| |

| |
| Names | |
|---|---|
| Other names
Thulium iodide
Thulium triiodide | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.052 |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| TmI3 | |
| Appearance | Yellow hexagonal crystal [1] |
| Melting point | 1015 °C [2] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Thulium(III) iodide is an iodide of thulium, with the chemical formula of TmI3. Thulium(III) iodide is used as a component of metal halide lamps. [3]
Preparation
Mercury(II) iodide can be heated with thulium to obtain thulium(III) iodide: [4]
- 2 Tm + 3 HgI2 → 2 TmI3 + 3 Hg
The mercury produced in the reaction can be removed by distillation. [5] [4]
The thulium(III) iodide hydrate crystallized from solution can be heated with ammonium iodide to obtain the anhydrous form. [6] [4]
Properties
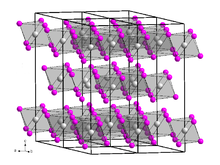
Thulium(III) iodide is a yellow, highly hygroscopic solid with a bismuth(III) iodide-type crystal structure. [4] It forms in the hexagonal crystal system. [6] Its hydrate can be heated in air to obtain TmOI. Its anhydrous form can be heated with thulium to obtain TmI2: [7]
- 2 TmI3 + Tm → 3 TmI2
References
- ^ 王世华,谷亚平,蒋盛邦.在Tm-HgI2体系中TmI2生成机理[J].物理化学学报,1987(03):334-336.
- ^ Jantsch, G.; Zeitschrift fuer Anorganische und Allgemeine Chemie 1933, V212, P65-83 CAPLUS
- ^ Flesch, Peter G. (2007). Light and Light Sources: High-Intensity Discharge Lamps. Springer. p. 45. ISBN 9783540326847.
- ^ a b c d Georg Brauer (Hrsg.), unter Mitarbeit von Marianne Baudler u. a.: Handbuch der Präparativen Anorganischen Chemie. 3., umgearbeitete Auflage. Band I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6, S. 1077.
- ^ Asprey, L. B.; Keenan, T. K.; Kruse, F. H. Preparation and crystal data for lanthanide and actinide triiodides. Inorg. Chem., 1964. 3 (8): 1137-1240
- ^ a b 无机化学丛书 第七卷 钪 稀土元素. 科学出版社. pp 211
- ^ L.B. Asprey, F.H. Kruse (1960). "Divalent thulium. Thulium di-iodide". Journal of Inorganic and Nuclear Chemistry. 13 (1–2): 32–35. doi: 10.1016/0022-1902(60)80232-1. Archived from the original on 2018-06-09. Retrieved 2018-04-13.(注:此文献指出Tm+HgI2的反应生成TmI2)

| |

| |
| Names | |
|---|---|
| Other names
Thulium iodide
Thulium triiodide | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.052 |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| TmI3 | |
| Appearance | Yellow hexagonal crystal [1] |
| Melting point | 1015 °C [2] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Thulium(III) iodide is an iodide of thulium, with the chemical formula of TmI3. Thulium(III) iodide is used as a component of metal halide lamps. [3]
Preparation
Mercury(II) iodide can be heated with thulium to obtain thulium(III) iodide: [4]
- 2 Tm + 3 HgI2 → 2 TmI3 + 3 Hg
The mercury produced in the reaction can be removed by distillation. [5] [4]
The thulium(III) iodide hydrate crystallized from solution can be heated with ammonium iodide to obtain the anhydrous form. [6] [4]
Properties
Thulium(III) iodide is a yellow, highly hygroscopic solid with a bismuth(III) iodide-type crystal structure. [4] It forms in the hexagonal crystal system. [6] Its hydrate can be heated in air to obtain TmOI. Its anhydrous form can be heated with thulium to obtain TmI2: [7]
- 2 TmI3 + Tm → 3 TmI2
References
- ^ 王世华,谷亚平,蒋盛邦.在Tm-HgI2体系中TmI2生成机理[J].物理化学学报,1987(03):334-336.
- ^ Jantsch, G.; Zeitschrift fuer Anorganische und Allgemeine Chemie 1933, V212, P65-83 CAPLUS
- ^ Flesch, Peter G. (2007). Light and Light Sources: High-Intensity Discharge Lamps. Springer. p. 45. ISBN 9783540326847.
- ^ a b c d Georg Brauer (Hrsg.), unter Mitarbeit von Marianne Baudler u. a.: Handbuch der Präparativen Anorganischen Chemie. 3., umgearbeitete Auflage. Band I, Ferdinand Enke, Stuttgart 1975, ISBN 3-432-02328-6, S. 1077.
- ^ Asprey, L. B.; Keenan, T. K.; Kruse, F. H. Preparation and crystal data for lanthanide and actinide triiodides. Inorg. Chem., 1964. 3 (8): 1137-1240
- ^ a b 无机化学丛书 第七卷 钪 稀土元素. 科学出版社. pp 211
- ^ L.B. Asprey, F.H. Kruse (1960). "Divalent thulium. Thulium di-iodide". Journal of Inorganic and Nuclear Chemistry. 13 (1–2): 32–35. doi: 10.1016/0022-1902(60)80232-1. Archived from the original on 2018-06-09. Retrieved 2018-04-13.(注:此文献指出Tm+HgI2的反应生成TmI2)