Transition metal crystal structures

Transition metals crystallize mainly in three types of lattice, the cubic BCC and FCC lattices and the hexagonal HCP lattice. The FCC and HCP lattices are close packed crystal structures. The BCC lattice has a lower atomic packing factor and is not close packed. The crystal structures of the transition metals are determined by their electronic band structure and the electronic configuration of their d-bands.
It is easier to determine the electronic structure of an atom in vacuum than to determine the electronic structure of a metal. Absorption and emission spectra give detailed information about the energy levels in a free atom. UPS photoemission spectra taken from solid metals are hard to analyze because they don't probe the electronic band structure directly. Instead they give a rather distorted picture of a mixture of the local density of states at the surface of the metal and some contributions from deeper layers. Though photo-emission spectra often show a lot of detail, they clearly show band widths and band edges, they probably provide us mainly with information about the properties of the surface layers of the metal but leave the electronic structure of the interior of the solid concealed.
It is more difficult to calculate the electronic band structures and binding or cohesion energies of transition metals with a high level of accuracy than performing a detailed atomic or molecular electronic structure calculation. A free atom has spherical symmetrical surrounding while the atom in the solid has to adjust to the potentials with the symmetry of the crystal lattice. In an atom or molecule the number of electrons is fixed and potentials and wave functions are confined to a limited and rather small area. In a metal the electronic states are extended and correlated and depend on the volume. In transition metals these correlation effects can't be neglected when an attempt is made to calculate the cohesion energy. Even though it is hard to calculate the cohesion energy of a transition metal, it is not difficult to explain the crystal structure and magnetic properties of the transition metals on the basis of a chemical LCAO or equivalent solid-state Tight-Binding model [1].
| Group | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
|---|---|---|---|---|---|---|---|---|---|---|
| Period 4 | Sc HCP | Ti HCP | V BCC | Cr BCC | Mn BCC | Fe BCC | Co HCP | Ni FCC | Cu FCC | Zn HCP |
| Period 5 | Y HCP | Zr HCP | Nb BCC | Mo BCC | Tc HCP | Ru HCP | Rh FCC | Pd FCC | Ag FCC | Cd HCP |
| Period 6 | Hf HCP | Ta BCC | W BCC | Re HCP | Os HCP | Ir FCC | Pt FCC | Au FCC | Hg RHOM |
The table shows that the transition metals at the beginning of the series, in group 3 and 4, have Hexagonal lattices. The metals towards the middle of the series, in groups 5, 6 of all have a BCC lattice. The metals at the end of the series, in groups 9, 10 and 11, all, except Cobalt, have a FCC lattice. In groups 7 and 8 the period 5 and 6 metals have a HCP crystal structure and the period 4 metals have a BCC crystal structure.
Point group symmetries of the lattices
-
Hexagonal
-
Body-centered cubic (I)
-
Face-centered cubic (F)
The crystal systems of the transition metals have different symmetries and the potentials surrounding the the atoms have different point group symmetries.
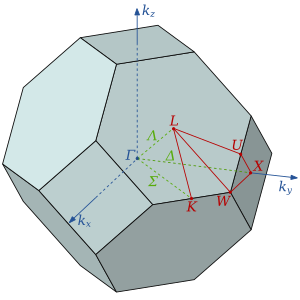
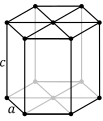
The atoms in a HCP crystal structure have twelve neighbors, six in the plane with hexagonal symmetry and three above and three below that plane. The plane with hexagonal symmetry can be rotated in several directions to cut through another set of atoms. The D3h point group of the atom has a 12 fold symmetry, with a three fold achiral dihedral symmetry.
The atoms in the close packed FCC crystal structure have a surrounding very similar to the atoms in a HCP crystal structure. The difference be tween the two structures is that the atoms in the layers above and below the hexagonal symmetric plane are rotated 60o. The Oh point group of the atom has a 48 fold octahedral symmetry.
The atoms in the BCC crystal structure are in the Th point group with a 24 fold pyritohedral symmetry.
Energies of the d-bands
The french physicist J. Friedel [1]
For thermodynamic reasons structures with a higher symmetry should be favored over structures with lower symmetry because of the higher entropy of structures with higher symmetry. An FCC structure has e higher entropy than a HCP structure and as a result it has a lower Gibbs free energy. In general the enrgy or enthalpy of a close packed structure should be lower than the energy or enthalpy of a BCC structure. There must be a reason for the fact that there are more transition metals with BCC and HCP crystal structures than FCC structures. In a tight binding bandstructure calculation of the BCC lattice the bottom of the d-bands ...
Helium
| Atom | |
|---|---|
 An illustration of the
helium atom, depicting the
nucleus (pink) and the
electron cloud distribution (black). The nucleus (upper right) in helium-4 is in reality spherically symmetric and closely resembles the electron cloud, although for more complicated nuclei this is not always the case. The black bar is one
angstrom (10−10 m or 100
pm). | |
| Classification | |
| Smallest recognized division of a chemical element | |
| Properties | |
| Mass range | 1.67×10−27 to 4.52×10−25 kg |
| Electric charge | zero (neutral), or ion charge |
| Diameter range | 62 pm ( He) to 520 pm ( Cs) ( data page) |
| Components | Electrons and a compact nucleus of protons and neutrons |
Helium ion
The He+-ion has one electron and a nucleus with two protons and two neutrons. The electronic ground state configuration of the He+-ion is 1s1. The angular part of the s wave function is the spherical harmonic . The radial wave function is a Slater-type orbital
The potential is given by
and the centrosymmetric Schrödinger equation is
with
Substitution of for the STO, for the Hamiltonian and for the potential gives a Schrödinger equation
Helium atom
The Helium atom has two electrons and a nucleus with two protons and two neutrons. The electronic ground state configuration of the He-atom is 1s2. The angular part of the s wave functions are for both electrons the same the spherical harmonic . The radial wave function is also the same for both electrons.
Kinetic energy of a Slater-orbital
The radial wave function is a Slater-type orbital
References
- ^ a b Walter Ashley Harrison (1989). Electronic Structure and the Properties of Solids. Dover Publications. ISBN 0-486-66021-4. Cite error: The named reference "Harrison" was defined multiple times with different content (see the help page).
Transition metal crystal structures

Transition metals crystallize mainly in three types of lattice, the cubic BCC and FCC lattices and the hexagonal HCP lattice. The FCC and HCP lattices are close packed crystal structures. The BCC lattice has a lower atomic packing factor and is not close packed. The crystal structures of the transition metals are determined by their electronic band structure and the electronic configuration of their d-bands.
It is easier to determine the electronic structure of an atom in vacuum than to determine the electronic structure of a metal. Absorption and emission spectra give detailed information about the energy levels in a free atom. UPS photoemission spectra taken from solid metals are hard to analyze because they don't probe the electronic band structure directly. Instead they give a rather distorted picture of a mixture of the local density of states at the surface of the metal and some contributions from deeper layers. Though photo-emission spectra often show a lot of detail, they clearly show band widths and band edges, they probably provide us mainly with information about the properties of the surface layers of the metal but leave the electronic structure of the interior of the solid concealed.
It is more difficult to calculate the electronic band structures and binding or cohesion energies of transition metals with a high level of accuracy than performing a detailed atomic or molecular electronic structure calculation. A free atom has spherical symmetrical surrounding while the atom in the solid has to adjust to the potentials with the symmetry of the crystal lattice. In an atom or molecule the number of electrons is fixed and potentials and wave functions are confined to a limited and rather small area. In a metal the electronic states are extended and correlated and depend on the volume. In transition metals these correlation effects can't be neglected when an attempt is made to calculate the cohesion energy. Even though it is hard to calculate the cohesion energy of a transition metal, it is not difficult to explain the crystal structure and magnetic properties of the transition metals on the basis of a chemical LCAO or equivalent solid-state Tight-Binding model [1].
| Group | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
|---|---|---|---|---|---|---|---|---|---|---|
| Period 4 | Sc HCP | Ti HCP | V BCC | Cr BCC | Mn BCC | Fe BCC | Co HCP | Ni FCC | Cu FCC | Zn HCP |
| Period 5 | Y HCP | Zr HCP | Nb BCC | Mo BCC | Tc HCP | Ru HCP | Rh FCC | Pd FCC | Ag FCC | Cd HCP |
| Period 6 | Hf HCP | Ta BCC | W BCC | Re HCP | Os HCP | Ir FCC | Pt FCC | Au FCC | Hg RHOM |
The table shows that the transition metals at the beginning of the series, in group 3 and 4, have Hexagonal lattices. The metals towards the middle of the series, in groups 5, 6 of all have a BCC lattice. The metals at the end of the series, in groups 9, 10 and 11, all, except Cobalt, have a FCC lattice. In groups 7 and 8 the period 5 and 6 metals have a HCP crystal structure and the period 4 metals have a BCC crystal structure.
Point group symmetries of the lattices
-
Hexagonal
-
Body-centered cubic (I)
-
Face-centered cubic (F)
The crystal systems of the transition metals have different symmetries and the potentials surrounding the the atoms have different point group symmetries.
The atoms in a HCP crystal structure have twelve neighbors, six in the plane with hexagonal symmetry and three above and three below that plane. The plane with hexagonal symmetry can be rotated in several directions to cut through another set of atoms. The D3h point group of the atom has a 12 fold symmetry, with a three fold achiral dihedral symmetry.
The atoms in the close packed FCC crystal structure have a surrounding very similar to the atoms in a HCP crystal structure. The difference be tween the two structures is that the atoms in the layers above and below the hexagonal symmetric plane are rotated 60o. The Oh point group of the atom has a 48 fold octahedral symmetry.
The atoms in the BCC crystal structure are in the Th point group with a 24 fold pyritohedral symmetry.

Energies of the d-bands
The french physicist J. Friedel [1]
For thermodynamic reasons structures with a higher symmetry should be favored over structures with lower symmetry because of the higher entropy of structures with higher symmetry. An FCC structure has e higher entropy than a HCP structure and as a result it has a lower Gibbs free energy. In general the enrgy or enthalpy of a close packed structure should be lower than the energy or enthalpy of a BCC structure. There must be a reason for the fact that there are more transition metals with BCC and HCP crystal structures than FCC structures. In a tight binding bandstructure calculation of the BCC lattice the bottom of the d-bands ...
Helium
| Atom | |
|---|---|
 An illustration of the
helium atom, depicting the
nucleus (pink) and the
electron cloud distribution (black). The nucleus (upper right) in helium-4 is in reality spherically symmetric and closely resembles the electron cloud, although for more complicated nuclei this is not always the case. The black bar is one
angstrom (10−10 m or 100
pm). | |
| Classification | |
| Smallest recognized division of a chemical element | |
| Properties | |
| Mass range | 1.67×10−27 to 4.52×10−25 kg |
| Electric charge | zero (neutral), or ion charge |
| Diameter range | 62 pm ( He) to 520 pm ( Cs) ( data page) |
| Components | Electrons and a compact nucleus of protons and neutrons |
Helium ion
The He+-ion has one electron and a nucleus with two protons and two neutrons. The electronic ground state configuration of the He+-ion is 1s1. The angular part of the s wave function is the spherical harmonic . The radial wave function is a Slater-type orbital
The potential is given by
and the centrosymmetric Schrödinger equation is
with
Substitution of for the STO, for the Hamiltonian and for the potential gives a Schrödinger equation
Helium atom
The Helium atom has two electrons and a nucleus with two protons and two neutrons. The electronic ground state configuration of the He-atom is 1s2. The angular part of the s wave functions are for both electrons the same the spherical harmonic . The radial wave function is also the same for both electrons.
Kinetic energy of a Slater-orbital
The radial wave function is a Slater-type orbital
References
- ^ a b Walter Ashley Harrison (1989). Electronic Structure and the Properties of Solids. Dover Publications. ISBN 0-486-66021-4. Cite error: The named reference "Harrison" was defined multiple times with different content (see the help page).






![{\displaystyle \left[-{\frac {\hbar ^{2}}{2\mu }}\left(\nabla ^{2}-{l(l+1) \over r^{2}}\right)+V(r)\right]R(r)=ER(r),}](https://wikimedia.org/api/rest_v1/media/math/render/svg/8a473e54268405cfa6470a46a9b9aca98620394c)

![{\displaystyle \nabla _{r}^{2}R_{n}(r)=\left[{n(n-1) \over r^{2}}-{2n\zeta \over r}+\zeta ^{2}\right]R_{n}(r)}](https://wikimedia.org/api/rest_v1/media/math/render/svg/5f2895465bf4b5e80e8b7a5499cc7d42dc769ac1)



![{\displaystyle \left[{\frac {\hbar ^{2}}{2\mu }}\left({2\zeta \over r}+\zeta ^{2}\right)-{\frac {1}{2\pi \epsilon _{0}}}{\frac {e^{2}}{r}}\right]=E}](https://wikimedia.org/api/rest_v1/media/math/render/svg/b30c180f115661d9128fd83f9400cdfc925ea1c5)
![{\displaystyle \left[{\frac {\zeta \hbar ^{2}}{\mu }}-{\frac {e^{2}}{2\pi \epsilon _{0}}}\right]{1 \over r}=E+{\frac {\zeta ^{2}\hbar ^{2}}{2\mu }}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/eb79bab314b2ab9e580e88ac01d45b8e578753d6)



![{\displaystyle {\partial R(r) \over \partial r}=\left[{\frac {(n-1)}{r}}-\zeta \right]R(r)}](https://wikimedia.org/api/rest_v1/media/math/render/svg/3b76d7bfaf4c27b3630d1a53578d1b8c7c6d83bc)
![{\displaystyle \left(r^{2}{\partial \over \partial r}\right)R(r)=\left[(n-1)r-\zeta r^{2}\right]R(r)}](https://wikimedia.org/api/rest_v1/media/math/render/svg/d8b38b36896ea1a89d4068b21bf6c2a913909f30)
![{\displaystyle \nabla ^{2}R(r)=\left({1 \over r^{2}}{\partial \over \partial r}\right)\left[(n-1)r-\zeta r^{2}\right]R(r)}](https://wikimedia.org/api/rest_v1/media/math/render/svg/7e88111514ed906e1dad4302c6e43088d4c796b8)
![{\displaystyle \nabla ^{2}R(r)={n-1 \over r^{2}}{\partial \over \partial r}\left[rR(r)\right]-{\zeta \over r^{2}}{\partial \over \partial r}\left[r^{2}R(r)\right]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/5ed84641a30d7185458c96af1ed0e2399fcbfd94)
![{\displaystyle \nabla ^{2}R(r)=\left[{n-1 \over r^{2}}-{2\zeta \over r}\right]R(r)+\left[{n-1 \over r}-\zeta \right]{\partial \over \partial r}R(r)}](https://wikimedia.org/api/rest_v1/media/math/render/svg/d462adea3a26a08c1f3870d2201cedb7054c2898)
![{\displaystyle \nabla ^{2}R(r)=\left[{n(n-1) \over r^{2}}-{2n\zeta \over r}+\zeta ^{2}\right]R(r)}](https://wikimedia.org/api/rest_v1/media/math/render/svg/06402e4029c897e7262d77226e05a339bb86defe)