Methylenedioxypyrovalerone
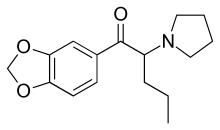
Proposed content for inclusion into main article.
Starting with piperonal, one would perform a Grignard alkylation with n-butylmagnesium bromide, oxidation of the resulting alcohol back into a ketone with pyridinium chlorochromate, followed by bromination and subsequent amination with pyrollidine.
One method for preparation
[1] involving only four steps would involve
alkylation of
piperonal via a
Grignard reaction using
n-butylmagnesium bromide,
oxidation of the resulting alcohol back into a ketone with
pyridinium chlorochromate, followed by
bromination and subsequent
amination with
pyrollidine.
[2]
One particularly academic route follows a Grignard-based strategy capable of yielding various substituted cathinones, by making the appropriate modifications.
MDPV involves a 4-step alkylation-oxidation-bromination-amination methodology.
[1]
Beginning with
alkylation of
piperonal in a
Grignard reaction using
n-butylmagnesium bromide or
1-bromobutane and magnesium metal in
diethyl ether under
anhydrous conditions using an inert atmosphere such as nitrogen to avoid moisture. The second step then involve
oxidation of the resulting alcohol back into a ketone by adding it (without the ether phase) to a suspension of
pyridinium chlorochromate in
chloroform. The use of an optional
organometallic scavenger such as nano-porous silica gel will make workup easier. After workup and redissolving residue in fresh chloroform, third-step addition of bromine will create a
haloketone with bromide at the
alpha position, and acts as the
leaving group during subsequent (fourth-step)
amination with
pyrollidine in ether, which should take 12 hours and proceed at room temperature (20°C). Several washes with cold water, keeping the ethereal phase, will draw off most of the excess pyrollidine. Dissolve in absolute ethanol and
salt with
HCl. Recrystallize from a mixture of ethanol and ether. M.P. 229-231°C
[2]

The α,α-di brominated or α-mono- brominated intermediates from the 3rd step bromination above are the most likely impurities to be present when using this method. [3] Bromination is likely to in excess, since excess pyrollidine amine will form a black precipitate that reported [2] to be difficult to separate. Workup should start with washing with water, and then several washes with saturated NaHCO3 [3] for this reason before proceeding with recrystallization. The Beilstein test is a qualitative test that can be used to test for the presence of halides. The Beilstein test is very sensitive and so may not be appropriate as a test for purity, but rather as an indicator that the compound may have been produced via this route.
Such brominated halo- aromatic ketones are of particular concern since it was known to have been abused as a recreational compound in the years leading up to 2011. Aside from the bromine ion being highly electronegative and reactive, the alkyl- bromine compounds often being alkylating agents, and brominated aromatic derivatives being implicated as hormone disruptors, there also exists the mechanism for bromine substituting for the methyl group in the nitrogenous base 5-methyluracil of DNA, creating the base-analog 5-bromouracil, which can be incorporated into DNA and induce a point mutation via base substitution. [4]
MDPV undergoes CYP450 2D6, 2C19 and COMT phase 1 metabolism (liver) into methylcatechol and pyrrolidine, which in turn are glucuronated ( uridine 5'-diphospho-glucuronosyl-transferase) allowing it to be excreted by the kidneys, with only a small fraction of the metabolites being excreted into the fecal matter. [5] No free pyrrolidine could be detected in the urine. [6]
First, methylenedioxypyrovalerone undergoes demethylenation via the CYP2D6 enzyme, followed by methylation of the aromatic ring via catechol-O-methyl transferase. Then hydroxylation of both the aromatic ring and side chain takes place followed by and oxidation of the pyrimidine ring to the corresponding lactam, with subsequent detachment and ring opening to the corresponding carboxylic acid. [7]

MDPV HCl has a granular particle size between 9 and 12 mesh. 10 mesh powdered sugar is approximately the same granule size and has a similar appearance and behavior. One might describe the appearance of MDPV by comparing it to powdered sugar.
- ^ a b Loo, P; 2010 Jun. 3. "Convenient Synthesis and Spectroscopic Data of Methcathinone Analogs". 4th Seminar of European Customs Chemists..
- ^ a b c Koppe H, Ludwig G, Karl Z; 1969 Nov. 11. 1-(3',4'-METHYLENEDIOXY-PHENYL)-2-PYRROLIDINO-ALKANONES-(1). United States Patent 3,478,050.
- ^ a b Brandt, S.; (2010) "Analysis of NRG ‘legal highs’ in the UK: Identification and formation of novel cathinones". Drug Test. Analysis. doi: 10.1002/dta.204. PMID: [ 21191917
-
^ Nester EW, Anderson DG and Roberts CE, Jr. (4 June 2009).
Loose Leaf Version of Microbiology: A Human Perspective. McGraw-Hill.
ISBN
9780077366476. Retrieved 25 June 2011.
{{ cite book}}: CS1 maint: multiple names: authors list ( link) - ^ Strano-Rossi, S; (2010) "Toxicological determination and in vitro metabolism of the designer drug methylenedioxypyrovalerone (MPDV) by gas chromatography/mass spectrometry and liquid chromatography/quadrupole time-of-flight mass spectrometry." Rapid Communications in Mass Spectrometry, 24: 2706–2714. doi: 10.1002/rcm.4692. PMID: 20814976.
- ^ Michaelis, W; (1970) "The metabolism of pyrovalerone hydrochloride". J Med Chem. 13(3): 497-503.
- ^ Meyer, M; (2010) "Studies on the metabolism of the α-pyrrolidinophenone designer drug methylenedioxy-pyrovalerone (MDPV) in rat and human urine and human liver microsomes using GC–MS and LC–high-resolution MS and its detectability in urine by GC–MS". Journal of Mass Spectrometry, 45: 1426–1442. doi: 10.1002/jms.1859. PMID: 21053377.
Methylenedioxypyrovalerone


Proposed content for inclusion into main article.
Starting with piperonal, one would perform a Grignard alkylation with n-butylmagnesium bromide, oxidation of the resulting alcohol back into a ketone with pyridinium chlorochromate, followed by bromination and subsequent amination with pyrollidine.
One method for preparation
[1] involving only four steps would involve
alkylation of
piperonal via a
Grignard reaction using
n-butylmagnesium bromide,
oxidation of the resulting alcohol back into a ketone with
pyridinium chlorochromate, followed by
bromination and subsequent
amination with
pyrollidine.
[2]
One particularly academic route follows a Grignard-based strategy capable of yielding various substituted cathinones, by making the appropriate modifications.
MDPV involves a 4-step alkylation-oxidation-bromination-amination methodology.
[1]
Beginning with
alkylation of
piperonal in a
Grignard reaction using
n-butylmagnesium bromide or
1-bromobutane and magnesium metal in
diethyl ether under
anhydrous conditions using an inert atmosphere such as nitrogen to avoid moisture. The second step then involve
oxidation of the resulting alcohol back into a ketone by adding it (without the ether phase) to a suspension of
pyridinium chlorochromate in
chloroform. The use of an optional
organometallic scavenger such as nano-porous silica gel will make workup easier. After workup and redissolving residue in fresh chloroform, third-step addition of bromine will create a
haloketone with bromide at the
alpha position, and acts as the
leaving group during subsequent (fourth-step)
amination with
pyrollidine in ether, which should take 12 hours and proceed at room temperature (20°C). Several washes with cold water, keeping the ethereal phase, will draw off most of the excess pyrollidine. Dissolve in absolute ethanol and
salt with
HCl. Recrystallize from a mixture of ethanol and ether. M.P. 229-231°C
[2]

The α,α-di brominated or α-mono- brominated intermediates from the 3rd step bromination above are the most likely impurities to be present when using this method. [3] Bromination is likely to in excess, since excess pyrollidine amine will form a black precipitate that reported [2] to be difficult to separate. Workup should start with washing with water, and then several washes with saturated NaHCO3 [3] for this reason before proceeding with recrystallization. The Beilstein test is a qualitative test that can be used to test for the presence of halides. The Beilstein test is very sensitive and so may not be appropriate as a test for purity, but rather as an indicator that the compound may have been produced via this route.
Such brominated halo- aromatic ketones are of particular concern since it was known to have been abused as a recreational compound in the years leading up to 2011. Aside from the bromine ion being highly electronegative and reactive, the alkyl- bromine compounds often being alkylating agents, and brominated aromatic derivatives being implicated as hormone disruptors, there also exists the mechanism for bromine substituting for the methyl group in the nitrogenous base 5-methyluracil of DNA, creating the base-analog 5-bromouracil, which can be incorporated into DNA and induce a point mutation via base substitution. [4]
MDPV undergoes CYP450 2D6, 2C19 and COMT phase 1 metabolism (liver) into methylcatechol and pyrrolidine, which in turn are glucuronated ( uridine 5'-diphospho-glucuronosyl-transferase) allowing it to be excreted by the kidneys, with only a small fraction of the metabolites being excreted into the fecal matter. [5] No free pyrrolidine could be detected in the urine. [6]
First, methylenedioxypyrovalerone undergoes demethylenation via the CYP2D6 enzyme, followed by methylation of the aromatic ring via catechol-O-methyl transferase. Then hydroxylation of both the aromatic ring and side chain takes place followed by and oxidation of the pyrimidine ring to the corresponding lactam, with subsequent detachment and ring opening to the corresponding carboxylic acid. [7]

MDPV HCl has a granular particle size between 9 and 12 mesh. 10 mesh powdered sugar is approximately the same granule size and has a similar appearance and behavior. One might describe the appearance of MDPV by comparing it to powdered sugar.
- ^ a b Loo, P; 2010 Jun. 3. "Convenient Synthesis and Spectroscopic Data of Methcathinone Analogs". 4th Seminar of European Customs Chemists..
- ^ a b c Koppe H, Ludwig G, Karl Z; 1969 Nov. 11. 1-(3',4'-METHYLENEDIOXY-PHENYL)-2-PYRROLIDINO-ALKANONES-(1). United States Patent 3,478,050.
- ^ a b Brandt, S.; (2010) "Analysis of NRG ‘legal highs’ in the UK: Identification and formation of novel cathinones". Drug Test. Analysis. doi: 10.1002/dta.204. PMID: [ 21191917
-
^ Nester EW, Anderson DG and Roberts CE, Jr. (4 June 2009).
Loose Leaf Version of Microbiology: A Human Perspective. McGraw-Hill.
ISBN
9780077366476. Retrieved 25 June 2011.
{{ cite book}}: CS1 maint: multiple names: authors list ( link) - ^ Strano-Rossi, S; (2010) "Toxicological determination and in vitro metabolism of the designer drug methylenedioxypyrovalerone (MPDV) by gas chromatography/mass spectrometry and liquid chromatography/quadrupole time-of-flight mass spectrometry." Rapid Communications in Mass Spectrometry, 24: 2706–2714. doi: 10.1002/rcm.4692. PMID: 20814976.
- ^ Michaelis, W; (1970) "The metabolism of pyrovalerone hydrochloride". J Med Chem. 13(3): 497-503.
- ^ Meyer, M; (2010) "Studies on the metabolism of the α-pyrrolidinophenone designer drug methylenedioxy-pyrovalerone (MDPV) in rat and human urine and human liver microsomes using GC–MS and LC–high-resolution MS and its detectability in urine by GC–MS". Journal of Mass Spectrometry, 45: 1426–1442. doi: 10.1002/jms.1859. PMID: 21053377.