| |

| |
| Names | |
|---|---|
|
IUPAC name
8-Methyl-8-azabicyclo[3.2.1]octan-3-one
| |
| Other names
3-Tropinone
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.007.756 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C8H13NO | |
| Molar mass | 139.195 g/mol |
| Appearance | Brown solid |
| Melting point | 42.5 °C (108.5 °F; 315.6 K) |
| Boiling point | (decomposes) |
| Hazards | |
| GHS labelling: | |

 [1]
[1]
| |
| Danger | |
| H302, H314 [1] | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Tropinone is an alkaloid, famously synthesised in 1917 by Robert Robinson as a synthetic precursor to atropine, a scarce commodity during World War I. [2] [3] Tropinone and the alkaloids cocaine and atropine all share the same tropane core structure. Its corresponding conjugate acid at pH 7.3 major species is known as tropiniumone. [4]
Synthesis
The first synthesis of tropinone was by Richard Willstätter in 1901. It started from the seemingly related cycloheptanone, but required many steps to introduce the nitrogen bridge; the overall yield for the synthesis path is only 0.75%. [5] Willstätter had previously synthesized cocaine from tropinone, in what was the first synthesis and elucidation of the structure of cocaine. [6]

Robinson's "double Mannich" reaction
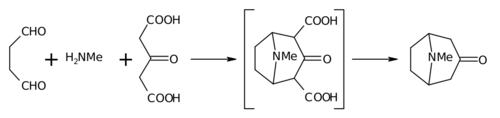
The 1917 synthesis by Robinson is considered a classic in total synthesis [8] due to its simplicity and biomimetic approach. Tropinone is a bicyclic molecule, but the reactants used in its preparation are fairly simple: succinaldehyde, methylamine and acetonedicarboxylic acid (or even acetone). The synthesis is a good example of a biomimetic reaction or biogenetic-type synthesis because biosynthesis makes use of the same building blocks. It also demonstrates a tandem reaction in a one-pot synthesis. Furthermore, the yield of the synthesis was 17% and with subsequent improvements exceeded 90%. [5]
This reaction is described as an intramolecular "double Mannich reaction" for obvious reasons. It is not unique in this regard, as others have also attempted it in piperidine synthesis. [9] [10]
In place of acetone, acetonedicarboxylic acid is known as the " synthetic equivalent" the 1,3-dicarboxylic acid groups are so-called " activating groups" to facilitate the ring forming reactions. The calcium salt is there as a " buffer" as it is claimed that higher yields are possible if the reaction is conducted at " physiological pH".
Reaction mechanism
The main features apparent from the reaction sequence below are:
- Nucleophilic addition of methylamine to succinaldehyde, followed by loss of water to create an imine
- Intramolecular addition of the imine to the second aldehyde unit and first ring closure
- Intermolecular Mannich reaction of the enolate of acetone dicarboxylate
- New enolate formation and new imine formation with loss of water for
- Second intramolecular Mannich reaction and second ring closure
- Loss of 2 carboxylic groups to tropinone
Some authors have actually tried to retain one of the CO2H groups. [11]
CO2R-tropinone has 4 stereoisomers, although the corresponding ecgonidine alkyl ester has only a pair of enantiomers.
From cycloheptanone
IBX dehydrogenation (oxidation) of cycloheptanone (suberone) to 2,6-cycloheptadienone [1192-93-4] followed by reaction with an amine is versatile a way of forming tropinones. [12] [13] The mechanism evoked is clearly delineated to be a double Michael reaction (i.e. conjugate addition).
Biochemistry method
|
| This section is empty. You can help by
adding to it. (April 2022) |
Reduction of tropinone
The reduction of tropinone is mediated by NADPH-dependent reductase enzymes, which have been characterized in multiple plant species. [15] These plant species all contain two types of the reductase enzymes, tropinone reductase I and tropinone reductase II. TRI produces tropine and TRII produces pseudotropine. Due to differing kinetic and pH/activity characteristics of the enzymes and by the 25-fold higher activity of TRI over TRII, the majority of the tropinone reduction is from TRI to form tropine. [16]

See also
- Benztropine
- Daturaolone
- 2-Carbomethoxytropinone (2-CMT) an intermediate in the creation of ecgonine cocaine analogues
- Ecgonidine
References
- ^ a b "Tropinone". Substance Information. ECHA.
- ^ Robinson R (1917). "LXIII. A Synthesis of Tropinone". Journal of the Chemical Society, Transactions. 111: 762–768. doi: 10.1039/CT9171100762.
- ^ Nicolaou KC, Vourloumis D, Winssinger N, Baran PS (2000). "The Art and Science of Total Synthesis at the Dawn of the Twenty-First Century". Angewandte Chemie International Edition. 39 (1): 44–122. doi: 10.1002/(SICI)1521-3773(20000103)39:1<44::AID-ANIE44>3.0.CO;2-L. PMID 10649349.
- ^ Chemical Entities of Biological Interest Identification code: ChEBI:57851 "tropiniumone"
- ^ a b Smit WA, Smit WA, Bochkov AF, Caple R (1998). Organic Synthesis. doi: 10.1039/9781847551573. ISBN 978-0-85404-544-0.
- ^ Humphrey AJ, O'Hagan D (2001). "Tropane alkaloid biosynthesis. A century old problem unresolved". Natural Product Reports. 18 (5). Royal Society of Chemistry: 494–502. doi: 10.1039/b001713m. PMID 11699882.
- ^ Doble M, Kruthiventi AK (2007). Green Chemistry and Engineering. Oxford: Elsevier. p. 34. ISBN 978-0-12-372532-5.
- ^ Birch AJ (1993). "Investigating a Scientific Legend: The Tropinone Synthesis of Sir Robert Robinson, F.R.S". Notes and Records of the Royal Society of London. 47 (2): 277–296. doi: 10.1098/rsnr.1993.0034. JSTOR 531792. S2CID 143267467.
- ^ Wang S, Sakamuri S, Enyedy IJ, Kozikowski AP, Deschaux O, Bandyopadhyay BC, Tella SR, Zaman WA, Johnson KM (2000). "Discovery of a novel dopamine transporter inhibitor, 4-hydroxy-1-methyl-4-(4-methylphenyl)-3-piperidyl 4-methylphenyl ketone, as a potential cocaine antagonist through 3D-database pharmacophore searching. Molecular modeling, structure-activity relationships, and behavioral pharmacological studies". Journal of Medicinal Chemistry. 43 (3): 351–360. doi: 10.1021/jm990516x. PMID 10669562.
- ^ Wang S, Sakamuri, Enyedy, Kozikowski, Zaman, Johnson (2001). "Molecular modeling, structure--activity relationships and functional antagonism studies of 4-hydroxy-1-methyl-4-(4-methylphenyl)-3-piperidyl 4-methylphenyl ketones as a novel class of dopamine transporter inhibitors". Bioorganic & Medicinal Chemistry. 9 (7): 1753–1764. doi: 10.1016/S0968-0896(01)00090-6. PMID 11425577.
- ^ Findlay SP (1957). "Concerning 2-Carbomethoxytropinone". Journal of Organic Chemistry. 22 (11): 1385–1394. doi: 10.1021/jo01362a022.
- ^ U.S. patent 8,609,690
- ^ Nicolaou KC, Montagnon T, Baran PS, Zhong YL (2002). "Iodine(V) reagents in organic synthesis. Part 4. O-Iodoxybenzoic acid as a chemospecific tool for single electron transfer-based oxidation processes". Journal of the American Chemical Society. 124 (10): 2245–58. doi: 10.1021/ja012127+. PMID 11878978.
- ^ Bedewitz MA, Jones AD, D'Auria JC, Barry CS (2018). "Tropinone synthesis via an atypical polyketide synthase and P450-mediated cyclization". Nature Communications. 9 (1): 5281. Bibcode: 2018NatCo...9.5281B. doi: 10.1038/s41467-018-07671-3. ISSN 2041-1723. PMC 6290073. PMID 30538251.
- ^ A. Portsteffen, B. Draeger, A. Nahrstedt (1992). "Two tropinone reducing enzymes from Datura stramonium transformed root cultures". Phytochemistry. 31 (4): 1135. Bibcode: 1992PChem..31.1135P. doi: 10.1016/0031-9422(92)80247-C.
- ^ Boswell HD, Dräger B, McLauchlan WR, et al. (November 1999). "Specificities of the enzymes of N-alkyltropane biosynthesis in Brugmansia and Datura". Phytochemistry. 52 (5): 871–8. Bibcode: 1999PChem..52..871B. doi: 10.1016/S0031-9422(99)00293-9. PMID 10626376.
External links

| |

| |
| Names | |
|---|---|
|
IUPAC name
8-Methyl-8-azabicyclo[3.2.1]octan-3-one
| |
| Other names
3-Tropinone
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.007.756 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C8H13NO | |
| Molar mass | 139.195 g/mol |
| Appearance | Brown solid |
| Melting point | 42.5 °C (108.5 °F; 315.6 K) |
| Boiling point | (decomposes) |
| Hazards | |
| GHS labelling: | |

 [1]
[1]
| |
| Danger | |
| H302, H314 [1] | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Tropinone is an alkaloid, famously synthesised in 1917 by Robert Robinson as a synthetic precursor to atropine, a scarce commodity during World War I. [2] [3] Tropinone and the alkaloids cocaine and atropine all share the same tropane core structure. Its corresponding conjugate acid at pH 7.3 major species is known as tropiniumone. [4]
Synthesis
The first synthesis of tropinone was by Richard Willstätter in 1901. It started from the seemingly related cycloheptanone, but required many steps to introduce the nitrogen bridge; the overall yield for the synthesis path is only 0.75%. [5] Willstätter had previously synthesized cocaine from tropinone, in what was the first synthesis and elucidation of the structure of cocaine. [6]

Robinson's "double Mannich" reaction
The 1917 synthesis by Robinson is considered a classic in total synthesis [8] due to its simplicity and biomimetic approach. Tropinone is a bicyclic molecule, but the reactants used in its preparation are fairly simple: succinaldehyde, methylamine and acetonedicarboxylic acid (or even acetone). The synthesis is a good example of a biomimetic reaction or biogenetic-type synthesis because biosynthesis makes use of the same building blocks. It also demonstrates a tandem reaction in a one-pot synthesis. Furthermore, the yield of the synthesis was 17% and with subsequent improvements exceeded 90%. [5]
This reaction is described as an intramolecular "double Mannich reaction" for obvious reasons. It is not unique in this regard, as others have also attempted it in piperidine synthesis. [9] [10]
In place of acetone, acetonedicarboxylic acid is known as the " synthetic equivalent" the 1,3-dicarboxylic acid groups are so-called " activating groups" to facilitate the ring forming reactions. The calcium salt is there as a " buffer" as it is claimed that higher yields are possible if the reaction is conducted at " physiological pH".
Reaction mechanism
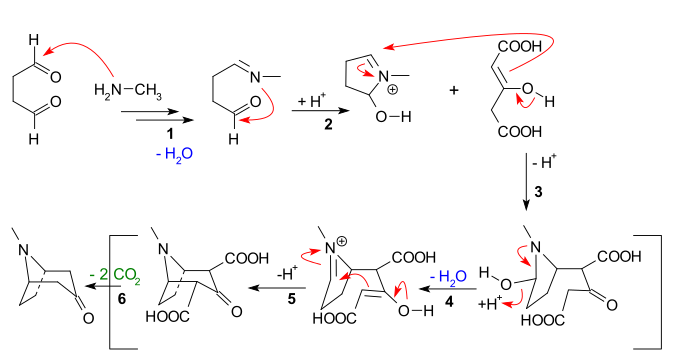
The main features apparent from the reaction sequence below are:
- Nucleophilic addition of methylamine to succinaldehyde, followed by loss of water to create an imine
- Intramolecular addition of the imine to the second aldehyde unit and first ring closure
- Intermolecular Mannich reaction of the enolate of acetone dicarboxylate
- New enolate formation and new imine formation with loss of water for
- Second intramolecular Mannich reaction and second ring closure
- Loss of 2 carboxylic groups to tropinone
Some authors have actually tried to retain one of the CO2H groups. [11]
CO2R-tropinone has 4 stereoisomers, although the corresponding ecgonidine alkyl ester has only a pair of enantiomers.
From cycloheptanone
IBX dehydrogenation (oxidation) of cycloheptanone (suberone) to 2,6-cycloheptadienone [1192-93-4] followed by reaction with an amine is versatile a way of forming tropinones. [12] [13] The mechanism evoked is clearly delineated to be a double Michael reaction (i.e. conjugate addition).
Biochemistry method
|
| This section is empty. You can help by
adding to it. (April 2022) |
Reduction of tropinone
The reduction of tropinone is mediated by NADPH-dependent reductase enzymes, which have been characterized in multiple plant species. [15] These plant species all contain two types of the reductase enzymes, tropinone reductase I and tropinone reductase II. TRI produces tropine and TRII produces pseudotropine. Due to differing kinetic and pH/activity characteristics of the enzymes and by the 25-fold higher activity of TRI over TRII, the majority of the tropinone reduction is from TRI to form tropine. [16]

See also
- Benztropine
- Daturaolone
- 2-Carbomethoxytropinone (2-CMT) an intermediate in the creation of ecgonine cocaine analogues
- Ecgonidine
References
- ^ a b "Tropinone". Substance Information. ECHA.
- ^ Robinson R (1917). "LXIII. A Synthesis of Tropinone". Journal of the Chemical Society, Transactions. 111: 762–768. doi: 10.1039/CT9171100762.
- ^ Nicolaou KC, Vourloumis D, Winssinger N, Baran PS (2000). "The Art and Science of Total Synthesis at the Dawn of the Twenty-First Century". Angewandte Chemie International Edition. 39 (1): 44–122. doi: 10.1002/(SICI)1521-3773(20000103)39:1<44::AID-ANIE44>3.0.CO;2-L. PMID 10649349.
- ^ Chemical Entities of Biological Interest Identification code: ChEBI:57851 "tropiniumone"
- ^ a b Smit WA, Smit WA, Bochkov AF, Caple R (1998). Organic Synthesis. doi: 10.1039/9781847551573. ISBN 978-0-85404-544-0.
- ^ Humphrey AJ, O'Hagan D (2001). "Tropane alkaloid biosynthesis. A century old problem unresolved". Natural Product Reports. 18 (5). Royal Society of Chemistry: 494–502. doi: 10.1039/b001713m. PMID 11699882.
- ^ Doble M, Kruthiventi AK (2007). Green Chemistry and Engineering. Oxford: Elsevier. p. 34. ISBN 978-0-12-372532-5.
- ^ Birch AJ (1993). "Investigating a Scientific Legend: The Tropinone Synthesis of Sir Robert Robinson, F.R.S". Notes and Records of the Royal Society of London. 47 (2): 277–296. doi: 10.1098/rsnr.1993.0034. JSTOR 531792. S2CID 143267467.
- ^ Wang S, Sakamuri S, Enyedy IJ, Kozikowski AP, Deschaux O, Bandyopadhyay BC, Tella SR, Zaman WA, Johnson KM (2000). "Discovery of a novel dopamine transporter inhibitor, 4-hydroxy-1-methyl-4-(4-methylphenyl)-3-piperidyl 4-methylphenyl ketone, as a potential cocaine antagonist through 3D-database pharmacophore searching. Molecular modeling, structure-activity relationships, and behavioral pharmacological studies". Journal of Medicinal Chemistry. 43 (3): 351–360. doi: 10.1021/jm990516x. PMID 10669562.
- ^ Wang S, Sakamuri, Enyedy, Kozikowski, Zaman, Johnson (2001). "Molecular modeling, structure--activity relationships and functional antagonism studies of 4-hydroxy-1-methyl-4-(4-methylphenyl)-3-piperidyl 4-methylphenyl ketones as a novel class of dopamine transporter inhibitors". Bioorganic & Medicinal Chemistry. 9 (7): 1753–1764. doi: 10.1016/S0968-0896(01)00090-6. PMID 11425577.
- ^ Findlay SP (1957). "Concerning 2-Carbomethoxytropinone". Journal of Organic Chemistry. 22 (11): 1385–1394. doi: 10.1021/jo01362a022.
- ^ U.S. patent 8,609,690
- ^ Nicolaou KC, Montagnon T, Baran PS, Zhong YL (2002). "Iodine(V) reagents in organic synthesis. Part 4. O-Iodoxybenzoic acid as a chemospecific tool for single electron transfer-based oxidation processes". Journal of the American Chemical Society. 124 (10): 2245–58. doi: 10.1021/ja012127+. PMID 11878978.
- ^ Bedewitz MA, Jones AD, D'Auria JC, Barry CS (2018). "Tropinone synthesis via an atypical polyketide synthase and P450-mediated cyclization". Nature Communications. 9 (1): 5281. Bibcode: 2018NatCo...9.5281B. doi: 10.1038/s41467-018-07671-3. ISSN 2041-1723. PMC 6290073. PMID 30538251.
- ^ A. Portsteffen, B. Draeger, A. Nahrstedt (1992). "Two tropinone reducing enzymes from Datura stramonium transformed root cultures". Phytochemistry. 31 (4): 1135. Bibcode: 1992PChem..31.1135P. doi: 10.1016/0031-9422(92)80247-C.
- ^ Boswell HD, Dräger B, McLauchlan WR, et al. (November 1999). "Specificities of the enzymes of N-alkyltropane biosynthesis in Brugmansia and Datura". Phytochemistry. 52 (5): 871–8. Bibcode: 1999PChem..52..871B. doi: 10.1016/S0031-9422(99)00293-9. PMID 10626376.