Triazenes are organic compounds that contain the functional group R1−N=N−NR2R3, where the R are each any of various types of substituent groups. [2] Some anti-cancer medications and dyes are triazenes. [3] Formally, the triazenes are related to the unstable chemical triazene, H2N−N=NH.
Production
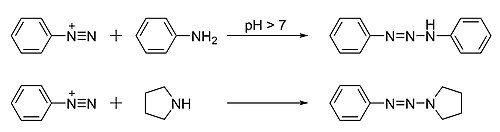
Triazenes are prepared from the N-coupling reaction between diazonium salts and primary or secondary amines. [4] The coupling reactions are typically mild, using a base such as sodium acetate, [5] sodium carbonate, [5] or sodium bicarbonate. [4]
The diazonium reagents are themselves available starting from amines. For symmetrical triazenes derived from primary amines, partial diazotization gives a mixture of the original amine and its diazo derivative that then couple with each other. For example, 1,3-diphenyltriazene (PhN=N−NHPh) can be made from aniline in a one-pot reaction. [5] [6] For asymmetrical triazenes, for example (phenyldiazenyl)pyrrolidine (PhN=N−NC4H8), the diazonium salt must be pre-made.
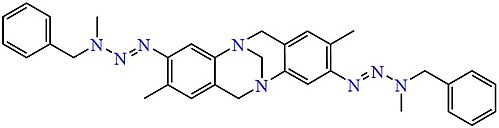
Analogues of Tröger's base containing a symmetric pair of asymmetric triazene side-chains have been obtained similarly. [7]
Reactions and applications
Triazenes derived from primary amines engage in tautomerism. In the case of symmetric triazenes, the tautomers are identical.
Triazenes can be converted to diazonium salts. [8]

Triazenes decompose in the presence of protonating or alkylating agents into quaternary amines and diazonium salts; as such triazenes have been used as an in situ source of diazonium that reacted with sodium sulfide to give the corresponding thiophenols. [5] A strategy for the protection and deprotection of sensitive secondary amines is based on this principle. [9]
Polymeric triazenes are applied as conductive and absorbent materials. [10] Triazenes have been used in the synthesis of cinnoline, functionalized lactams, and coumarins. [9] [8]
References
- ^ "Dacarbazine". The American Society of Health-System Pharmacists. Archived from the original on 11 September 2017. Retrieved 8 December 2016.
- ^ Suleymanov, Abdusalom A.; Severin, Kay (2021-03-22). "Vinyl and Alkynyl Triazenes: Synthesis, Reactivity, and Applications". Angewandte Chemie International Edition. 60 (13): 6879–6889. doi: 10.1002/anie.202011031. ISSN 1433-7851.
- ^ Berneth, Horst (2008). "Methine Dyes and Pigments". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a16_487.pub2. ISBN 978-3527306732.
- ^ a b Sengupta, Saumitra; Sadhukhan, Subir K. (2002). "Synthesis of Symmetrical trans-Stilbenes by a Double Heck Reaction of (Arylazo)amines with Vinyltriethoxysilane: trans-4,4′-Dibromostilbene". Organic Syntheses. 79: 52. doi: 10.15227/orgsyn.079.0052.
- ^ a b c d Kazem-Rostami, M.; Khazaei, A.; Moosavi-Zare, A. R.; Bayat, M.; Saednia, S. (2012). "Novel One-Pot Synthesis of Thiophenols from Related Triazenes under Mild Conditions". Synlett. 23 (13): 1893–1896. doi: 10.1055/s-0032-1316557. S2CID 196805424.
-
^ Hartman, W. W.; Dickey, J. B. (1934). "Diazoaminobenzene". Organic Syntheses. 14: 24.
doi:
10.15227/orgsyn.014.0024.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Kazemostami, Masoud (2017). "Facile preparation of Ʌ-shaped building blocks: Hünlich-base derivatization". Synlett. 28: 1641–1645. doi: 10.1055/s-0036-1588180. S2CID 99294625.
- ^ a b Kimball, D. B.; Haley, M. M. (2002). "Triazenes: A Versatile Tool in Organic Synthesis". Angewandte Chemie International Edition. 41 (18): 3338–51. doi: 10.1002/1521-3773(20020916)41:18<3338::AID-ANIE3338>3.0.CO;2-7. PMID 12298030.
- ^ a b Lazny, R.; Poplawski, J.; Köbberling, J.; Enders, D.; Bräse, S. (1999). "Triazenes: A Useful Protecting Strategy for Sensitive Secondary Amines". Synlett. 1999 (8): 1304–1306. doi: 10.1055/s-1999-2803.
- ^ Khazaei, A.; Zare, A.; Moosavi-Zare, A. R.; Sadeghpour, M.; Afkhami, A. (2013). "Synthesis, characterization, and application of a triazene-based polysulfone as a dye adsorbent". Journal of Applied Polymer Science. 129 (6): 3439–3446. doi: 10.1002/app.39069.

Triazenes are organic compounds that contain the functional group R1−N=N−NR2R3, where the R are each any of various types of substituent groups. [2] Some anti-cancer medications and dyes are triazenes. [3] Formally, the triazenes are related to the unstable chemical triazene, H2N−N=NH.
Production
Triazenes are prepared from the N-coupling reaction between diazonium salts and primary or secondary amines. [4] The coupling reactions are typically mild, using a base such as sodium acetate, [5] sodium carbonate, [5] or sodium bicarbonate. [4]
The diazonium reagents are themselves available starting from amines. For symmetrical triazenes derived from primary amines, partial diazotization gives a mixture of the original amine and its diazo derivative that then couple with each other. For example, 1,3-diphenyltriazene (PhN=N−NHPh) can be made from aniline in a one-pot reaction. [5] [6] For asymmetrical triazenes, for example (phenyldiazenyl)pyrrolidine (PhN=N−NC4H8), the diazonium salt must be pre-made.
Analogues of Tröger's base containing a symmetric pair of asymmetric triazene side-chains have been obtained similarly. [7]
Reactions and applications
Triazenes derived from primary amines engage in tautomerism. In the case of symmetric triazenes, the tautomers are identical.
Triazenes can be converted to diazonium salts. [8]

Triazenes decompose in the presence of protonating or alkylating agents into quaternary amines and diazonium salts; as such triazenes have been used as an in situ source of diazonium that reacted with sodium sulfide to give the corresponding thiophenols. [5] A strategy for the protection and deprotection of sensitive secondary amines is based on this principle. [9]
Polymeric triazenes are applied as conductive and absorbent materials. [10] Triazenes have been used in the synthesis of cinnoline, functionalized lactams, and coumarins. [9] [8]
References
- ^ "Dacarbazine". The American Society of Health-System Pharmacists. Archived from the original on 11 September 2017. Retrieved 8 December 2016.
- ^ Suleymanov, Abdusalom A.; Severin, Kay (2021-03-22). "Vinyl and Alkynyl Triazenes: Synthesis, Reactivity, and Applications". Angewandte Chemie International Edition. 60 (13): 6879–6889. doi: 10.1002/anie.202011031. ISSN 1433-7851.
- ^ Berneth, Horst (2008). "Methine Dyes and Pigments". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a16_487.pub2. ISBN 978-3527306732.
- ^ a b Sengupta, Saumitra; Sadhukhan, Subir K. (2002). "Synthesis of Symmetrical trans-Stilbenes by a Double Heck Reaction of (Arylazo)amines with Vinyltriethoxysilane: trans-4,4′-Dibromostilbene". Organic Syntheses. 79: 52. doi: 10.15227/orgsyn.079.0052.
- ^ a b c d Kazem-Rostami, M.; Khazaei, A.; Moosavi-Zare, A. R.; Bayat, M.; Saednia, S. (2012). "Novel One-Pot Synthesis of Thiophenols from Related Triazenes under Mild Conditions". Synlett. 23 (13): 1893–1896. doi: 10.1055/s-0032-1316557. S2CID 196805424.
-
^ Hartman, W. W.; Dickey, J. B. (1934). "Diazoaminobenzene". Organic Syntheses. 14: 24.
doi:
10.15227/orgsyn.014.0024.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Kazemostami, Masoud (2017). "Facile preparation of Ʌ-shaped building blocks: Hünlich-base derivatization". Synlett. 28: 1641–1645. doi: 10.1055/s-0036-1588180. S2CID 99294625.
- ^ a b Kimball, D. B.; Haley, M. M. (2002). "Triazenes: A Versatile Tool in Organic Synthesis". Angewandte Chemie International Edition. 41 (18): 3338–51. doi: 10.1002/1521-3773(20020916)41:18<3338::AID-ANIE3338>3.0.CO;2-7. PMID 12298030.
- ^ a b Lazny, R.; Poplawski, J.; Köbberling, J.; Enders, D.; Bräse, S. (1999). "Triazenes: A Useful Protecting Strategy for Sensitive Secondary Amines". Synlett. 1999 (8): 1304–1306. doi: 10.1055/s-1999-2803.
- ^ Khazaei, A.; Zare, A.; Moosavi-Zare, A. R.; Sadeghpour, M.; Afkhami, A. (2013). "Synthesis, characterization, and application of a triazene-based polysulfone as a dye adsorbent". Journal of Applied Polymer Science. 129 (6): 3439–3446. doi: 10.1002/app.39069.