|
| This It is of interest to the following WikiProjects: | ||||||||||
| |||||||||||
|
| This article contains a translation of Farmacocinética from es.wikipedia. |
Extended content
|
|---|
|
This is an important topic that deserces an article of its own. However, copying the abstradt of a review article verbatim in Wikipedia is against the rules (copyright violation) and not useful, because these docoments are too long and too technical. The editor is welcome to write a short encyclopedic excerpt, taking into account other sources like WHO, NIH etc. Andreas 17:13, 9 January 2006 (UTC) I have been given the assignment of giving a brief lecture at my church on this very topic. I will web-research the topic and write my findings to this article. As a retired, but still licensed registered nurse, I have limited access to hospital medical libraries. I will also make my best effort at including references and links. User:W8IMP 0837, 15 January 2007]] (UTC) Why is this article titled "Pharmacokinetics (section), when there is another article, not a "talk" section devoted to this topic. I would gladly re-title the article as "Drug-drug and drug-herbal interactions", as soon as I have more data, and someone tells me to change the title. User:W8IMP 08:51, 15 January 2007]] (UTC) |
The constant which relates the exponent of decay (k) and half-life is easily determined by first principles from the equation for exponential decay. Mentioning circuit analysis seems totally crazy to me; no pharmacokineticist would know anything about circuits or ever need to. If this is important then I think that the maths only should be mentioned. ben 06:50, 23 May 2006 (UTC)
- You forget that biomedical engineers (which usually have some understanding of circuits) are involved in the design of medical devices, such as insulin pumps (which have ICs). Pharmacokinetics is not such a narrow field. The research that lead to transdermal patches was done by a chemical engineer -- see Robert S. Langer. [1] Nephron T| C 20:24, 14 July 2006 (UTC)
Shouldn't there be something here about two (or more) compartment models, also non compartmental models? Let me hasten to add, I'm not the person to write it ... Daen 15:22, 25 September 2006 (UTC)
- You are right. Just some remarks:
The upper limit in practical use are three compartments, because the more compartments you add to your model, the more parameters you have to estimate from the data (two parameters for each compartment). Generally we have only samples from a limited number of compartments (plasma and urine – if we are lucky). Though it’s possible to get solutions from our curve fitting (i.e., nonlinear regression), the parameters are not identifiable for higher-order models any more. Not identifiable means that it is not possible to tell whether a given rate constant is attributable to a transfer process between two compartments, or elimination from the central compartment, e.t.c.
Non compartmental models don’t exist (that’s a contradiction). The term used for example in bioequivalence testing is Non Compartmental Analysis (NCA). Parameters (or metrics) of NCA are e.g., the area under the curve AUC (calculated by some kind of numerical integration algorithm, mostly the trapezoidal rule), the peak concentration Cmax and the time point of its occurence tmax,… All these values are directly obtained from the data assuming no underlying PK model. -- Alfie66 ( talk) 00:41, 10 June 2008 (UTC)
Actually, there are PK model assumptions made for the typical implementation of numerical integration, A.K.A. noncompartmental methods. Those assumptions become important when they are incorrect, which may actually be almost all of the time. Those assumptions commonly, but not always, include (1) that between samples of a monotonically decreasing blood concentration (i.e., a washout model of plasma or whole blood depending on the drug's hydrophilicity and lipophilicity), that the path of concentration is exponential. (2) That one can extrapolate exponentials to earlier times than the first sample time without error. (3) That the last and one or two prior samples can be used to extrapolate the AUC to infinity implying (3a) that the half-life is terminal at that time and (3b) that half-lives only arise from exponential functions. Granted, there are other ways of doing numerical integration, and the selection of extrapolating functions is not trivial. Let us review these assumptions. Ad (1) and (2), there is no evidence to that effect, and early concentration of a renal filtered non metabolized drug has been shown to be more a linear concentration and logarithm of time function than a logarithm of concentration linear time function [1]. Indeed, the former implies an initial zero volume of drug and the later implies an unrealistic instant mixing in an appreciable volume (see below). The dearth of testing on this subject appalls me. Ad (3a), the half-life for the last samples is typically not terminal. Indeed, the terminal function may occur very late in time and may be a power function [2], what the terminal function does not appear to be is an exponential [3], which reference also offers an explanation of drug volumes in time. I welcome comments on this, and have not altered the main text due to a conflict of interest on my part. I need other minds to review this. CarlWesolowski ( talk) 16:08, 23 August 2016 (UTC).
BTW, there are multiple circumstances in which compartmental models are not "the best" at anything. Rather than give a whole bunch of references here regarding the need to correct one compartmental clearances by 15% or so, the superiority of other methods like constant infusion and saturated systems for measuring clearance, adaptive gamma variate fitting for bolus experiment clearances, Michaelis-Menten for saturable receptor clearances and so forth, I just changed the text to be more honest, i.e., from best to most frequently used. Also, there are broken links in the text some of which I have fixed, HELP!!! This isn't my article, so would someone else take care of it, too, please? And, if you don't agree with my (minor) changes, do let's talk, please. CarlWesolowski ( talk) 20:20, 9 February 2017 (UTC)
It may be more useful to refer to Enzyme kinetics. At the very least, there should be a reference to it somewhere in the article. 136.165.191.251 ( talk) 16:07, 27 April 2009 (UTC)
References
Variable volume pharmacokinetic models is now linked to this article. Linking back is a consideration, so is thinking about it. Talk to me. CarlWesolowski ( talk) 14:20, 23 August 2016 (UTC) CarlWesolowski ( talk) 03:44, 9 September 2016 (UTC) No one talked to me. The linking article was an orphan so that linking to it was needed. And for completeness, it should be listed anyway. CarlWesolowski ( talk) 21:53, 17 November 2016 (UTC)
- Variable volume pharmacokinetic models was deleted because of "contribution rules." Variable volume models were correctly stated as an alternative to compartmental modelling by S. Niazi "Volume of Distribution as a Function of Time" Journal of Pharmaceutical Sciences 1976 Vol. 65 Issue 3 Pages 452-454 https://doi.org/10.1002/jps.2600650339. This was later generalized to all PK models by C. A. Wesolowski (full disclosure, me) et al. "Time Varying Apparent Volume of Distribution and Drug Half-Lives Following Intravenous Bolus Injections" Plos One 2016, https://doi.org/10.1371/journal.pone.0158798, and applied in subsequent papers. Content contribution rules being somewhat arcane here, someone else would have to link these to the current article instead of the now dead link, or alternatively provide text once again to the now defunct "Variable volume pharmacokinetic models" link. With time, this may become increasingly important as alternative models are shown to work better than the same old stuff. CarlWesolowski ( talk) 01:01, 17 November 2022 (UTC)
Who "invented" the "Pharmacokinetics"? —Preceding unsigned comment added by 217.86.119.8 ( talk) 21:22, 3 October 2006 (UTC)
- Nobody – or like Newton 'invented' 'Gravity'… ;-)
The term ( de:Pharmakokinetik) was first used in the 1950ies by the German Professor for Pediatrics Friedrich Hartmut Dost (*1910 †1985). [1] I would translate the reference with “Blood level: Kinetics of the Course of Concentrations in the Systemic Circulation”.
“Dost’s law of corresponding areas” (sometimes called Superposition Principle) describes the fact that in a linear pharmacokinetic system the area under the (plasma drug concentration-time) curve after a single dose (t=0-∞) equals to the AUC within a dosage interval (t=0-τ) in steady-state. -- Alfie ( talk) 00:15, 10 June 2008 (UTC)
References
- ^ Dost, Friedrich Hartmut (1953). Der Blutspiegel: Kinetik der Konzentrationsabläufe in der Kreislaufflüssigkeit (in German). Leipzig: Thieme-Verlag. p. 244.
Would it be useful to have a list of the standard PK parameters, with some brief explanation? Examples: Cmax, Tmax, AUC, etc. —Preceding unsigned comment added by 216.158.41.81 ( talk) 18:35, 27 June 2007 (UTC)
It would be helpful to me! I would definitely read that section carefully. Mlbish ( talk) 21:32, 21 February 2008 (UTC)
- Agree. Unfortunatelly I’m a little short in time… In the meantime have a look at a document [1] published by the German ‘Association for Applied Human Pharmacology’ (AGAH). -- Alfie ( talk) 00:50, 2 July 2008 (UTC)
- I have made an initial effort at adding a Pharmacokinetics#Parameters section. Thanks for the reference which I have added to the article. Cheers. Boghog ( talk) 16:15, 6 June 2010 (UTC)
References
- ^ "Collection of terms, symbols, equations, and explanations of common pharmacokinetic and pharmacodynamic parameters and some statistical functions" (PDF). AGAH working group PHARMACOKINETICS. 16 February 2004. Retrieved 2009-09-18.
Drug properties that influence its pharmacokinetics
Several drug properties can often influence the pharmacokinetics of many drugs.
Organ partitioning
Partition coefficient
The partition or distribution coefficient (KD) is the ratio of concentrations of a compound in the two phases of a mixture of two immiscible solvents at equilibrium. [1] It can influence the ADME properties (Absorption, Distribution, Metabolism, and Excretion) of the drug. When orally administered drugs are absorbed they must first pass through lipid bilayers in the intestinal epithelium (a process known as transcellular transport). For efficient transport the drug must be hydrophobic enough to partition into the lipid bilayer but not so hydrophobic that once it is in the bilayer it will not partition out again. [2] The partition coefficient is dependent on the hydrophobic or hydrophilic properties of the drug.
References
-
^ Leo A, Hansch C, and Elkins D (1971). "Partition coefficients and their uses". Chem Rev. 71 (6): 525–616.
doi:
10.1021/cr60274a001.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Kubinyi H (1979). "Nonlinear dependence of biological activity on hydrophobic character: the bilinear model". Farmaco [Sci]. 34 (3): 248–76. PMID 43264.
Drug transporters
Drug transporters are transmembrane proteins on the surface of cells and are responsible for facilitating or hindering the intracellular and paracellular transport of nutrients and other substances. Some known drug transporters include Breast Cancer Resistance Protein (BCRP, also known as ABCG2) and human intestinal peptide transporter (hPepT1). BCRP is a type of ATP-binding cassette transporter that can decrease the efficacy of chemotherapeutic agents in breast cancer by exporting agents out of the tumor cells, thus making them resistant to chemotherapy. Valacyclovir uses the hPepT1 transporter to increase the intestinal absorption of valacyclovir compared to acyclovir. Drug transporters can increase or decrease the absorption of drugs into the body as well as limit or facilitate the exposure of certain organs.
Perfusion
Perfusion or flow of blood to different organs, affects rate of presentation of drugs to different parts of the body and often affects the pharmacokinetics of many drugs. While the partition coefficient can affect the distribution of drugs from the blood stream to the organs, perfusion affects how fast a drug is presented to the organs. Different organs receive a vast supply of drugs while others receive minor amounts. For example, the kidneys receive vast quantities of blood, especially considering the relatively small size of the kidneys. Adipose tissue, on the other hand, receives a minor supply blood. Organs with a rich blood supply would be presented drug at a higher rate than organs with a lower blood supply.
Biophys ( talk) 01:56, 5 September 2008 (UTC)
Article Reassessment for WikiProject Pharmacology
Hello. I am a member of WikiProject Pharmacology, a Wikipedia wide project that maintains and improves articles that fall under the scope of pharmacology. Since your article has fallen under our scope, I have placed the correct template(s) on this talk page for verification. Upon review of the article, I'd like to make a few points, as shown:
- Ressessed article with class and importance factors
- It was not a B-class article, yes, so it has been lowered to C. I also put this article for mid importance due to the fact that it is specifically taught in classes at multiple universities and required by state pharmacy boards, such as the South Dakota Board of Pharmacy.
I'm glad this article could fall within our scope, and I hope to see it grow large! Many thanks! Renaissancee (talk) 02:21, 3 June 2009 (UTC)
- I have changed pharmacology project importance rating from mid to top; along with pharmacodynamics, pharmacokinetics is a core pharmacology topic! Boghog ( talk) 16:24, 6 June 2010 (UTC)
This article is quite poor to be in the English version of Wikipedia; I compared it with other versions like the spanish one, and I noticed there is an important lack of information here. An improvement of this article should be done. —Preceding unsigned comment added by 83.44.20.68 ( talk • contribs) 01:20, 20 September 2009 (UTC)
- Please add new sections to talk pages at their bottom (I moved your contribution). Well, from a quick look at the Spanish version, I guess you are right. You are are cordially invited to start working on an adaption. — Alfie ±Talk 13:08, 20 September 2009 (UTC)
Request for review of Plateau Principle entry
I have added a review of the development of the Plateau Principle in pharmacokinetics and the wide applicability of the mathematical model to other areas in biological sciences. It would be very helpful to obtain an expert review of this new entry. Please provide comments in the talk section of Plateau Principleand delete this section when a review has been done. Many thanks. Jhargrov ( talk) 14:03, 10 October 2009 (UTC)James Hargrove Jhargrov ( talk) 14:03, 10 October 2009 (UTC)
- Hi James! Wow, you've done a lot of work! I will have a deeper look, time allowing (there are some minor flaws in the TEX-markup). To be honest, this term is rarely (whenever) used in pharmacokinetics - steady state is used instead. I cann't find the term in standard textbooks (like Dost 1953, Gibaldi/Perrier 1982, Wagner 1993, Gabrielsson/Weiner 2006) explaining the Superposition Principle. In German the term Plateauphase was used years ago, but was abandoned due to the sexual connotation (see Plateau phase); in German the English term 'steady state' is solely in use as a loanword for some time now. — Alfie ±Talk 11:22, 12 October 2009 (UTC)
It's been a while since I've dealt with topics in pharmacokinetics. Is "liberation" a new and recognized stage of drug kinetics? If so, please provide citation. -- Bobthefish2 ( talk) 04:53, 7 August 2011 (UTC)
- A search of LADME in
PubMed yields only
3 hits while ADME yields
774 hits. Hence LADME does not as yet appear to be a widely used term. Hence the previous discussion about LADME in this article appears to give
undue weight to this term. I have therefore modified this section to first define what ADME is, and then mention LADME supported by a reliable source.
Boghog (
talk) 22:30, 7 August 2011 (UTC)
- A caveat about your pubmed analysis is that you should've limited the date to maybe Jan 2009 - Aug 2011. ADME has been around for a VERY long time. Suppose LADME has gained prevalence recently (which it hasn't, at the moment), its hits will only prevail over that of ADME for the time period that we'd consider as recent (which is approximately 2-3 years for biological literature). --
Bobthefish2 (
talk) 02:20, 8 August 2011 (UTC)
- Limiting the search to the last two years decreases the number of ADME hits from 774 to 227 vs 3 for LADME. The conclusion remains the same.
Boghog (
talk) 02:27, 8 August 2011 (UTC)
- That's good. By the way, the pharmacodynamics article is in bad-shape. Feel free to clean that up too. I am thinking of making a few minor changes, but I am not even sure where to start. -- Bobthefish2 ( talk) 02:50, 8 August 2011 (UTC)
- Limiting the search to the last two years decreases the number of ADME hits from 774 to 227 vs 3 for LADME. The conclusion remains the same.
Boghog (
talk) 02:27, 8 August 2011 (UTC)
- A caveat about your pubmed analysis is that you should've limited the date to maybe Jan 2009 - Aug 2011. ADME has been around for a VERY long time. Suppose LADME has gained prevalence recently (which it hasn't, at the moment), its hits will only prevail over that of ADME for the time period that we'd consider as recent (which is approximately 2-3 years for biological literature). --
Bobthefish2 (
talk) 02:20, 8 August 2011 (UTC)
- Hi to both of you! Well the term is not that new… See
the cover of Heinrich P. Koch; Wolfgang A. Ritschel (1986).
Synopsis der Biopharmazie und Pharmakokinetik (in German). Ecomed.
ISBN
978-3-609-64970-2.
These guys are not amateurs. ;-) Go to PubMed and paste the following into the search field:- (Koch HP[Author]) OR (Ritschel WA[Author]) OR (Wolfgang Ritschel[Full Author Name])
- See also the article on PK in the Spanish WP (which is the only one of all language versions rated as good).
Alfie
↑↓
© 11:26, 17 August 2011 (UTC)
- Thanks for the citation. I have
added the citation to the text and deleted the word "recent". Were Koch and/or Ritschel (or perhaps
Leopold) the first ones to coin the phrase "LADME"? Liberation is of course a prerequisite for adsorption. Nevertheless the ADME acronym is far more widely used than LADME. Hence I think it is appropriate that ADME first be defined followed by LADME. I agree that there is a lot of good material in the Spanish article could be useful in expanding the English version.
Boghog (
talk) 17:52, 17 August 2011 (UTC)
- Agreed. Again, I am a few years outdated with regards to knowledge about pharmacokinetics, but we should be careful about giving LADME too much emphasis. Even though are literature that endorse this model, we can probably find a great distribution of good and bad non-mainstream ideas if we look hard enough anyway. I think the current version of that section would suffice until LADME goes mainstream (i.e. get into prominent textbooks and used in big research articles). --
Bobthefish2 (
talk) 21:08, 17 August 2011 (UTC)
- Hi to both of you! The first reference giving "LADME" I have is in a chapter in Wolfgang A. Ritschel (1980). "Liberation, Absorption, Distribution, Metabolism, Elimination".
Handbook of Basic Pharmacokinetics. Hamilton, Ill.: Drug Intelligence Publications. p. 16.
ISBN
978-0-914768-34-0. Another old reference: Ernst Glaser (1985). "Pharmakokinetik".
Pharmakokinetik: Grundlagen, lineare Modelle, Rechenverfahren, Auswertemethoden (in German). PMI Verlag. p. 2.
ISBN
978-3-89119-011-1. A recent reference is given in Amie Brooks; Steven T. Boyd; Karen Nagel-Edwards (2008). "Biopharmaceutics".
Kaplan NAPLEX Review: The Complete Guide to Licensing Exam Certification for Pharmacists. New York, NY: Kaplan Publishing. pp. 401–405.
ISBN
978-1-4277-9576-2.
{{ cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) ( help) Some points to note: Liberation is synonymous to drug release (which is by far more commonly used) and is further divided into desintegration and dissolution. There is a wealth of information available. Earlier references on LADME (from the 1970ies) are in German – which is not surprising, since a lot of work in the field at that time was done in German-speaking countries (Germany, Switzerland, Austria; the term pharmacokinetics was translated from Pharmakokinetik – a neologism introduced by Friedrich Hartmut Dost in 1953). I would keep the statement about LADME in the article as it is – until somebody (te-heh) advances PK to a better shape. ;-) Alfie ↑↓ © 13:03, 18 August 2011 (UTC)
- Hi to both of you! The first reference giving "LADME" I have is in a chapter in Wolfgang A. Ritschel (1980). "Liberation, Absorption, Distribution, Metabolism, Elimination".
Handbook of Basic Pharmacokinetics. Hamilton, Ill.: Drug Intelligence Publications. p. 16.
ISBN
978-0-914768-34-0. Another old reference: Ernst Glaser (1985). "Pharmakokinetik".
Pharmakokinetik: Grundlagen, lineare Modelle, Rechenverfahren, Auswertemethoden (in German). PMI Verlag. p. 2.
ISBN
978-3-89119-011-1. A recent reference is given in Amie Brooks; Steven T. Boyd; Karen Nagel-Edwards (2008). "Biopharmaceutics".
Kaplan NAPLEX Review: The Complete Guide to Licensing Exam Certification for Pharmacists. New York, NY: Kaplan Publishing. pp. 401–405.
ISBN
978-1-4277-9576-2.
- Agreed. Again, I am a few years outdated with regards to knowledge about pharmacokinetics, but we should be careful about giving LADME too much emphasis. Even though are literature that endorse this model, we can probably find a great distribution of good and bad non-mainstream ideas if we look hard enough anyway. I think the current version of that section would suffice until LADME goes mainstream (i.e. get into prominent textbooks and used in big research articles). --
Bobthefish2 (
talk) 21:08, 17 August 2011 (UTC)
- Thanks for the citation. I have
added the citation to the text and deleted the word "recent". Were Koch and/or Ritschel (or perhaps
Leopold) the first ones to coin the phrase "LADME"? Liberation is of course a prerequisite for adsorption. Nevertheless the ADME acronym is far more widely used than LADME. Hence I think it is appropriate that ADME first be defined followed by LADME. I agree that there is a lot of good material in the Spanish article could be useful in expanding the English version.
Boghog (
talk) 17:52, 17 August 2011 (UTC)
Following copied from User_talk:Boghog#Alpha.2FBeta_phase_of_i.v.:
Hi Boghog, I saw your new section and I think it's well-written. One additional thing that'd be helpful is to include an example chart of concentration vs. time with arrows pointing to the phase where passive diffusion dominates and the phase where elimination dominates (By the way, do you mean elimination as in "excretion" or elimination as in "metabolism" + "excretion"? Some pharmacologists use the term "elimination" in the same way as "excretion"). -- Bobthefish2 ( talk) 18:13, 26 August 2011 (UTC)
- Hi Bobthefish. Thanks for your kind words. I think the meaning of "elimination" intended here is "metabolism" + "excretion". I need to do more background reading (disclaimer: I am not a pharmacokineticist although I do work in the pharmaceutical industry.) I will see what I can do about the graphic. Cheers.
Boghog (
talk) 21:01, 26 August 2011 (UTC)
- No worries, I am much less a pharmacokineticist than you are - namely, I only have a undergraduate level of understanding in medicinal chemistry. If the graph is too much of a hassle, you don't need to bother. I was under the impression that these graphs are easy to find. Maybe I will try to find one in a Pharmacology 101 lecture document. --
Bobthefish2 (
talk) 00:08, 27 August 2011 (UTC)
- Found it. Image: [2], website: [3]. Now, the question is how to import the image into WP. I don't have any experience with that. -- Bobthefish2 ( talk) 07:47, 27 August 2011 (UTC)
- No worries, I am much less a pharmacokineticist than you are - namely, I only have a undergraduate level of understanding in medicinal chemistry. If the graph is too much of a hassle, you don't need to bother. I was under the impression that these graphs are easy to find. Maybe I will try to find one in a Pharmacology 101 lecture document. --
Bobthefish2 (
talk) 00:08, 27 August 2011 (UTC)

- Good work. That website also has other good images. I will see if I can redraw some of them like you did and put them up there. -- Bobthefish2 ( talk) 19:33, 27 August 2011 (UTC)
- Hi guys; sorry for cutting in. ;-) We must not forget that the phases after i.v. administration always imply an underlying pharmacokinetic model. We shouldn't make a general statement about alpha/beta (=2 compartment open model) giving readers the false impression that this is 'carved from stone'. Especially at low doses only one phase is captured due to limitations in bioanalytical methods, whereas with 'better' methods and/or at higher doses more phases (gamma and sometimes even delta) are commonly seen. I'm also not happy with the image. The concentration is in log-scale which is good for educational purposes – but might be confusing if compared to the p.o. image below (linear scale). I would suggest to either give an explanation in the image itself or at least in the image's caption.
Alfie
↑↓
© 12:25, 31 August 2011 (UTC)
- Thanks for the suggestions. I think both the figure and the text can be tweaked to address your concerns. Starting with the figure, I have uploaded a new version (displayed to the right) that specifies that the y-axis is on a log scale. For an encyclopedia aimed at both a wide audience, but also specialists, I suggest that we not complicate the figure any further by adding additional phases. But these should be mentioned in the text. Do you have any good sources that document gamma and delta phases?
Boghog (
talk) 20:35, 31 August 2011 (UTC)
- The "phases" shown are from the biexponential model, not the data. For a triexponential there are three "phases," and for the sum of four exponential functions, there are four "phases." Think of it this way, each exponential is a local function whose influence on the total sum of the model drops significantly in time to become insignificant after several "half-lives" of that individual term. BTW, this type of reasoning is restricted to strictly log convex functions and sums of exponential terms may converge in the complex field alone, i.e., this type of model is not robust, and for biexponentials at least 6 samples are needed for a failure to achieve a real valued solution of circa 2%, although Bayesian, Tikhonov or other inverse method for treatment of ill-posed problems of the Fredholm integral of the first kind /info/en/?search=Fredholm_integral_equation#Equation_of_the_first_kind can be used for regularization. Also see https://jnm.snmjournals.org/content/43/6/762.short and https://journals.lww.com/nuclearmedicinecomm/Fulltext/2016/01000/Accurate_and_precise_plasma_clearance_measurement.12.aspx CarlWesolowski ( talk) 02:46, 23 December 2022 (UTC)
- Thanks for the suggestions. I think both the figure and the text can be tweaked to address your concerns. Starting with the figure, I have uploaded a new version (displayed to the right) that specifies that the y-axis is on a log scale. For an encyclopedia aimed at both a wide audience, but also specialists, I suggest that we not complicate the figure any further by adding additional phases. But these should be mentioned in the text. Do you have any good sources that document gamma and delta phases?
Boghog (
talk) 20:35, 31 August 2011 (UTC)
End of insert Alfie ↑↓ © 00:07, 1 September 2011 (UTC)
- Hi Boghog! Yes, the new image is a good starter. Well, I do have about one meter (sorry for my metric background) of textbooks on PK and easily 2,000 hardcopies of published papers ;-)
Pharmacokineticists use two types of notations: If a model is defined by compartments they are simply indexed, whereas the central compartment (e.g., plasma or serum) is given #1. 'One-way'-compartments (i.e., the GI-tract in oral administration or urine) get #0. Peripheral compartments (e.g., tissue or erythrocytes) get higher indices (#2, #3,…). Compartments with indices >0 have volumes of distribution (V1, V2,…). Rate constants connecting compartments get two indices. Example for a two-compartment model after an oral dose and renal elimination: k01 is the absorption rate constant (GIT→central), k12 the disposition rate constant (central→peripheral), k21 the disposition rate constant (peripheral→central), and k10 the elimination rate constant (central→urine). To make things more complicated some authors use ka instead of k01, kel instead of k10, and Vc instead of V1 (in any combination).
The second notation is based on the fact that any mammillary PK model can be rewriten as a sum of exponential terms. - By convention the terms are ordered from fast (large) to slow (small) by the exponents. Now it gets really nasty. Some authors use the indices as above (with the special case of λz denoting the last (=slowest) phase, whilst others come up with something like (again a two-compartment oral):
- It's a mess because the same drug given i.v. would simply be described by:
- (note the 'missing' absorption phase)
- Confusion is perfect, because the distribution phase (p.o.: = i.v.: ) and the elimination phase (p.o.: = i.v.: )…
Right now I have no idea how the rewrite the section in the article – maybe anybody else has more intellectual horsepower than me. That's why I have taken the discussion over here. Alfie ↑↓ © 00:07, 1 September 2011 (UTC)- Can't really help you with that. I am more used to traditional kinetic models and haven't actually worked with the sum of exponential decays that you showed. --
Bobthefish2 (
talk) 02:07, 1 September 2011 (UTC)
- This is way beyond my capabilities. I just
added a short mention to the article that additional phases are sometimes seen with a supporting citation. Feel free to expand.
Boghog (
talk) 08:55, 3 September 2011 (UTC)
- Is this the most common way of mathematically expressing drug kinetics though? I thought differential equations like these are more main stream
[4] (but then again, I have little experience with drug-lifetime-kinetic models) --
Bobthefish2 (
talk) 17:37, 3 September 2011 (UTC)
- Does not make a difference as typically used. The solution to a suitable differential equation with the correct initial conditions yields a function of time, and that function of time can be written as a differential equation. There is some art involved in doing that, but in the simple case of mammalian compartmental models, a model having n exponential terms is equivalent to an n-th order differential equation with constant coefficients. Not so easy to do with some other models, and if I had to pick between them, I would choose the solution model rather than differential equation model. CarlWesolowski ( talk) 11:24, 23 December 2022 (UTC)
- Is this the most common way of mathematically expressing drug kinetics though? I thought differential equations like these are more main stream
[4] (but then again, I have little experience with drug-lifetime-kinetic models) --
Bobthefish2 (
talk) 17:37, 3 September 2011 (UTC)
- This is way beyond my capabilities. I just
added a short mention to the article that additional phases are sometimes seen with a supporting citation. Feel free to expand.
Boghog (
talk) 08:55, 3 September 2011 (UTC)
- Can't really help you with that. I am more used to traditional kinetic models and haven't actually worked with the sum of exponential decays that you showed. --
Bobthefish2 (
talk) 02:07, 1 September 2011 (UTC)
- Hi Boghog! Yes, the new image is a good starter. Well, I do have about one meter (sorry for my metric background) of textbooks on PK and easily 2,000 hardcopies of published papers ;-)
Under the "Simulation" subsection of the "Software" section it states: "All model based software above." Is that all of the software listed above, or certain members listed above? Aren't all of the names mentioned "model based"? Could someone check on this? Maybe the statement should be: "Any of the software listed above." -- 74.179.120.70 ( talk) 17:57, 13 September 2011 (UTC)
- Hi IP! I’m not a native speaker, maybe you can do better: Actually for simulation you can use any software except the ones in the first group (Noncompartmental ≡ not based on a PK model). Since I’m using a good part of them myself I’m routine-blinded. ;-) Alfie ↑↓ © 00:31, 14 September 2011 (UTC)
1. Ref 13 links to an empty page.
2. Also in the list of pharmacokinetic metrics, the rate of elimination has been missed out. Could this be included too? Elimination rate constant is different to rate of elimination and including both would prevent confusion for readers. Thanks
Earl Moss (
talk) 13:47, 19 December 2012 (UTC)
- Hi Earl Moss! No 1: Not to an empty page, but reading "The electronic version of this document is currently unavailable". Unfortunately the document is not available at www.archive.org. I added a dead-link template to the ref. in the meantime. The position paper is mentioned here. I receive a daily feed from EMA giving their new guidelines. As soon as something new is published I will update the ref.
- No 2: I don't understand what you mean by "Elimination rate constant is different to rate of elimination". Can you paraphrase? Alfie ↑↓ © 14:26, 22 December 2012 (UTC)
Hello Alfie. Thank you very much for reading my comment and trying to act on it.
Regarding No 2: Sure, I can try (btw I'm just a student, no expert). The "Elimination rate constant" (time-1) describes the fraction of drug eliminated from the body in a given time. However the "Rate of elimination" (mass time-1) describes the mass of drug eliminated from the body in a given time. The "Rate of elimination" shares the same units as "Infusion rate". (Example of a sentence using "Rate of elimination": The amount of drug in the body remains constant when the rate of elimination equals the rate of infusion) The example would not make sense if you replaced the phrase "rate of elimination" with "elimination rate constant". I hope that has helped to explain what I meant.
So they are different variables, which leaves the question about whether there is a need to include it in the list. From my lectures, it seems that "Rate of elimination" is often talked about in education and is also used clinically. Added to the fact that some people may confuse the two variables (like me when I first read it), I think that both should be in the table. Feel free to ask for clarification or anything else. I'm happy to discuss it if you disagree. Merry Christmas/Seasons greetings!!
Earl Moss (
talk) 22:17, 24 December 2012 (UTC)
Why does this article lead off with a graph of Michaelis Menten Kinetics? Its a beautiful graph, but is not mentioned until the section on multi-compartment models. I associate the MM curve more with biochemistry than pharmacokinetics, the latter having more to do with drug levels in vivo. So as an introductory figure it kind of misses the point. Additionally, its labeled in Spanish.
This figure should be relabeled in English and moved to the middle portion of the article. The 3rd figure (Farmacocinética_no_lineal.svg) should also be relabeled in English.
I think a better introductory graph would be the IV time vs. concentration curve. (Iv time conc curve.svg) mattelfesso ( talk) 20:58, 29 April 2014 (UTC)
— Preceding unsigned comment added by 70.62.125.212 ( talk) 15:54, 7 August 2014 (UTC)
The last paragraph of the Bioavailability section says "When two drugs have the same bioavailability, they are said to be biological equivalents or bioequivalents. This concept of bioequivalence is important because it is currently used as a yardstick in the authorization of generic drugs in many countries."
This makes it sound like the article is saying bioavailability (BA) and bioequivalence (BE) are the same thing, which is not true. I'm guessing it's trying to say that BA contributes to BE. I don't feel confident enough to edit this sentence (I know the basics, but really just the basics), but I'm hoping someone with more knowledge that me can. Thank you to anyone who can help. — Preceding unsigned comment added by 70.62.125.212 ( talk) 15:58, 7 August 2014 (UTC)
- Your comments are spot on. An accurate definition of bioequivalence is found here that makes clear that bioequivalence depends not only on bioavailability as measured as area under the curve, but also on Cmax. Bioavailability relates to the fraction of drug absorbed after oral administration and is estimated from ratio of area under the curve after oral vs. intravenous administration. Bioequivalence in addition requires that the rate of absorption as measured by Cmax are similar. Boghog ( talk) 19:24, 7 August 2014 (UTC)
- Hi IP and Boghog! AFAIK the FDA introduced the terms extent and rate of biovailability in the early 1980s, whereas the former is given by the AUC and the latter by Cmax. At that time the pharmacokinetic community protested, since according to all textbooks BA = AUC. Still many colleagues call the term extent of BA plain nonsense. Since I’m not a native speaker of English any suggestions to change the paragraph are welcome. In an article about PK I would stick to the definition of BA as given in textbooks and make clear that the regulatory definition is different. Alfie ↑↓ © 14:09, 11 August 2014 (UTC)
Hello fellow Wikipedians,
I have just modified 3 external links on Pharmacokinetics. Please take a moment to review my edit. If you have any questions, or need the bot to ignore the links, or the page altogether, please visit this simple FaQ for additional information. I made the following changes:
- Added
{{ dead link}}tag to http://www.revistanefrologia.com/mostrarfile.asp?ID=1566 - Added archive https://web.archive.org/web/20150407143248/http://www.mango-solutions.com/wp/products-services/products/rapidnca/ to http://www.mango-solutions.com/wp/products-services/products/rapidnca/
- Added archive https://web.archive.org/web/20080530085228/http://www.simulations-plus.com/Products.aspx?grpID=3&cID=16&pID=11 to http://www.simulations-plus.com/Products.aspx?grpID=3&cID=16&pID=11
- Added archive https://web.archive.org/web/20110201042414/http://www.pharsight.com/products/prod_phoenix_nlme_home.php to http://www.pharsight.com/products/prod_phoenix_nlme_home.php
When you have finished reviewing my changes, you may follow the instructions on the template below to fix any issues with the URLs.
This message was posted before February 2018.
After February 2018, "External links modified" talk page sections are no longer generated or monitored by InternetArchiveBot. No special action is required regarding these talk page notices, other than
regular verification using the archive tool instructions below. Editors
have permission to delete these "External links modified" talk page sections if they want to de-clutter talk pages, but see the
RfC before doing mass systematic removals. This message is updated dynamically through the template {{
source check}} (last update: 5 June 2024).
- If you have discovered URLs which were erroneously considered dead by the bot, you can report them with this tool.
- If you found an error with any archives or the URLs themselves, you can fix them with this tool.
Cheers.— InternetArchiveBot ( Report bug) 09:05, 21 September 2017 (UTC)
At [5] there is a need for a section under "Pharmacokinetic data" pertaining to the following:
- Antibiotic resistance
- Medication Tolerance
If anyone is interested in this topic, please aid in the development of its proper titles, details (for all drugs), most importantly, the drug statistics on the population involving their likelihood for a human body to not respond to it. Hopefully, we can approximate dose quantities that lead to these issues in the long term. Twillisjr ( talk) 21:27, 4 September 2018 (UTC)

I tried making diagram, to simplify the description of the relationship between the Kinetics and Dynamics. What do you guys think? Claes Lindhardt ( talk) 20:20, 16 January 2023 (UTC)
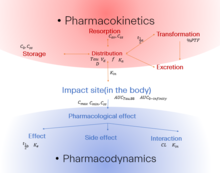
I found it confusing which metrics are used where I tried making this diagram where I tried associating the diffrent units with the place where at first glance it sounds like they might belong. But I would really appriacte any help to correct it :) I feel like it is very difficult to keep the overview of all the interplay when everything is only on list form. I can quickly adapt any change suggestions. Thank you in advance Claes Lindhardt ( talk) 20:40, 16 January 2023 (UTC)
- Hi Claes,
- I moved your two proposals to this new section.
- A picture is not always worth a thousand words. To get an idea how complicated it really is, see this presentation (slide 5) [1] – and that’s mainly the PK part…
- I appreciate your efforts but don’t think that your first image provides additional value to the article. Sorry. The labels you used in the second image are entirely wrong. All are PK metrics.
Alfie
↑↓
© 11:18, 17 January 2023 (UTC)
- Thank you very much for the feedback and specefied referance :). If you have the time to tell be where the thing in the second picture would ideally be. I would be more then happy to move them there.
- I will try to make a new version based on the linked slide. I think the connection between the diffrenet concepts under PK is not described in an accessible way. An i think some good diagrams could really help. Especially for people beeing presented to the theme for the first time. Claes Lindhardt ( talk) 19:59, 19 January 2023 (UTC)
References
- ^ Schütz, Helmut (December 15, 2021). "Bioequivalence. An Old Area with some Uncharted Territories" (PDF). Biometrical Colloquium. Retrieved January 17, 2013.
- C-Class vital articles
- Wikipedia level-4 vital articles
- Wikipedia vital articles in Biology and health sciences
- C-Class level-4 vital articles
- Wikipedia level-4 vital articles in Biology and health sciences
- C-Class vital articles in Biology and health sciences
- C-Class pharmacology articles
- Top-importance pharmacology articles
- WikiProject Pharmacology articles
- Pages translated from Spanish Wikipedia
|
| This It is of interest to the following WikiProjects: | ||||||||||
| |||||||||||
|
| This article contains a translation of Farmacocinética from es.wikipedia. |
Extended content
|
|---|
|
This is an important topic that deserces an article of its own. However, copying the abstradt of a review article verbatim in Wikipedia is against the rules (copyright violation) and not useful, because these docoments are too long and too technical. The editor is welcome to write a short encyclopedic excerpt, taking into account other sources like WHO, NIH etc. Andreas 17:13, 9 January 2006 (UTC) I have been given the assignment of giving a brief lecture at my church on this very topic. I will web-research the topic and write my findings to this article. As a retired, but still licensed registered nurse, I have limited access to hospital medical libraries. I will also make my best effort at including references and links. User:W8IMP 0837, 15 January 2007]] (UTC) Why is this article titled "Pharmacokinetics (section), when there is another article, not a "talk" section devoted to this topic. I would gladly re-title the article as "Drug-drug and drug-herbal interactions", as soon as I have more data, and someone tells me to change the title. User:W8IMP 08:51, 15 January 2007]] (UTC) |
The constant which relates the exponent of decay (k) and half-life is easily determined by first principles from the equation for exponential decay. Mentioning circuit analysis seems totally crazy to me; no pharmacokineticist would know anything about circuits or ever need to. If this is important then I think that the maths only should be mentioned. ben 06:50, 23 May 2006 (UTC)
- You forget that biomedical engineers (which usually have some understanding of circuits) are involved in the design of medical devices, such as insulin pumps (which have ICs). Pharmacokinetics is not such a narrow field. The research that lead to transdermal patches was done by a chemical engineer -- see Robert S. Langer. [1] Nephron T| C 20:24, 14 July 2006 (UTC)
Shouldn't there be something here about two (or more) compartment models, also non compartmental models? Let me hasten to add, I'm not the person to write it ... Daen 15:22, 25 September 2006 (UTC)
- You are right. Just some remarks:
The upper limit in practical use are three compartments, because the more compartments you add to your model, the more parameters you have to estimate from the data (two parameters for each compartment). Generally we have only samples from a limited number of compartments (plasma and urine – if we are lucky). Though it’s possible to get solutions from our curve fitting (i.e., nonlinear regression), the parameters are not identifiable for higher-order models any more. Not identifiable means that it is not possible to tell whether a given rate constant is attributable to a transfer process between two compartments, or elimination from the central compartment, e.t.c.
Non compartmental models don’t exist (that’s a contradiction). The term used for example in bioequivalence testing is Non Compartmental Analysis (NCA). Parameters (or metrics) of NCA are e.g., the area under the curve AUC (calculated by some kind of numerical integration algorithm, mostly the trapezoidal rule), the peak concentration Cmax and the time point of its occurence tmax,… All these values are directly obtained from the data assuming no underlying PK model. -- Alfie66 ( talk) 00:41, 10 June 2008 (UTC)
Actually, there are PK model assumptions made for the typical implementation of numerical integration, A.K.A. noncompartmental methods. Those assumptions become important when they are incorrect, which may actually be almost all of the time. Those assumptions commonly, but not always, include (1) that between samples of a monotonically decreasing blood concentration (i.e., a washout model of plasma or whole blood depending on the drug's hydrophilicity and lipophilicity), that the path of concentration is exponential. (2) That one can extrapolate exponentials to earlier times than the first sample time without error. (3) That the last and one or two prior samples can be used to extrapolate the AUC to infinity implying (3a) that the half-life is terminal at that time and (3b) that half-lives only arise from exponential functions. Granted, there are other ways of doing numerical integration, and the selection of extrapolating functions is not trivial. Let us review these assumptions. Ad (1) and (2), there is no evidence to that effect, and early concentration of a renal filtered non metabolized drug has been shown to be more a linear concentration and logarithm of time function than a logarithm of concentration linear time function [1]. Indeed, the former implies an initial zero volume of drug and the later implies an unrealistic instant mixing in an appreciable volume (see below). The dearth of testing on this subject appalls me. Ad (3a), the half-life for the last samples is typically not terminal. Indeed, the terminal function may occur very late in time and may be a power function [2], what the terminal function does not appear to be is an exponential [3], which reference also offers an explanation of drug volumes in time. I welcome comments on this, and have not altered the main text due to a conflict of interest on my part. I need other minds to review this. CarlWesolowski ( talk) 16:08, 23 August 2016 (UTC).
BTW, there are multiple circumstances in which compartmental models are not "the best" at anything. Rather than give a whole bunch of references here regarding the need to correct one compartmental clearances by 15% or so, the superiority of other methods like constant infusion and saturated systems for measuring clearance, adaptive gamma variate fitting for bolus experiment clearances, Michaelis-Menten for saturable receptor clearances and so forth, I just changed the text to be more honest, i.e., from best to most frequently used. Also, there are broken links in the text some of which I have fixed, HELP!!! This isn't my article, so would someone else take care of it, too, please? And, if you don't agree with my (minor) changes, do let's talk, please. CarlWesolowski ( talk) 20:20, 9 February 2017 (UTC)
It may be more useful to refer to Enzyme kinetics. At the very least, there should be a reference to it somewhere in the article. 136.165.191.251 ( talk) 16:07, 27 April 2009 (UTC)
References
Variable volume pharmacokinetic models is now linked to this article. Linking back is a consideration, so is thinking about it. Talk to me. CarlWesolowski ( talk) 14:20, 23 August 2016 (UTC) CarlWesolowski ( talk) 03:44, 9 September 2016 (UTC) No one talked to me. The linking article was an orphan so that linking to it was needed. And for completeness, it should be listed anyway. CarlWesolowski ( talk) 21:53, 17 November 2016 (UTC)
- Variable volume pharmacokinetic models was deleted because of "contribution rules." Variable volume models were correctly stated as an alternative to compartmental modelling by S. Niazi "Volume of Distribution as a Function of Time" Journal of Pharmaceutical Sciences 1976 Vol. 65 Issue 3 Pages 452-454 https://doi.org/10.1002/jps.2600650339. This was later generalized to all PK models by C. A. Wesolowski (full disclosure, me) et al. "Time Varying Apparent Volume of Distribution and Drug Half-Lives Following Intravenous Bolus Injections" Plos One 2016, https://doi.org/10.1371/journal.pone.0158798, and applied in subsequent papers. Content contribution rules being somewhat arcane here, someone else would have to link these to the current article instead of the now dead link, or alternatively provide text once again to the now defunct "Variable volume pharmacokinetic models" link. With time, this may become increasingly important as alternative models are shown to work better than the same old stuff. CarlWesolowski ( talk) 01:01, 17 November 2022 (UTC)
Who "invented" the "Pharmacokinetics"? —Preceding unsigned comment added by 217.86.119.8 ( talk) 21:22, 3 October 2006 (UTC)
- Nobody – or like Newton 'invented' 'Gravity'… ;-)
The term ( de:Pharmakokinetik) was first used in the 1950ies by the German Professor for Pediatrics Friedrich Hartmut Dost (*1910 †1985). [1] I would translate the reference with “Blood level: Kinetics of the Course of Concentrations in the Systemic Circulation”.
“Dost’s law of corresponding areas” (sometimes called Superposition Principle) describes the fact that in a linear pharmacokinetic system the area under the (plasma drug concentration-time) curve after a single dose (t=0-∞) equals to the AUC within a dosage interval (t=0-τ) in steady-state. -- Alfie ( talk) 00:15, 10 June 2008 (UTC)
References
- ^ Dost, Friedrich Hartmut (1953). Der Blutspiegel: Kinetik der Konzentrationsabläufe in der Kreislaufflüssigkeit (in German). Leipzig: Thieme-Verlag. p. 244.
Would it be useful to have a list of the standard PK parameters, with some brief explanation? Examples: Cmax, Tmax, AUC, etc. —Preceding unsigned comment added by 216.158.41.81 ( talk) 18:35, 27 June 2007 (UTC)
It would be helpful to me! I would definitely read that section carefully. Mlbish ( talk) 21:32, 21 February 2008 (UTC)
- Agree. Unfortunatelly I’m a little short in time… In the meantime have a look at a document [1] published by the German ‘Association for Applied Human Pharmacology’ (AGAH). -- Alfie ( talk) 00:50, 2 July 2008 (UTC)
- I have made an initial effort at adding a Pharmacokinetics#Parameters section. Thanks for the reference which I have added to the article. Cheers. Boghog ( talk) 16:15, 6 June 2010 (UTC)
References
- ^ "Collection of terms, symbols, equations, and explanations of common pharmacokinetic and pharmacodynamic parameters and some statistical functions" (PDF). AGAH working group PHARMACOKINETICS. 16 February 2004. Retrieved 2009-09-18.
Drug properties that influence its pharmacokinetics
Several drug properties can often influence the pharmacokinetics of many drugs.
Organ partitioning
Partition coefficient
The partition or distribution coefficient (KD) is the ratio of concentrations of a compound in the two phases of a mixture of two immiscible solvents at equilibrium. [1] It can influence the ADME properties (Absorption, Distribution, Metabolism, and Excretion) of the drug. When orally administered drugs are absorbed they must first pass through lipid bilayers in the intestinal epithelium (a process known as transcellular transport). For efficient transport the drug must be hydrophobic enough to partition into the lipid bilayer but not so hydrophobic that once it is in the bilayer it will not partition out again. [2] The partition coefficient is dependent on the hydrophobic or hydrophilic properties of the drug.
References
-
^ Leo A, Hansch C, and Elkins D (1971). "Partition coefficients and their uses". Chem Rev. 71 (6): 525–616.
doi:
10.1021/cr60274a001.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ Kubinyi H (1979). "Nonlinear dependence of biological activity on hydrophobic character: the bilinear model". Farmaco [Sci]. 34 (3): 248–76. PMID 43264.
Drug transporters
Drug transporters are transmembrane proteins on the surface of cells and are responsible for facilitating or hindering the intracellular and paracellular transport of nutrients and other substances. Some known drug transporters include Breast Cancer Resistance Protein (BCRP, also known as ABCG2) and human intestinal peptide transporter (hPepT1). BCRP is a type of ATP-binding cassette transporter that can decrease the efficacy of chemotherapeutic agents in breast cancer by exporting agents out of the tumor cells, thus making them resistant to chemotherapy. Valacyclovir uses the hPepT1 transporter to increase the intestinal absorption of valacyclovir compared to acyclovir. Drug transporters can increase or decrease the absorption of drugs into the body as well as limit or facilitate the exposure of certain organs.
Perfusion
Perfusion or flow of blood to different organs, affects rate of presentation of drugs to different parts of the body and often affects the pharmacokinetics of many drugs. While the partition coefficient can affect the distribution of drugs from the blood stream to the organs, perfusion affects how fast a drug is presented to the organs. Different organs receive a vast supply of drugs while others receive minor amounts. For example, the kidneys receive vast quantities of blood, especially considering the relatively small size of the kidneys. Adipose tissue, on the other hand, receives a minor supply blood. Organs with a rich blood supply would be presented drug at a higher rate than organs with a lower blood supply.
Biophys ( talk) 01:56, 5 September 2008 (UTC)
Article Reassessment for WikiProject Pharmacology
Hello. I am a member of WikiProject Pharmacology, a Wikipedia wide project that maintains and improves articles that fall under the scope of pharmacology. Since your article has fallen under our scope, I have placed the correct template(s) on this talk page for verification. Upon review of the article, I'd like to make a few points, as shown:
- Ressessed article with class and importance factors
- It was not a B-class article, yes, so it has been lowered to C. I also put this article for mid importance due to the fact that it is specifically taught in classes at multiple universities and required by state pharmacy boards, such as the South Dakota Board of Pharmacy.
I'm glad this article could fall within our scope, and I hope to see it grow large! Many thanks! Renaissancee (talk) 02:21, 3 June 2009 (UTC)
- I have changed pharmacology project importance rating from mid to top; along with pharmacodynamics, pharmacokinetics is a core pharmacology topic! Boghog ( talk) 16:24, 6 June 2010 (UTC)
This article is quite poor to be in the English version of Wikipedia; I compared it with other versions like the spanish one, and I noticed there is an important lack of information here. An improvement of this article should be done. —Preceding unsigned comment added by 83.44.20.68 ( talk • contribs) 01:20, 20 September 2009 (UTC)
- Please add new sections to talk pages at their bottom (I moved your contribution). Well, from a quick look at the Spanish version, I guess you are right. You are are cordially invited to start working on an adaption. — Alfie ±Talk 13:08, 20 September 2009 (UTC)
Request for review of Plateau Principle entry
I have added a review of the development of the Plateau Principle in pharmacokinetics and the wide applicability of the mathematical model to other areas in biological sciences. It would be very helpful to obtain an expert review of this new entry. Please provide comments in the talk section of Plateau Principleand delete this section when a review has been done. Many thanks. Jhargrov ( talk) 14:03, 10 October 2009 (UTC)James Hargrove Jhargrov ( talk) 14:03, 10 October 2009 (UTC)
- Hi James! Wow, you've done a lot of work! I will have a deeper look, time allowing (there are some minor flaws in the TEX-markup). To be honest, this term is rarely (whenever) used in pharmacokinetics - steady state is used instead. I cann't find the term in standard textbooks (like Dost 1953, Gibaldi/Perrier 1982, Wagner 1993, Gabrielsson/Weiner 2006) explaining the Superposition Principle. In German the term Plateauphase was used years ago, but was abandoned due to the sexual connotation (see Plateau phase); in German the English term 'steady state' is solely in use as a loanword for some time now. — Alfie ±Talk 11:22, 12 October 2009 (UTC)
It's been a while since I've dealt with topics in pharmacokinetics. Is "liberation" a new and recognized stage of drug kinetics? If so, please provide citation. -- Bobthefish2 ( talk) 04:53, 7 August 2011 (UTC)
- A search of LADME in
PubMed yields only
3 hits while ADME yields
774 hits. Hence LADME does not as yet appear to be a widely used term. Hence the previous discussion about LADME in this article appears to give
undue weight to this term. I have therefore modified this section to first define what ADME is, and then mention LADME supported by a reliable source.
Boghog (
talk) 22:30, 7 August 2011 (UTC)
- A caveat about your pubmed analysis is that you should've limited the date to maybe Jan 2009 - Aug 2011. ADME has been around for a VERY long time. Suppose LADME has gained prevalence recently (which it hasn't, at the moment), its hits will only prevail over that of ADME for the time period that we'd consider as recent (which is approximately 2-3 years for biological literature). --
Bobthefish2 (
talk) 02:20, 8 August 2011 (UTC)
- Limiting the search to the last two years decreases the number of ADME hits from 774 to 227 vs 3 for LADME. The conclusion remains the same.
Boghog (
talk) 02:27, 8 August 2011 (UTC)
- That's good. By the way, the pharmacodynamics article is in bad-shape. Feel free to clean that up too. I am thinking of making a few minor changes, but I am not even sure where to start. -- Bobthefish2 ( talk) 02:50, 8 August 2011 (UTC)
- Limiting the search to the last two years decreases the number of ADME hits from 774 to 227 vs 3 for LADME. The conclusion remains the same.
Boghog (
talk) 02:27, 8 August 2011 (UTC)
- A caveat about your pubmed analysis is that you should've limited the date to maybe Jan 2009 - Aug 2011. ADME has been around for a VERY long time. Suppose LADME has gained prevalence recently (which it hasn't, at the moment), its hits will only prevail over that of ADME for the time period that we'd consider as recent (which is approximately 2-3 years for biological literature). --
Bobthefish2 (
talk) 02:20, 8 August 2011 (UTC)
- Hi to both of you! Well the term is not that new… See
the cover of Heinrich P. Koch; Wolfgang A. Ritschel (1986).
Synopsis der Biopharmazie und Pharmakokinetik (in German). Ecomed.
ISBN
978-3-609-64970-2.
These guys are not amateurs. ;-) Go to PubMed and paste the following into the search field:- (Koch HP[Author]) OR (Ritschel WA[Author]) OR (Wolfgang Ritschel[Full Author Name])
- See also the article on PK in the Spanish WP (which is the only one of all language versions rated as good).
Alfie
↑↓
© 11:26, 17 August 2011 (UTC)
- Thanks for the citation. I have
added the citation to the text and deleted the word "recent". Were Koch and/or Ritschel (or perhaps
Leopold) the first ones to coin the phrase "LADME"? Liberation is of course a prerequisite for adsorption. Nevertheless the ADME acronym is far more widely used than LADME. Hence I think it is appropriate that ADME first be defined followed by LADME. I agree that there is a lot of good material in the Spanish article could be useful in expanding the English version.
Boghog (
talk) 17:52, 17 August 2011 (UTC)
- Agreed. Again, I am a few years outdated with regards to knowledge about pharmacokinetics, but we should be careful about giving LADME too much emphasis. Even though are literature that endorse this model, we can probably find a great distribution of good and bad non-mainstream ideas if we look hard enough anyway. I think the current version of that section would suffice until LADME goes mainstream (i.e. get into prominent textbooks and used in big research articles). --
Bobthefish2 (
talk) 21:08, 17 August 2011 (UTC)
- Hi to both of you! The first reference giving "LADME" I have is in a chapter in Wolfgang A. Ritschel (1980). "Liberation, Absorption, Distribution, Metabolism, Elimination".
Handbook of Basic Pharmacokinetics. Hamilton, Ill.: Drug Intelligence Publications. p. 16.
ISBN
978-0-914768-34-0. Another old reference: Ernst Glaser (1985). "Pharmakokinetik".
Pharmakokinetik: Grundlagen, lineare Modelle, Rechenverfahren, Auswertemethoden (in German). PMI Verlag. p. 2.
ISBN
978-3-89119-011-1. A recent reference is given in Amie Brooks; Steven T. Boyd; Karen Nagel-Edwards (2008). "Biopharmaceutics".
Kaplan NAPLEX Review: The Complete Guide to Licensing Exam Certification for Pharmacists. New York, NY: Kaplan Publishing. pp. 401–405.
ISBN
978-1-4277-9576-2.
{{ cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) ( help) Some points to note: Liberation is synonymous to drug release (which is by far more commonly used) and is further divided into desintegration and dissolution. There is a wealth of information available. Earlier references on LADME (from the 1970ies) are in German – which is not surprising, since a lot of work in the field at that time was done in German-speaking countries (Germany, Switzerland, Austria; the term pharmacokinetics was translated from Pharmakokinetik – a neologism introduced by Friedrich Hartmut Dost in 1953). I would keep the statement about LADME in the article as it is – until somebody (te-heh) advances PK to a better shape. ;-) Alfie ↑↓ © 13:03, 18 August 2011 (UTC)
- Hi to both of you! The first reference giving "LADME" I have is in a chapter in Wolfgang A. Ritschel (1980). "Liberation, Absorption, Distribution, Metabolism, Elimination".
Handbook of Basic Pharmacokinetics. Hamilton, Ill.: Drug Intelligence Publications. p. 16.
ISBN
978-0-914768-34-0. Another old reference: Ernst Glaser (1985). "Pharmakokinetik".
Pharmakokinetik: Grundlagen, lineare Modelle, Rechenverfahren, Auswertemethoden (in German). PMI Verlag. p. 2.
ISBN
978-3-89119-011-1. A recent reference is given in Amie Brooks; Steven T. Boyd; Karen Nagel-Edwards (2008). "Biopharmaceutics".
Kaplan NAPLEX Review: The Complete Guide to Licensing Exam Certification for Pharmacists. New York, NY: Kaplan Publishing. pp. 401–405.
ISBN
978-1-4277-9576-2.
- Agreed. Again, I am a few years outdated with regards to knowledge about pharmacokinetics, but we should be careful about giving LADME too much emphasis. Even though are literature that endorse this model, we can probably find a great distribution of good and bad non-mainstream ideas if we look hard enough anyway. I think the current version of that section would suffice until LADME goes mainstream (i.e. get into prominent textbooks and used in big research articles). --
Bobthefish2 (
talk) 21:08, 17 August 2011 (UTC)
- Thanks for the citation. I have
added the citation to the text and deleted the word "recent". Were Koch and/or Ritschel (or perhaps
Leopold) the first ones to coin the phrase "LADME"? Liberation is of course a prerequisite for adsorption. Nevertheless the ADME acronym is far more widely used than LADME. Hence I think it is appropriate that ADME first be defined followed by LADME. I agree that there is a lot of good material in the Spanish article could be useful in expanding the English version.
Boghog (
talk) 17:52, 17 August 2011 (UTC)
Following copied from User_talk:Boghog#Alpha.2FBeta_phase_of_i.v.:
Hi Boghog, I saw your new section and I think it's well-written. One additional thing that'd be helpful is to include an example chart of concentration vs. time with arrows pointing to the phase where passive diffusion dominates and the phase where elimination dominates (By the way, do you mean elimination as in "excretion" or elimination as in "metabolism" + "excretion"? Some pharmacologists use the term "elimination" in the same way as "excretion"). -- Bobthefish2 ( talk) 18:13, 26 August 2011 (UTC)
- Hi Bobthefish. Thanks for your kind words. I think the meaning of "elimination" intended here is "metabolism" + "excretion". I need to do more background reading (disclaimer: I am not a pharmacokineticist although I do work in the pharmaceutical industry.) I will see what I can do about the graphic. Cheers.
Boghog (
talk) 21:01, 26 August 2011 (UTC)
- No worries, I am much less a pharmacokineticist than you are - namely, I only have a undergraduate level of understanding in medicinal chemistry. If the graph is too much of a hassle, you don't need to bother. I was under the impression that these graphs are easy to find. Maybe I will try to find one in a Pharmacology 101 lecture document. --
Bobthefish2 (
talk) 00:08, 27 August 2011 (UTC)
- Found it. Image: [2], website: [3]. Now, the question is how to import the image into WP. I don't have any experience with that. -- Bobthefish2 ( talk) 07:47, 27 August 2011 (UTC)
- No worries, I am much less a pharmacokineticist than you are - namely, I only have a undergraduate level of understanding in medicinal chemistry. If the graph is too much of a hassle, you don't need to bother. I was under the impression that these graphs are easy to find. Maybe I will try to find one in a Pharmacology 101 lecture document. --
Bobthefish2 (
talk) 00:08, 27 August 2011 (UTC)

- Good work. That website also has other good images. I will see if I can redraw some of them like you did and put them up there. -- Bobthefish2 ( talk) 19:33, 27 August 2011 (UTC)
- Hi guys; sorry for cutting in. ;-) We must not forget that the phases after i.v. administration always imply an underlying pharmacokinetic model. We shouldn't make a general statement about alpha/beta (=2 compartment open model) giving readers the false impression that this is 'carved from stone'. Especially at low doses only one phase is captured due to limitations in bioanalytical methods, whereas with 'better' methods and/or at higher doses more phases (gamma and sometimes even delta) are commonly seen. I'm also not happy with the image. The concentration is in log-scale which is good for educational purposes – but might be confusing if compared to the p.o. image below (linear scale). I would suggest to either give an explanation in the image itself or at least in the image's caption.
Alfie
↑↓
© 12:25, 31 August 2011 (UTC)
- Thanks for the suggestions. I think both the figure and the text can be tweaked to address your concerns. Starting with the figure, I have uploaded a new version (displayed to the right) that specifies that the y-axis is on a log scale. For an encyclopedia aimed at both a wide audience, but also specialists, I suggest that we not complicate the figure any further by adding additional phases. But these should be mentioned in the text. Do you have any good sources that document gamma and delta phases?
Boghog (
talk) 20:35, 31 August 2011 (UTC)
- The "phases" shown are from the biexponential model, not the data. For a triexponential there are three "phases," and for the sum of four exponential functions, there are four "phases." Think of it this way, each exponential is a local function whose influence on the total sum of the model drops significantly in time to become insignificant after several "half-lives" of that individual term. BTW, this type of reasoning is restricted to strictly log convex functions and sums of exponential terms may converge in the complex field alone, i.e., this type of model is not robust, and for biexponentials at least 6 samples are needed for a failure to achieve a real valued solution of circa 2%, although Bayesian, Tikhonov or other inverse method for treatment of ill-posed problems of the Fredholm integral of the first kind /info/en/?search=Fredholm_integral_equation#Equation_of_the_first_kind can be used for regularization. Also see https://jnm.snmjournals.org/content/43/6/762.short and https://journals.lww.com/nuclearmedicinecomm/Fulltext/2016/01000/Accurate_and_precise_plasma_clearance_measurement.12.aspx CarlWesolowski ( talk) 02:46, 23 December 2022 (UTC)
- Thanks for the suggestions. I think both the figure and the text can be tweaked to address your concerns. Starting with the figure, I have uploaded a new version (displayed to the right) that specifies that the y-axis is on a log scale. For an encyclopedia aimed at both a wide audience, but also specialists, I suggest that we not complicate the figure any further by adding additional phases. But these should be mentioned in the text. Do you have any good sources that document gamma and delta phases?
Boghog (
talk) 20:35, 31 August 2011 (UTC)
End of insert Alfie ↑↓ © 00:07, 1 September 2011 (UTC)
- Hi Boghog! Yes, the new image is a good starter. Well, I do have about one meter (sorry for my metric background) of textbooks on PK and easily 2,000 hardcopies of published papers ;-)
Pharmacokineticists use two types of notations: If a model is defined by compartments they are simply indexed, whereas the central compartment (e.g., plasma or serum) is given #1. 'One-way'-compartments (i.e., the GI-tract in oral administration or urine) get #0. Peripheral compartments (e.g., tissue or erythrocytes) get higher indices (#2, #3,…). Compartments with indices >0 have volumes of distribution (V1, V2,…). Rate constants connecting compartments get two indices. Example for a two-compartment model after an oral dose and renal elimination: k01 is the absorption rate constant (GIT→central), k12 the disposition rate constant (central→peripheral), k21 the disposition rate constant (peripheral→central), and k10 the elimination rate constant (central→urine). To make things more complicated some authors use ka instead of k01, kel instead of k10, and Vc instead of V1 (in any combination).
The second notation is based on the fact that any mammillary PK model can be rewriten as a sum of exponential terms. - By convention the terms are ordered from fast (large) to slow (small) by the exponents. Now it gets really nasty. Some authors use the indices as above (with the special case of λz denoting the last (=slowest) phase, whilst others come up with something like (again a two-compartment oral):
- It's a mess because the same drug given i.v. would simply be described by:
- (note the 'missing' absorption phase)
- Confusion is perfect, because the distribution phase (p.o.: = i.v.: ) and the elimination phase (p.o.: = i.v.: )…
Right now I have no idea how the rewrite the section in the article – maybe anybody else has more intellectual horsepower than me. That's why I have taken the discussion over here. Alfie ↑↓ © 00:07, 1 September 2011 (UTC)- Can't really help you with that. I am more used to traditional kinetic models and haven't actually worked with the sum of exponential decays that you showed. --
Bobthefish2 (
talk) 02:07, 1 September 2011 (UTC)
- This is way beyond my capabilities. I just
added a short mention to the article that additional phases are sometimes seen with a supporting citation. Feel free to expand.
Boghog (
talk) 08:55, 3 September 2011 (UTC)
- Is this the most common way of mathematically expressing drug kinetics though? I thought differential equations like these are more main stream
[4] (but then again, I have little experience with drug-lifetime-kinetic models) --
Bobthefish2 (
talk) 17:37, 3 September 2011 (UTC)
- Does not make a difference as typically used. The solution to a suitable differential equation with the correct initial conditions yields a function of time, and that function of time can be written as a differential equation. There is some art involved in doing that, but in the simple case of mammalian compartmental models, a model having n exponential terms is equivalent to an n-th order differential equation with constant coefficients. Not so easy to do with some other models, and if I had to pick between them, I would choose the solution model rather than differential equation model. CarlWesolowski ( talk) 11:24, 23 December 2022 (UTC)
- Is this the most common way of mathematically expressing drug kinetics though? I thought differential equations like these are more main stream
[4] (but then again, I have little experience with drug-lifetime-kinetic models) --
Bobthefish2 (
talk) 17:37, 3 September 2011 (UTC)
- This is way beyond my capabilities. I just
added a short mention to the article that additional phases are sometimes seen with a supporting citation. Feel free to expand.
Boghog (
talk) 08:55, 3 September 2011 (UTC)
- Can't really help you with that. I am more used to traditional kinetic models and haven't actually worked with the sum of exponential decays that you showed. --
Bobthefish2 (
talk) 02:07, 1 September 2011 (UTC)
- Hi Boghog! Yes, the new image is a good starter. Well, I do have about one meter (sorry for my metric background) of textbooks on PK and easily 2,000 hardcopies of published papers ;-)
Under the "Simulation" subsection of the "Software" section it states: "All model based software above." Is that all of the software listed above, or certain members listed above? Aren't all of the names mentioned "model based"? Could someone check on this? Maybe the statement should be: "Any of the software listed above." -- 74.179.120.70 ( talk) 17:57, 13 September 2011 (UTC)
- Hi IP! I’m not a native speaker, maybe you can do better: Actually for simulation you can use any software except the ones in the first group (Noncompartmental ≡ not based on a PK model). Since I’m using a good part of them myself I’m routine-blinded. ;-) Alfie ↑↓ © 00:31, 14 September 2011 (UTC)
1. Ref 13 links to an empty page.
2. Also in the list of pharmacokinetic metrics, the rate of elimination has been missed out. Could this be included too? Elimination rate constant is different to rate of elimination and including both would prevent confusion for readers. Thanks
Earl Moss (
talk) 13:47, 19 December 2012 (UTC)
- Hi Earl Moss! No 1: Not to an empty page, but reading "The electronic version of this document is currently unavailable". Unfortunately the document is not available at www.archive.org. I added a dead-link template to the ref. in the meantime. The position paper is mentioned here. I receive a daily feed from EMA giving their new guidelines. As soon as something new is published I will update the ref.
- No 2: I don't understand what you mean by "Elimination rate constant is different to rate of elimination". Can you paraphrase? Alfie ↑↓ © 14:26, 22 December 2012 (UTC)
Hello Alfie. Thank you very much for reading my comment and trying to act on it.
Regarding No 2: Sure, I can try (btw I'm just a student, no expert). The "Elimination rate constant" (time-1) describes the fraction of drug eliminated from the body in a given time. However the "Rate of elimination" (mass time-1) describes the mass of drug eliminated from the body in a given time. The "Rate of elimination" shares the same units as "Infusion rate". (Example of a sentence using "Rate of elimination": The amount of drug in the body remains constant when the rate of elimination equals the rate of infusion) The example would not make sense if you replaced the phrase "rate of elimination" with "elimination rate constant". I hope that has helped to explain what I meant.
So they are different variables, which leaves the question about whether there is a need to include it in the list. From my lectures, it seems that "Rate of elimination" is often talked about in education and is also used clinically. Added to the fact that some people may confuse the two variables (like me when I first read it), I think that both should be in the table. Feel free to ask for clarification or anything else. I'm happy to discuss it if you disagree. Merry Christmas/Seasons greetings!!
Earl Moss (
talk) 22:17, 24 December 2012 (UTC)
Why does this article lead off with a graph of Michaelis Menten Kinetics? Its a beautiful graph, but is not mentioned until the section on multi-compartment models. I associate the MM curve more with biochemistry than pharmacokinetics, the latter having more to do with drug levels in vivo. So as an introductory figure it kind of misses the point. Additionally, its labeled in Spanish.
This figure should be relabeled in English and moved to the middle portion of the article. The 3rd figure (Farmacocinética_no_lineal.svg) should also be relabeled in English.
I think a better introductory graph would be the IV time vs. concentration curve. (Iv time conc curve.svg) mattelfesso ( talk) 20:58, 29 April 2014 (UTC)
— Preceding unsigned comment added by 70.62.125.212 ( talk) 15:54, 7 August 2014 (UTC)
The last paragraph of the Bioavailability section says "When two drugs have the same bioavailability, they are said to be biological equivalents or bioequivalents. This concept of bioequivalence is important because it is currently used as a yardstick in the authorization of generic drugs in many countries."
This makes it sound like the article is saying bioavailability (BA) and bioequivalence (BE) are the same thing, which is not true. I'm guessing it's trying to say that BA contributes to BE. I don't feel confident enough to edit this sentence (I know the basics, but really just the basics), but I'm hoping someone with more knowledge that me can. Thank you to anyone who can help. — Preceding unsigned comment added by 70.62.125.212 ( talk) 15:58, 7 August 2014 (UTC)
- Your comments are spot on. An accurate definition of bioequivalence is found here that makes clear that bioequivalence depends not only on bioavailability as measured as area under the curve, but also on Cmax. Bioavailability relates to the fraction of drug absorbed after oral administration and is estimated from ratio of area under the curve after oral vs. intravenous administration. Bioequivalence in addition requires that the rate of absorption as measured by Cmax are similar. Boghog ( talk) 19:24, 7 August 2014 (UTC)
- Hi IP and Boghog! AFAIK the FDA introduced the terms extent and rate of biovailability in the early 1980s, whereas the former is given by the AUC and the latter by Cmax. At that time the pharmacokinetic community protested, since according to all textbooks BA = AUC. Still many colleagues call the term extent of BA plain nonsense. Since I’m not a native speaker of English any suggestions to change the paragraph are welcome. In an article about PK I would stick to the definition of BA as given in textbooks and make clear that the regulatory definition is different. Alfie ↑↓ © 14:09, 11 August 2014 (UTC)
Hello fellow Wikipedians,
I have just modified 3 external links on Pharmacokinetics. Please take a moment to review my edit. If you have any questions, or need the bot to ignore the links, or the page altogether, please visit this simple FaQ for additional information. I made the following changes:
- Added
{{ dead link}}tag to http://www.revistanefrologia.com/mostrarfile.asp?ID=1566 - Added archive https://web.archive.org/web/20150407143248/http://www.mango-solutions.com/wp/products-services/products/rapidnca/ to http://www.mango-solutions.com/wp/products-services/products/rapidnca/
- Added archive https://web.archive.org/web/20080530085228/http://www.simulations-plus.com/Products.aspx?grpID=3&cID=16&pID=11 to http://www.simulations-plus.com/Products.aspx?grpID=3&cID=16&pID=11
- Added archive https://web.archive.org/web/20110201042414/http://www.pharsight.com/products/prod_phoenix_nlme_home.php to http://www.pharsight.com/products/prod_phoenix_nlme_home.php
When you have finished reviewing my changes, you may follow the instructions on the template below to fix any issues with the URLs.
This message was posted before February 2018.
After February 2018, "External links modified" talk page sections are no longer generated or monitored by InternetArchiveBot. No special action is required regarding these talk page notices, other than
regular verification using the archive tool instructions below. Editors
have permission to delete these "External links modified" talk page sections if they want to de-clutter talk pages, but see the
RfC before doing mass systematic removals. This message is updated dynamically through the template {{
source check}} (last update: 5 June 2024).
- If you have discovered URLs which were erroneously considered dead by the bot, you can report them with this tool.
- If you found an error with any archives or the URLs themselves, you can fix them with this tool.
Cheers.— InternetArchiveBot ( Report bug) 09:05, 21 September 2017 (UTC)
At [5] there is a need for a section under "Pharmacokinetic data" pertaining to the following:
- Antibiotic resistance
- Medication Tolerance
If anyone is interested in this topic, please aid in the development of its proper titles, details (for all drugs), most importantly, the drug statistics on the population involving their likelihood for a human body to not respond to it. Hopefully, we can approximate dose quantities that lead to these issues in the long term. Twillisjr ( talk) 21:27, 4 September 2018 (UTC)

I tried making diagram, to simplify the description of the relationship between the Kinetics and Dynamics. What do you guys think? Claes Lindhardt ( talk) 20:20, 16 January 2023 (UTC)

I found it confusing which metrics are used where I tried making this diagram where I tried associating the diffrent units with the place where at first glance it sounds like they might belong. But I would really appriacte any help to correct it :) I feel like it is very difficult to keep the overview of all the interplay when everything is only on list form. I can quickly adapt any change suggestions. Thank you in advance Claes Lindhardt ( talk) 20:40, 16 January 2023 (UTC)
- Hi Claes,
- I moved your two proposals to this new section.
- A picture is not always worth a thousand words. To get an idea how complicated it really is, see this presentation (slide 5) [1] – and that’s mainly the PK part…
- I appreciate your efforts but don’t think that your first image provides additional value to the article. Sorry. The labels you used in the second image are entirely wrong. All are PK metrics.
Alfie
↑↓
© 11:18, 17 January 2023 (UTC)
- Thank you very much for the feedback and specefied referance :). If you have the time to tell be where the thing in the second picture would ideally be. I would be more then happy to move them there.
- I will try to make a new version based on the linked slide. I think the connection between the diffrenet concepts under PK is not described in an accessible way. An i think some good diagrams could really help. Especially for people beeing presented to the theme for the first time. Claes Lindhardt ( talk) 19:59, 19 January 2023 (UTC)
References
- ^ Schütz, Helmut (December 15, 2021). "Bioequivalence. An Old Area with some Uncharted Territories" (PDF). Biometrical Colloquium. Retrieved January 17, 2013.
- C-Class vital articles
- Wikipedia level-4 vital articles
- Wikipedia vital articles in Biology and health sciences
- C-Class level-4 vital articles
- Wikipedia level-4 vital articles in Biology and health sciences
- C-Class vital articles in Biology and health sciences
- C-Class pharmacology articles
- Top-importance pharmacology articles
- WikiProject Pharmacology articles
- Pages translated from Spanish Wikipedia






