The Human Cognome Project is an effort to reverse engineer the human brain by studying both its structure and function, in order to fully understand mental processes, also known as cognition. The project has many parallels to the Human Genome Project. The human cognome project spans various scientific fields, including neuroscience, cognitive science, artificial intelligence, and psychology. A better understanding of the cognome can illuminate how the brain perceives and responds to the environment, thereby augmenting artificial intelligence technology [1]. It also has many important implications for the study of disease progression by observing changes in cognition to localized damage. A map of the cognome will also increase mechanistic understandings of the brain.
Project
Robert Horn, an American political scientist best known for his development of information mapping, conceived the Human Cognome Project as an effort to elucidate social messes and convert human cognition to computing. He suggested the project to the National Science Foundation in 2002. He thought that by mapping the cognome genetically and cognitively, thought processing could be transformed to computing and confer a huge evolutionary leap in cognition and technology. Horn also suggested that the project explore visual language as a tool for human prostheses. He suggests that mapping out the human cognome could help unfold mechanisms of chunking and an understanding of the cognitive processes of the human brain. [2]
In 2003 Paul Allen pledged 100 million dollars to cofound the Allen Institute for Brain Science whose main purpose was to discover how the brain works. Researchers used the mouse model to map 20,000 genes down to their cellular functions. The mouse model was used as an experimental model because of the simplicity of working with mice in the laboratory as well similarity to human DNA. In 2008, Allen began a similar initiative exploring the mouse spinal cord gene map coined "Allen Spinal Cord Atlas." Both initiatives are aimed at clarifying neurological disorders through mapping the genes involved in the central nervous system. [3] [4]
On November 3, 2011, the Allen Institute for Brain Science launched a new brain atlas resource called the Allen Mouse Brain Connectivity Atlas, which, unlike the institute's previous mapping efforts, focuses more on neural circuitry than gene expression. [5] This new brain atlas allows the user to interact with the axonal projections that connect different brain areas. These “information highways” form the circuits responsible for brain functions like behavior and perception. [5] This three-dimensional, high-resolution map of neural connections will help scientists better understand how the brain is wired, which can lead to new understanding of brain function and neurological disorders and diseases.
State of Completion
The Human Cognome Project has not to this date been completed. Ongoing research in the fields of cognitive science, psychology, and neuroscience will help move the project forward.
Development of brain morphology
Brain morphology is the structure of the brain and its distinctive structural features. These features are determined during development, when the cells are differentiating into their respective types that become the brain's well-known structure. Neurogenesis usually populates the growing brain, however the process can continue into adulthood in the hippocampus and subventricular zone. Declining neurogenesis correlates with a decrease in cognitive performance [6]. The understanding of morphology provides a connection to cognition, which aids in the study of the cognome.
Glial monorail

Neurons migrate to specific regions of the brain using glia, specifically astroglia. The structure of the brain centers on the neural and glial networks of which it is composed. The structure and interactions of these cells form the morphology of the brain. Early neuronal migrations are guided by radial glia ( astrocytes) that serve as scaffolds to deliver neurons to their proper layered location [7] . All neurons do not necessarily use the glial scaffold, only the cells that are layered [8]. This includes cells in the cortex, hippocampus, cerebellum, and spinal cord. Radial glia are the first cells that can be distinguished from neuroepithelium and are the first cells to appear during neurogenesis [9]. In the adult brain, neural precursor cells in the ventricular zone and subventricular zone migrate to their final destinations to maintain brain morphology [8].
Inductive factors
The early morphology of the brain is dictated by transcription and growth factors. Inductive signals promoting both neuronal and glial development include retinoic acid, fibroblast growth factor, bone morphogenetic proteins, and sonic hedgehog [9]. These factors regulate the Hox genes that promote development of the major brain regions, thus contributing to brain morphology. Hox genes have also been implicated in directing neural cell fate [10]. Cell fate refers to the cell type that will develop later in differentiation--whether it be a neuron or glial cell in this case specifically. Early in development, astrocytes strongly support axon growth. As the brain ages, this ability decreases (as seen by astrocyte scars that inhibit axonal growth) [8]. Early brain formation and neuronal regeneration into adulthood is controlled by many complex, interacting factors.
Other transcription factors affect the basic structure of the neuron, giving it its characteristic polarized morphology. The polarized morphology refers to the differentiation between dendrites present on the cell body and the axon which propagates the action potential away from the cell body. This polarization is essential to generating neuronal circuits in the developing brain. Factors that affect the development of the axon include molecular scaffolds, Rho-GTPases and their regulators, protein kinases, kinesin motors, and microtubule-associated proteins (MAPs) [11]. Extracellular signals promoting axon development include neurotrophin BDNF and the growth factor TGF-β that act through the protein kinases SAD-A/B and the Par complex [11]. Other signals promote the development of dendritic spines. Semaphorin 3A promotes the recruitment of dendritic spines to nascent neurons [11]. Foxo transcription factors have also been implicated as major regulators of the development of neuronal polarity [11]. The human cognome project will help uncover the complex relationships between transcription factors and their implicated mechanisms in neuron development, location, and morphology.
Cognitive Science
Brain mapping
Functional MRI (fMRI) has been utilized in the mapping of different functional regions of the human cortex by recording changes in brain hemodynamics [12]. This ability to directly observe brain function can lead to new understanding of brain organization. Despite its relative infancy, fMRI has already been used to identify abnormal functional brain activity, study the modulatory effects of genetic risk factors for neurological disease on brain activation, predict the course of a disease, and reveal processes related to neuroplasticity [12]. As more advances in this technology are discovered, new applications for fMRI may be utilized which could provide insights into the function of fine-scale neural circuitry of the human brain in health and disease [12].
fMRI and early differential diagnosis
There is a growing body of evidence that suggests alterations in synaptic function contribute to the pathophysiological process of Alzheimer’s disease (AD) and related disorders [12]. fMRI may be useful in detecting these alterations in brain function long before clinical symptoms and specific neuropathology develop. FMRI detects alterations in the pattern and degree of regional brain activation during task performance, which is particularly advantageous in the early discovery and differential diagnosis of disorders causing cognitive impairment [12]. For instance, fMRI may be helpful in distinguishing a neurodegenerative process from depression in elderly individuals with cognitive symptoms. Studies using fMRI in this setting showed that hippocampal activation during memory tasks was decreased in AD patients compared with controls and depressed patients. On the other hand, orbitofrontal and cingulated activation were greater in depressed patients than in AD subjects and controls [12]. This ability to distinguish one cognitive process from another provides further insight into brain function that is particularly useful for the Human Cognome Project.
MRI and autism
The use of brain mapping and MRI have helped shed light on previously elusive neurological disorders. Analyzing the autistic brain using MRI and fMRI have helped uncover disease mechanisms and connectivity problems implicated in autism. Recent studies have delved into elucidating the structural connectivity issues implicated in autism. Using MRI and fMRI researchers have found decelerated white matter in the frontal, temporal, parietal, and occipital lobes [13]. The impaired growth of white matter in these regions provides a link between the characteristic social impairments associated with autism [13]. Connectivity in cortical networks was also explored using fMRI. White matter was observed to be depleted using fMRI in brain regions involved with social behaviors and language [13]. The sources of demyelination in these areas remains an ongoing research topic. The use of fMRI in the Human Cognome Project will help elucidate brain disease mechanisms and deficiencies in neurological disorders.
MRI and schizophrenia
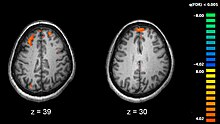
Schizophrenia is linked to dysfunction in the insular cortex region of the brain. In a recent study, real time fMRI ( rtfMRII) was used to track the blood oxygen level in this region by patients who were trained to exert control over their insular region [14]. The insula has been linked to emotion recognition, an ability lacking in many schizophrenic patients [15]. Previous studies have shown that the insula is depleted in schizophrenic patients. rtfMRI can be used to enhance neural networks and support behavioral modifications in schizophrenic populations. Mapping neurological disorders using fMRI can help the Human Cognome project uncover the mechanisms underlying schizophrenia and other diseases.
Cognitive Disorders
Cognitive disorders are those that disrupt normal human thought processes by their nature. Dementia, delirium, Down's syndrome, and amnesia are all cognitive disorders. Other diseases, such as schizophrenia and progressive multifocal leukoencephalopathy can have cognitive implications as they progress [16]. The Human Cognome Project will provide an approach to studying and understanding these diseases, by first understanding the basic principles behind human cognition [17]. ** This citation is incorrect**
Brain Atlases
A brain atlas is a reference consisting of images of the brain, mapped-out for a specific purpose. The Allen Brain Atlas shows both brain anatomy and gene expression information using several imaging technologies, including MRI. [18] Regulation of gene expression in the brain determines morphogenesis and adaptability of an organism, and therefore leads to cognitive function. The Allen Institute’s atlas is an important step in understanding the human cognome.
Benefits
Success in mapping the human cognome would allow all people to better understand how their brain works. Additionally, understanding the connections that the brain makes would allow scientists to create technologies that are well-matched with human control, and can therefore perform tasks more in a more efficient and successful manner. [19]
Current Research
Allen Institute for Brain Science
On November 3, 2011, the Allen Institute for Brain Science launched a new brain atlas resource called the Allen Mouse Brain Connectivity Atlas, which, unlike the institute's previous mapping efforts, focuses more on neural circuitry than gene expression.[5] This new brain atlas allows the user to interact with the axonal projections that connect different brain areas. These “information highways” form the circuits responsible for brain functions like behavior and perception.[5] This three-dimensional, high-resolution map of neural connections will help scientists better understand how the brain is wired, which can lead to new understanding of brain function and neurological disorders and diseases.
Pluripotent Stem Cells

The brain is composed of numerous neural subtypes, which are determined by a neuron's position along the axis of the central nervous system, age, genes, and surrounding neurons [20]. Pluripotent stem cells have given researchers the opportunity to create multifarious populations of neurons in vitro. These neurons are genetically manipulated, which provides data to examine neurodevelopment in mammals. Genetic alterations cause changes in neuronal function, and therefore illuminate that gene's role in the cell. Stem cells that are induced into neurons found in the prosencephalon become either cortical projection neurons or ventral forebrain neurons. Cerebellar neurons are found in the hindbrain. Pluripotent stem cells are also induced to become neurons in the mesencephalon and spinal cord [20]. The neuron is the basic structure of the brain, and the discovery of specific neuronal functions in each of these main sections of the brain is a key to understanding the cognome. [20].
Diagnostics
There are four indices in the Wechsler Adult Intelligence Scale (WAIS), verbal comprehension (VCI), perceptual organization (POI), working memory (WMI), and processing speed (PSI). Significant relationships were found in researches between brain deficits and lesion regions. Lesions in left inferior frontal cortex impair performance in verbal comprehension (VCI), left frontal and parietal cortex in working memory (WMI), and right parietal cortex in perceptual organization (POI). Deficit in processing speed (PSI) was not correlated with lesions within a single location, but across both hemispheres. The correlation between dysfunctional brain areas and brain deficits make brain mapping a potentially very powerful diagnostic tool [21].
External links
- The Redwood Center for Theoretical Neuroscience (founded by Jeff Hawkins)
- This Is Your Brain Online "Paul Allen’s $100 million for mapping the brain will produce the largest trove of biological data ever." Allen Brain Atlas
- BrainMaps.org High-Resolution Brain Maps and Interactive Brain Atlases
- The Brain Mind Institute in Switzerland, affiliated with the University of Geneva, the University of Lausanne, and others.
References
- ^ Horn, Robert E. (2006). "To Think Bigger Thoughts: Why the Human Cognome Project Requires Visual Language Tools to Address Social Messes". ANNALS. 1013: 212–220. doi: 10.1196/annals.1305.015. PMID 15194616.
- ^ Horn, Robert E. (April 4, 2002). "Beginning to conceptualize the Human Cognome Project" (PDF). Retrieved November 27, 2011.
-
^ Mehta, Aalok (6 December 2011).
"First-Ever Brain 'Atlas' Completed".
National Geographic News.
{{ cite news}}: Cite has empty unknown parameter:|coauthors=( help) -
^ Lein, Ed S.; et al. (2007). "Genome-wide atlas of gene expression in the adult mouse brain".
Nature. 445 (7124): 168–176.
doi:
10.1038/nature05453.
PMID
17151600.
{{ cite journal}}: CS1 maint: date and year ( link) - ^ a b "New Allen Mouse Brain Connectivity Atlas". News-Medical.net. 2011-11-14. Retrieved 2011-12-07.
-
^ Lee, Star W.; Clemenson, Gregory D.; Gage, Fred H. (2011 18 October).
"New neurons in an aged brain". Behavioral Brain Research. 227 (2): 497–507.
doi:
10.1016/j.bbr.2011.10.009.
PMC
3264739.
PMID
22024433.
{{ cite journal}}: Check date values in:|date=( help) - ^ Hatten, Mary E. (1990). "Riding the glial monorail: a common mechanism for glial- guided neuronal migration in different regions of the developing mammalian brain". Trends in Neuroscience. 13 (5): 179–184. doi: 10.1016/0166-2236(90)90044-B. PMID 1693236.
- ^
a
b
c Verkhratsky, Alexei (2007).
Glial Neurobiology.
John Wiley & Sons, Ltd.
ISBN
978-0-470-51740-6.
{{ cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) ( help) - ^ a b Doetsch, Fiona (2003). "The glial identity of neural stem cells". Nature Neuroscience. 6 (11): 1127–1134. doi: 10.1038/nn1144. PMID 14583753.
- ^ Bami, Myrto; Episkopou, Vasso; Gavalas, Anthony; Gouti, Mina (2011). "Directed Neural Differentiation of Mouse Embryonic Stem Cells Is a Sensitive System for the Identification of Novel Hox Gene Effectors". PLOS ONE. 6 (5): 5. doi: 10.1371/journal.pone.0020197. PMC 3102681. PMID 21637844.
- ^ a b c d de la Torre-Ubieta, Luis; Bonni, Azad (2011). "Transcriptional Regulation of Neuronal Polarity and Morphogenesis in the Mammalian Brain". Neuron. 72 (1): 22–40. doi: 10.1016/j.neuron.2011.09.018. PMC 3391704. PMID 21982366.
- ^ a b c d e f Dickerson, Bradford C. (July 2007). "Advances in Functional Magnetic Resonance Imaging: Technology and Clinical Applications". ScienceDirect. 4 (3): 360–370. Retrieved 2011-10-26.
- ^ a b c Hua, Xue; Thompson, Paul M.; Leow, Alex D.; Madsen, Sarah K.; Caplan, Rochelle (2011). "Brain Growth Rate Abnormalities Visualized in Adolescents with Autism". Human Brain Mapping. 34 (2): 425–436. doi: 10.1002/hbm.21441. PMC 4144412. PMID 22021093.
- ^ Ruiz, Sergio; Lee, Sangkyun; Soekadar, Surjo R.; Cario, Andrea; Veit, Ralf; Kircher, Tilo; Birbaumer, Niels; Sitaram, Ranganatha (2011). "Acquired Self-control of Insula Cortex Modulates Emotion Recognition and Brain Network Connectivity in Schizophrenia". Human Brain Mapping. 34 (1): 200–212. doi: 10.1002/hbm.21427. PMC 6869886. PMID 22021045.
- ^ Makris, Nikos; Goldstein, Jill M.; Kennedy, David; Hodge, Steven M.; Caviness, Verne S.; Faraone, Stephen V.; Tsuang, Ming T.; Seidman, Larry J. (2006). "Decreased volume of left and total anterior insular lobule in schizophrenia". Schizophrenia Research. 83 (2–3): 155–171. doi: 10.1016/j.schres.2005.11.020. PMID 16448806.
- ^ Moczynski, Nancy (2007). "Cognitive Disorders" (PDF). Tufts University. Retrieved October 31, 2011.
- ^ Choi, W.S. "Expanding Human Cognition and Communication" (PDF). Retrieved December 8, 2011.
- ^ Allen, Paul (2011). "About the Allen Human Brain Atlas". Allen Institute for Brain Science. Retrieved November 1, 2011.
- ^ Bainbridge, Charles Q. "Expanding Human Cognition and Communication" (PDF). Retrieved December 8, 2011.
- ^
a
b
c Petros, Timothy J.; Tyson, Jennifer A.; Anderson, Stewart A. (12 October).
"Pluripotent stem cells for the study of CNS development". Frontiers in Molecular Science. 4: 30.
doi:
10.3389/fnmol.2011.00030.
PMC
3191505.
PMID
22016722.
{{ cite journal}}: Check date values in:|date=and|year=/|date=mismatch ( help) -
^ Gläscher, Jan.; Tyson, Jennifer A.; Paul, Lynn K. (12 March).
"Lesion Mapping of Cognitive Abilities Linked to Intelligence". Neuron. 61 (5): 681–691.
doi:
10.1016/j.neuron.2009.01.026.
PMC
2728583.
PMID
19285465.
{{ cite journal}}: Check date values in:|date=and|year=/|date=mismatch ( help)
The Human Cognome Project is an effort to reverse engineer the human brain by studying both its structure and function, in order to fully understand mental processes, also known as cognition. The project has many parallels to the Human Genome Project. The human cognome project spans various scientific fields, including neuroscience, cognitive science, artificial intelligence, and psychology. A better understanding of the cognome can illuminate how the brain perceives and responds to the environment, thereby augmenting artificial intelligence technology [1]. It also has many important implications for the study of disease progression by observing changes in cognition to localized damage. A map of the cognome will also increase mechanistic understandings of the brain.

Project
Robert Horn, an American political scientist best known for his development of information mapping, conceived the Human Cognome Project as an effort to elucidate social messes and convert human cognition to computing. He suggested the project to the National Science Foundation in 2002. He thought that by mapping the cognome genetically and cognitively, thought processing could be transformed to computing and confer a huge evolutionary leap in cognition and technology. Horn also suggested that the project explore visual language as a tool for human prostheses. He suggests that mapping out the human cognome could help unfold mechanisms of chunking and an understanding of the cognitive processes of the human brain. [2]
In 2003 Paul Allen pledged 100 million dollars to cofound the Allen Institute for Brain Science whose main purpose was to discover how the brain works. Researchers used the mouse model to map 20,000 genes down to their cellular functions. The mouse model was used as an experimental model because of the simplicity of working with mice in the laboratory as well similarity to human DNA. In 2008, Allen began a similar initiative exploring the mouse spinal cord gene map coined "Allen Spinal Cord Atlas." Both initiatives are aimed at clarifying neurological disorders through mapping the genes involved in the central nervous system. [3] [4]
On November 3, 2011, the Allen Institute for Brain Science launched a new brain atlas resource called the Allen Mouse Brain Connectivity Atlas, which, unlike the institute's previous mapping efforts, focuses more on neural circuitry than gene expression. [5] This new brain atlas allows the user to interact with the axonal projections that connect different brain areas. These “information highways” form the circuits responsible for brain functions like behavior and perception. [5] This three-dimensional, high-resolution map of neural connections will help scientists better understand how the brain is wired, which can lead to new understanding of brain function and neurological disorders and diseases.
State of Completion
The Human Cognome Project has not to this date been completed. Ongoing research in the fields of cognitive science, psychology, and neuroscience will help move the project forward.
Development of brain morphology
Brain morphology is the structure of the brain and its distinctive structural features. These features are determined during development, when the cells are differentiating into their respective types that become the brain's well-known structure. Neurogenesis usually populates the growing brain, however the process can continue into adulthood in the hippocampus and subventricular zone. Declining neurogenesis correlates with a decrease in cognitive performance [6]. The understanding of morphology provides a connection to cognition, which aids in the study of the cognome.
Glial monorail

Neurons migrate to specific regions of the brain using glia, specifically astroglia. The structure of the brain centers on the neural and glial networks of which it is composed. The structure and interactions of these cells form the morphology of the brain. Early neuronal migrations are guided by radial glia ( astrocytes) that serve as scaffolds to deliver neurons to their proper layered location [7] . All neurons do not necessarily use the glial scaffold, only the cells that are layered [8]. This includes cells in the cortex, hippocampus, cerebellum, and spinal cord. Radial glia are the first cells that can be distinguished from neuroepithelium and are the first cells to appear during neurogenesis [9]. In the adult brain, neural precursor cells in the ventricular zone and subventricular zone migrate to their final destinations to maintain brain morphology [8].
Inductive factors

The early morphology of the brain is dictated by transcription and growth factors. Inductive signals promoting both neuronal and glial development include retinoic acid, fibroblast growth factor, bone morphogenetic proteins, and sonic hedgehog [9]. These factors regulate the Hox genes that promote development of the major brain regions, thus contributing to brain morphology. Hox genes have also been implicated in directing neural cell fate [10]. Cell fate refers to the cell type that will develop later in differentiation--whether it be a neuron or glial cell in this case specifically. Early in development, astrocytes strongly support axon growth. As the brain ages, this ability decreases (as seen by astrocyte scars that inhibit axonal growth) [8]. Early brain formation and neuronal regeneration into adulthood is controlled by many complex, interacting factors.
Other transcription factors affect the basic structure of the neuron, giving it its characteristic polarized morphology. The polarized morphology refers to the differentiation between dendrites present on the cell body and the axon which propagates the action potential away from the cell body. This polarization is essential to generating neuronal circuits in the developing brain. Factors that affect the development of the axon include molecular scaffolds, Rho-GTPases and their regulators, protein kinases, kinesin motors, and microtubule-associated proteins (MAPs) [11]. Extracellular signals promoting axon development include neurotrophin BDNF and the growth factor TGF-β that act through the protein kinases SAD-A/B and the Par complex [11]. Other signals promote the development of dendritic spines. Semaphorin 3A promotes the recruitment of dendritic spines to nascent neurons [11]. Foxo transcription factors have also been implicated as major regulators of the development of neuronal polarity [11]. The human cognome project will help uncover the complex relationships between transcription factors and their implicated mechanisms in neuron development, location, and morphology.
Cognitive Science
Brain mapping
Functional MRI (fMRI) has been utilized in the mapping of different functional regions of the human cortex by recording changes in brain hemodynamics [12]. This ability to directly observe brain function can lead to new understanding of brain organization. Despite its relative infancy, fMRI has already been used to identify abnormal functional brain activity, study the modulatory effects of genetic risk factors for neurological disease on brain activation, predict the course of a disease, and reveal processes related to neuroplasticity [12]. As more advances in this technology are discovered, new applications for fMRI may be utilized which could provide insights into the function of fine-scale neural circuitry of the human brain in health and disease [12].
fMRI and early differential diagnosis
There is a growing body of evidence that suggests alterations in synaptic function contribute to the pathophysiological process of Alzheimer’s disease (AD) and related disorders [12]. fMRI may be useful in detecting these alterations in brain function long before clinical symptoms and specific neuropathology develop. FMRI detects alterations in the pattern and degree of regional brain activation during task performance, which is particularly advantageous in the early discovery and differential diagnosis of disorders causing cognitive impairment [12]. For instance, fMRI may be helpful in distinguishing a neurodegenerative process from depression in elderly individuals with cognitive symptoms. Studies using fMRI in this setting showed that hippocampal activation during memory tasks was decreased in AD patients compared with controls and depressed patients. On the other hand, orbitofrontal and cingulated activation were greater in depressed patients than in AD subjects and controls [12]. This ability to distinguish one cognitive process from another provides further insight into brain function that is particularly useful for the Human Cognome Project.
MRI and autism
The use of brain mapping and MRI have helped shed light on previously elusive neurological disorders. Analyzing the autistic brain using MRI and fMRI have helped uncover disease mechanisms and connectivity problems implicated in autism. Recent studies have delved into elucidating the structural connectivity issues implicated in autism. Using MRI and fMRI researchers have found decelerated white matter in the frontal, temporal, parietal, and occipital lobes [13]. The impaired growth of white matter in these regions provides a link between the characteristic social impairments associated with autism [13]. Connectivity in cortical networks was also explored using fMRI. White matter was observed to be depleted using fMRI in brain regions involved with social behaviors and language [13]. The sources of demyelination in these areas remains an ongoing research topic. The use of fMRI in the Human Cognome Project will help elucidate brain disease mechanisms and deficiencies in neurological disorders.
MRI and schizophrenia

Schizophrenia is linked to dysfunction in the insular cortex region of the brain. In a recent study, real time fMRI ( rtfMRII) was used to track the blood oxygen level in this region by patients who were trained to exert control over their insular region [14]. The insula has been linked to emotion recognition, an ability lacking in many schizophrenic patients [15]. Previous studies have shown that the insula is depleted in schizophrenic patients. rtfMRI can be used to enhance neural networks and support behavioral modifications in schizophrenic populations. Mapping neurological disorders using fMRI can help the Human Cognome project uncover the mechanisms underlying schizophrenia and other diseases.
Cognitive Disorders
Cognitive disorders are those that disrupt normal human thought processes by their nature. Dementia, delirium, Down's syndrome, and amnesia are all cognitive disorders. Other diseases, such as schizophrenia and progressive multifocal leukoencephalopathy can have cognitive implications as they progress [16]. The Human Cognome Project will provide an approach to studying and understanding these diseases, by first understanding the basic principles behind human cognition [17]. ** This citation is incorrect**
Brain Atlases
A brain atlas is a reference consisting of images of the brain, mapped-out for a specific purpose. The Allen Brain Atlas shows both brain anatomy and gene expression information using several imaging technologies, including MRI. [18] Regulation of gene expression in the brain determines morphogenesis and adaptability of an organism, and therefore leads to cognitive function. The Allen Institute’s atlas is an important step in understanding the human cognome.
Benefits
Success in mapping the human cognome would allow all people to better understand how their brain works. Additionally, understanding the connections that the brain makes would allow scientists to create technologies that are well-matched with human control, and can therefore perform tasks more in a more efficient and successful manner. [19]
Current Research
Allen Institute for Brain Science
On November 3, 2011, the Allen Institute for Brain Science launched a new brain atlas resource called the Allen Mouse Brain Connectivity Atlas, which, unlike the institute's previous mapping efforts, focuses more on neural circuitry than gene expression.[5] This new brain atlas allows the user to interact with the axonal projections that connect different brain areas. These “information highways” form the circuits responsible for brain functions like behavior and perception.[5] This three-dimensional, high-resolution map of neural connections will help scientists better understand how the brain is wired, which can lead to new understanding of brain function and neurological disorders and diseases.
Pluripotent Stem Cells

The brain is composed of numerous neural subtypes, which are determined by a neuron's position along the axis of the central nervous system, age, genes, and surrounding neurons [20]. Pluripotent stem cells have given researchers the opportunity to create multifarious populations of neurons in vitro. These neurons are genetically manipulated, which provides data to examine neurodevelopment in mammals. Genetic alterations cause changes in neuronal function, and therefore illuminate that gene's role in the cell. Stem cells that are induced into neurons found in the prosencephalon become either cortical projection neurons or ventral forebrain neurons. Cerebellar neurons are found in the hindbrain. Pluripotent stem cells are also induced to become neurons in the mesencephalon and spinal cord [20]. The neuron is the basic structure of the brain, and the discovery of specific neuronal functions in each of these main sections of the brain is a key to understanding the cognome. [20].
Diagnostics
There are four indices in the Wechsler Adult Intelligence Scale (WAIS), verbal comprehension (VCI), perceptual organization (POI), working memory (WMI), and processing speed (PSI). Significant relationships were found in researches between brain deficits and lesion regions. Lesions in left inferior frontal cortex impair performance in verbal comprehension (VCI), left frontal and parietal cortex in working memory (WMI), and right parietal cortex in perceptual organization (POI). Deficit in processing speed (PSI) was not correlated with lesions within a single location, but across both hemispheres. The correlation between dysfunctional brain areas and brain deficits make brain mapping a potentially very powerful diagnostic tool [21].
External links
- The Redwood Center for Theoretical Neuroscience (founded by Jeff Hawkins)
- This Is Your Brain Online "Paul Allen’s $100 million for mapping the brain will produce the largest trove of biological data ever." Allen Brain Atlas
- BrainMaps.org High-Resolution Brain Maps and Interactive Brain Atlases
- The Brain Mind Institute in Switzerland, affiliated with the University of Geneva, the University of Lausanne, and others.
References
- ^ Horn, Robert E. (2006). "To Think Bigger Thoughts: Why the Human Cognome Project Requires Visual Language Tools to Address Social Messes". ANNALS. 1013: 212–220. doi: 10.1196/annals.1305.015. PMID 15194616.
- ^ Horn, Robert E. (April 4, 2002). "Beginning to conceptualize the Human Cognome Project" (PDF). Retrieved November 27, 2011.
-
^ Mehta, Aalok (6 December 2011).
"First-Ever Brain 'Atlas' Completed".
National Geographic News.
{{ cite news}}: Cite has empty unknown parameter:|coauthors=( help) -
^ Lein, Ed S.; et al. (2007). "Genome-wide atlas of gene expression in the adult mouse brain".
Nature. 445 (7124): 168–176.
doi:
10.1038/nature05453.
PMID
17151600.
{{ cite journal}}: CS1 maint: date and year ( link) - ^ a b "New Allen Mouse Brain Connectivity Atlas". News-Medical.net. 2011-11-14. Retrieved 2011-12-07.
-
^ Lee, Star W.; Clemenson, Gregory D.; Gage, Fred H. (2011 18 October).
"New neurons in an aged brain". Behavioral Brain Research. 227 (2): 497–507.
doi:
10.1016/j.bbr.2011.10.009.
PMC
3264739.
PMID
22024433.
{{ cite journal}}: Check date values in:|date=( help) - ^ Hatten, Mary E. (1990). "Riding the glial monorail: a common mechanism for glial- guided neuronal migration in different regions of the developing mammalian brain". Trends in Neuroscience. 13 (5): 179–184. doi: 10.1016/0166-2236(90)90044-B. PMID 1693236.
- ^
a
b
c Verkhratsky, Alexei (2007).
Glial Neurobiology.
John Wiley & Sons, Ltd.
ISBN
978-0-470-51740-6.
{{ cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) ( help) - ^ a b Doetsch, Fiona (2003). "The glial identity of neural stem cells". Nature Neuroscience. 6 (11): 1127–1134. doi: 10.1038/nn1144. PMID 14583753.
- ^ Bami, Myrto; Episkopou, Vasso; Gavalas, Anthony; Gouti, Mina (2011). "Directed Neural Differentiation of Mouse Embryonic Stem Cells Is a Sensitive System for the Identification of Novel Hox Gene Effectors". PLOS ONE. 6 (5): 5. doi: 10.1371/journal.pone.0020197. PMC 3102681. PMID 21637844.
- ^ a b c d de la Torre-Ubieta, Luis; Bonni, Azad (2011). "Transcriptional Regulation of Neuronal Polarity and Morphogenesis in the Mammalian Brain". Neuron. 72 (1): 22–40. doi: 10.1016/j.neuron.2011.09.018. PMC 3391704. PMID 21982366.
- ^ a b c d e f Dickerson, Bradford C. (July 2007). "Advances in Functional Magnetic Resonance Imaging: Technology and Clinical Applications". ScienceDirect. 4 (3): 360–370. Retrieved 2011-10-26.
- ^ a b c Hua, Xue; Thompson, Paul M.; Leow, Alex D.; Madsen, Sarah K.; Caplan, Rochelle (2011). "Brain Growth Rate Abnormalities Visualized in Adolescents with Autism". Human Brain Mapping. 34 (2): 425–436. doi: 10.1002/hbm.21441. PMC 4144412. PMID 22021093.
- ^ Ruiz, Sergio; Lee, Sangkyun; Soekadar, Surjo R.; Cario, Andrea; Veit, Ralf; Kircher, Tilo; Birbaumer, Niels; Sitaram, Ranganatha (2011). "Acquired Self-control of Insula Cortex Modulates Emotion Recognition and Brain Network Connectivity in Schizophrenia". Human Brain Mapping. 34 (1): 200–212. doi: 10.1002/hbm.21427. PMC 6869886. PMID 22021045.
- ^ Makris, Nikos; Goldstein, Jill M.; Kennedy, David; Hodge, Steven M.; Caviness, Verne S.; Faraone, Stephen V.; Tsuang, Ming T.; Seidman, Larry J. (2006). "Decreased volume of left and total anterior insular lobule in schizophrenia". Schizophrenia Research. 83 (2–3): 155–171. doi: 10.1016/j.schres.2005.11.020. PMID 16448806.
- ^ Moczynski, Nancy (2007). "Cognitive Disorders" (PDF). Tufts University. Retrieved October 31, 2011.
- ^ Choi, W.S. "Expanding Human Cognition and Communication" (PDF). Retrieved December 8, 2011.
- ^ Allen, Paul (2011). "About the Allen Human Brain Atlas". Allen Institute for Brain Science. Retrieved November 1, 2011.
- ^ Bainbridge, Charles Q. "Expanding Human Cognition and Communication" (PDF). Retrieved December 8, 2011.
- ^
a
b
c Petros, Timothy J.; Tyson, Jennifer A.; Anderson, Stewart A. (12 October).
"Pluripotent stem cells for the study of CNS development". Frontiers in Molecular Science. 4: 30.
doi:
10.3389/fnmol.2011.00030.
PMC
3191505.
PMID
22016722.
{{ cite journal}}: Check date values in:|date=and|year=/|date=mismatch ( help) -
^ Gläscher, Jan.; Tyson, Jennifer A.; Paul, Lynn K. (12 March).
"Lesion Mapping of Cognitive Abilities Linked to Intelligence". Neuron. 61 (5): 681–691.
doi:
10.1016/j.neuron.2009.01.026.
PMC
2728583.
PMID
19285465.
{{ cite journal}}: Check date values in:|date=and|year=/|date=mismatch ( help)