^ "FDA approves Plan B One-Step emergency contraceptive for use without a prescription for all women of child-bearing potential" (Press release). June 20, 2013. Archived from the original on 14 January 2016. Retrieved 2 February 2016.
- ^ "Levonorgestrel - Drug Usage Statistics". ClinCalc. Archived from the original on 2 July 2020. Retrieved 11 April 2020.