| |
| Names | |
|---|---|
|
IUPAC name
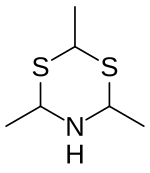
2,4,6-Trimethyl-1,3,5-dithiazinane
| |
| Other names
Dihydro-2,4,6-trimethyl-4H-1,3,5-dithiazine
| |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.010.295 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H13NS2 | |
| Molar mass | 163.30 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Thialdine is a basic heterocyclic chemical compound with the molecular formula C6H13NS2.
Thialdine was first synthesized by Justus von Liebig and Friedrich Wöhler in 1847 by passing hydrogen sulfide through a solution of acetaldehyde ammonia trimer, with thialdine crystallizing from the solution. [1]
Acetaldehyde ammonia trimer is the cyclic trimer formed in the condensation reaction of acetaldehyde with ammonia:
- 3 CH3CHO + 3 NH3 → (CH3CHNH)3 + 3 H2O
The aldehyde ammonia does not need to be isolated as an intermediate.
The configuration of the three methyl groups in thialdine can differ, so that multiple cis-trans isomers are possible. However, X-ray diffraction has shown that in practice thialdine occurs in the all-cis configuration. [2]
Thialdine is used as a flavoring agent in foods. It is included in the list of flavoring substances authorized in the European Union [3] and is considered generally recognized as safe (GRAS) in the United States. [4] The FEMA number of thialdine is 4018. Thialdine has a roasted meat smell and is therefore used as a flavoring in meat. [5]
Salts of thialdine have been used as an antioxidant additive in lubricating oils. [6]
Thialdine and similar compounds have been proposed as rubber vulcanization accelerators. [7]
- ^ Wöhler, Liebig (1847). "Ueber das Thialdin und Selenaldin, zwei künstlich darstellbare organische Basen". Annalen der Chemie und Pharmacie. 61: 1. doi: 10.1002/jlac.18470610102.
- ^ Day, Cynthia S.; Hansen, Thomsen J.; Keefer, Larry K. (1982). "Stereochemistry of thialdine". Journal of Heterocyclic Chemistry. 19 (6): 1301–1304. doi: 10.1002/jhet.5570190608.
- ^ Lijst van aromastoffen als bedoeld in Verordening 2232/96, tot opname van die lijst in bijlage I bij Verordening 1334/2008. (number 15.109)
-
^ R.L. Smith, J. Doull, V.J. Feron, J.I. Goodman, I.C. Munro, P.M. Newberne, P.S. Portoghese, W.J. Waddell, B.M. Wagner, T.B. Adams, and M.M. McGowen (December 2001).
"GRAS Flavoring Substances 20" (PDF). Food Technology. 55 (12).
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ The Good Scents Company: Thialdine
- ^ Hydrocarbon oil additive, U.S. Patent 2712526, July 5, 1955, Exxon Research Engineering Co.
- ^ Rubber vulcanization accelerator, U.S. Patent 1664481, April 3, 1928, Rubber Service Lab Co.

| |
| Names | |
|---|---|
|
IUPAC name
2,4,6-Trimethyl-1,3,5-dithiazinane
| |
| Other names
Dihydro-2,4,6-trimethyl-4H-1,3,5-dithiazine
| |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.010.295 |
| EC Number |
|
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C6H13NS2 | |
| Molar mass | 163.30 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Thialdine is a basic heterocyclic chemical compound with the molecular formula C6H13NS2.
Thialdine was first synthesized by Justus von Liebig and Friedrich Wöhler in 1847 by passing hydrogen sulfide through a solution of acetaldehyde ammonia trimer, with thialdine crystallizing from the solution. [1]
Acetaldehyde ammonia trimer is the cyclic trimer formed in the condensation reaction of acetaldehyde with ammonia:
- 3 CH3CHO + 3 NH3 → (CH3CHNH)3 + 3 H2O
The aldehyde ammonia does not need to be isolated as an intermediate.
The configuration of the three methyl groups in thialdine can differ, so that multiple cis-trans isomers are possible. However, X-ray diffraction has shown that in practice thialdine occurs in the all-cis configuration. [2]
Thialdine is used as a flavoring agent in foods. It is included in the list of flavoring substances authorized in the European Union [3] and is considered generally recognized as safe (GRAS) in the United States. [4] The FEMA number of thialdine is 4018. Thialdine has a roasted meat smell and is therefore used as a flavoring in meat. [5]
Salts of thialdine have been used as an antioxidant additive in lubricating oils. [6]
Thialdine and similar compounds have been proposed as rubber vulcanization accelerators. [7]
- ^ Wöhler, Liebig (1847). "Ueber das Thialdin und Selenaldin, zwei künstlich darstellbare organische Basen". Annalen der Chemie und Pharmacie. 61: 1. doi: 10.1002/jlac.18470610102.
- ^ Day, Cynthia S.; Hansen, Thomsen J.; Keefer, Larry K. (1982). "Stereochemistry of thialdine". Journal of Heterocyclic Chemistry. 19 (6): 1301–1304. doi: 10.1002/jhet.5570190608.
- ^ Lijst van aromastoffen als bedoeld in Verordening 2232/96, tot opname van die lijst in bijlage I bij Verordening 1334/2008. (number 15.109)
-
^ R.L. Smith, J. Doull, V.J. Feron, J.I. Goodman, I.C. Munro, P.M. Newberne, P.S. Portoghese, W.J. Waddell, B.M. Wagner, T.B. Adams, and M.M. McGowen (December 2001).
"GRAS Flavoring Substances 20" (PDF). Food Technology. 55 (12).
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ The Good Scents Company: Thialdine
- ^ Hydrocarbon oil additive, U.S. Patent 2712526, July 5, 1955, Exxon Research Engineering Co.
- ^ Rubber vulcanization accelerator, U.S. Patent 1664481, April 3, 1928, Rubber Service Lab Co.

