Tryptamine Information
Tryptamine is an indolamine metabolite of the essential amino acid, tryptophan. [1] [2] The chemical structure is defined by an indole - a fused benzene and pyrrole ring, and a 2-aminoethyl group at the third carbon. [1] The structure of tryptamine is a shared feature of certain aminergic neuromodulators including melatonin, serotonin, bufotenin and psychedelic derivatives such as dimethyltryptamine (DMT), psilocybin, psilocin and others. [1] [3] [4] [5] Various amounts of tryptamine and related indolamine alkaloids are present in plants, fungi and animals. [6] Tryptamine has been shown to activate trace amine-associated receptors expressed in the mammalian brain, and regulates the activity of of dopaminergic, serotonergic and glutamatergic systems. [7] [8] In the human gut, symbiotic bacteria convert dietary tryptophan to tryptamine, which activates 5-HT4 receptors and regulates gastrointestinal motility. [2] [9] [10] Multiple tryptamine-derived drugs have been developed to treat migraines, while trace amine-associated receptors are being explored as a potential treatment target for neuropsychiatric disorders. [11] [12] [13] [6]
For a list of tryptamine derivatives, see: List of substituted tryptamines.

Natural Occurrences
For a list of plants, fungi and animals containing tryptamines, see: List of psychoactive plants and List of naturally occurring tryptamines.
Mammalian Brain
Endogenous levels of tryptamine in the mammalian brain are less than 100ng per gram of tissue. [4] [8] However, elevated levels of trace amines have been observed in neuropsychiatric disorders, such as bipolar depression and schizophrenia. [14]
Mammalian Gut Microbiome
Tryptamine is relatively abundant in the gut and feces of humans and rodents. [2] [9] Commensal bacteria, including Ruminococcus gnavus and Clostridium sporogenes in the gastrointestinal tract, possess the enzyme tryptophan decarboxylase, which aids in the conversion of dietary tryptophan to tryptamine. [2] Tryptamine is a ligand for gut epithelial serotonin type 4 (5-HT4) receptors and regulates gastrointestinal electrolyte balance through colonic secretions. [9]
Metabolism of Tryptamine
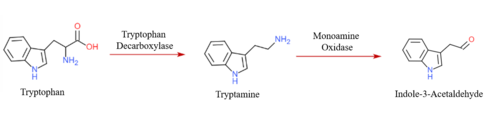
Biosynthesis
To yield tryptamine in vivo, tryptophan decarboxylase removes the carboxylic acid group on the α-carbon of tryptophan. [4] Synthetic modifications to tryptamine can produce serotonin and melatonin, however it is not the main pathway of endogenous neurotransmitter synthesis. [15]
Catabolism
Monoamine oxidases A and B are the primary enzymes involved in tryptamine metabolism to produce indole-3-acetaldehyde, however it is unclear which isoform is specific to tryptamine degradation. [16]
Mechanism of Action and Biological Effects
Neuromodulation
Tryptamine can weakly activate the trace amine-associated receptor, TAAR1 (hTAAR1 in humans). [17] [7] [18] Limited studies have considered tryptamine to be a trace neuromodulator capable of regulating the activity of neuronal cell responses without binding to the associated postsynaptic receptors. [18] [14]
hTAAR1
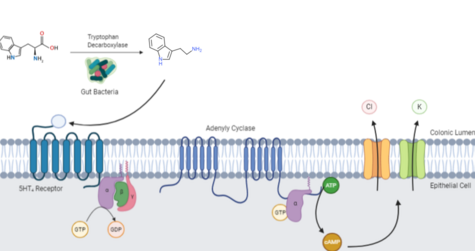
hTAAR1 is a stimulatory G-protein coupled receptor (GPCR) that is weakly expressed in the intracellular compartment of both pre- and postsynaptic neurons. [8] Tryptamine and other hTAAR1 agonists can increase neuronal firing by inhibiting neurotransmitter recycling through cAMP-dependent phosphorylation of the monoamine reuptake transporter. [19] [14] This mechanism increases the amount of neurotransmitter in the synaptic cleft, subsequently increasing postsynaptic receptor binding and neuronal activation. [14] Conversely, when hTAAR1 are colocalized with G protein-coupled inwardly-rectifying potassium channels (GIRKs), receptor activation reduces neuronal firing by facilitating membrane hyperpolarization through the efflux of potassium ions. [14] The balance between the inhibitory and excitatory activity of hTAAR1 activation highlights the role of tryptamine in the regulation of neural activity. [20]
Activation of hTAAR1 is under investigation as a novel treatment for depression, addiction and schizophrenia. [21] hTAAR1 is primarily expressed in brain structures associated with dopamine systems, such as the ventral tegmental area (VTA) and serotonin systems in the dorsal raphe nuclei (DRN). [21] Additionally, the hTAAR1 gene is localized at 6q23.2 on the human chromosome, which is a susceptibility locus for mood disorders and schizophrenia. [22] Activation of TAAR1 suggests a potential novel treatment for neuropsychiatric disorders, as TAAR1 agonists produce anti-depressive activity, increased cognition, reduced stress and anti-addiction effects. [20] [22]
Gastrointestinal Motility
Tryptamine produced by mutualistic bacteria in the human gut activates serotonin GPCRs ubiquitously expressed along the colonic epithelium. [9] Upon tryptamine binding, the activated 5-HT4 receptor undergoes a conformational change which allows its Gs alpha subunit to exchange GDP for GTP, and its liberation from the 5-HT4 receptor and βγ subunit. [9] GTP-bound Gs activates adenylyl cyclase, which catalyzes the conversion of ATP into cyclic adenosine monophosphate (cAMP). [9] cAMP opens chloride and potassium ion channels to drive colonic electrolyte secretion and promote intestinal motility. [10] [23]
Pharmacokinetics
| Tryptamine | Human TAAR1 | Mouse TAAR1 | Rat TAAR | |||
|---|---|---|---|---|---|---|
| EC50 | Ki | EC50 | Ki | EC50 | Ki | |
| Tryptamine | 21 | N/A | 2.7 | 1.4 | 0.41 | 0.13 |
| Serotonin | >50 | N/A | >50 | N/A | 5.2 | N/A |
| Psilocin | >30 | N/A | 2.7 | 17 | 0.92 | 1.4 |
| DMT | >10 | N/A | 1.2 | 3.3 | 1.5 | 22 |
|
EC50 and Ki values are in micromolar (μM).
EC50 reflects the amount
of tryptamine required to elicit 50% of the maximum TAAR1 response. The smaller the Ki value, the stronger the tryptamine binds to the receptor. | ||||||
Tryptamine-Based Therapeutics
| Drug | Mechanism | Treatment | Effect | Structure |
|---|---|---|---|---|
| Sumatriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Rizatriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Zolmitriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Almotriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Eletriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Frovatriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Naratriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
See also
- Tryptophan
- Substituted tryptamines
- Trace amines
- Serotonin receptor agonist
- Human trace amine associated receptor 1
- Neuromodulation
 | This is a user sandbox of
Kwingfield. You can use it for testing or practicing edits. This is not the sandbox where you should draft your assigned article for a dashboard.wikiedu.org course. To find the right sandbox for your assignment, visit your Dashboard course page and follow the Sandbox Draft link for your assigned article in the My Articles section. |
- ^ a b c PubChem. "Tryptamine". pubchem.ncbi.nlm.nih.gov. Retrieved 2020-12-01.
- ^
a
b
c
d Jenkins, Trisha A.; Nguyen, Jason C. D.; Polglaze, Kate E.; Bertrand, Paul P. (2016-01-20).
"Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis". Nutrients. 8 (1).
doi:
10.3390/nu8010056.
ISSN
2072-6643.
PMC
4728667.
PMID
26805875.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Tylš, Filip; Páleníček, Tomáš; Horáček, Jiří (2014-03-01). "Psilocybin – Summary of knowledge and new perspectives". European Neuropsychopharmacology. 24 (3): 342–356. doi: 10.1016/j.euroneuro.2013.12.006. ISSN 0924-977X.
- ^ a b c Tittarelli, Roberta; Mannocchi, Giulio; Pantano, Flaminia; Romolo, Francesco Saverio (2015). "Recreational Use, Analysis and Toxicity of Tryptamines". Current Neuropharmacology. 13 (1): 26–46. doi: 10.2174/1570159X13666141210222409. ISSN 1570-159X. PMC 4462041. PMID 26074742.
- ^ "The Ayahuasca Phenomenon". MAPS. Retrieved 2020-10-03.
- ^
a
b Kousara, Shazia; Anjuma, Sadia Noreen; Jaleela, Farrukh; Khana, Jallat; Naseema, Sidra (2017).
"Biomedical Significance of Tryptamine: A Review". Journal of Pharmacovigilance. 5 (5).
doi:
10.4172/2329-6887.1000239.
ISSN
2329-6887.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ a b Khan, Muhammad Zahid; Nawaz, Waqas (2016-10-01). "The emerging roles of human trace amines and human trace amine-associated receptors (hTAARs) in central nervous system". Biomedicine & Pharmacotherapy. 83: 439–449. doi: 10.1016/j.biopha.2016.07.002. ISSN 0753-3322.
- ^ a b c "Pharmacology of human trace amine-associated receptors: Therapeutic opportunities and challenges". Pharmacology & Therapeutics. 180: 161–180. 2017-12-01. doi: 10.1016/j.pharmthera.2017.07.002. ISSN 0163-7258.
- ^ a b c d e f Bhattarai, Yogesh; Williams, Brianna B.; Battaglioli, Eric J.; Whitaker, Weston R.; Till, Lisa; Grover, Madhusudan; Linden, David R.; Akiba, Yasutada; Kandimalla, Karunya K.; Zachos, Nicholas C.; Kaunitz, Jonathan D. (2018-06-13). "Gut Microbiota-Produced Tryptamine Activates an Epithelial G-Protein-Coupled Receptor to Increase Colonic Secretion". Cell Host & Microbe. 23 (6): 775–785.e5. doi: 10.1016/j.chom.2018.05.004. ISSN 1931-3128. PMID 29902441.
- ^ a b Field, Michael (2003). "Intestinal ion transport and the pathophysiology of diarrhea". Journal of Clinical Investigation. 111 (7): 931–943. doi: 10.1172/JCI200318326. ISSN 0021-9738. PMID 12671039.
- ^ a b c d e f g h "Serotonin Receptor Agonists (Triptans)", LiverTox: Clinical and Research Information on Drug-Induced Liver Injury, Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases, 2012, PMID 31644023, retrieved 2020-10-15
- ^ "New Compound Related to Psychedelic Ibogaine Could Treat Addiction, Depression". UC Davis. 2020-12-09. Retrieved 2020-12-11.
-
^ ServiceDec. 9, Robert F.; 2020; Am, 11:45 (2020-12-09).
"Chemists re-engineer a psychedelic to treat depression and addiction in rodents". Science | AAAS. Retrieved 2020-12-11.
{{ cite web}}:|last2=has numeric name ( help)CS1 maint: numeric names: authors list ( link) - ^ a b c d e Miller, Gregory M. (2011). "The Emerging Role of Trace Amine Associated Receptor 1 in the Functional Regulation of Monoamine Transporters and Dopaminergic Activity". Journal of neurochemistry. 116 (2): 164–176. doi: 10.1111/j.1471-4159.2010.07109.x. ISSN 0022-3042. PMC 3005101. PMID 21073468.
-
^
"Serotonin Synthesis and Metabolism". Sigma Aldrich. 2020.
{{ cite web}}: CS1 maint: url-status ( link) - ^ "MetaCyc L-tryptophan degradation VI (via tryptamine)". biocyc.org. Retrieved 2020-12-11.
- ^ Yu, Ai-Ming; Granvil, Camille P.; Haining, Robert L.; Krausz, Kristopher W.; Corchero, Javier; Küpfer, Adrian; Idle, Jeffrey R.; Gonzalez, Frank J. (2003-02-01). "The Relative Contribution of Monoamine Oxidase and Cytochrome P450 Isozymes to the Metabolic Deamination of the Trace Amine Tryptamine". Journal of Pharmacology and Experimental Therapeutics. 304 (2): 539–546. doi: 10.1124/jpet.102.043786. ISSN 0022-3565. PMID 12538805.
- ^ a b Zucchi, R; Chiellini, G; Scanlan, T S; Grandy, D K (2006). "Trace amine-associated receptors and their ligands". British Journal of Pharmacology. 149 (8): 967–978. doi: 10.1038/sj.bjp.0706948. ISSN 0007-1188. PMC 2014643. PMID 17088868.
- ^ Jing, Li; Li, Jun-Xu (2015-08-15). "Trace amine-associated receptor 1: a promising target for the treatment of psychostimulant addiction". European journal of pharmacology. 761: 345–352. doi: 10.1016/j.ejphar.2015.06.019. ISSN 0014-2999. PMC 4532615. PMID 26092759.
- ^ a b Grandy, David K.; Miller, Gregory M.; Li, Jun-Xu (2016-02-01). ""TAARgeting Addiction" The Alamo Bears Witness to Another Revolution". Drug and alcohol dependence. 159: 9–16. doi: 10.1016/j.drugalcdep.2015.11.014. ISSN 0376-8716. PMC 4724540. PMID 26644139.
- ^ a b "Pharmacology of human trace amine-associated receptors: Therapeutic opportunities and challenges". Pharmacology & Therapeutics. 180: 161–180. 2017-12-01. doi: 10.1016/j.pharmthera.2017.07.002. ISSN 0163-7258.
- ^ a b c Gainetdinov, Raul R.; Hoener, Marius C.; Berry, Mark D. (2018-07-01). "Trace Amines and Their Receptors". Pharmacological Reviews. 70 (3): 549–620. doi: 10.1124/pr.117.015305. ISSN 0031-6997. PMID 29941461.
- ^ "Microbiome-Lax May Relieve Constipation". GEN - Genetic Engineering and Biotechnology News. 2018-06-15. Retrieved 2020-12-11.
Tryptamine Information
Tryptamine is an indolamine metabolite of the essential amino acid, tryptophan. [1] [2] The chemical structure is defined by an indole - a fused benzene and pyrrole ring, and a 2-aminoethyl group at the third carbon. [1] The structure of tryptamine is a shared feature of certain aminergic neuromodulators including melatonin, serotonin, bufotenin and psychedelic derivatives such as dimethyltryptamine (DMT), psilocybin, psilocin and others. [1] [3] [4] [5] Various amounts of tryptamine and related indolamine alkaloids are present in plants, fungi and animals. [6] Tryptamine has been shown to activate trace amine-associated receptors expressed in the mammalian brain, and regulates the activity of of dopaminergic, serotonergic and glutamatergic systems. [7] [8] In the human gut, symbiotic bacteria convert dietary tryptophan to tryptamine, which activates 5-HT4 receptors and regulates gastrointestinal motility. [2] [9] [10] Multiple tryptamine-derived drugs have been developed to treat migraines, while trace amine-associated receptors are being explored as a potential treatment target for neuropsychiatric disorders. [11] [12] [13] [6]
For a list of tryptamine derivatives, see: List of substituted tryptamines.

Natural Occurrences
For a list of plants, fungi and animals containing tryptamines, see: List of psychoactive plants and List of naturally occurring tryptamines.
Mammalian Brain
Endogenous levels of tryptamine in the mammalian brain are less than 100ng per gram of tissue. [4] [8] However, elevated levels of trace amines have been observed in neuropsychiatric disorders, such as bipolar depression and schizophrenia. [14]
Mammalian Gut Microbiome
Tryptamine is relatively abundant in the gut and feces of humans and rodents. [2] [9] Commensal bacteria, including Ruminococcus gnavus and Clostridium sporogenes in the gastrointestinal tract, possess the enzyme tryptophan decarboxylase, which aids in the conversion of dietary tryptophan to tryptamine. [2] Tryptamine is a ligand for gut epithelial serotonin type 4 (5-HT4) receptors and regulates gastrointestinal electrolyte balance through colonic secretions. [9]
Metabolism of Tryptamine

Biosynthesis
To yield tryptamine in vivo, tryptophan decarboxylase removes the carboxylic acid group on the α-carbon of tryptophan. [4] Synthetic modifications to tryptamine can produce serotonin and melatonin, however it is not the main pathway of endogenous neurotransmitter synthesis. [15]
Catabolism
Monoamine oxidases A and B are the primary enzymes involved in tryptamine metabolism to produce indole-3-acetaldehyde, however it is unclear which isoform is specific to tryptamine degradation. [16]
Mechanism of Action and Biological Effects
Neuromodulation
Tryptamine can weakly activate the trace amine-associated receptor, TAAR1 (hTAAR1 in humans). [17] [7] [18] Limited studies have considered tryptamine to be a trace neuromodulator capable of regulating the activity of neuronal cell responses without binding to the associated postsynaptic receptors. [18] [14]
hTAAR1

hTAAR1 is a stimulatory G-protein coupled receptor (GPCR) that is weakly expressed in the intracellular compartment of both pre- and postsynaptic neurons. [8] Tryptamine and other hTAAR1 agonists can increase neuronal firing by inhibiting neurotransmitter recycling through cAMP-dependent phosphorylation of the monoamine reuptake transporter. [19] [14] This mechanism increases the amount of neurotransmitter in the synaptic cleft, subsequently increasing postsynaptic receptor binding and neuronal activation. [14] Conversely, when hTAAR1 are colocalized with G protein-coupled inwardly-rectifying potassium channels (GIRKs), receptor activation reduces neuronal firing by facilitating membrane hyperpolarization through the efflux of potassium ions. [14] The balance between the inhibitory and excitatory activity of hTAAR1 activation highlights the role of tryptamine in the regulation of neural activity. [20]
Activation of hTAAR1 is under investigation as a novel treatment for depression, addiction and schizophrenia. [21] hTAAR1 is primarily expressed in brain structures associated with dopamine systems, such as the ventral tegmental area (VTA) and serotonin systems in the dorsal raphe nuclei (DRN). [21] Additionally, the hTAAR1 gene is localized at 6q23.2 on the human chromosome, which is a susceptibility locus for mood disorders and schizophrenia. [22] Activation of TAAR1 suggests a potential novel treatment for neuropsychiatric disorders, as TAAR1 agonists produce anti-depressive activity, increased cognition, reduced stress and anti-addiction effects. [20] [22]
Gastrointestinal Motility
Tryptamine produced by mutualistic bacteria in the human gut activates serotonin GPCRs ubiquitously expressed along the colonic epithelium. [9] Upon tryptamine binding, the activated 5-HT4 receptor undergoes a conformational change which allows its Gs alpha subunit to exchange GDP for GTP, and its liberation from the 5-HT4 receptor and βγ subunit. [9] GTP-bound Gs activates adenylyl cyclase, which catalyzes the conversion of ATP into cyclic adenosine monophosphate (cAMP). [9] cAMP opens chloride and potassium ion channels to drive colonic electrolyte secretion and promote intestinal motility. [10] [23]
Pharmacokinetics
| Tryptamine | Human TAAR1 | Mouse TAAR1 | Rat TAAR | |||
|---|---|---|---|---|---|---|
| EC50 | Ki | EC50 | Ki | EC50 | Ki | |
| Tryptamine | 21 | N/A | 2.7 | 1.4 | 0.41 | 0.13 |
| Serotonin | >50 | N/A | >50 | N/A | 5.2 | N/A |
| Psilocin | >30 | N/A | 2.7 | 17 | 0.92 | 1.4 |
| DMT | >10 | N/A | 1.2 | 3.3 | 1.5 | 22 |
|
EC50 and Ki values are in micromolar (μM).
EC50 reflects the amount
of tryptamine required to elicit 50% of the maximum TAAR1 response. The smaller the Ki value, the stronger the tryptamine binds to the receptor. | ||||||
Tryptamine-Based Therapeutics
| Drug | Mechanism | Treatment | Effect | Structure |
|---|---|---|---|---|
| Sumatriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Rizatriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Zolmitriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Almotriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Eletriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Frovatriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
| Naratriptan [11] | 5-HT1B and 5-HT1D agonist | Migraine Headaches | Vasoconstriction of brain blood vessels |  |
See also
- Tryptophan
- Substituted tryptamines
- Trace amines
- Serotonin receptor agonist
- Human trace amine associated receptor 1
- Neuromodulation
 | This is a user sandbox of
Kwingfield. You can use it for testing or practicing edits. This is not the sandbox where you should draft your assigned article for a dashboard.wikiedu.org course. To find the right sandbox for your assignment, visit your Dashboard course page and follow the Sandbox Draft link for your assigned article in the My Articles section. |
- ^ a b c PubChem. "Tryptamine". pubchem.ncbi.nlm.nih.gov. Retrieved 2020-12-01.
- ^
a
b
c
d Jenkins, Trisha A.; Nguyen, Jason C. D.; Polglaze, Kate E.; Bertrand, Paul P. (2016-01-20).
"Influence of Tryptophan and Serotonin on Mood and Cognition with a Possible Role of the Gut-Brain Axis". Nutrients. 8 (1).
doi:
10.3390/nu8010056.
ISSN
2072-6643.
PMC
4728667.
PMID
26805875.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Tylš, Filip; Páleníček, Tomáš; Horáček, Jiří (2014-03-01). "Psilocybin – Summary of knowledge and new perspectives". European Neuropsychopharmacology. 24 (3): 342–356. doi: 10.1016/j.euroneuro.2013.12.006. ISSN 0924-977X.
- ^ a b c Tittarelli, Roberta; Mannocchi, Giulio; Pantano, Flaminia; Romolo, Francesco Saverio (2015). "Recreational Use, Analysis and Toxicity of Tryptamines". Current Neuropharmacology. 13 (1): 26–46. doi: 10.2174/1570159X13666141210222409. ISSN 1570-159X. PMC 4462041. PMID 26074742.
- ^ "The Ayahuasca Phenomenon". MAPS. Retrieved 2020-10-03.
- ^
a
b Kousara, Shazia; Anjuma, Sadia Noreen; Jaleela, Farrukh; Khana, Jallat; Naseema, Sidra (2017).
"Biomedical Significance of Tryptamine: A Review". Journal of Pharmacovigilance. 5 (5).
doi:
10.4172/2329-6887.1000239.
ISSN
2329-6887.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ a b Khan, Muhammad Zahid; Nawaz, Waqas (2016-10-01). "The emerging roles of human trace amines and human trace amine-associated receptors (hTAARs) in central nervous system". Biomedicine & Pharmacotherapy. 83: 439–449. doi: 10.1016/j.biopha.2016.07.002. ISSN 0753-3322.
- ^ a b c "Pharmacology of human trace amine-associated receptors: Therapeutic opportunities and challenges". Pharmacology & Therapeutics. 180: 161–180. 2017-12-01. doi: 10.1016/j.pharmthera.2017.07.002. ISSN 0163-7258.
- ^ a b c d e f Bhattarai, Yogesh; Williams, Brianna B.; Battaglioli, Eric J.; Whitaker, Weston R.; Till, Lisa; Grover, Madhusudan; Linden, David R.; Akiba, Yasutada; Kandimalla, Karunya K.; Zachos, Nicholas C.; Kaunitz, Jonathan D. (2018-06-13). "Gut Microbiota-Produced Tryptamine Activates an Epithelial G-Protein-Coupled Receptor to Increase Colonic Secretion". Cell Host & Microbe. 23 (6): 775–785.e5. doi: 10.1016/j.chom.2018.05.004. ISSN 1931-3128. PMID 29902441.
- ^ a b Field, Michael (2003). "Intestinal ion transport and the pathophysiology of diarrhea". Journal of Clinical Investigation. 111 (7): 931–943. doi: 10.1172/JCI200318326. ISSN 0021-9738. PMID 12671039.
- ^ a b c d e f g h "Serotonin Receptor Agonists (Triptans)", LiverTox: Clinical and Research Information on Drug-Induced Liver Injury, Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases, 2012, PMID 31644023, retrieved 2020-10-15
- ^ "New Compound Related to Psychedelic Ibogaine Could Treat Addiction, Depression". UC Davis. 2020-12-09. Retrieved 2020-12-11.
-
^ ServiceDec. 9, Robert F.; 2020; Am, 11:45 (2020-12-09).
"Chemists re-engineer a psychedelic to treat depression and addiction in rodents". Science | AAAS. Retrieved 2020-12-11.
{{ cite web}}:|last2=has numeric name ( help)CS1 maint: numeric names: authors list ( link) - ^ a b c d e Miller, Gregory M. (2011). "The Emerging Role of Trace Amine Associated Receptor 1 in the Functional Regulation of Monoamine Transporters and Dopaminergic Activity". Journal of neurochemistry. 116 (2): 164–176. doi: 10.1111/j.1471-4159.2010.07109.x. ISSN 0022-3042. PMC 3005101. PMID 21073468.
-
^
"Serotonin Synthesis and Metabolism". Sigma Aldrich. 2020.
{{ cite web}}: CS1 maint: url-status ( link) - ^ "MetaCyc L-tryptophan degradation VI (via tryptamine)". biocyc.org. Retrieved 2020-12-11.
- ^ Yu, Ai-Ming; Granvil, Camille P.; Haining, Robert L.; Krausz, Kristopher W.; Corchero, Javier; Küpfer, Adrian; Idle, Jeffrey R.; Gonzalez, Frank J. (2003-02-01). "The Relative Contribution of Monoamine Oxidase and Cytochrome P450 Isozymes to the Metabolic Deamination of the Trace Amine Tryptamine". Journal of Pharmacology and Experimental Therapeutics. 304 (2): 539–546. doi: 10.1124/jpet.102.043786. ISSN 0022-3565. PMID 12538805.
- ^ a b Zucchi, R; Chiellini, G; Scanlan, T S; Grandy, D K (2006). "Trace amine-associated receptors and their ligands". British Journal of Pharmacology. 149 (8): 967–978. doi: 10.1038/sj.bjp.0706948. ISSN 0007-1188. PMC 2014643. PMID 17088868.
- ^ Jing, Li; Li, Jun-Xu (2015-08-15). "Trace amine-associated receptor 1: a promising target for the treatment of psychostimulant addiction". European journal of pharmacology. 761: 345–352. doi: 10.1016/j.ejphar.2015.06.019. ISSN 0014-2999. PMC 4532615. PMID 26092759.
- ^ a b Grandy, David K.; Miller, Gregory M.; Li, Jun-Xu (2016-02-01). ""TAARgeting Addiction" The Alamo Bears Witness to Another Revolution". Drug and alcohol dependence. 159: 9–16. doi: 10.1016/j.drugalcdep.2015.11.014. ISSN 0376-8716. PMC 4724540. PMID 26644139.
- ^ a b "Pharmacology of human trace amine-associated receptors: Therapeutic opportunities and challenges". Pharmacology & Therapeutics. 180: 161–180. 2017-12-01. doi: 10.1016/j.pharmthera.2017.07.002. ISSN 0163-7258.
- ^ a b c Gainetdinov, Raul R.; Hoener, Marius C.; Berry, Mark D. (2018-07-01). "Trace Amines and Their Receptors". Pharmacological Reviews. 70 (3): 549–620. doi: 10.1124/pr.117.015305. ISSN 0031-6997. PMID 29941461.
- ^ "Microbiome-Lax May Relieve Constipation". GEN - Genetic Engineering and Biotechnology News. 2018-06-15. Retrieved 2020-12-11.