
Pancreatic agenesis is a rare type of
congenital
genetic disorder, which arises from innate
pancreas
malformation and the absence of pancreatic
tissues.
[3] Pancreatic agenesis is classified into four types, and the signs and symptoms vary among the affected individuals.
[3]
[4] The exact cause of pancreatic agenesis is not well studied due to the limited cases reported worldwide.
[5] Nevertheless, the most recognized mechanisms for pancreatic agenesis are
genetic mutations in the
PDX1 gene and
PTF1A gene, which lead to incomplete pancreatic development in the
fetus, subsequently inducing
agenesis.
[3] There is no standard method to diagnose pancreatic agenesis, but
medical imaging has been widely used in many medical case reports due to its ability to detect structural abnormalities in patients' pancreas.
[4] If symptoms appear, treatments are provided to alleviate the pain and prevent the worsening of tissues and
organ dysfunction.Cite error: A <ref> tag is missing the closing </ref> (see the
help page). Reports suggest that the most common type of pancreatic agenesis is partial
dorsal pancreatic agenesis, while the less common types are partial
ventral pancreatic agenesis and complete pancreatic agenesis, due to the incompatibility for humans to live.
[4]
[6]
[7]

Individuals with dorsal pancreatic agenesis will experience either a partial or complete failure in pancreatic development, referred to as partial dorsal pancreatic agenesis and complete dorsal pancreatic agenesis, respectively. [3] [4] The same theory applies to ventral pancreatic agenesis. [9]
Table 1. Types of pancreatic agenesis based on the degree and location of pancreatic malformation. [3] [5] [4] [7] [10]
| Degree of pancreatic malformation | Location of pancreatic malformation | Types of pancreatic agenesis |
|---|---|---|
| Complete | Dorsal
Ventral |
Complete dorsal pancreatic agenesis
Complete ventral pancreatic agenesis |
| Partial | Dorsal
Ventral |
Partial dorsal pancreatic agenesis
Partial ventral pancreatic agenesis |
Signs and symptoms of pancreatic agenesis vary among individuals due to different levels of disease severity. [3] The disease severity is determined by the degree of congenital malformation and the damaged pancreatic tissues. [3] [11] A greater pancreatic malformation indicates a more severe impairment of the pancreatic tissues, and thus, causing a deterioration in the normal digestive functions which further induces digestive diseases. [12]
Common symptoms associated with pancreatic agenesis are pancreatitis, diabetes mellitus, hyperglycemia, bile duct obstruction, abdominal pain. [12] [13] Recent research has suggested that 50% of the affected individuals developed hyperglycemia or the aforementioned symptoms, while the remaining half did not develop any symptom throughout their lifetime. [12] [14] [7] Other rare diseases associated with pancreatic agenesis are polysplenia and heterotaxy syndrome. [6] [15]
Medical complications refer to the accompanying symptoms found in affected individuals, which leads to further worsening of health conditions. [16] The common complications of pancreatic agenesis are pancreatitis, diabetes, tumors, and organ malformations. [17] Upon the published studies, most of these complications are not confirmed to have a causal relationship with pancreatic agenesis. [17]
- Pancreatitis refers to pancreatic inflammation caused by enzyme's autodigestion of tissues, resulting in pancreatic structural damage and dysfunction. [18] Pancreatitis is featured with abdominal symptoms like abdominal pain and bloating. [19] Acute pancreatitis appears suddenly and lasts for several days, while chronic pancreatitis usually onsets after several times of acute pancreatitis. [19] Factions of pancreatic agenesis patients have abdominal pain, which is later confirmed to be pancreatitis-derived rather than agenesis-caused. [17] Several cases demonstrate chronic pancreatitis occurs after treatment to pancreatic agenesis by identifying recurrent alcohol-use associated abdominal pain. [17]
- Hyperglycemia (high blood sugar level) and hypoglycemia (low blood sugar level) are associated with pancreas agenesis. [10] Despite proper medications, the unexplained fluctuation of blood sugar levels is frequently observable in pancreas agenesis patients. [20] Pancreatic agenesis patients with hyperglycemia are subjected to type 1 diabetes due to limited insulin production by the damaged pancreas, and these patients usually do not have abdominal symptoms. [21] Commonly, pancreatic agenesis patients develop hypoglycemia as the result of insulin therapy and other diabetes medications. [22]
- Both pancreatic tumors and some non-pancreatic tumors are found along with pancreatic agenesis. [23] According to published studies, these non-pancreatic tumors include ampullary tumors, solid pseudopapillary tumors, cystic adenocarcinoma, neuroendocrine tumors, etc. [23]
- The non-pancreas organ malformation is identified as the accompanying disease with pancreatic agenesis. The organ malformations are multiple splenic deformities, polycystic kidney disease, biliary atresia, and congenital choledochal cysts. [24] [25]
Pancreatic agenesis is the failure of pancreatic development due to the absence of primordial germ cells in the embryonic stage. [26] [27] Nonetheless, the exact causes of pancreatic agenesis are not fully known. [6] [28] Many research studies proposed that the pathophysiological mechanisms are associated with genetic mutations and genetic inheritance. [3] [14] In research laboratories, the more well-known genetic mutations related to pancreatic agenesis are the PDX1 gene and PTF1A gene, while the less studied genetic mutations are the GATA4 gene, GATA6 gene, and Mnx1 gene. [5] [6] [11] [15] [17]

A mutation in the Pancreatic Duodenal Homeobox-1 (PDX1) gene is associated with pancreatic agenesis. [3] [29] [30] The PDX1 gene codes for the proliferation of the pancreas in the embryonic stage. [31] The PDX1 gene develops signals during the first three to eight weeks of embryogenesis (embryonic development), which is received by the pancreatic bud. [11] [32] [33] Upon release of signals, the pancreatic bud will differentiate into a complete pancreas composed of a neck, body, and tail. [32] [34] However, a mutation in the PDX1 gene will cause abnormal development of the pancreatic bud into an incomplete pancreas, and thus, leading to structural abnormalities and pancreatic dysfunctions. [29] [35]
Additionally, the PDX1 gene is suspected to be associated with various metabolic diseases in pancreatic agenesis patients. [34] [36] One commonly associated metabolic disease is diabetes mellitus. [3] [29] The PDX1 gene is a major transcription factor for the insulin gene, which controls the absorbing ability of pancreatic beta-cells and serves to maintain the body's blood glucose level. [11] [35] [37] A mutation of the PDX1 gene depletes the functional pancreatic beta-cells that sustain the body's blood glucose level in the normal range. [14] [29] Thus, the mutation induces hyperglycemia (high blood sugar level) and diabetes mellitus. [14] [29]
A missense mutation in the Pancreas Associated Transcription Factor 1a (PTF1A) gene is associated with the incidence of pancreatic agenesis. [3] A missense mutation is a specific type of genetic mutation in which a single based nucleotide in the DNA is changed, and results in a substitution of an amino acid that codes for a different protein. [38]
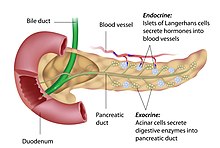
The PTF1A gene promotes the development of embryonic foregut endoderm to form a proper pancreas. [40] At an early stage, the PTF1A genes are expressed in progenitors of pancreatic ducts, endocrine, and exocrine cells of the pancreas. [41] At a later stage, the PTF1A gene controls the expression of the PDX1 gene for pancreatic development in the embryo[9]. When the PTF1A gene mutates, the embryonic foregut endoderm will fail to develop into a complete pancreas, and therefore, inducing pancreatic agenesis. [42] [43]
Furthermore, a missense mutation in the PTF1A gene is responsible for the inheritance of neonatal diabetes mellitus, which is associated with pancreatic agenesis. [3] [43] [44] Neonatal diabetes mellitus is an autosomal recessive syndrome, and it is a type of diabetes onset in the first six months in infancy. [44] If neonatal diabetes mellitus remains untreated, symptoms such as dehydration, muscle weakness, learning disabilities, and other health complications may develop. [45] Nonetheless, the occurrence of both neonatal diabetes mellitus and pancreatic agenesis is uncommon. [46]
Pancreatic agenesis is inherited by autosomal recessive or X-linked dominant mode at the neonatal or infancy stage. [3] [6] Research suggested that individuals who carry these affected genes may temporarily exhibit no symptoms or a historical record of pancreatic agenesis. [3] [6]
The diagnosis of pancreatic agenesis is not well-established due to the limited cases reported worldwide. Although there is no standard method to diagnose this disease, many clinicians use imaging examinations as the primary diagnostic tool for detecting pancreatic structural abnormalities. [47] [48] The choice of imaging techniques is based on the specific symptoms and clinical availability. [47] [48] In some cases, clinicians apply multiple imaging techniques to confirm the diagnosis of pancreatic agenesis. [13] [47] [48]
Imaging examination is commonly used by physicians to detect abdomen symptoms. [49] Recent research recommends to confirm pancreatic agenesis diagnosis by integrating one or more imaging examinations. [50] Despite limited clinical reports, endoscopic retrograde cholangiopancreatography (ERCP) is found to give the highest diagnostic values for pancreatic agenesis. [50]
- Ultrasound scan: also known as sonography. [51] It is a medical test that captures the pancreatic image through the use of high-frequency sound waves. [51] This method is commonly used to detect pancreatic pathology. [52] However, the detection of pancreatic agenesis becomes inaccurate when intestinal gas blocks the view of the partial pancreas near the splenic vessels. [49]
- Computed tomography (CT) & Magnetic resonance imaging (MRI): CT mainly uses X-ray to capture cross-sectional images while MRI is magnet-based. [53] These two diagnostic tools are usually applied to display the internal structure of the pancreas. [53] However, both CT and MRI remain arguable in diagnosing pancreatic agenesis. Some clinical reports used to state that both CT or MRI can reveal the integrity and partiality of the pancreatic head, body, and tail. [54] Conversely, recent clinical reports have declared the CT's and MRI's inability to fully display the pancreatic body and tail. [55]

- Endoscopic retrograde cholangiopancreatography (ERCP) & Magnetic resonance cholangiopancreatography (MRCP): ERCP is used for detailed examination on the pancreatic duct by inserting a flexible plastic tube (endoscopic) into the stomach and duodenum. [56] MRCP is a non-invasive imaging technique that combines magnetic and radioactive information to evaluate the pancreas and pancreatic duct. [57] Both ERCP and MRCP can expose the detailed shape of the main and accessory pancreatic ducts. The detection of a short pancreatic duct indicates the absence of pancreatic tissues. [55] [58] Therefore, the two methods complement the information of the pancreatic duct that is missed by other imaging techniques. [55] [58]
Differential diagnostic tools are applied to differentiate pancreatic agenesis from diseases with overlapping symptoms. These diseases include pancreatic fat infiltration, chronic pancreatitis, atrophy in pancreatic body and tail. [17] For example, pancreatic fat infiltration bears a higher fat signal fraction (FSF) in MRI images and unique echo characteristics in ultrasound examination. [59] Chronic pancreatitis, the common mimicker to pancreatic agenesis, is featured by pancreas dividum and autodigestion despite it possesses similar structural abnormality under imaging examinations. [60] Recently, published research shows that chronic pancreatitis owns dilated pancreatic ducts compared with pancreatic agenesis. [61]
There is no universal agreement regarding the standard treatment of pancreatic agenesis. Most of the management measures and treatments focus on relieving symptoms and preventing further damage to pancreatic tissues. [17] Recent studies suggest simple pancreatic agenesis requires no special treatment, but severe complications need medical treatments, such as the top two common complications: hyperglycemia (up to 50%) and pancreatitis. [62] [63]
- Pancreatitis: Most acute pancreatitis patients are administered with an oxygen supply and intravenous transfusion of medication fluids with nutrition. [63] Bowel rest is recommended that no food or liquid intake from the mouth. [63] For better recovery, stomach juices is removed by a nasogastric tube for further resting the intestine. [63] In cases of chronic pancreatitis, medication for pain or nausea is prescribed to alleviate symptoms. [17]
- Hyperglycemia: Management of hyperglycemia varies across patients with different causes. [64] For pancreatic agenesis patients, insulin deficiency is caused by damaged pancreatic tissues, including insulin-secreting beta-cells. [64] A pancreatic agenesis patient may present both endocrine and exocrine dysfunctions, requiring supplementation of pancreatic enzymes and insulin. [65] Half of the pancreatic agenesis patients with hyperglycemia have received insulin therapy. [65] At the same time, exercise is beneficial to lower high blood sugar level when urine ketone is absent. In the condition of moderate to large amounts of urine ketone, exercise will exacerbate blood sugar level. [66] Diet is modulated by the dietitian to keep the sugar level in a normal range. [63]
Pancreas agenesis is a rare congenital anomaly inherited by autosomal recessive alleles or X-linked dominant mode. [3] [9] Up to now, only around 50-100 cases of partial pancreas agenesis are reported, and the disease prevalence is estimated to be 1 in 1,000,000. [9]
- ^ Belaval, Vinay V. "Dorsal pancreatic agenesis | Radiology Case | Radiopaedia.org". Radiopaedia. Retrieved 2021-04-14.
-
^
"Pancreatitis". www.stritch.luc.edu. Retrieved 2021-04-14.
{{ cite web}}: CS1 maint: url-status ( link) - ^
a
b
c
d
e
f
g
h
i
j
k
l
m
n
o RESERVED, INSERM US14-- ALL RIGHTS.
"Orphanet: Partial pancreatic agenesis". www.orpha.net. Retrieved 2021-04-01.
{{ cite web}}: CS1 maint: numeric names: authors list ( link) - ^ a b c d e Weerakkody, Yuranga. "Dorsal pancreatic agenesis | Radiology Reference Article | Radiopaedia.org". Radiopaedia. Retrieved 2021-04-01.
- ^
a
b
c Erotokritou, A.; Gerharz, C. D.; Sagir, A. (2018-06-30).
"Agenesis of dorsal pancreas associated with pancreatic neuroendocrine tumor: a case report and review of the literature". Journal of Medical Case Reports. 12 (1): 185.
doi:
10.1186/s13256-018-1733-9.
ISSN
1752-1947.
PMC
6026343.
PMID
29958547.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^
a
b
c
d
e
f Cite error: The named reference
:2was invoked but never defined (see the help page). - ^
a
b
c Thakur, Shruti; Jhobta, Anupam; Sharma, Dinesh; Thakur, Charu S (2014).
"MR in complete dorsal pancreatic agenesis: Case report and review of literature". The Indian Journal of Radiology & Imaging. 24 (2): 156–159.
doi:
10.4103/0971-3026.134401.
ISSN
0971-3026.
PMC
4094968.
PMID
25024525.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) -
^ Kim, Seung Soo; Shin, Hyeong Cheol; Hwang, Jeong Ah (2019-06-28).
"Various congenital abnormalities and anatomic variants of the pancreas: A pictorial review". Journal of the Belgian Society of Radiology. 103 (1): 39.
doi:
10.5334/jbsr.1780.
ISSN
2514-8281.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^
a
b
c RESERVED, INSERM US14-- ALL RIGHTS.
"Orphanet: Partial pancreatic agenesis". www.orpha.net. Retrieved 2021-04-13.
{{ cite web}}: CS1 maint: numeric names: authors list ( link) - ^
a
b Cite error: The named reference
:19was invoked but never defined (see the help page). - ^ a b c d "Malformações congênitas do pâncreas: um caso clínico". Revista da Associação Médica Brasileira. 59 (1): 35–39. 2013-01-01. doi: 10.1590/S0104-42302013000100009. ISSN 0104-4230.
- ^ a b c "Agenesis of the dorsal pancreas | Genetic and Rare Diseases Information Center (GARD) – an NCATS Program". rarediseases.info.nih.gov. Retrieved 2021-04-01.
- ^ a b Valiyeva, Sayali; Romano, Lucia; Schietroma, Mario; Carlei, Francesco; Giuliani, Antonio (2020-10-10). "Partial agenesis of dorsal pancreas. Report of two cases". International Journal of Surgery Case Reports. 77 (Suppl): S17–S20. doi: 10.1016/j.ijscr.2020.10.029. ISSN 2210-2612. PMC 7876736. PMID 33168455.
- ^ a b c d Liang, Kaiyong; Ou, Xiaojuan; Huang, Xukai; Lan, Qunfang (2018-03-02). "Agenesis of the dorsal pancreas: a rare cause of insulin-dependent diabetes without abdominal pain". Medicine. 97 (9). doi: 10.1097/MD.0000000000010046. ISSN 0025-7974. PMC 5851771. PMID 29489657.
- ^ a b Kapa, Suraj; Vege, Santhi Swaroop (2007). "Dorsal Pancreas Agenesis and Polysplenia/Heterotaxy Syndrome: A Novel Association with Aortic Coarctation and a Review of the Literature". JOP. Journal of the Pancreas. 8 (4). ISSN 1590-8577.
- ^ "Definition of Complication". MedicineNet. Retrieved 2021-04-13.
- ^
a
b
c
d
e
f
g
h Mei, Wentong; Cao, Feng; Li, Fei (2020-04-06).
"Two cases of agenesis of the dorsal pancreas and a review of the literature". BMC Gastroenterology. 20 (1): 94.
doi:
10.1186/s12876-020-01245-8.
ISSN
1471-230X.
PMC
7137410.
PMID
32252649.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^ "Pancreatitis - Symptoms and causes". Mayo Clinic. Retrieved 2021-04-13.
- ^ a b "Pancreatitis". medlineplus.gov. Retrieved 2021-04-13.
- ^ "Hyperglycemia: Symptoms, causes, and treatments". www.medicalnewstoday.com. 2019-05-07. Retrieved 2021-04-13.
- ^ Riguetto, Cínthia Minatel; Pelichek, Samantha; Moura, Arnaldo (2019-05-25). "Heterotaxy syndrome with agenesis of dorsal pancreas and diabetes mellitus: case report and review of the literature". Archives of Endocrinology and Metabolism. doi: 10.20945/2359-3997000000142. ISSN 2359-3997.
- ^ "Hypoglycemia - Symptoms and causes". Mayo Clinic. Retrieved 2021-04-13.
- ^
a
b Erotokritou, A.; Gerharz, C. D.; Sagir, A. (2018-06-30).
"Agenesis of dorsal pancreas associated with pancreatic neuroendocrine tumor: a case report and review of the literature". Journal of Medical Case Reports. 12 (1): 185.
doi:
10.1186/s13256-018-1733-9.
ISSN
1752-1947.
PMC
6026343.
PMID
29958547.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^ Demir, Mustafa Kemal; Furuncuoglu, Yavuz (2017-06-06). "Coincidence of Polysplenia, Kartagener Syndrome, Dorsal Pancreas Agenesis, and Polycystic Kidney Disease in an Adult". The Eurasian Journal of Medicine. 49 (2): 152–154. doi: 10.5152/eurasianjmed.2017.17067. ISSN 1308-8734.
- ^ Jeong, Jae Hoon; Kim, Gwang Ha; Song, Geun Am; Lee, Dong Gun; Moon, Ji Yoon; Cheong, Jae Hoon; Kim, Suk (2012). "Polysplenia Syndrome with Congenital Agenesis of Dorsal Pancreas Presenting as Acute Pancreatitis and the Role of Endoscopic Ultrasonography in Its Diagnosis". The Korean Journal of Gastroenterology. 60 (1): 47. doi: 10.4166/kjg.2012.60.1.47. ISSN 1598-9992.
- ^ "Agenesis - Medical Dictionary / Glossary | Medindia". www.medindia.net. Retrieved 2021-04-14.
-
^ Yang, Tian; Yang, Xudan; Wang, Luping; Mo, Jun (2019-11-11).
"Agenesis of the dorsal pancreas presenting with diabetic ketoacidosis – a case report and literature review". BMC Endocrine Disorders. 19 (1): 120.
doi:
10.1186/s12902-019-0449-1.
ISSN
1472-6823.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Sannappa, Raghu M.; Buragohain, Jadunath; Sarma, Dipti; Saikia, Uma Kaimal; Choudhury, Bipul Kumar (2014). "Agenesis of Dorsal Pancreas Associated with Periampullary Pancreaticobiliary Type Adenocarcinoma". JOP. Journal of the Pancreas. 15 (5). ISSN 1590-8577.
- ^
a
b
c
d
e
"DUODENUM HOMEOBOX PROTEIN 1". www.omim.org. Retrieved 2021-04-02.
{{ cite web}}: CS1 maint: url-status ( link) - ^ Teo, Adrian Kee Keong; Tsuneyoshi, Norihiro; Hoon, Shawn; Tan, Ee Kim; Stanton, Lawrence W.; Wright, Christopher V. E.; Dunn, N. Ray (2015-04-14). "PDX1 Binds and Represses Hepatic Genes to Ensure Robust Pancreatic Commitment in Differentiating Human Embryonic Stem Cells". Stem Cell Reports. 4 (4): 578–590. doi: 10.1016/j.stemcr.2015.02.015. ISSN 2213-6711. PMID 25843046.
-
^ Zhu, Yaxi; Liu, Qian; Zhou, Zhiguang; Ikeda, Yasuhiro (2017-11-02).
"PDX1, Neurogenin-3, and MAFA: critical transcription regulators for beta cell development and regeneration". Stem Cell Research & Therapy. 8 (1): 240.
doi:
10.1186/s13287-017-0694-z.
ISSN
1757-6512.
PMC
5667467.
PMID
29096722.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^ a b Marty-Santos, Leilani; Cleaver, Ondine (2016-01-01). "Pdx1 regulates pancreas tubulogenesis and E-cadherin expression". Development. 143 (1): 101–112. doi: 10.1242/dev.126755. ISSN 0950-1991. PMID 26657766.
- ^ "What is Embryogenesis?". News-Medical.net. 2011-02-24. Retrieved 2021-04-01.
- ^
a
b Fujimoto, K.; Polonsky, K. S. (2009).
"Pdx1 and other factors that regulate pancreatic β-cell survival". Diabetes, Obesity and Metabolism. 11 (s4): 30–37.
doi:
10.1111/j.1463-1326.2009.01121.x.
ISSN
1463-1326.
PMC
2802270.
PMID
19817786.
{{ cite journal}}: CS1 maint: PMC format ( link) - ^ a b "PDX1 pancreatic and duodenal homeobox 1 [Homo sapiens (human)] - Gene - NCBI". www.ncbi.nlm.nih.gov. Retrieved 2021-04-14.
- ^ Caetano, L. A.; Santana, L. S.; Costa‐Riquetto, A. D.; Lerario, A. M.; Nery, M.; Nogueira, G. F.; Ortega, C. D.; Rocha, M. S.; Jorge, A. a. L.; Teles, M. G. (2018). "PDX1 -MODY and dorsal pancreatic agenesis: New phenotype of a rare disease". Clinical Genetics. 93 (2): 382–386. doi: 10.1111/cge.13044. ISSN 1399-0004.
-
^ Editor (2019-01-15).
"Beta cells are unique cells in the pancreas that produce, store and release the hormone insulin". Diabetes. Retrieved 2021-04-01.
{{ cite web}}:|last=has generic name ( help) - ^ "Missense Mutation". Genome.gov. Retrieved 2021-04-01.
- ^ Foundation, J. E. S. "J.E.S Foundation for Pancreatic Cancer". www.thejesfoundation.com. Retrieved 2021-04-14.
- ^ "PTF1A Gene - GeneCards | PTF1A Protein | PTF1A Antibody". www.genecards.org. Retrieved 2021-04-02.
- ^ Dong, P. Duc Si; Provost, Elayne; Leach, Steven D.; Stainier, Didier Y. R. (2008-06-01). "Graded levels of Ptf1a differentially regulate endocrine and exocrine fates in the developing pancreas". Genes & Development. 22 (11): 1445–1450. doi: 10.1101/gad.1663208. ISSN 0890-9369. PMID 18519637.
- ^ Houghton, Jayne A. L.; Swift, Galvin H.; Shaw-Smith, Charles; Flanagan, Sarah E.; Franco, Elisa de; Caswell, Richard; Hussain, Khalid; Mohamed, Sarar; Abdulrasoul, Majedah; Hattersley, Andrew T.; MacDonald, Raymond J. (2016-09-01). "Isolated Pancreatic Aplasia Due to a Hypomorphic PTF1A Mutation". Diabetes. 65 (9): 2810–2815. doi: 10.2337/db15-1666. ISSN 0012-1797. PMID 27284104.
- ^
a
b RESERVED, INSERM US14-- ALL RIGHTS.
"Orphanet: pancreas associated transcription factor 1a PTF1A". www.orpha.net. Retrieved 2021-04-02.
{{ cite web}}: CS1 maint: numeric names: authors list ( link) - ^ a b "Permanent neonatal diabetes mellitus: MedlinePlus Genetics". medlineplus.gov. Retrieved 2021-04-01.
-
^ Editor (2019-01-15).
"Neonatal diabetes is a rare form of diabetes that is usually diagnosed in children under 6 months of age". Diabetes. Retrieved 2021-04-01.
{{ cite web}}:|last=has generic name ( help) - ^ Salina, Alessandro; Pasquali, Lorenzo; Aloi, Concetta; Lugani, Francesca; d'Annunzio, Giuseppe; Lorini, Renata (2010-08-01). "Neonatal Diabetes Caused by Pancreatic Agenesia: Which other genes should be used for diagnosis?". Diabetes Care. 33 (8): e112–e112. doi: 10.2337/dc10-0876. ISSN 0149-5992. PMID 20668147.
- ^ a b c "(PDF) Dorsal Pancreatic Agenesis". ResearchGate. Retrieved 2021-04-14.
- ^ a b c Bissonnette, Bruno; Luginbuehl, Igor; Engelhardt, Thomas (2019), "Congenital Pancreatic Agenesis", Syndromes: Rapid Recognition and Perioperative Implications (2 ed.), New York, NY: McGraw-Hill Education, retrieved 2021-04-14
- ^ a b "WHO | Medical imaging". WHO. Retrieved 2021-04-13.
- ^ a b Schnedl, Wolfgang J.; Reisinger, Emil C.; Schreiber, Florian; Pieber, Thomas R.; Lipp, Rainer W.; Krejs, Guenter J. (1995-11-01). "Complete and partial agenesis of the dorsal pancreas within one family". Gastrointestinal Endoscopy. 42 (5): 485–487. doi: 10.1016/S0016-5107(95)70055-2. ISSN 0016-5107.
- ^ a b "Ultrasound: Purpose, Procedure, and Preparation". Healthline. 2016-05-05. Retrieved 2021-04-13.
- ^ Czarniecki, Marcin. "Pancreatic ultrasound | Radiology Reference Article | Radiopaedia.org". Radiopaedia. Retrieved 2021-04-13.
- ^ a b "What's the Difference Between an MRI and a CT?". Center For Diagnostic Imaging (CDI). 2016-03-30. Retrieved 2021-04-13.
-
^ Busireddy, Kiran K; AlObaidy, Mamdoh; Ramalho, Miguel; Kalubowila, Janaka; Baodong, Liu; Santagostino, Ilaria; Semelka, Richard C (2014-08-15).
"Pancreatitis-imaging approach". World Journal of Gastrointestinal Pathophysiology. 5 (3): 252–270.
doi:
10.4291/wjgp.v5.i3.252.
ISSN
2150-5330.
PMC
4133524.
PMID
25133027.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^
a
b
c Pasaoglu, Lale; Vural, Murat; Hatipoglu, Hatice Gul; Tereklioglu, Gokce; Koparal, Suha (2008-05-14).
"Agenesis of the dorsal pancreas". World Journal of Gastroenterology : WJG. 14 (18): 2915–2916.
doi:
10.3748/wjg.14.2915.
ISSN
1007-9327.
PMC
2710738.
PMID
18473421.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ "ERCP (Endoscopic Retrograde Cholangio-Pancreatography) Patient Information from SAGES". SAGES. Retrieved 2021-04-13.
- ^ Radiology (ACR), Radiological Society of North America (RSNA) and American College of. "Magnetic Resonance Cholangiopancreatography (MRCP)". Radiologyinfo.org. Retrieved 2021-04-13.
- ^
a
b Vijayaraghavan, SBoopathy; Gouru, Swapna; Senthil, Sathiya (2013).
"Sonographic features of agenesis of dorsal pancreas". Indian Journal of Radiology and Imaging. 23 (2): 179.
doi:
10.4103/0971-3026.116570.
ISSN
0971-3026.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) -
^ Hong, Cheng William; Fazeli Dehkordy, Soudabeh; Hooker, Jonathan C.; Hamilton, Gavin; Sirlin, Claude B. (2017-12-XX).
"Fat Quantification in the Abdomen". Topics in Magnetic Resonance Imaging. 26 (6): 221–227.
doi:
10.1097/RMR.0000000000000141.
ISSN
1536-1004.
{{ cite journal}}: Check date values in:|date=( help) - ^ "Dorsal Pancreatic Agenesis". scholar.googleusercontent.com. Retrieved 2021-04-01.
-
^ Gutta, Aditya; Fogel, Evan; Sherman, Stuart (2019-11-02).
"Identification and management of pancreas divisum". Expert Review of Gastroenterology & Hepatology. 13 (11): 1089–1105.
doi:
10.1080/17474124.2019.1685871.
ISSN
1747-4124.
PMC
6872911.
PMID
31663403.
{{ cite journal}}: CS1 maint: PMC format ( link) - ^ Weerakkody, Yuranga. "Dorsal pancreatic agenesis | Radiology Reference Article | Radiopaedia.org". Radiopaedia. Retrieved 2021-04-01.
- ^ a b c d e "Agenesis of the dorsal pancreas | Genetic and Rare Diseases Information Center (GARD) – an NCATS Program". rarediseases.info.nih.gov. Retrieved 2021-04-13.
- ^ a b "Hyperglycemia: Symptoms, causes, and treatments". www.medicalnewstoday.com. 2019-05-07. Retrieved 2021-04-01.
- ^ a b Sonkar, Satyendra Kumar; Kumar, Satish; Singh, Neeraj Kumar (2018-02-14). "Agenesis of dorsal pancreas in a young adult: a rare cause of diabetes mellitus". BMJ Case Reports: bcr–2017-223301. doi: 10.1136/bcr-2017-223301. ISSN 1757-790X.
- ^ "Five Things to Know About Ketones | ADA". www.diabetes.org. Retrieved 2021-04-13.
Warning: Default sort key "Pancreatic Agenesis" overrides earlier default sort key "Pancreatic agenesis".

Pancreatic agenesis is a rare type of
congenital
genetic disorder, which arises from innate
pancreas
malformation and the absence of pancreatic
tissues.
[3] Pancreatic agenesis is classified into four types, and the signs and symptoms vary among the affected individuals.
[3]
[4] The exact cause of pancreatic agenesis is not well studied due to the limited cases reported worldwide.
[5] Nevertheless, the most recognized mechanisms for pancreatic agenesis are
genetic mutations in the
PDX1 gene and
PTF1A gene, which lead to incomplete pancreatic development in the
fetus, subsequently inducing
agenesis.
[3] There is no standard method to diagnose pancreatic agenesis, but
medical imaging has been widely used in many medical case reports due to its ability to detect structural abnormalities in patients' pancreas.
[4] If symptoms appear, treatments are provided to alleviate the pain and prevent the worsening of tissues and
organ dysfunction.Cite error: A <ref> tag is missing the closing </ref> (see the
help page). Reports suggest that the most common type of pancreatic agenesis is partial
dorsal pancreatic agenesis, while the less common types are partial
ventral pancreatic agenesis and complete pancreatic agenesis, due to the incompatibility for humans to live.
[4]
[6]
[7]

Individuals with dorsal pancreatic agenesis will experience either a partial or complete failure in pancreatic development, referred to as partial dorsal pancreatic agenesis and complete dorsal pancreatic agenesis, respectively. [3] [4] The same theory applies to ventral pancreatic agenesis. [9]
Table 1. Types of pancreatic agenesis based on the degree and location of pancreatic malformation. [3] [5] [4] [7] [10]
| Degree of pancreatic malformation | Location of pancreatic malformation | Types of pancreatic agenesis |
|---|---|---|
| Complete | Dorsal
Ventral |
Complete dorsal pancreatic agenesis
Complete ventral pancreatic agenesis |
| Partial | Dorsal
Ventral |
Partial dorsal pancreatic agenesis
Partial ventral pancreatic agenesis |
Signs and symptoms of pancreatic agenesis vary among individuals due to different levels of disease severity. [3] The disease severity is determined by the degree of congenital malformation and the damaged pancreatic tissues. [3] [11] A greater pancreatic malformation indicates a more severe impairment of the pancreatic tissues, and thus, causing a deterioration in the normal digestive functions which further induces digestive diseases. [12]
Common symptoms associated with pancreatic agenesis are pancreatitis, diabetes mellitus, hyperglycemia, bile duct obstruction, abdominal pain. [12] [13] Recent research has suggested that 50% of the affected individuals developed hyperglycemia or the aforementioned symptoms, while the remaining half did not develop any symptom throughout their lifetime. [12] [14] [7] Other rare diseases associated with pancreatic agenesis are polysplenia and heterotaxy syndrome. [6] [15]
Medical complications refer to the accompanying symptoms found in affected individuals, which leads to further worsening of health conditions. [16] The common complications of pancreatic agenesis are pancreatitis, diabetes, tumors, and organ malformations. [17] Upon the published studies, most of these complications are not confirmed to have a causal relationship with pancreatic agenesis. [17]
- Pancreatitis refers to pancreatic inflammation caused by enzyme's autodigestion of tissues, resulting in pancreatic structural damage and dysfunction. [18] Pancreatitis is featured with abdominal symptoms like abdominal pain and bloating. [19] Acute pancreatitis appears suddenly and lasts for several days, while chronic pancreatitis usually onsets after several times of acute pancreatitis. [19] Factions of pancreatic agenesis patients have abdominal pain, which is later confirmed to be pancreatitis-derived rather than agenesis-caused. [17] Several cases demonstrate chronic pancreatitis occurs after treatment to pancreatic agenesis by identifying recurrent alcohol-use associated abdominal pain. [17]
- Hyperglycemia (high blood sugar level) and hypoglycemia (low blood sugar level) are associated with pancreas agenesis. [10] Despite proper medications, the unexplained fluctuation of blood sugar levels is frequently observable in pancreas agenesis patients. [20] Pancreatic agenesis patients with hyperglycemia are subjected to type 1 diabetes due to limited insulin production by the damaged pancreas, and these patients usually do not have abdominal symptoms. [21] Commonly, pancreatic agenesis patients develop hypoglycemia as the result of insulin therapy and other diabetes medications. [22]
- Both pancreatic tumors and some non-pancreatic tumors are found along with pancreatic agenesis. [23] According to published studies, these non-pancreatic tumors include ampullary tumors, solid pseudopapillary tumors, cystic adenocarcinoma, neuroendocrine tumors, etc. [23]
- The non-pancreas organ malformation is identified as the accompanying disease with pancreatic agenesis. The organ malformations are multiple splenic deformities, polycystic kidney disease, biliary atresia, and congenital choledochal cysts. [24] [25]
Pancreatic agenesis is the failure of pancreatic development due to the absence of primordial germ cells in the embryonic stage. [26] [27] Nonetheless, the exact causes of pancreatic agenesis are not fully known. [6] [28] Many research studies proposed that the pathophysiological mechanisms are associated with genetic mutations and genetic inheritance. [3] [14] In research laboratories, the more well-known genetic mutations related to pancreatic agenesis are the PDX1 gene and PTF1A gene, while the less studied genetic mutations are the GATA4 gene, GATA6 gene, and Mnx1 gene. [5] [6] [11] [15] [17]

A mutation in the Pancreatic Duodenal Homeobox-1 (PDX1) gene is associated with pancreatic agenesis. [3] [29] [30] The PDX1 gene codes for the proliferation of the pancreas in the embryonic stage. [31] The PDX1 gene develops signals during the first three to eight weeks of embryogenesis (embryonic development), which is received by the pancreatic bud. [11] [32] [33] Upon release of signals, the pancreatic bud will differentiate into a complete pancreas composed of a neck, body, and tail. [32] [34] However, a mutation in the PDX1 gene will cause abnormal development of the pancreatic bud into an incomplete pancreas, and thus, leading to structural abnormalities and pancreatic dysfunctions. [29] [35]
Additionally, the PDX1 gene is suspected to be associated with various metabolic diseases in pancreatic agenesis patients. [34] [36] One commonly associated metabolic disease is diabetes mellitus. [3] [29] The PDX1 gene is a major transcription factor for the insulin gene, which controls the absorbing ability of pancreatic beta-cells and serves to maintain the body's blood glucose level. [11] [35] [37] A mutation of the PDX1 gene depletes the functional pancreatic beta-cells that sustain the body's blood glucose level in the normal range. [14] [29] Thus, the mutation induces hyperglycemia (high blood sugar level) and diabetes mellitus. [14] [29]
A missense mutation in the Pancreas Associated Transcription Factor 1a (PTF1A) gene is associated with the incidence of pancreatic agenesis. [3] A missense mutation is a specific type of genetic mutation in which a single based nucleotide in the DNA is changed, and results in a substitution of an amino acid that codes for a different protein. [38]

The PTF1A gene promotes the development of embryonic foregut endoderm to form a proper pancreas. [40] At an early stage, the PTF1A genes are expressed in progenitors of pancreatic ducts, endocrine, and exocrine cells of the pancreas. [41] At a later stage, the PTF1A gene controls the expression of the PDX1 gene for pancreatic development in the embryo[9]. When the PTF1A gene mutates, the embryonic foregut endoderm will fail to develop into a complete pancreas, and therefore, inducing pancreatic agenesis. [42] [43]
Furthermore, a missense mutation in the PTF1A gene is responsible for the inheritance of neonatal diabetes mellitus, which is associated with pancreatic agenesis. [3] [43] [44] Neonatal diabetes mellitus is an autosomal recessive syndrome, and it is a type of diabetes onset in the first six months in infancy. [44] If neonatal diabetes mellitus remains untreated, symptoms such as dehydration, muscle weakness, learning disabilities, and other health complications may develop. [45] Nonetheless, the occurrence of both neonatal diabetes mellitus and pancreatic agenesis is uncommon. [46]
Pancreatic agenesis is inherited by autosomal recessive or X-linked dominant mode at the neonatal or infancy stage. [3] [6] Research suggested that individuals who carry these affected genes may temporarily exhibit no symptoms or a historical record of pancreatic agenesis. [3] [6]
The diagnosis of pancreatic agenesis is not well-established due to the limited cases reported worldwide. Although there is no standard method to diagnose this disease, many clinicians use imaging examinations as the primary diagnostic tool for detecting pancreatic structural abnormalities. [47] [48] The choice of imaging techniques is based on the specific symptoms and clinical availability. [47] [48] In some cases, clinicians apply multiple imaging techniques to confirm the diagnosis of pancreatic agenesis. [13] [47] [48]
Imaging examination is commonly used by physicians to detect abdomen symptoms. [49] Recent research recommends to confirm pancreatic agenesis diagnosis by integrating one or more imaging examinations. [50] Despite limited clinical reports, endoscopic retrograde cholangiopancreatography (ERCP) is found to give the highest diagnostic values for pancreatic agenesis. [50]
- Ultrasound scan: also known as sonography. [51] It is a medical test that captures the pancreatic image through the use of high-frequency sound waves. [51] This method is commonly used to detect pancreatic pathology. [52] However, the detection of pancreatic agenesis becomes inaccurate when intestinal gas blocks the view of the partial pancreas near the splenic vessels. [49]
- Computed tomography (CT) & Magnetic resonance imaging (MRI): CT mainly uses X-ray to capture cross-sectional images while MRI is magnet-based. [53] These two diagnostic tools are usually applied to display the internal structure of the pancreas. [53] However, both CT and MRI remain arguable in diagnosing pancreatic agenesis. Some clinical reports used to state that both CT or MRI can reveal the integrity and partiality of the pancreatic head, body, and tail. [54] Conversely, recent clinical reports have declared the CT's and MRI's inability to fully display the pancreatic body and tail. [55]

- Endoscopic retrograde cholangiopancreatography (ERCP) & Magnetic resonance cholangiopancreatography (MRCP): ERCP is used for detailed examination on the pancreatic duct by inserting a flexible plastic tube (endoscopic) into the stomach and duodenum. [56] MRCP is a non-invasive imaging technique that combines magnetic and radioactive information to evaluate the pancreas and pancreatic duct. [57] Both ERCP and MRCP can expose the detailed shape of the main and accessory pancreatic ducts. The detection of a short pancreatic duct indicates the absence of pancreatic tissues. [55] [58] Therefore, the two methods complement the information of the pancreatic duct that is missed by other imaging techniques. [55] [58]
Differential diagnostic tools are applied to differentiate pancreatic agenesis from diseases with overlapping symptoms. These diseases include pancreatic fat infiltration, chronic pancreatitis, atrophy in pancreatic body and tail. [17] For example, pancreatic fat infiltration bears a higher fat signal fraction (FSF) in MRI images and unique echo characteristics in ultrasound examination. [59] Chronic pancreatitis, the common mimicker to pancreatic agenesis, is featured by pancreas dividum and autodigestion despite it possesses similar structural abnormality under imaging examinations. [60] Recently, published research shows that chronic pancreatitis owns dilated pancreatic ducts compared with pancreatic agenesis. [61]
There is no universal agreement regarding the standard treatment of pancreatic agenesis. Most of the management measures and treatments focus on relieving symptoms and preventing further damage to pancreatic tissues. [17] Recent studies suggest simple pancreatic agenesis requires no special treatment, but severe complications need medical treatments, such as the top two common complications: hyperglycemia (up to 50%) and pancreatitis. [62] [63]
- Pancreatitis: Most acute pancreatitis patients are administered with an oxygen supply and intravenous transfusion of medication fluids with nutrition. [63] Bowel rest is recommended that no food or liquid intake from the mouth. [63] For better recovery, stomach juices is removed by a nasogastric tube for further resting the intestine. [63] In cases of chronic pancreatitis, medication for pain or nausea is prescribed to alleviate symptoms. [17]
- Hyperglycemia: Management of hyperglycemia varies across patients with different causes. [64] For pancreatic agenesis patients, insulin deficiency is caused by damaged pancreatic tissues, including insulin-secreting beta-cells. [64] A pancreatic agenesis patient may present both endocrine and exocrine dysfunctions, requiring supplementation of pancreatic enzymes and insulin. [65] Half of the pancreatic agenesis patients with hyperglycemia have received insulin therapy. [65] At the same time, exercise is beneficial to lower high blood sugar level when urine ketone is absent. In the condition of moderate to large amounts of urine ketone, exercise will exacerbate blood sugar level. [66] Diet is modulated by the dietitian to keep the sugar level in a normal range. [63]
Pancreas agenesis is a rare congenital anomaly inherited by autosomal recessive alleles or X-linked dominant mode. [3] [9] Up to now, only around 50-100 cases of partial pancreas agenesis are reported, and the disease prevalence is estimated to be 1 in 1,000,000. [9]
- ^ Belaval, Vinay V. "Dorsal pancreatic agenesis | Radiology Case | Radiopaedia.org". Radiopaedia. Retrieved 2021-04-14.
-
^
"Pancreatitis". www.stritch.luc.edu. Retrieved 2021-04-14.
{{ cite web}}: CS1 maint: url-status ( link) - ^
a
b
c
d
e
f
g
h
i
j
k
l
m
n
o RESERVED, INSERM US14-- ALL RIGHTS.
"Orphanet: Partial pancreatic agenesis". www.orpha.net. Retrieved 2021-04-01.
{{ cite web}}: CS1 maint: numeric names: authors list ( link) - ^ a b c d e Weerakkody, Yuranga. "Dorsal pancreatic agenesis | Radiology Reference Article | Radiopaedia.org". Radiopaedia. Retrieved 2021-04-01.
- ^
a
b
c Erotokritou, A.; Gerharz, C. D.; Sagir, A. (2018-06-30).
"Agenesis of dorsal pancreas associated with pancreatic neuroendocrine tumor: a case report and review of the literature". Journal of Medical Case Reports. 12 (1): 185.
doi:
10.1186/s13256-018-1733-9.
ISSN
1752-1947.
PMC
6026343.
PMID
29958547.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^
a
b
c
d
e
f Cite error: The named reference
:2was invoked but never defined (see the help page). - ^
a
b
c Thakur, Shruti; Jhobta, Anupam; Sharma, Dinesh; Thakur, Charu S (2014).
"MR in complete dorsal pancreatic agenesis: Case report and review of literature". The Indian Journal of Radiology & Imaging. 24 (2): 156–159.
doi:
10.4103/0971-3026.134401.
ISSN
0971-3026.
PMC
4094968.
PMID
25024525.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) -
^ Kim, Seung Soo; Shin, Hyeong Cheol; Hwang, Jeong Ah (2019-06-28).
"Various congenital abnormalities and anatomic variants of the pancreas: A pictorial review". Journal of the Belgian Society of Radiology. 103 (1): 39.
doi:
10.5334/jbsr.1780.
ISSN
2514-8281.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^
a
b
c RESERVED, INSERM US14-- ALL RIGHTS.
"Orphanet: Partial pancreatic agenesis". www.orpha.net. Retrieved 2021-04-13.
{{ cite web}}: CS1 maint: numeric names: authors list ( link) - ^
a
b Cite error: The named reference
:19was invoked but never defined (see the help page). - ^ a b c d "Malformações congênitas do pâncreas: um caso clínico". Revista da Associação Médica Brasileira. 59 (1): 35–39. 2013-01-01. doi: 10.1590/S0104-42302013000100009. ISSN 0104-4230.
- ^ a b c "Agenesis of the dorsal pancreas | Genetic and Rare Diseases Information Center (GARD) – an NCATS Program". rarediseases.info.nih.gov. Retrieved 2021-04-01.
- ^ a b Valiyeva, Sayali; Romano, Lucia; Schietroma, Mario; Carlei, Francesco; Giuliani, Antonio (2020-10-10). "Partial agenesis of dorsal pancreas. Report of two cases". International Journal of Surgery Case Reports. 77 (Suppl): S17–S20. doi: 10.1016/j.ijscr.2020.10.029. ISSN 2210-2612. PMC 7876736. PMID 33168455.
- ^ a b c d Liang, Kaiyong; Ou, Xiaojuan; Huang, Xukai; Lan, Qunfang (2018-03-02). "Agenesis of the dorsal pancreas: a rare cause of insulin-dependent diabetes without abdominal pain". Medicine. 97 (9). doi: 10.1097/MD.0000000000010046. ISSN 0025-7974. PMC 5851771. PMID 29489657.
- ^ a b Kapa, Suraj; Vege, Santhi Swaroop (2007). "Dorsal Pancreas Agenesis and Polysplenia/Heterotaxy Syndrome: A Novel Association with Aortic Coarctation and a Review of the Literature". JOP. Journal of the Pancreas. 8 (4). ISSN 1590-8577.
- ^ "Definition of Complication". MedicineNet. Retrieved 2021-04-13.
- ^
a
b
c
d
e
f
g
h Mei, Wentong; Cao, Feng; Li, Fei (2020-04-06).
"Two cases of agenesis of the dorsal pancreas and a review of the literature". BMC Gastroenterology. 20 (1): 94.
doi:
10.1186/s12876-020-01245-8.
ISSN
1471-230X.
PMC
7137410.
PMID
32252649.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^ "Pancreatitis - Symptoms and causes". Mayo Clinic. Retrieved 2021-04-13.
- ^ a b "Pancreatitis". medlineplus.gov. Retrieved 2021-04-13.
- ^ "Hyperglycemia: Symptoms, causes, and treatments". www.medicalnewstoday.com. 2019-05-07. Retrieved 2021-04-13.
- ^ Riguetto, Cínthia Minatel; Pelichek, Samantha; Moura, Arnaldo (2019-05-25). "Heterotaxy syndrome with agenesis of dorsal pancreas and diabetes mellitus: case report and review of the literature". Archives of Endocrinology and Metabolism. doi: 10.20945/2359-3997000000142. ISSN 2359-3997.
- ^ "Hypoglycemia - Symptoms and causes". Mayo Clinic. Retrieved 2021-04-13.
- ^
a
b Erotokritou, A.; Gerharz, C. D.; Sagir, A. (2018-06-30).
"Agenesis of dorsal pancreas associated with pancreatic neuroendocrine tumor: a case report and review of the literature". Journal of Medical Case Reports. 12 (1): 185.
doi:
10.1186/s13256-018-1733-9.
ISSN
1752-1947.
PMC
6026343.
PMID
29958547.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^ Demir, Mustafa Kemal; Furuncuoglu, Yavuz (2017-06-06). "Coincidence of Polysplenia, Kartagener Syndrome, Dorsal Pancreas Agenesis, and Polycystic Kidney Disease in an Adult". The Eurasian Journal of Medicine. 49 (2): 152–154. doi: 10.5152/eurasianjmed.2017.17067. ISSN 1308-8734.
- ^ Jeong, Jae Hoon; Kim, Gwang Ha; Song, Geun Am; Lee, Dong Gun; Moon, Ji Yoon; Cheong, Jae Hoon; Kim, Suk (2012). "Polysplenia Syndrome with Congenital Agenesis of Dorsal Pancreas Presenting as Acute Pancreatitis and the Role of Endoscopic Ultrasonography in Its Diagnosis". The Korean Journal of Gastroenterology. 60 (1): 47. doi: 10.4166/kjg.2012.60.1.47. ISSN 1598-9992.
- ^ "Agenesis - Medical Dictionary / Glossary | Medindia". www.medindia.net. Retrieved 2021-04-14.
-
^ Yang, Tian; Yang, Xudan; Wang, Luping; Mo, Jun (2019-11-11).
"Agenesis of the dorsal pancreas presenting with diabetic ketoacidosis – a case report and literature review". BMC Endocrine Disorders. 19 (1): 120.
doi:
10.1186/s12902-019-0449-1.
ISSN
1472-6823.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ Sannappa, Raghu M.; Buragohain, Jadunath; Sarma, Dipti; Saikia, Uma Kaimal; Choudhury, Bipul Kumar (2014). "Agenesis of Dorsal Pancreas Associated with Periampullary Pancreaticobiliary Type Adenocarcinoma". JOP. Journal of the Pancreas. 15 (5). ISSN 1590-8577.
- ^
a
b
c
d
e
"DUODENUM HOMEOBOX PROTEIN 1". www.omim.org. Retrieved 2021-04-02.
{{ cite web}}: CS1 maint: url-status ( link) - ^ Teo, Adrian Kee Keong; Tsuneyoshi, Norihiro; Hoon, Shawn; Tan, Ee Kim; Stanton, Lawrence W.; Wright, Christopher V. E.; Dunn, N. Ray (2015-04-14). "PDX1 Binds and Represses Hepatic Genes to Ensure Robust Pancreatic Commitment in Differentiating Human Embryonic Stem Cells". Stem Cell Reports. 4 (4): 578–590. doi: 10.1016/j.stemcr.2015.02.015. ISSN 2213-6711. PMID 25843046.
-
^ Zhu, Yaxi; Liu, Qian; Zhou, Zhiguang; Ikeda, Yasuhiro (2017-11-02).
"PDX1, Neurogenin-3, and MAFA: critical transcription regulators for beta cell development and regeneration". Stem Cell Research & Therapy. 8 (1): 240.
doi:
10.1186/s13287-017-0694-z.
ISSN
1757-6512.
PMC
5667467.
PMID
29096722.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^ a b Marty-Santos, Leilani; Cleaver, Ondine (2016-01-01). "Pdx1 regulates pancreas tubulogenesis and E-cadherin expression". Development. 143 (1): 101–112. doi: 10.1242/dev.126755. ISSN 0950-1991. PMID 26657766.
- ^ "What is Embryogenesis?". News-Medical.net. 2011-02-24. Retrieved 2021-04-01.
- ^
a
b Fujimoto, K.; Polonsky, K. S. (2009).
"Pdx1 and other factors that regulate pancreatic β-cell survival". Diabetes, Obesity and Metabolism. 11 (s4): 30–37.
doi:
10.1111/j.1463-1326.2009.01121.x.
ISSN
1463-1326.
PMC
2802270.
PMID
19817786.
{{ cite journal}}: CS1 maint: PMC format ( link) - ^ a b "PDX1 pancreatic and duodenal homeobox 1 [Homo sapiens (human)] - Gene - NCBI". www.ncbi.nlm.nih.gov. Retrieved 2021-04-14.
- ^ Caetano, L. A.; Santana, L. S.; Costa‐Riquetto, A. D.; Lerario, A. M.; Nery, M.; Nogueira, G. F.; Ortega, C. D.; Rocha, M. S.; Jorge, A. a. L.; Teles, M. G. (2018). "PDX1 -MODY and dorsal pancreatic agenesis: New phenotype of a rare disease". Clinical Genetics. 93 (2): 382–386. doi: 10.1111/cge.13044. ISSN 1399-0004.
-
^ Editor (2019-01-15).
"Beta cells are unique cells in the pancreas that produce, store and release the hormone insulin". Diabetes. Retrieved 2021-04-01.
{{ cite web}}:|last=has generic name ( help) - ^ "Missense Mutation". Genome.gov. Retrieved 2021-04-01.
- ^ Foundation, J. E. S. "J.E.S Foundation for Pancreatic Cancer". www.thejesfoundation.com. Retrieved 2021-04-14.
- ^ "PTF1A Gene - GeneCards | PTF1A Protein | PTF1A Antibody". www.genecards.org. Retrieved 2021-04-02.
- ^ Dong, P. Duc Si; Provost, Elayne; Leach, Steven D.; Stainier, Didier Y. R. (2008-06-01). "Graded levels of Ptf1a differentially regulate endocrine and exocrine fates in the developing pancreas". Genes & Development. 22 (11): 1445–1450. doi: 10.1101/gad.1663208. ISSN 0890-9369. PMID 18519637.
- ^ Houghton, Jayne A. L.; Swift, Galvin H.; Shaw-Smith, Charles; Flanagan, Sarah E.; Franco, Elisa de; Caswell, Richard; Hussain, Khalid; Mohamed, Sarar; Abdulrasoul, Majedah; Hattersley, Andrew T.; MacDonald, Raymond J. (2016-09-01). "Isolated Pancreatic Aplasia Due to a Hypomorphic PTF1A Mutation". Diabetes. 65 (9): 2810–2815. doi: 10.2337/db15-1666. ISSN 0012-1797. PMID 27284104.
- ^
a
b RESERVED, INSERM US14-- ALL RIGHTS.
"Orphanet: pancreas associated transcription factor 1a PTF1A". www.orpha.net. Retrieved 2021-04-02.
{{ cite web}}: CS1 maint: numeric names: authors list ( link) - ^ a b "Permanent neonatal diabetes mellitus: MedlinePlus Genetics". medlineplus.gov. Retrieved 2021-04-01.
-
^ Editor (2019-01-15).
"Neonatal diabetes is a rare form of diabetes that is usually diagnosed in children under 6 months of age". Diabetes. Retrieved 2021-04-01.
{{ cite web}}:|last=has generic name ( help) - ^ Salina, Alessandro; Pasquali, Lorenzo; Aloi, Concetta; Lugani, Francesca; d'Annunzio, Giuseppe; Lorini, Renata (2010-08-01). "Neonatal Diabetes Caused by Pancreatic Agenesia: Which other genes should be used for diagnosis?". Diabetes Care. 33 (8): e112–e112. doi: 10.2337/dc10-0876. ISSN 0149-5992. PMID 20668147.
- ^ a b c "(PDF) Dorsal Pancreatic Agenesis". ResearchGate. Retrieved 2021-04-14.
- ^ a b c Bissonnette, Bruno; Luginbuehl, Igor; Engelhardt, Thomas (2019), "Congenital Pancreatic Agenesis", Syndromes: Rapid Recognition and Perioperative Implications (2 ed.), New York, NY: McGraw-Hill Education, retrieved 2021-04-14
- ^ a b "WHO | Medical imaging". WHO. Retrieved 2021-04-13.
- ^ a b Schnedl, Wolfgang J.; Reisinger, Emil C.; Schreiber, Florian; Pieber, Thomas R.; Lipp, Rainer W.; Krejs, Guenter J. (1995-11-01). "Complete and partial agenesis of the dorsal pancreas within one family". Gastrointestinal Endoscopy. 42 (5): 485–487. doi: 10.1016/S0016-5107(95)70055-2. ISSN 0016-5107.
- ^ a b "Ultrasound: Purpose, Procedure, and Preparation". Healthline. 2016-05-05. Retrieved 2021-04-13.
- ^ Czarniecki, Marcin. "Pancreatic ultrasound | Radiology Reference Article | Radiopaedia.org". Radiopaedia. Retrieved 2021-04-13.
- ^ a b "What's the Difference Between an MRI and a CT?". Center For Diagnostic Imaging (CDI). 2016-03-30. Retrieved 2021-04-13.
-
^ Busireddy, Kiran K; AlObaidy, Mamdoh; Ramalho, Miguel; Kalubowila, Janaka; Baodong, Liu; Santagostino, Ilaria; Semelka, Richard C (2014-08-15).
"Pancreatitis-imaging approach". World Journal of Gastrointestinal Pathophysiology. 5 (3): 252–270.
doi:
10.4291/wjgp.v5.i3.252.
ISSN
2150-5330.
PMC
4133524.
PMID
25133027.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^
a
b
c Pasaoglu, Lale; Vural, Murat; Hatipoglu, Hatice Gul; Tereklioglu, Gokce; Koparal, Suha (2008-05-14).
"Agenesis of the dorsal pancreas". World Journal of Gastroenterology : WJG. 14 (18): 2915–2916.
doi:
10.3748/wjg.14.2915.
ISSN
1007-9327.
PMC
2710738.
PMID
18473421.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) - ^ "ERCP (Endoscopic Retrograde Cholangio-Pancreatography) Patient Information from SAGES". SAGES. Retrieved 2021-04-13.
- ^ Radiology (ACR), Radiological Society of North America (RSNA) and American College of. "Magnetic Resonance Cholangiopancreatography (MRCP)". Radiologyinfo.org. Retrieved 2021-04-13.
- ^
a
b Vijayaraghavan, SBoopathy; Gouru, Swapna; Senthil, Sathiya (2013).
"Sonographic features of agenesis of dorsal pancreas". Indian Journal of Radiology and Imaging. 23 (2): 179.
doi:
10.4103/0971-3026.116570.
ISSN
0971-3026.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) -
^ Hong, Cheng William; Fazeli Dehkordy, Soudabeh; Hooker, Jonathan C.; Hamilton, Gavin; Sirlin, Claude B. (2017-12-XX).
"Fat Quantification in the Abdomen". Topics in Magnetic Resonance Imaging. 26 (6): 221–227.
doi:
10.1097/RMR.0000000000000141.
ISSN
1536-1004.
{{ cite journal}}: Check date values in:|date=( help) - ^ "Dorsal Pancreatic Agenesis". scholar.googleusercontent.com. Retrieved 2021-04-01.
-
^ Gutta, Aditya; Fogel, Evan; Sherman, Stuart (2019-11-02).
"Identification and management of pancreas divisum". Expert Review of Gastroenterology & Hepatology. 13 (11): 1089–1105.
doi:
10.1080/17474124.2019.1685871.
ISSN
1747-4124.
PMC
6872911.
PMID
31663403.
{{ cite journal}}: CS1 maint: PMC format ( link) - ^ Weerakkody, Yuranga. "Dorsal pancreatic agenesis | Radiology Reference Article | Radiopaedia.org". Radiopaedia. Retrieved 2021-04-01.
- ^ a b c d e "Agenesis of the dorsal pancreas | Genetic and Rare Diseases Information Center (GARD) – an NCATS Program". rarediseases.info.nih.gov. Retrieved 2021-04-13.
- ^ a b "Hyperglycemia: Symptoms, causes, and treatments". www.medicalnewstoday.com. 2019-05-07. Retrieved 2021-04-01.
- ^ a b Sonkar, Satyendra Kumar; Kumar, Satish; Singh, Neeraj Kumar (2018-02-14). "Agenesis of dorsal pancreas in a young adult: a rare cause of diabetes mellitus". BMJ Case Reports: bcr–2017-223301. doi: 10.1136/bcr-2017-223301. ISSN 1757-790X.
- ^ "Five Things to Know About Ketones | ADA". www.diabetes.org. Retrieved 2021-04-13.
Warning: Default sort key "Pancreatic Agenesis" overrides earlier default sort key "Pancreatic agenesis".