| |
| Names | |
|---|---|
|
Preferred IUPAC name
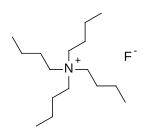
N,N,N-Tributylbutan-1-aminium fluoride | |
| Other names
Tetrabutylammonium fluoride; TBAF; n-Bu4NF
| |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.006.417 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| (C4H9)4NF | |
| Molar mass | 261.46 g/mol |
| Melting point | 58 to 60 °C (136 to 140 °F; 331 to 333 K) (trihydrate) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Tetra-n-butylammonium fluoride, commonly abbreviated to TBAF and n-Bu4NF, is a quaternary ammonium salt with the chemical formula (CH3CH2CH2CH2)4N+F−. It is commercially available as the white solid trihydrate and as a solution in tetrahydrofuran. TBAF is used as a source of fluoride ion in organic solvents. [1]
Preparation and properties
TBAF can be prepared by passing hydrofluoric acid through an ion-exchange resin, followed by tetrabutylammonium bromide. Upon evaporation of the water, TBAF can be collected as an oil in quantitative yield. [1]
Preparing anhydrous samples is of interest as the basicity of fluoride increases by more than 20 pK units on passing from aqueous to aprotic solvent.[ citation needed] However, heating samples of the hydrated material to 77 °C under vacuum causes decomposition to the hydrogen difluoride salt. [2] Similarly, samples dried at 40 °C under high vacuum still contain 10-30 mol% of water and some 10% of difluoride. [3] Instead, anhydrous TBAF has been prepared by the reaction of hexafluorobenzene and tetrabutylammonium cyanide. Solutions of the salt in acetonitrile and dimethyl sulfoxide are stable. [4]
Reactions and uses
Because the fluoride ion is such a strong hydrogen bond acceptor, its salts tend to be hydrated and of limited solubility in organic solvents. As a fluoride ion source, TBAF solves this problem, although the nature of the fluoride is uncertain because TBAF samples are almost always hydrated, resulting in the formation of bifluoride (HF2−) hydroxide (OH−) as well as fluoride. Many applications tolerate heterogeneous or ill-defined fluoride sources.
As a fluoride source in organic solvents, TBAF is used to remove silyl ether protecting groups. It is also used as a phase transfer catalyst and as a mild base. As a deprotecting agent, TBAF in DMSO will convert O-silylated enolates into carbonyls. With C-Si bonds, TBAF gives carbanions that can be trapped with electrophiles or undergo protonolysis. [1] [5]
References
- ^ a b c Li, Hui-Yin; Sun, Haoran; DiMagno, Stephen G. (2007). "Tetrabutylammonium Fluoride". In Paquette, Leo A. (ed.). Encyclopedia of Reagents for Organic Synthesis. John Wiley & Sons. doi: 10.1002/9780470842898.rt015.pub2. ISBN 978-0471936237.
- ^ Ramesh K. Sharma; James L. Fry (1983). "Instability of anhydrous tetra-n-alkylammonium fluorides". Journal of Organic Chemistry. 48 (12): 2112–4. doi: 10.1021/jo00160a041.
- ^ D. Phillip Cox; Jacek Terpinski; Witold Lawrynowicz (1984). "'Anhydrous' tetrabutylammonium fluoride: a mild but highly efficient source of nucleophilic fluoride ion". Journal of Organic Chemistry. 49 (17): 3216–9. doi: 10.1021/jo00191a035.
- ^ Haoran Sun & Stephen G. DiMagno (2005). "Anhydrous Tetrabutylammonium Fluoride". Journal of the American Chemical Society. 127 (7): 2050–1. doi: 10.1021/ja0440497. PMID 15713075.
- ^ Nina Gommermann and Paul Knochel "N,N-Dibenzyl-N-[1-cyclohexyl-3-(trimethylsilyl)-2-propynyl]-amine from Cyclohexanecarbaldehyde, Trimethylsilylacetylene and Dibenzylamine" Org. Synth. 2007, 84, 1. doi: 10.15227/orgsyn.084.0001
Further reading
- K. Hiroya; R. Jouka; M. Kameda; A. Yasuhara & T. Sakamoto (2001). "Cyclization reactions of 2-alkynylbenzyl alcohol and 2-alkynylbenzylamine derivatives promoted by tetrabutylammonium fluoride". Tetrahedron. 57 (48): 9697–710. doi: 10.1016/S0040-4020(01)00991-7..

| |
| Names | |
|---|---|
|
Preferred IUPAC name
N,N,N-Tributylbutan-1-aminium fluoride | |
| Other names
Tetrabutylammonium fluoride; TBAF; n-Bu4NF
| |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.006.417 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| (C4H9)4NF | |
| Molar mass | 261.46 g/mol |
| Melting point | 58 to 60 °C (136 to 140 °F; 331 to 333 K) (trihydrate) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Tetra-n-butylammonium fluoride, commonly abbreviated to TBAF and n-Bu4NF, is a quaternary ammonium salt with the chemical formula (CH3CH2CH2CH2)4N+F−. It is commercially available as the white solid trihydrate and as a solution in tetrahydrofuran. TBAF is used as a source of fluoride ion in organic solvents. [1]
Preparation and properties
TBAF can be prepared by passing hydrofluoric acid through an ion-exchange resin, followed by tetrabutylammonium bromide. Upon evaporation of the water, TBAF can be collected as an oil in quantitative yield. [1]
Preparing anhydrous samples is of interest as the basicity of fluoride increases by more than 20 pK units on passing from aqueous to aprotic solvent.[ citation needed] However, heating samples of the hydrated material to 77 °C under vacuum causes decomposition to the hydrogen difluoride salt. [2] Similarly, samples dried at 40 °C under high vacuum still contain 10-30 mol% of water and some 10% of difluoride. [3] Instead, anhydrous TBAF has been prepared by the reaction of hexafluorobenzene and tetrabutylammonium cyanide. Solutions of the salt in acetonitrile and dimethyl sulfoxide are stable. [4]
Reactions and uses
Because the fluoride ion is such a strong hydrogen bond acceptor, its salts tend to be hydrated and of limited solubility in organic solvents. As a fluoride ion source, TBAF solves this problem, although the nature of the fluoride is uncertain because TBAF samples are almost always hydrated, resulting in the formation of bifluoride (HF2−) hydroxide (OH−) as well as fluoride. Many applications tolerate heterogeneous or ill-defined fluoride sources.
As a fluoride source in organic solvents, TBAF is used to remove silyl ether protecting groups. It is also used as a phase transfer catalyst and as a mild base. As a deprotecting agent, TBAF in DMSO will convert O-silylated enolates into carbonyls. With C-Si bonds, TBAF gives carbanions that can be trapped with electrophiles or undergo protonolysis. [1] [5]
References
- ^ a b c Li, Hui-Yin; Sun, Haoran; DiMagno, Stephen G. (2007). "Tetrabutylammonium Fluoride". In Paquette, Leo A. (ed.). Encyclopedia of Reagents for Organic Synthesis. John Wiley & Sons. doi: 10.1002/9780470842898.rt015.pub2. ISBN 978-0471936237.
- ^ Ramesh K. Sharma; James L. Fry (1983). "Instability of anhydrous tetra-n-alkylammonium fluorides". Journal of Organic Chemistry. 48 (12): 2112–4. doi: 10.1021/jo00160a041.
- ^ D. Phillip Cox; Jacek Terpinski; Witold Lawrynowicz (1984). "'Anhydrous' tetrabutylammonium fluoride: a mild but highly efficient source of nucleophilic fluoride ion". Journal of Organic Chemistry. 49 (17): 3216–9. doi: 10.1021/jo00191a035.
- ^ Haoran Sun & Stephen G. DiMagno (2005). "Anhydrous Tetrabutylammonium Fluoride". Journal of the American Chemical Society. 127 (7): 2050–1. doi: 10.1021/ja0440497. PMID 15713075.
- ^ Nina Gommermann and Paul Knochel "N,N-Dibenzyl-N-[1-cyclohexyl-3-(trimethylsilyl)-2-propynyl]-amine from Cyclohexanecarbaldehyde, Trimethylsilylacetylene and Dibenzylamine" Org. Synth. 2007, 84, 1. doi: 10.15227/orgsyn.084.0001
Further reading
- K. Hiroya; R. Jouka; M. Kameda; A. Yasuhara & T. Sakamoto (2001). "Cyclization reactions of 2-alkynylbenzyl alcohol and 2-alkynylbenzylamine derivatives promoted by tetrabutylammonium fluoride". Tetrahedron. 57 (48): 9697–710. doi: 10.1016/S0040-4020(01)00991-7..