|
| This It is of interest to the following WikiProjects: | ||||||||||
| |||||||||||
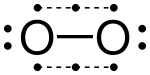
The article mentions that ozone does not have an octet...does this refer to one of the less stable resonance structures? I am assuming this is the case for carbon monoxide.-- GregRM 19:42, 26 March 2006 (UTC)
- This is wrong. I think the author is confused with the fact that atoms in those molecules have formal charge. But they definitely do follow the octet rule. -- Spoon! 20:08, 29 August 2006 (UTC)
http://elftor.com/elftor.php?number=189 he just made it up without knowing that it exists, right? -- Ruben 15:10, 27 June 2006 (UTC)
- Knowing Elftor, the author probably knew what the Octet rule and Grimm's Law were. However, as the strip has absolutely nothing to do with the Octet rule (or Grimm's Law), I don't think it really bears mention. - User:69.110.129.129 (Talk) 11 November 2006
Could anyone list a sound physical reason why atoms tend to react until their valence shell is full? I know I understood this once, probably after taking an QM class years ago, but I can't remember or find this information. -- User:67.169.18.217 (talk) 1 July 2006
- In short, electron pairs, acting as little spinning magnets, are favored due to "electron spin coupling" effects, in which 2, 4, 6, 8, 10, 12, etc., are all favorable shell filling numbers; however, the "crowding effect" of having too many negative charges, which are attracted to the positive nucleus, located in one area creates a break-up tendency such that a crowd of 8 is optimal, kind of similar to the 150 of the Dunbar's number phenomenon or similar to why elements past atomic number 92, i.e. uranium, become unstable. I hope this helps? -- Sadi Carnot 15:48, 20 December 2006 (UTC)
- This is simply untrue. Electrons infact unpair in atoms to form states with the highest spin (Hund's rule) e.g. Phosphurus with three electrons in its 3p shell has the three unpaired electrons (not two paired). Note (as my comment below) Hund's rule is not an explanation just a statement about the sign of the coupling between the electron spins (i.e. its ferromagnetic not antiferromagnetic). There are plenty of other examples in physics where antiferromagnetic couplings are the rule. For an explanation of Hund's rule look into so-called Fermi holes and Fermi heaps. Bcbccouk ( talk) 12:21, 2 July 2011 (UTC)
The article starts of well by saying the octet rule is a rule of thumb (i.e. not an explanation). But then, phrases like "A consequence of the octet rule" ruin the article for me. The rule is not based on physics but rather empirical observations. One should really be considering electron affinities and ionisation energies. For this reason "an element's valence shell is full and most stable when it contains eight electrons" is simply nonsense. For a start, stable compared to what? Bcbccouk ( talk) 12:21, 2 July 2011 (UTC)
- The rule started as a rule of thumb based on empirical observations (see History section), but is explained approximately by quantum mechanics. "Stable" in this context means stable with respect to gaining or losing an electron. An examination of ionization energies and electron affinities makes the discussion more precise but also more complicated.
- Yes, the rule is simple-minded and there are exceptions (noted in the article), but it does work well enough to be a rough guide to organic and main-group chemistry, which is why we still teach it. Dirac66 ( talk) 20:00, 2 July 2011 (UTC)
- I think that since the article is already going into discussion of p and s orbitals (for God knows what reason, I don't see how it helps), talk of IEs and EAs wouldn't be unwarranted (it's a simpler concept) --- its a more proper way of saying basically whats alrady being said. For (terrible) example, the first ionisation energy of Sodium is sufficiently small that this energy cost is recovered on forming a bond say with a chloride ion. In this sense its not "stable with respect to gaining or losing an electron" (energy needs to be put in to ionise Na), but rather stable with respect to the products of a reaction. What should be discussed in the article, and isn't, is the notion that further ionisation (or electron gain) is a prohibitively high energy process. The article should also make it more clear that there is nothing inherently "stable" about a full electron shell. Bcbccouk ( talk) 19:30, 8 July 2011 (UTC)
- I'm editing this article to make it factually correct. I will not make wholesale changes for the moment. I do think however some of the article is however misconceived and serves to proliferate misunderstanding. — Preceding
unsigned comment added by
Bcbccouk (
talk •
contribs)
20:05, 15 October 2011 (UTC)
- I partially agree with your changes. The use of the word "stable" to describe the valence shell was incorrect. Instead stable compounds of certain elements tend to contain valence shells with an octet of valence electrons, and explaining the energetics (thermodynamics) of this is an improvement.
- However I do not agree with removing all mention of s and p orbitals. It is true that Lewis and Langmuir originally formulated the rule empirically before quantum mechanics. At this time eight was just a mysterious magic number. However the modern understanding of the rule (and its exceptions) is based on the fact that it corresponds to an s2p6 configuration, as is now taught in introductory chemistry. So I think this should be restored to the article, though probably in a separate section after the energetics.
Dirac66 (
talk)
01:42, 17 October 2011 (UTC)
- I have sympathy for your position, allow me to comment. My main objection to the previous article was the notion of stability of filled shells - something which has been incorrectly taught to less able students in the UK for a long time. The express mention of filling of shells and the configurations is to the detriment of increasing understanding, if the article is not very carefully worded. It is not the filled shell which is important per say but where additionally ionised (added) electrons reside (will reside). There is nothing therefore magic about an s2p6 configuration. I agree talk of sub-orbitals does explain why its the octet (and not another multiplet) rule, but only in a superficial sense: why is there one s-orbital and three p-orbitals? This is best discussed elsewhere. Simply stating the maximum number of electrons in each shell (shell as understood by periods in the PT, not principle quantum number) seems the best thing to do - particularly given the likely level of readers of this article. Links can be made to other articles. I will nevertheless have a think about where it can be best placed.
Bcbccouk (
talk)
14:56, 17 October 2011 (UTC)
- The level of readers is probably quite variable, since the page view statistics show 17K views of the article in September. So I think at least some of them would benefit from a simple explanation of the relation of the octet rule to the next level of abstraction which is electron configurations. This can be placed in a separate section after (not before) a thermodynamically correct statement of the experimental facts (or tendencies). And we should take the time to word it carefully and avoid the previous mis-statements, which is why I have not simply added a hastily-written paragraph. As for why there are one s and three p orbitals, at this level I think we have to just say that it can be shown from the quantum theory, since clearly we don't want the Schrödinger equation in THIS article. But as long we mention electron configuration, the interested minority can follow the links from that article and eventually find more information about quantum mechanics. Dirac66 ( talk) 19:56, 17 October 2011 (UTC)
- I have sympathy for your position, allow me to comment. My main objection to the previous article was the notion of stability of filled shells - something which has been incorrectly taught to less able students in the UK for a long time. The express mention of filling of shells and the configurations is to the detriment of increasing understanding, if the article is not very carefully worded. It is not the filled shell which is important per say but where additionally ionised (added) electrons reside (will reside). There is nothing therefore magic about an s2p6 configuration. I agree talk of sub-orbitals does explain why its the octet (and not another multiplet) rule, but only in a superficial sense: why is there one s-orbital and three p-orbitals? This is best discussed elsewhere. Simply stating the maximum number of electrons in each shell (shell as understood by periods in the PT, not principle quantum number) seems the best thing to do - particularly given the likely level of readers of this article. Links can be made to other articles. I will nevertheless have a think about where it can be best placed.
Bcbccouk (
talk)
14:56, 17 October 2011 (UTC)
- I'm editing this article to make it factually correct. I will not make wholesale changes for the moment. I do think however some of the article is however misconceived and serves to proliferate misunderstanding. — Preceding
unsigned comment added by
Bcbccouk (
talk •
contribs)
20:05, 15 October 2011 (UTC)
Within the past few weeks, Bcbccouk has oversimplified the article by removing all mention of subshells. I don't think the octet rule can really be understood correctly, without understanding how subshells fit into the matter. As an example of how it doesn't work, consider the recently-introduced text "The second and third shells can contain at most 8 electrons. The third shell can contain 18 electrons." The two sentences not only contradict each other, but the problem can't be fixed by adjusting either one of the two contradictory sentences. Saying that the 3 shell can contain at most 8 electrons is incorrect, but saying that the 3 shell can contain 18 electrons gives no insight into understanding why 8 electrons in the 3 shell would be preferred. It really is helpful to bring up subshells, and explain that the 4s subshell fills before the 3d subshell.
To counter this, I am restoring the "Explanation in quantum theory" and "Exceptions" sections to what they were before Bcbccouk's first edit a few weeks ago, since I think the earlier versions of those sections, which don't shy away from discussing subshells, were better than what those sections had been edited into in recent weeks. Red Act ( talk) 00:30, 4 November 2011 (UTC)
- I agree that subshells are essential to this article. However Bcbccouk did have a valid point that the notion of a "stable" configuration is poorly defined. So after much thought I have now rewritten the section to refer instead to a "closed shell", with appropriate changes in the explanations. Dirac66 ( talk) 02:23, 21 November 2011 (UTC)
- Here's a quick few ramblings on the comments above, and the current state of the article. I made a couple of typos which has clearly confused the issue. I had meant to say the first shell can contain 2 electrons, the second and third 8, and the fourth 18. (As there are elements in the periodic table). This use of shell is that commonly understood at a secondary school level in the UK, not that formed by considering hydrogenic orbitals.
- I will except that talk of subshells is warranted, but only after establishing the basic empirical observations. As Dirac66 said previously it should go later in the article; being first as currently is, reinvorces (I believe) the notion that filled shells are inherently stabilised by some mysterious effect. This is simply not true and as I've said before I don't believe we should be proliferating such nonsense. Talk of subshells can be later in a sort of "modern justification" section which could then link into the exceptions to the rule. The exceptions are really just breakdowns of the idea that all shells have 8 electrons.
- As the article stands there is also a harsh juxtaposition between a very old, and in some sense outdated, observation/theory (the introduction) and a modern explanation. Perhaps the history section should come first?
- The statement "However the currently preferred model uses three-center four-electron bonding" in the exceptions section needs to be removed. Who is this preferred by for a start? These differing simple descriptions ultimately stem from a desire to simplify and interpret electronic structure calculations. I've not done such calculations myself but I suspect what is meant by "currently preferred" is that the d orbitals have a small coefficient in the bonding molecular orbitals, and as such the "use of d orbitals in bonding" is not in this sense a correct description.
Bcbccouk (
talk)
10:37, 30 November 2011 (UTC)
- Yes, I did suggest placing the quantum explanation later in the article. I now propose to re-order the existing sections as follows: Intro 1. Example NaCl (easy to understand on its own) 2. History 3. Quantum theory (after history to place it in context) 4. Exceptions (which require subshells for understanding).
- For 3c-4e bonding however, I think the explanation given goes far enough at the level of this article. The reader is invited to See also the article on Hypervalent molecule, where a more advanced explanation is given with mention of ab initio calculations and a reference to a 1990 JACS article.
Dirac66 (
talk)
04:22, 1 December 2011 (UTC)
- Those changes sound eminently sensible. I still think "the currently preferred model" is a subjective statement. The best description would be (forgive the flippancy, but you'll understand my point), a bunch of electrons associated with a bunch of atomic nuclei --- nature does not care for how we like to partition systems. I would suggest saying "another model" in place of the above. Bcbccouk ( talk) 20:09, 2 December 2011 (UTC)
- The statement "However the currently preferred model uses three-center four-electron bonding" in the exceptions section needs to be removed. Who is this preferred by for a start? These differing simple descriptions ultimately stem from a desire to simplify and interpret electronic structure calculations. I've not done such calculations myself but I suspect what is meant by "currently preferred" is that the d orbitals have a small coefficient in the bonding molecular orbitals, and as such the "use of d orbitals in bonding" is not in this sense a correct description.
Bcbccouk (
talk)
10:37, 30 November 2011 (UTC)
I have a real problem with the statement that PCl5 conforms to the octet rule. This claim in the "Disputed' section REQUIRES a valid reference. Why? Because you can't multiply 5 by 2 and get 8; that is: on its face, Anything bonded to 5 Cl atoms can NOT obey the Octet rule - as a matter of fact Any-one-Atom bonded to 5 ANYTHING fails since a 'bond' is, by defintion, sharing 2 electrons. Next time I am passing this way, if the offending section is not cleaned up/justified, I will remove it. ... I also have a problem with the lede that claims the rule is explicitly limited to "atoms of low (<20) atomic number". It is NOT. It often fails as At. No. increases, so what? It remains the Best 'first guess'. Upon closer reading, this whole article is a mess. It mentions a dodectet rule, but there is no such thing. (That is, unless a single reference to an article should be weighed equally with decades of references to ONLY the octet rule?) It fails COMPLETELY to provide ANY good references to any of the myriad of text books that define the rule. What happened to the old article? I know that this used to be better than this current crud. Wikipedia seems to be declining in quality. The octet rule hasn't changed for decades. We now better understand where it is applicable and where it fails, but this is the context of the Rule, not the rule itself. It is, unless things have changed dramatically since I was an undergrad (?), a SIMPLE way to sketch out the Lewis Structure of a molecule or ion. If you need to run a computational model in order to determine the sketch, then whatever you have done is not the Octet Rule. (Although may be consistent with it). To summarize, this article has lost sight of the purpose of the rule, which is PREDICTION. Abitslow ( talk) 18:47, 16 May 2014 (UTC)
- It is true that the sections you have criticized need some rewording and clarification, as well as references. However an encyclopedia article should not only present the original rule, but also assess its validity in the light of current knowledge. The point for PCl5 is that the structure with five covalent bonds is now known to be a poor description of the molecule. Better descriptions use resonance structures which each have four covalent and one ionic bond, or else three-centre four-electron bonds. I will try to find time to improve the article in order to explain this properly and add more references. Dirac66 ( talk) 00:43, 17 May 2014 (UTC)
The following claim in this article is problematic: "Stable molecular radicals (e.g. nitric oxide, NO) do obtain octet configurations by means of a three-electron bond which contributes one shared and one unshared electron to the octet of each bonded atom." The Wikipedia article on "Covalent bond" adds "Another example of a molecule containing a 3-electron bond, in addition to two 2-electron bonds, is nitric oxide, NO." If the atoms (N and O) thus have 2 + 2 + 3 = 7 electrons in the bonds, there should be one non-bonding electron on the nitrogen and three non-bonding electrons on the oxygen of NO, to give them the octet. This sounds absurd. I am not sure how one can divide 11 valence electrons of NO in two octets, and there is no electron-dot diagram for NO in the article to illustrate this. Furthermore, there are no 3-electron bonds in Lewis structures. The octet rule is an expectation that the electrons in a Lewis structure can be arranged to give every p-block atom 8 e- in bonds & lone pairs. This is impossible in the Lewis structure of NO. Dmitri Goussev (Dmitry G. Gusev), Professor of Chemistry, Dept. of Chemistry and Biochemistry, Wilfrid Laurier University, Waterloo, Ontario, CANADA, Researcher ID: H-3775-2013.

- To divide 11 valence electrons into two octets, you need to count 5 electrons twice. For the 2-electron C-H bond, the two shared electrons are of course counted for both C and H. For the 3-electron N-O bond, the electron on the left is counted for N only, the electron on the right is counted for O only, and the shared electron is counted for both N and O. So these 3 electrons correspond to 2 on N and 2 on O, with 1 electron double-counted.

- Then as shown in the second diagram here (from the article Nitric oxide), there are two normal covalent bonds (solid lines) in addition to the 3-electron bond (dotted line). The three bonds correspond to six electrons on each atom (two from each bond), but the total number of electrons shown is only 2+2+3 = 7 and we have counted 5 electrons twice.
- Finally the two octets are completed by placing 2 electrons (a lone pair) on each atom, not 1 on N and 3 on O. This makes a total of 7+2+2 = 11 valence electrons. I agree that a more complete diagram showing all 11 valence electrons would be preferable.
- As to whether there are 3-electron bonds in Lewis structures, this is a question of definition. I do not know whether G.N. Lewis personally included them in any diagrams, but if not we can consider diagrams with 3-electron bonds as generalized Lewis structures.
Dirac66 (
talk)
02:00, 7 April 2016 (UTC)
- Added reference for the use of the 3-electron bond in Lewis structures. They are however referred to as valence-bond structures in the book. I think I first read that idea (that I made the image for) in the first edition of the same book.--
Officer781 (
talk)
14:12, 11 April 2016 (UTC)
- I have now explained the electron counting for NO and also O2 in the article, in the new section about 3-electron bonds added by Officer781 (which I have renamed).
Dirac66 (
talk)
02:45, 14 April 2016 (UTC)
- @ Dirac66: I know this is long overdue, but I've added simple "Lewis-like" images showing the three-electron bonds in NO and O2:
- I have now explained the electron counting for NO and also O2 in the article, in the new section about 3-electron bonds added by Officer781 (which I have renamed).
Dirac66 (
talk)
02:45, 14 April 2016 (UTC)
- Added reference for the use of the 3-electron bond in Lewis structures. They are however referred to as valence-bond structures in the book. I think I first read that idea (that I made the image for) in the first edition of the same book.--
Officer781 (
talk)
14:12, 11 April 2016 (UTC)
- If you have any suggestions for the two images please let me know. Thanks!--
Officer781 (
talk)
13:31, 19 February 2019 (UTC)
- I think the two added images are good. As we now have 3 different types of image in this section, perhaps we should add a few words to distinguish the 3 types: one-bond Lewis diagrams, one-bond MO diagrams, and all-valence-electron Lewis diagrams.
Dirac66 (
talk)
01:55, 20 February 2019 (UTC)
- @
Dirac66: Should I change the examples in the one-bond diagrams to H2 and He2+ instead so it becomes all-valence-electron diagrams effectively? I note that a number of sources use these two molecules to introduce the respective bonds.--
Officer781 (
talk)
14:30, 20 February 2019 (UTC)
- Perhaps it would be better to start with H2 and He2+ as the simplest examples, and then show the NO and CO diagrams with a comment that the 3-electron bond in NO is similar to the one in He2+.
Dirac66 (
talk)
21:04, 20 February 2019 (UTC)
- I felt that that might be a bit too complicated for this article and I assume readers will be able to isolate a single bond from the molecule? I've changed the image caption to say that Lewis and MO diagrams are involved.--
Officer781 (
talk)
00:44, 21 February 2019 (UTC)
- OK, thanks. I have made very minor changes only - 1 link and 1 plural. Dirac66 ( talk) 01:26, 21 February 2019 (UTC)
- I felt that that might be a bit too complicated for this article and I assume readers will be able to isolate a single bond from the molecule? I've changed the image caption to say that Lewis and MO diagrams are involved.--
Officer781 (
talk)
00:44, 21 February 2019 (UTC)
- Perhaps it would be better to start with H2 and He2+ as the simplest examples, and then show the NO and CO diagrams with a comment that the 3-electron bond in NO is similar to the one in He2+.
Dirac66 (
talk)
21:04, 20 February 2019 (UTC)
- @
Dirac66: Should I change the examples in the one-bond diagrams to H2 and He2+ instead so it becomes all-valence-electron diagrams effectively? I note that a number of sources use these two molecules to introduce the respective bonds.--
Officer781 (
talk)
14:30, 20 February 2019 (UTC)
- I think the two added images are good. As we now have 3 different types of image in this section, perhaps we should add a few words to distinguish the 3 types: one-bond Lewis diagrams, one-bond MO diagrams, and all-valence-electron Lewis diagrams.
Dirac66 (
talk)
01:55, 20 February 2019 (UTC)
- If you have any suggestions for the two images please let me know. Thanks!--
Officer781 (
talk)
13:31, 19 February 2019 (UTC)
- The following discussion is closed. Please do not modify it. Subsequent comments should be made in a new section. A summary of the conclusions reached follows.
- The result of this discussion was merge and change to disambiguation. Officer781 ( talk) 03:01, 28 January 2019 (UTC)
@ Dirac66: @ Bduke: @ Alsosaid1987: Isn't noble gas configuration really just the octet rule?-- Officer781 ( talk) 07:52, 26 January 2019 (UTC)
Yes, it is a historical name used for the octet rule. As explained in the article Noble gas configuration, it was the phrase used in 1916 by Lewis and Kossel.
Support. I would agree to a merger into Octet rule, with the extra content of Noble gas configuration added to the History section, and a redirect from Noble gas configuration. Dirac66 ( talk) 20:25, 26 January 2019 (UTC)
- On second thought I would merge the content into octet rule but turn the page into a disambiguation that also refers to the 18-electron rule since that page also references noble gases.-- Officer781 ( talk) 01:11, 27 January 2019 (UTC)
This article's "History" section attributes this quote:
"During the formation of a chemical bond, atoms combine together by gaining, losing or sharing electrons in such a way that they acquire nearest noble gas configuration."
to: Lewis, Gilbert N. (1916). "The Atom and the Molecule". Journal of the American Chemical Society. 38 (4): 762–785.
However, this quote appears nowhere in that article by Lewis.
The quote could not have come from Walther Kossel's article because Kossel's article was written in German.
VexorAbVikipædia ( talk) 23:24, 28 July 2019 (UTC)
Octet rule 119.2.125.162 ( talk) 14:38, 5 August 2022 (UTC)
- Start-Class vital articles
- Wikipedia level-5 vital articles
- Wikipedia vital articles in Physical sciences
- Start-Class level-5 vital articles
- Wikipedia level-5 vital articles in Physical sciences
- Start-Class vital articles in Physical sciences
- Start-Class Chemistry articles
- High-importance Chemistry articles
- WikiProject Chemistry articles
|
| This It is of interest to the following WikiProjects: | ||||||||||
| |||||||||||
The article mentions that ozone does not have an octet...does this refer to one of the less stable resonance structures? I am assuming this is the case for carbon monoxide.-- GregRM 19:42, 26 March 2006 (UTC)
- This is wrong. I think the author is confused with the fact that atoms in those molecules have formal charge. But they definitely do follow the octet rule. -- Spoon! 20:08, 29 August 2006 (UTC)
http://elftor.com/elftor.php?number=189 he just made it up without knowing that it exists, right? -- Ruben 15:10, 27 June 2006 (UTC)
- Knowing Elftor, the author probably knew what the Octet rule and Grimm's Law were. However, as the strip has absolutely nothing to do with the Octet rule (or Grimm's Law), I don't think it really bears mention. - User:69.110.129.129 (Talk) 11 November 2006
Could anyone list a sound physical reason why atoms tend to react until their valence shell is full? I know I understood this once, probably after taking an QM class years ago, but I can't remember or find this information. -- User:67.169.18.217 (talk) 1 July 2006
- In short, electron pairs, acting as little spinning magnets, are favored due to "electron spin coupling" effects, in which 2, 4, 6, 8, 10, 12, etc., are all favorable shell filling numbers; however, the "crowding effect" of having too many negative charges, which are attracted to the positive nucleus, located in one area creates a break-up tendency such that a crowd of 8 is optimal, kind of similar to the 150 of the Dunbar's number phenomenon or similar to why elements past atomic number 92, i.e. uranium, become unstable. I hope this helps? -- Sadi Carnot 15:48, 20 December 2006 (UTC)
- This is simply untrue. Electrons infact unpair in atoms to form states with the highest spin (Hund's rule) e.g. Phosphurus with three electrons in its 3p shell has the three unpaired electrons (not two paired). Note (as my comment below) Hund's rule is not an explanation just a statement about the sign of the coupling between the electron spins (i.e. its ferromagnetic not antiferromagnetic). There are plenty of other examples in physics where antiferromagnetic couplings are the rule. For an explanation of Hund's rule look into so-called Fermi holes and Fermi heaps. Bcbccouk ( talk) 12:21, 2 July 2011 (UTC)
The article starts of well by saying the octet rule is a rule of thumb (i.e. not an explanation). But then, phrases like "A consequence of the octet rule" ruin the article for me. The rule is not based on physics but rather empirical observations. One should really be considering electron affinities and ionisation energies. For this reason "an element's valence shell is full and most stable when it contains eight electrons" is simply nonsense. For a start, stable compared to what? Bcbccouk ( talk) 12:21, 2 July 2011 (UTC)
- The rule started as a rule of thumb based on empirical observations (see History section), but is explained approximately by quantum mechanics. "Stable" in this context means stable with respect to gaining or losing an electron. An examination of ionization energies and electron affinities makes the discussion more precise but also more complicated.
- Yes, the rule is simple-minded and there are exceptions (noted in the article), but it does work well enough to be a rough guide to organic and main-group chemistry, which is why we still teach it. Dirac66 ( talk) 20:00, 2 July 2011 (UTC)
- I think that since the article is already going into discussion of p and s orbitals (for God knows what reason, I don't see how it helps), talk of IEs and EAs wouldn't be unwarranted (it's a simpler concept) --- its a more proper way of saying basically whats alrady being said. For (terrible) example, the first ionisation energy of Sodium is sufficiently small that this energy cost is recovered on forming a bond say with a chloride ion. In this sense its not "stable with respect to gaining or losing an electron" (energy needs to be put in to ionise Na), but rather stable with respect to the products of a reaction. What should be discussed in the article, and isn't, is the notion that further ionisation (or electron gain) is a prohibitively high energy process. The article should also make it more clear that there is nothing inherently "stable" about a full electron shell. Bcbccouk ( talk) 19:30, 8 July 2011 (UTC)
- I'm editing this article to make it factually correct. I will not make wholesale changes for the moment. I do think however some of the article is however misconceived and serves to proliferate misunderstanding. — Preceding
unsigned comment added by
Bcbccouk (
talk •
contribs)
20:05, 15 October 2011 (UTC)
- I partially agree with your changes. The use of the word "stable" to describe the valence shell was incorrect. Instead stable compounds of certain elements tend to contain valence shells with an octet of valence electrons, and explaining the energetics (thermodynamics) of this is an improvement.
- However I do not agree with removing all mention of s and p orbitals. It is true that Lewis and Langmuir originally formulated the rule empirically before quantum mechanics. At this time eight was just a mysterious magic number. However the modern understanding of the rule (and its exceptions) is based on the fact that it corresponds to an s2p6 configuration, as is now taught in introductory chemistry. So I think this should be restored to the article, though probably in a separate section after the energetics.
Dirac66 (
talk)
01:42, 17 October 2011 (UTC)
- I have sympathy for your position, allow me to comment. My main objection to the previous article was the notion of stability of filled shells - something which has been incorrectly taught to less able students in the UK for a long time. The express mention of filling of shells and the configurations is to the detriment of increasing understanding, if the article is not very carefully worded. It is not the filled shell which is important per say but where additionally ionised (added) electrons reside (will reside). There is nothing therefore magic about an s2p6 configuration. I agree talk of sub-orbitals does explain why its the octet (and not another multiplet) rule, but only in a superficial sense: why is there one s-orbital and three p-orbitals? This is best discussed elsewhere. Simply stating the maximum number of electrons in each shell (shell as understood by periods in the PT, not principle quantum number) seems the best thing to do - particularly given the likely level of readers of this article. Links can be made to other articles. I will nevertheless have a think about where it can be best placed.
Bcbccouk (
talk)
14:56, 17 October 2011 (UTC)
- The level of readers is probably quite variable, since the page view statistics show 17K views of the article in September. So I think at least some of them would benefit from a simple explanation of the relation of the octet rule to the next level of abstraction which is electron configurations. This can be placed in a separate section after (not before) a thermodynamically correct statement of the experimental facts (or tendencies). And we should take the time to word it carefully and avoid the previous mis-statements, which is why I have not simply added a hastily-written paragraph. As for why there are one s and three p orbitals, at this level I think we have to just say that it can be shown from the quantum theory, since clearly we don't want the Schrödinger equation in THIS article. But as long we mention electron configuration, the interested minority can follow the links from that article and eventually find more information about quantum mechanics. Dirac66 ( talk) 19:56, 17 October 2011 (UTC)
- I have sympathy for your position, allow me to comment. My main objection to the previous article was the notion of stability of filled shells - something which has been incorrectly taught to less able students in the UK for a long time. The express mention of filling of shells and the configurations is to the detriment of increasing understanding, if the article is not very carefully worded. It is not the filled shell which is important per say but where additionally ionised (added) electrons reside (will reside). There is nothing therefore magic about an s2p6 configuration. I agree talk of sub-orbitals does explain why its the octet (and not another multiplet) rule, but only in a superficial sense: why is there one s-orbital and three p-orbitals? This is best discussed elsewhere. Simply stating the maximum number of electrons in each shell (shell as understood by periods in the PT, not principle quantum number) seems the best thing to do - particularly given the likely level of readers of this article. Links can be made to other articles. I will nevertheless have a think about where it can be best placed.
Bcbccouk (
talk)
14:56, 17 October 2011 (UTC)
- I'm editing this article to make it factually correct. I will not make wholesale changes for the moment. I do think however some of the article is however misconceived and serves to proliferate misunderstanding. — Preceding
unsigned comment added by
Bcbccouk (
talk •
contribs)
20:05, 15 October 2011 (UTC)
Within the past few weeks, Bcbccouk has oversimplified the article by removing all mention of subshells. I don't think the octet rule can really be understood correctly, without understanding how subshells fit into the matter. As an example of how it doesn't work, consider the recently-introduced text "The second and third shells can contain at most 8 electrons. The third shell can contain 18 electrons." The two sentences not only contradict each other, but the problem can't be fixed by adjusting either one of the two contradictory sentences. Saying that the 3 shell can contain at most 8 electrons is incorrect, but saying that the 3 shell can contain 18 electrons gives no insight into understanding why 8 electrons in the 3 shell would be preferred. It really is helpful to bring up subshells, and explain that the 4s subshell fills before the 3d subshell.
To counter this, I am restoring the "Explanation in quantum theory" and "Exceptions" sections to what they were before Bcbccouk's first edit a few weeks ago, since I think the earlier versions of those sections, which don't shy away from discussing subshells, were better than what those sections had been edited into in recent weeks. Red Act ( talk) 00:30, 4 November 2011 (UTC)
- I agree that subshells are essential to this article. However Bcbccouk did have a valid point that the notion of a "stable" configuration is poorly defined. So after much thought I have now rewritten the section to refer instead to a "closed shell", with appropriate changes in the explanations. Dirac66 ( talk) 02:23, 21 November 2011 (UTC)
- Here's a quick few ramblings on the comments above, and the current state of the article. I made a couple of typos which has clearly confused the issue. I had meant to say the first shell can contain 2 electrons, the second and third 8, and the fourth 18. (As there are elements in the periodic table). This use of shell is that commonly understood at a secondary school level in the UK, not that formed by considering hydrogenic orbitals.
- I will except that talk of subshells is warranted, but only after establishing the basic empirical observations. As Dirac66 said previously it should go later in the article; being first as currently is, reinvorces (I believe) the notion that filled shells are inherently stabilised by some mysterious effect. This is simply not true and as I've said before I don't believe we should be proliferating such nonsense. Talk of subshells can be later in a sort of "modern justification" section which could then link into the exceptions to the rule. The exceptions are really just breakdowns of the idea that all shells have 8 electrons.
- As the article stands there is also a harsh juxtaposition between a very old, and in some sense outdated, observation/theory (the introduction) and a modern explanation. Perhaps the history section should come first?
- The statement "However the currently preferred model uses three-center four-electron bonding" in the exceptions section needs to be removed. Who is this preferred by for a start? These differing simple descriptions ultimately stem from a desire to simplify and interpret electronic structure calculations. I've not done such calculations myself but I suspect what is meant by "currently preferred" is that the d orbitals have a small coefficient in the bonding molecular orbitals, and as such the "use of d orbitals in bonding" is not in this sense a correct description.
Bcbccouk (
talk)
10:37, 30 November 2011 (UTC)
- Yes, I did suggest placing the quantum explanation later in the article. I now propose to re-order the existing sections as follows: Intro 1. Example NaCl (easy to understand on its own) 2. History 3. Quantum theory (after history to place it in context) 4. Exceptions (which require subshells for understanding).
- For 3c-4e bonding however, I think the explanation given goes far enough at the level of this article. The reader is invited to See also the article on Hypervalent molecule, where a more advanced explanation is given with mention of ab initio calculations and a reference to a 1990 JACS article.
Dirac66 (
talk)
04:22, 1 December 2011 (UTC)
- Those changes sound eminently sensible. I still think "the currently preferred model" is a subjective statement. The best description would be (forgive the flippancy, but you'll understand my point), a bunch of electrons associated with a bunch of atomic nuclei --- nature does not care for how we like to partition systems. I would suggest saying "another model" in place of the above. Bcbccouk ( talk) 20:09, 2 December 2011 (UTC)
- The statement "However the currently preferred model uses three-center four-electron bonding" in the exceptions section needs to be removed. Who is this preferred by for a start? These differing simple descriptions ultimately stem from a desire to simplify and interpret electronic structure calculations. I've not done such calculations myself but I suspect what is meant by "currently preferred" is that the d orbitals have a small coefficient in the bonding molecular orbitals, and as such the "use of d orbitals in bonding" is not in this sense a correct description.
Bcbccouk (
talk)
10:37, 30 November 2011 (UTC)
I have a real problem with the statement that PCl5 conforms to the octet rule. This claim in the "Disputed' section REQUIRES a valid reference. Why? Because you can't multiply 5 by 2 and get 8; that is: on its face, Anything bonded to 5 Cl atoms can NOT obey the Octet rule - as a matter of fact Any-one-Atom bonded to 5 ANYTHING fails since a 'bond' is, by defintion, sharing 2 electrons. Next time I am passing this way, if the offending section is not cleaned up/justified, I will remove it. ... I also have a problem with the lede that claims the rule is explicitly limited to "atoms of low (<20) atomic number". It is NOT. It often fails as At. No. increases, so what? It remains the Best 'first guess'. Upon closer reading, this whole article is a mess. It mentions a dodectet rule, but there is no such thing. (That is, unless a single reference to an article should be weighed equally with decades of references to ONLY the octet rule?) It fails COMPLETELY to provide ANY good references to any of the myriad of text books that define the rule. What happened to the old article? I know that this used to be better than this current crud. Wikipedia seems to be declining in quality. The octet rule hasn't changed for decades. We now better understand where it is applicable and where it fails, but this is the context of the Rule, not the rule itself. It is, unless things have changed dramatically since I was an undergrad (?), a SIMPLE way to sketch out the Lewis Structure of a molecule or ion. If you need to run a computational model in order to determine the sketch, then whatever you have done is not the Octet Rule. (Although may be consistent with it). To summarize, this article has lost sight of the purpose of the rule, which is PREDICTION. Abitslow ( talk) 18:47, 16 May 2014 (UTC)
- It is true that the sections you have criticized need some rewording and clarification, as well as references. However an encyclopedia article should not only present the original rule, but also assess its validity in the light of current knowledge. The point for PCl5 is that the structure with five covalent bonds is now known to be a poor description of the molecule. Better descriptions use resonance structures which each have four covalent and one ionic bond, or else three-centre four-electron bonds. I will try to find time to improve the article in order to explain this properly and add more references. Dirac66 ( talk) 00:43, 17 May 2014 (UTC)
The following claim in this article is problematic: "Stable molecular radicals (e.g. nitric oxide, NO) do obtain octet configurations by means of a three-electron bond which contributes one shared and one unshared electron to the octet of each bonded atom." The Wikipedia article on "Covalent bond" adds "Another example of a molecule containing a 3-electron bond, in addition to two 2-electron bonds, is nitric oxide, NO." If the atoms (N and O) thus have 2 + 2 + 3 = 7 electrons in the bonds, there should be one non-bonding electron on the nitrogen and three non-bonding electrons on the oxygen of NO, to give them the octet. This sounds absurd. I am not sure how one can divide 11 valence electrons of NO in two octets, and there is no electron-dot diagram for NO in the article to illustrate this. Furthermore, there are no 3-electron bonds in Lewis structures. The octet rule is an expectation that the electrons in a Lewis structure can be arranged to give every p-block atom 8 e- in bonds & lone pairs. This is impossible in the Lewis structure of NO. Dmitri Goussev (Dmitry G. Gusev), Professor of Chemistry, Dept. of Chemistry and Biochemistry, Wilfrid Laurier University, Waterloo, Ontario, CANADA, Researcher ID: H-3775-2013.

- To divide 11 valence electrons into two octets, you need to count 5 electrons twice. For the 2-electron C-H bond, the two shared electrons are of course counted for both C and H. For the 3-electron N-O bond, the electron on the left is counted for N only, the electron on the right is counted for O only, and the shared electron is counted for both N and O. So these 3 electrons correspond to 2 on N and 2 on O, with 1 electron double-counted.

- Then as shown in the second diagram here (from the article Nitric oxide), there are two normal covalent bonds (solid lines) in addition to the 3-electron bond (dotted line). The three bonds correspond to six electrons on each atom (two from each bond), but the total number of electrons shown is only 2+2+3 = 7 and we have counted 5 electrons twice.
- Finally the two octets are completed by placing 2 electrons (a lone pair) on each atom, not 1 on N and 3 on O. This makes a total of 7+2+2 = 11 valence electrons. I agree that a more complete diagram showing all 11 valence electrons would be preferable.
- As to whether there are 3-electron bonds in Lewis structures, this is a question of definition. I do not know whether G.N. Lewis personally included them in any diagrams, but if not we can consider diagrams with 3-electron bonds as generalized Lewis structures.
Dirac66 (
talk)
02:00, 7 April 2016 (UTC)
- Added reference for the use of the 3-electron bond in Lewis structures. They are however referred to as valence-bond structures in the book. I think I first read that idea (that I made the image for) in the first edition of the same book.--
Officer781 (
talk)
14:12, 11 April 2016 (UTC)
- I have now explained the electron counting for NO and also O2 in the article, in the new section about 3-electron bonds added by Officer781 (which I have renamed).
Dirac66 (
talk)
02:45, 14 April 2016 (UTC)
- @ Dirac66: I know this is long overdue, but I've added simple "Lewis-like" images showing the three-electron bonds in NO and O2:
- I have now explained the electron counting for NO and also O2 in the article, in the new section about 3-electron bonds added by Officer781 (which I have renamed).
Dirac66 (
talk)
02:45, 14 April 2016 (UTC)
- Added reference for the use of the 3-electron bond in Lewis structures. They are however referred to as valence-bond structures in the book. I think I first read that idea (that I made the image for) in the first edition of the same book.--
Officer781 (
talk)
14:12, 11 April 2016 (UTC)
- If you have any suggestions for the two images please let me know. Thanks!--
Officer781 (
talk)
13:31, 19 February 2019 (UTC)
- I think the two added images are good. As we now have 3 different types of image in this section, perhaps we should add a few words to distinguish the 3 types: one-bond Lewis diagrams, one-bond MO diagrams, and all-valence-electron Lewis diagrams.
Dirac66 (
talk)
01:55, 20 February 2019 (UTC)
- @
Dirac66: Should I change the examples in the one-bond diagrams to H2 and He2+ instead so it becomes all-valence-electron diagrams effectively? I note that a number of sources use these two molecules to introduce the respective bonds.--
Officer781 (
talk)
14:30, 20 February 2019 (UTC)
- Perhaps it would be better to start with H2 and He2+ as the simplest examples, and then show the NO and CO diagrams with a comment that the 3-electron bond in NO is similar to the one in He2+.
Dirac66 (
talk)
21:04, 20 February 2019 (UTC)
- I felt that that might be a bit too complicated for this article and I assume readers will be able to isolate a single bond from the molecule? I've changed the image caption to say that Lewis and MO diagrams are involved.--
Officer781 (
talk)
00:44, 21 February 2019 (UTC)
- OK, thanks. I have made very minor changes only - 1 link and 1 plural. Dirac66 ( talk) 01:26, 21 February 2019 (UTC)
- I felt that that might be a bit too complicated for this article and I assume readers will be able to isolate a single bond from the molecule? I've changed the image caption to say that Lewis and MO diagrams are involved.--
Officer781 (
talk)
00:44, 21 February 2019 (UTC)
- Perhaps it would be better to start with H2 and He2+ as the simplest examples, and then show the NO and CO diagrams with a comment that the 3-electron bond in NO is similar to the one in He2+.
Dirac66 (
talk)
21:04, 20 February 2019 (UTC)
- @
Dirac66: Should I change the examples in the one-bond diagrams to H2 and He2+ instead so it becomes all-valence-electron diagrams effectively? I note that a number of sources use these two molecules to introduce the respective bonds.--
Officer781 (
talk)
14:30, 20 February 2019 (UTC)
- I think the two added images are good. As we now have 3 different types of image in this section, perhaps we should add a few words to distinguish the 3 types: one-bond Lewis diagrams, one-bond MO diagrams, and all-valence-electron Lewis diagrams.
Dirac66 (
talk)
01:55, 20 February 2019 (UTC)
- If you have any suggestions for the two images please let me know. Thanks!--
Officer781 (
talk)
13:31, 19 February 2019 (UTC)
- The following discussion is closed. Please do not modify it. Subsequent comments should be made in a new section. A summary of the conclusions reached follows.
- The result of this discussion was merge and change to disambiguation. Officer781 ( talk) 03:01, 28 January 2019 (UTC)
@ Dirac66: @ Bduke: @ Alsosaid1987: Isn't noble gas configuration really just the octet rule?-- Officer781 ( talk) 07:52, 26 January 2019 (UTC)
Yes, it is a historical name used for the octet rule. As explained in the article Noble gas configuration, it was the phrase used in 1916 by Lewis and Kossel.
Support. I would agree to a merger into Octet rule, with the extra content of Noble gas configuration added to the History section, and a redirect from Noble gas configuration. Dirac66 ( talk) 20:25, 26 January 2019 (UTC)
- On second thought I would merge the content into octet rule but turn the page into a disambiguation that also refers to the 18-electron rule since that page also references noble gases.-- Officer781 ( talk) 01:11, 27 January 2019 (UTC)
This article's "History" section attributes this quote:
"During the formation of a chemical bond, atoms combine together by gaining, losing or sharing electrons in such a way that they acquire nearest noble gas configuration."
to: Lewis, Gilbert N. (1916). "The Atom and the Molecule". Journal of the American Chemical Society. 38 (4): 762–785.
However, this quote appears nowhere in that article by Lewis.
The quote could not have come from Walther Kossel's article because Kossel's article was written in German.
VexorAbVikipædia ( talk) 23:24, 28 July 2019 (UTC)
Octet rule 119.2.125.162 ( talk) 14:38, 5 August 2022 (UTC)
- Start-Class vital articles
- Wikipedia level-5 vital articles
- Wikipedia vital articles in Physical sciences
- Start-Class level-5 vital articles
- Wikipedia level-5 vital articles in Physical sciences
- Start-Class vital articles in Physical sciences
- Start-Class Chemistry articles
- High-importance Chemistry articles
- WikiProject Chemistry articles