Formyl cation, [HCO]+. Abundant in space, intermediate in the Gattermann-Koch reaction [1]. Can generate from formyl fluoride and antimony pentafluoride: [2]
- HC(O)F + SbF5 → [HCO]+ + [SbF6−
Can consider as protonated carbon monoxide (H+ + CO → HCO+), or formaldehyde from which a hydride ion has been abstracted (H2CO → H− + HCO+).
Structure
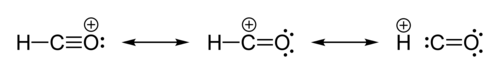
 |
 |
 |
| Species | Formula | CO bond order | r(C-O) / Å | r(C-H) / Å | technique | ref |
|---|---|---|---|---|---|---|
| carbon monoxide | CO | 3 | 1.128 | - | ? | G&E |
| formyl cation | HCO+ | 3 | 1.1107 | 1.0933 | MW | [3] |
| formyl radical | HCO | 2 | 1.1712 | 1.1110 | MW | [5] |
| formaldehyde | H2CO | 2 | 1.209 | 1.116 | MW | [5] |
| methanol | H3COH | 1 | 1.4246 | 1.0936 | MW | [5] |
Problem: bond lengths indicate C-O triple bond, but Lewis structure with C-O triple bond has wrong charges: +ve O and H, neutral C, whereas HF indicates neutral O, +ve C and H. Not just a simple matter of superimposing Lewis structures' wavefunctions - or is it?
Refs
- ^ J. Org. Chem. (1995) 60, 3846–3850
- ^ J. Phys. Chem. B (2001) 105, 8212–8219
- ^ a b J. Chem. Phys. (2008) 129, 164311
- ^ Astrophys. J. (1976) 205, L97-L100
- ^ a b c Lide, David R. (2008). CRC Handbook of Chemistry and Physics, 89th Edition. CRC Press. p. 9-36. ISBN 978-0849304880.
Formyl cation, [HCO]+. Abundant in space, intermediate in the Gattermann-Koch reaction [1]. Can generate from formyl fluoride and antimony pentafluoride: [2]
- HC(O)F + SbF5 → [HCO]+ + [SbF6−
Can consider as protonated carbon monoxide (H+ + CO → HCO+), or formaldehyde from which a hydride ion has been abstracted (H2CO → H− + HCO+).
Structure

 |
 |
 |
| Species | Formula | CO bond order | r(C-O) / Å | r(C-H) / Å | technique | ref |
|---|---|---|---|---|---|---|
| carbon monoxide | CO | 3 | 1.128 | - | ? | G&E |
| formyl cation | HCO+ | 3 | 1.1107 | 1.0933 | MW | [3] |
| formyl radical | HCO | 2 | 1.1712 | 1.1110 | MW | [5] |
| formaldehyde | H2CO | 2 | 1.209 | 1.116 | MW | [5] |
| methanol | H3COH | 1 | 1.4246 | 1.0936 | MW | [5] |
Problem: bond lengths indicate C-O triple bond, but Lewis structure with C-O triple bond has wrong charges: +ve O and H, neutral C, whereas HF indicates neutral O, +ve C and H. Not just a simple matter of superimposing Lewis structures' wavefunctions - or is it?
Refs
- ^ J. Org. Chem. (1995) 60, 3846–3850
- ^ J. Phys. Chem. B (2001) 105, 8212–8219
- ^ a b J. Chem. Phys. (2008) 129, 164311
- ^ Astrophys. J. (1976) 205, L97-L100
- ^ a b c Lide, David R. (2008). CRC Handbook of Chemistry and Physics, 89th Edition. CRC Press. p. 9-36. ISBN 978-0849304880.