Week 3 Tasks - Info for Chromium(III) AcetateSuperscriptExampleSubScriptExample

| |
| Names | |
|---|---|
|
IUPAC name
Chromium (iii) Acetate Hydrate
| |
| Identifiers | |
PubChem
CID
|
|
| Properties | |
| C6H9O6Cr | |
| Molar mass | 229.1g/mol |
| Appearance | Blue Crystals |
| Melting point | N/A |
| Boiling point | N/A |
| 675g/L (20C, pH 5) | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
-
Example picture. [1] (Example external link)
Aromaticity (Example internal link)
Example math formulas
Properties of Chromium(III) Acetate [2] [3] [4]
- Molecular formula - C6H9O6Cr
- Molar mass - 229.1 g/mol
- Melting point - N/A
- Boiling point - N/A
- Solubility (In water: 20oC, pH 5) - 675 g/L
Chromium(III) Acetate
| IR Peaks (nm) | Chemical Properties |
| 2900 | Molar mass: 229.1 g/mol |
| 1450 | Solubility: 675g/L |
| 650 |
Article Examples
"Interspecies Homology of Nitrogenase genes" [6]
"Isolation of an Iron-molybdenum cofactor from Nitrogenase" [7]
"Evidence for Interstitial Carbon in Nitrogenase FeMo cofactor" [8]
Carbon Monoxide Dehydrogenase (Early Contribution Revision+400 Word Equivalent)
The chemical process catalyzed by Carbon Monoxide Dehydrogenase is referred to as a water-gas shift reaction.
CO2 reduction requires quick and efficient electron transfers to deliver necessary electrons to a redox catalyst. Therefore, a variety of electron donors/receivers (Shown as "A" and "AH2" in the reaction equation above) are observed in micro-organisms which utilize CODH. Several examples of electron transfer cofactors include Ferredoxin, NADP+/NADPH and flavoprotein complexes like flavin adenine dinucleotide (FAD). [9] [10] [11]
Microbial organisms (Both Aerobic and Anaerobic) encode and synthesize CODH for the purpose of carbon fixation (CO oxidation and CO2 reduction). Depending on attached accessory proteins (A,B,C,D-Clusters), serve a variety of catalytic functions, including reduction of [4Fe-4S] clusters and insertion of Nickel. [12]
Classes (Adding onto the wiki article's section)
Based on the microbe's environmental conditions, different metal complex centers are synthesized. Aerobic carboxydotrophic bacteria utilize Copper (Cu),Molybdenum (Mo) and Iron (Fe) based flavoenzymes. Anaerobic bacteria utilize Nickel (Ni) and Iron (Fe) based CODHs due to their oxygen sensitive nature. [13]
Structure (Adding onto the wiki article's section)
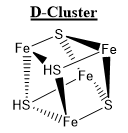
Homodimeric Ni-CODH consists of five metal complexes referred to as clusters. ..., each differing in individual coordination geometry, presence of Nickel, and location of the active site in either sub-unit α or β. [14]
Industrial Use (New Wiki Article Subheading)
Carbon monoxide dehydrogenase's or bio-mimetic complexes based on its catalytic potential in the formation of CO from CO2, shows potential future catalytic uses in industrial processes. Potential candidates include the production of CH3OH ( Acetic acid) from CO in processes found in the Cativa process and Monsanto process . Production of CO as a precursor for liquid hydrocarbons, in the Fischer-Trophsch process. [15]
Environmental relevance (Adding onto the wiki article's section)
Carbon Monoxide dehydrogenase is closely associated with the regulation of atmospheric CO and CO2 levels, maintaining optimal CO levels suitable for other forms of life. [16] [17] Microbial organisms rely on these enzymes for both energy conservation along with CO2 fixation. Often encoding for and synthesizing multiple unique forms of CODH for designated use. Further research into specific types of CODH show CO being used and condensed with CH3 ( Methyl groups) to form Acetyl-CoA. [18] Anaerobic micro-organisms like Acetogens undergo the Wood-Ljungdahl Pathway, relying on CODH to produce CO by reduction of CO2 needed for the synthesis of Acetyl-CoA from a methyl, coenzyme a (CoA) and corrinoid iron-sulfur protein. [19] Other types show CODH being utilized to generate a proton motive force for the purposes of energy generation. CODH is used for the CO oxidation, producing two protons which are subsequently reduced to form dihydrogen (H2, known colloquially as molecular hydrogen), providing the necessary free energy to drive ATP generation. [20]


 | This is a user sandbox of
ChemThings&Etc.. You can use it for testing or practicing edits. This is not the sandbox where you should draft your assigned article for a dashboard.wikiedu.org course. To find the right sandbox for your assignment, visit your Dashboard course page and follow the Sandbox Draft link for your assigned article in the My Articles section. |

- ^ "Benzene 401765". C6H6. Retrieved 2019-09-23.
-
^
"Chromium (iii) Acetate". Spectral Database for Organic Compounds. March 31st, 1999. Retrieved September 25th,2019.
{{ cite web}}: Check date values in:|access-date=and|date=( help)CS1 maint: url-status ( link) -
^
"Chromium (iii) Acetate". TOXNET: Toxicology Data Network. Retrieved September 25th,2019.
{{ cite web}}: Check date values in:|access-date=( help)CS1 maint: url-status ( link) - ^ "Chromium(III) acetate hydroxide, Blue crystals, Alfa Aesar | Fisher Scientific". www.fishersci.com. Retrieved 2019-09-26.
- ^ "SDBS-IR-NIDA-71297". sdbs.db.aist.go.jp. Retrieved 2019-09-26.
- ^ Spatzal, Thomas; Aksoyoglu, Müge; Zhang, Limei; Andrade, Susana L. A.; Schleicher, Erik; Weber, Stefan; Rees, Douglas C.; Einsle, Oliver (2011-11-18). "Evidence for Interstitial Carbon in Nitrogenase FeMo Cofactor". Science. 334 (6058): 940–940. doi: 10.1126/science.1214025. ISSN 0036-8075. PMID 22096190.
- ^ Shah, Vinod K.; Brill, Winston J. (1977-08-01). "Isolation of an iron-molybdenum cofactor from nitrogenase". Proceedings of the National Academy of Sciences. 74 (8): 3249–3253. doi: 10.1073/pnas.74.8.3249. ISSN 0027-8424. PMID 410019.
- ^ Ruvkun, G. B.; Ausubel, F. M. (1980-01-01). "Interspecies homology of nitrogenase genes". Proceedings of the National Academy of Sciences. 77 (1): 191–195. doi: 10.1073/pnas.77.1.191. ISSN 0027-8424. PMID 6987649.
-
^ Buckel, Wolfgang; Thauer, Rudolf K. (2018).
"Flavin-Based Electron Bifurcation, Ferredoxin, Flavodoxin, and Anaerobic Respiration With Protons (Ech) or NAD+ (Rnf) as Electron Acceptors: A Historical Review". Frontiers in Microbiology. 9.
doi:
10.3389/fmicb.2018.00401.
ISSN
1664-302X.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) -
^ Kracke, Frauke; Virdis, Bernardino; Bernhardt, Paul V.; Rabaey, Korneel; Krömer, Jens O. (2016-12).
"Redox dependent metabolic shift in Clostridium autoethanogenum by extracellular electron supply". Biotechnology for Biofuels. 9 (1): 249.
doi:
10.1186/s13068-016-0663-2.
ISSN
1754-6834.
PMC
5112729.
PMID
27882076.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^ Berg, Willy A. M. van den; Hagen, Wilfred R.; Dongen, Walter M. A. M. van (2000). "The hybrid-cluster protein ('prismane protein') from Escherichia coli". European Journal of Biochemistry. 267 (3): 666–676. doi: 10.1046/j.1432-1327.2000.01032.x. ISSN 1432-1033.
- ^ Hadj-Saïd, Jessica; Pandelia, Maria-Eirini; Léger, Christophe; Fourmond, Vincent; Dementin, Sébastien (2015-12-01). "The Carbon Monoxide Dehydrogenase from Desulfovibrio vulgaris". Biochimica et Biophysica Acta (BBA) - Bioenergetics. 1847 (12): 1574–1583. doi: 10.1016/j.bbabio.2015.08.002. ISSN 0005-2728.
-
^ Jeoung, Jae-Hun; Dobbek, Holger (November 30th,2007).
"Carbon Dioxide Activation at the Ni,Fe-Cluster of Anaerobic Carbon Monoxide Dehydrogenase". Science. 318 (5855). American Association for the Advancement of Science: 1461–1464 – via JSTOR.
{{ cite journal}}: Check date values in:|date=( help); line feed character in|title=at position 33 ( help) -
^ Wittenborn, Elizabeth C; Merrouch, Mériem; Ueda, Chie; Fradale, Laura; Léger, Christophe; Fourmond, Vincent; Pandelia, Maria-Eirini; Dementin, Sébastien; Drennan, Catherine L (2018-10-02).
"Redox-dependent rearrangements of the NiFeS cluster of carbon monoxide dehydrogenase". eLife. 7: e39451.
doi:
10.7554/eLife.39451.
ISSN
2050-084X.
PMC
6168284.
PMID
30277213.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^ Jeoung, Jae-Hun; Martins, Berta M.; Dobbek, Holger (2019), Hu, Yilin (ed.), "Carbon Monoxide Dehydrogenases", Metalloproteins, vol. 1876, Springer New York, pp. 37–54, doi: 10.1007/978-1-4939-8864-8_3, ISBN 9781493988631, retrieved 2019-10-09
- ^ Hu, Zhengguo; Spangler, Nathan J.; Anderson, Mark E.; Xia, Jinqiang; Ludden, Paul W.; Lindahl, Paul A.; Münck, Eckard (1996-01-01). "Nature of the C-Cluster in Ni-Containing Carbon Monoxide Dehydrogenases". Journal of the American Chemical Society. 118 (4): 830–845. doi: 10.1021/ja9528386. ISSN 0002-7863.
- ^ Bartholomew, G.W.; Alexander, M. (1979). "Microbial metabolism of carbon monoxide in culture and in soil". Applied and Environmental Microbiology. 37 (5): 932–7. PMC 243327. PMID 485139. Retrieved 2010-05-15.
-
^ Hadj-Saïd, Jessica; Pandelia, Maria-Eirini; Léger, Christophe; Fourmond, Vincent; Dementin, Sébastien (2015-12).
"The Carbon Monoxide Dehydrogenase from Desulfovibrio vulgaris". Biochimica et Biophysica Acta (BBA) - Bioenergetics. 1847 (12): 1574–1583.
doi:
10.1016/j.bbabio.2015.08.002.
{{ cite journal}}: Check date values in:|date=( help) -
^ Ragsdale, Stephen W.; Pierce, Elizabeth (2008-12).
"Acetogenesis and the Wood–Ljungdahl pathway of CO2 fixation". Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics. 1784 (12): 1873–1898.
doi:
10.1016/j.bbapap.2008.08.012.
PMC
2646786.
PMID
18801467.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) - ^ Ensign, S. A.; Ludden, P. W. (1991-09-25). "Characterization of the CO oxidation/H2 evolution system of Rhodospirillum rubrum. Role of a 22-kDa iron-sulfur protein in mediating electron transfer between carbon monoxide dehydrogenase and hydrogenase". The Journal of Biological Chemistry. 266 (27): 18395–18403. ISSN 0021-9258. PMID 1917963.
Week 3 Tasks - Info for Chromium(III) AcetateSuperscriptExampleSubScriptExample

| |
| Names | |
|---|---|
|
IUPAC name
Chromium (iii) Acetate Hydrate
| |
| Identifiers | |
PubChem
CID
|
|
| Properties | |
| C6H9O6Cr | |
| Molar mass | 229.1g/mol |
| Appearance | Blue Crystals |
| Melting point | N/A |
| Boiling point | N/A |
| 675g/L (20C, pH 5) | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
-
Example picture. [1] (Example external link)
Aromaticity (Example internal link)
Example math formulas
Properties of Chromium(III) Acetate [2] [3] [4]
- Molecular formula - C6H9O6Cr
- Molar mass - 229.1 g/mol
- Melting point - N/A
- Boiling point - N/A
- Solubility (In water: 20oC, pH 5) - 675 g/L
Chromium(III) Acetate
| IR Peaks (nm) | Chemical Properties |
| 2900 | Molar mass: 229.1 g/mol |
| 1450 | Solubility: 675g/L |
| 650 |
Article Examples
"Interspecies Homology of Nitrogenase genes" [6]
"Isolation of an Iron-molybdenum cofactor from Nitrogenase" [7]
"Evidence for Interstitial Carbon in Nitrogenase FeMo cofactor" [8]
Carbon Monoxide Dehydrogenase (Early Contribution Revision+400 Word Equivalent)
The chemical process catalyzed by Carbon Monoxide Dehydrogenase is referred to as a water-gas shift reaction.
CO2 reduction requires quick and efficient electron transfers to deliver necessary electrons to a redox catalyst. Therefore, a variety of electron donors/receivers (Shown as "A" and "AH2" in the reaction equation above) are observed in micro-organisms which utilize CODH. Several examples of electron transfer cofactors include Ferredoxin, NADP+/NADPH and flavoprotein complexes like flavin adenine dinucleotide (FAD). [9] [10] [11]
Microbial organisms (Both Aerobic and Anaerobic) encode and synthesize CODH for the purpose of carbon fixation (CO oxidation and CO2 reduction). Depending on attached accessory proteins (A,B,C,D-Clusters), serve a variety of catalytic functions, including reduction of [4Fe-4S] clusters and insertion of Nickel. [12]
Classes (Adding onto the wiki article's section)
Based on the microbe's environmental conditions, different metal complex centers are synthesized. Aerobic carboxydotrophic bacteria utilize Copper (Cu),Molybdenum (Mo) and Iron (Fe) based flavoenzymes. Anaerobic bacteria utilize Nickel (Ni) and Iron (Fe) based CODHs due to their oxygen sensitive nature. [13]
Structure (Adding onto the wiki article's section)
Homodimeric Ni-CODH consists of five metal complexes referred to as clusters. ..., each differing in individual coordination geometry, presence of Nickel, and location of the active site in either sub-unit α or β. [14]
Industrial Use (New Wiki Article Subheading)
Carbon monoxide dehydrogenase's or bio-mimetic complexes based on its catalytic potential in the formation of CO from CO2, shows potential future catalytic uses in industrial processes. Potential candidates include the production of CH3OH ( Acetic acid) from CO in processes found in the Cativa process and Monsanto process . Production of CO as a precursor for liquid hydrocarbons, in the Fischer-Trophsch process. [15]
Environmental relevance (Adding onto the wiki article's section)
Carbon Monoxide dehydrogenase is closely associated with the regulation of atmospheric CO and CO2 levels, maintaining optimal CO levels suitable for other forms of life. [16] [17] Microbial organisms rely on these enzymes for both energy conservation along with CO2 fixation. Often encoding for and synthesizing multiple unique forms of CODH for designated use. Further research into specific types of CODH show CO being used and condensed with CH3 ( Methyl groups) to form Acetyl-CoA. [18] Anaerobic micro-organisms like Acetogens undergo the Wood-Ljungdahl Pathway, relying on CODH to produce CO by reduction of CO2 needed for the synthesis of Acetyl-CoA from a methyl, coenzyme a (CoA) and corrinoid iron-sulfur protein. [19] Other types show CODH being utilized to generate a proton motive force for the purposes of energy generation. CODH is used for the CO oxidation, producing two protons which are subsequently reduced to form dihydrogen (H2, known colloquially as molecular hydrogen), providing the necessary free energy to drive ATP generation. [20]



 | This is a user sandbox of
ChemThings&Etc.. You can use it for testing or practicing edits. This is not the sandbox where you should draft your assigned article for a dashboard.wikiedu.org course. To find the right sandbox for your assignment, visit your Dashboard course page and follow the Sandbox Draft link for your assigned article in the My Articles section. |

- ^ "Benzene 401765". C6H6. Retrieved 2019-09-23.
-
^
"Chromium (iii) Acetate". Spectral Database for Organic Compounds. March 31st, 1999. Retrieved September 25th,2019.
{{ cite web}}: Check date values in:|access-date=and|date=( help)CS1 maint: url-status ( link) -
^
"Chromium (iii) Acetate". TOXNET: Toxicology Data Network. Retrieved September 25th,2019.
{{ cite web}}: Check date values in:|access-date=( help)CS1 maint: url-status ( link) - ^ "Chromium(III) acetate hydroxide, Blue crystals, Alfa Aesar | Fisher Scientific". www.fishersci.com. Retrieved 2019-09-26.
- ^ "SDBS-IR-NIDA-71297". sdbs.db.aist.go.jp. Retrieved 2019-09-26.
- ^ Spatzal, Thomas; Aksoyoglu, Müge; Zhang, Limei; Andrade, Susana L. A.; Schleicher, Erik; Weber, Stefan; Rees, Douglas C.; Einsle, Oliver (2011-11-18). "Evidence for Interstitial Carbon in Nitrogenase FeMo Cofactor". Science. 334 (6058): 940–940. doi: 10.1126/science.1214025. ISSN 0036-8075. PMID 22096190.
- ^ Shah, Vinod K.; Brill, Winston J. (1977-08-01). "Isolation of an iron-molybdenum cofactor from nitrogenase". Proceedings of the National Academy of Sciences. 74 (8): 3249–3253. doi: 10.1073/pnas.74.8.3249. ISSN 0027-8424. PMID 410019.
- ^ Ruvkun, G. B.; Ausubel, F. M. (1980-01-01). "Interspecies homology of nitrogenase genes". Proceedings of the National Academy of Sciences. 77 (1): 191–195. doi: 10.1073/pnas.77.1.191. ISSN 0027-8424. PMID 6987649.
-
^ Buckel, Wolfgang; Thauer, Rudolf K. (2018).
"Flavin-Based Electron Bifurcation, Ferredoxin, Flavodoxin, and Anaerobic Respiration With Protons (Ech) or NAD+ (Rnf) as Electron Acceptors: A Historical Review". Frontiers in Microbiology. 9.
doi:
10.3389/fmicb.2018.00401.
ISSN
1664-302X.
{{ cite journal}}: CS1 maint: unflagged free DOI ( link) -
^ Kracke, Frauke; Virdis, Bernardino; Bernhardt, Paul V.; Rabaey, Korneel; Krömer, Jens O. (2016-12).
"Redox dependent metabolic shift in Clostridium autoethanogenum by extracellular electron supply". Biotechnology for Biofuels. 9 (1): 249.
doi:
10.1186/s13068-016-0663-2.
ISSN
1754-6834.
PMC
5112729.
PMID
27882076.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^ Berg, Willy A. M. van den; Hagen, Wilfred R.; Dongen, Walter M. A. M. van (2000). "The hybrid-cluster protein ('prismane protein') from Escherichia coli". European Journal of Biochemistry. 267 (3): 666–676. doi: 10.1046/j.1432-1327.2000.01032.x. ISSN 1432-1033.
- ^ Hadj-Saïd, Jessica; Pandelia, Maria-Eirini; Léger, Christophe; Fourmond, Vincent; Dementin, Sébastien (2015-12-01). "The Carbon Monoxide Dehydrogenase from Desulfovibrio vulgaris". Biochimica et Biophysica Acta (BBA) - Bioenergetics. 1847 (12): 1574–1583. doi: 10.1016/j.bbabio.2015.08.002. ISSN 0005-2728.
-
^ Jeoung, Jae-Hun; Dobbek, Holger (November 30th,2007).
"Carbon Dioxide Activation at the Ni,Fe-Cluster of Anaerobic Carbon Monoxide Dehydrogenase". Science. 318 (5855). American Association for the Advancement of Science: 1461–1464 – via JSTOR.
{{ cite journal}}: Check date values in:|date=( help); line feed character in|title=at position 33 ( help) -
^ Wittenborn, Elizabeth C; Merrouch, Mériem; Ueda, Chie; Fradale, Laura; Léger, Christophe; Fourmond, Vincent; Pandelia, Maria-Eirini; Dementin, Sébastien; Drennan, Catherine L (2018-10-02).
"Redox-dependent rearrangements of the NiFeS cluster of carbon monoxide dehydrogenase". eLife. 7: e39451.
doi:
10.7554/eLife.39451.
ISSN
2050-084X.
PMC
6168284.
PMID
30277213.
{{ cite journal}}: CS1 maint: PMC format ( link) CS1 maint: unflagged free DOI ( link) - ^ Jeoung, Jae-Hun; Martins, Berta M.; Dobbek, Holger (2019), Hu, Yilin (ed.), "Carbon Monoxide Dehydrogenases", Metalloproteins, vol. 1876, Springer New York, pp. 37–54, doi: 10.1007/978-1-4939-8864-8_3, ISBN 9781493988631, retrieved 2019-10-09
- ^ Hu, Zhengguo; Spangler, Nathan J.; Anderson, Mark E.; Xia, Jinqiang; Ludden, Paul W.; Lindahl, Paul A.; Münck, Eckard (1996-01-01). "Nature of the C-Cluster in Ni-Containing Carbon Monoxide Dehydrogenases". Journal of the American Chemical Society. 118 (4): 830–845. doi: 10.1021/ja9528386. ISSN 0002-7863.
- ^ Bartholomew, G.W.; Alexander, M. (1979). "Microbial metabolism of carbon monoxide in culture and in soil". Applied and Environmental Microbiology. 37 (5): 932–7. PMC 243327. PMID 485139. Retrieved 2010-05-15.
-
^ Hadj-Saïd, Jessica; Pandelia, Maria-Eirini; Léger, Christophe; Fourmond, Vincent; Dementin, Sébastien (2015-12).
"The Carbon Monoxide Dehydrogenase from Desulfovibrio vulgaris". Biochimica et Biophysica Acta (BBA) - Bioenergetics. 1847 (12): 1574–1583.
doi:
10.1016/j.bbabio.2015.08.002.
{{ cite journal}}: Check date values in:|date=( help) -
^ Ragsdale, Stephen W.; Pierce, Elizabeth (2008-12).
"Acetogenesis and the Wood–Ljungdahl pathway of CO2 fixation". Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics. 1784 (12): 1873–1898.
doi:
10.1016/j.bbapap.2008.08.012.
PMC
2646786.
PMID
18801467.
{{ cite journal}}: Check date values in:|date=( help)CS1 maint: PMC format ( link) - ^ Ensign, S. A.; Ludden, P. W. (1991-09-25). "Characterization of the CO oxidation/H2 evolution system of Rhodospirillum rubrum. Role of a 22-kDa iron-sulfur protein in mediating electron transfer between carbon monoxide dehydrogenase and hydrogenase". The Journal of Biological Chemistry. 266 (27): 18395–18403. ISSN 0021-9258. PMID 1917963.
![Example picture.[1] (Example external link)](https://upload.wikimedia.org/wikipedia/commons/thumb/4/42/Benzene-Kekule-2D-skeletal.png/106px-Benzene-Kekule-2D-skeletal.png)


